Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Puberty in humans is defined as the period of first becoming capable of reproducing and is distinguished by maturation of the genital organs, development of secondary sex characteristics, acceleration in linear growth velocity, changes in affect, and the occurrence of menarche in the female. In humans, the transition into puberty is driven by two physiological processes: gonadarche and adrenarche. Gonadarche comprises the growth and maturation of the gonads and is associated with increased secretion of sex steroids and with the initiation of folliculogenesis and ovulation in the female and spermatogenesis in the male. Gonadarche is responsible for thelarche and menarche in girls and testicular enlargement in boys.
Adrenarche , which typically precedes gonadarche, is associated with increased secretion of adrenal androgens and leads to the appearance of sexual hair (pubarche). The major adrenal bioactive C19 androgens include dehydroepiandrosterone (DHEA), dehydroepiandrosterone sulfate (DHEAS), and androstenedione. Adrenarche occurring late in prepubertal development appears to be unique to our own species and to the great apes. Additionally, in humans, the absence of adrenarche does not prevent gonadarche or the attainment of fertility.
The age at onset of puberty and the tempo at which puberty progresses depends on many factors. In girls, increased ovarian and adrenal sex steroid secretion leads to the physical manifestations of puberty, thelarche, and pubarche. In general, these changes occur between 8 and 13 years of age. The mean age at menarche among multiple ethnic groups is between 12 and 13 years old. In boys, the earliest physical manifestation of puberty is an increase in testicular volume, and this usually occurs between 9 and 14 years of age.
The chronological age at the onset of puberty may be associated with health consequences later in adulthood. Younger age at menarche generally means a longer period of exposure to estrogen. Adverse consequences of this long exposure to estrogen include increased risks for endometrial and breast cancers. Younger age at menarche has also been associated with increased risk for development of the metabolic syndrome. In the longitudinal 25-year Coronary Artery Risk Development in Young Adults (CARDIA) study, which included both Caucasian and African-American women, the excess adiposity associated with earlier age at menarche was sustained through adulthood. In the 1966 Finland Birth Cohort study involving 2417 males and 2641 females, pubertal timing as estimated by pubertal height growth influenced adult metabolic outcome markers, such as LDL cholesterol, fasting insulin, and fasting glucose. Prepubertal growth explained 19% to 25% of adult body mass index (BMI) variation in this cohort of men and women. The large cross-sectional UK Biobank study reported associations between pubertal timing and subsequent health outcomes. These data showing associations among prepubertal growth, pubertal timing, and adult metabolic outcomes suggest that mechanisms advancing puberty may also contribute to adult metabolic disorders. Additional studies are needed to validate these epidemiological associations.
Not surprisingly, later age at menarche is associated with increased risks for osteopenia and osteoporotic fractures. Possible explanations are that a shorter duration of estrogen exposure predisposes to decreased bone mineral density or that girls with lower body mass tend to experience both later menarche and decreased bone mineral accrual. Areal bone mineral density (BMD) was already decreased during the prepubertal years in girls experiencing later puberty, suggesting that common genetic variants influence both BMD and the timing of puberty. , A greater understanding of the mechanisms influencing the timing of puberty might lead to the development of specific prevention strategies for disorders such as osteopenia/osteoporosis.
Traditionally, the diagnosis of precocious puberty is considered when signs of puberty develop prior to 8 years of age in girls and 9.5 years in boys, but these criteria should be used as guidelines to complement the evaluation of individual patients. , For girls, the absence of thelarche or menarche by age 13 and 16 years, respectively, is considered to be delayed puberty. For boys, delayed puberty is defined as the absence of testicular enlargement by age 14. These ages represent 2.5 to 3 standard deviations (SDs) below and above the mean age of puberty as defined by population studies.
While the mechanism underlying the onset of adrenarche remains to be elucidated, it is now established that gonadarche results from the resurgence of activity in the hypothalamic-pituitary axis, after a relatively quiescent period following the minipuberty of infancy. The neuroendocrine regulation of gonadarche in humans is similar to that observed in other higher primates. , Nonhuman primates (in particular, the rhesus monkey) have been extensively employed as paradigms for the study of human puberty. Subsequently, our discussion of the control of the onset of gonadarche will be based on both the human and nonhuman primate literature.
A secular trend for earlier age at menarche in European and North American girls has been observed.
A similar trend for earlier pubertal development, albeit with a smaller magnitude, has been observed also in boys.
The National Health and Nutrition Examination Survey (NHANES) III study based on data collected between 1988 and 1994 showed that among American girls, the mean ages for breast development and menarche were 9.5 and 12.1 for non-Hispanic black (NHB); 9.8 and 12.2 for Mexican-American (MXAM) girls; and 10.3 and 12.7 years for non-Hispanic white (NHW) girls.
For boys in NHANES III, the median estimated ages for genital stage 2 were 9.3 for NHB boys, 10.4 for MXAM boys, and 10.1 for NHW youths.
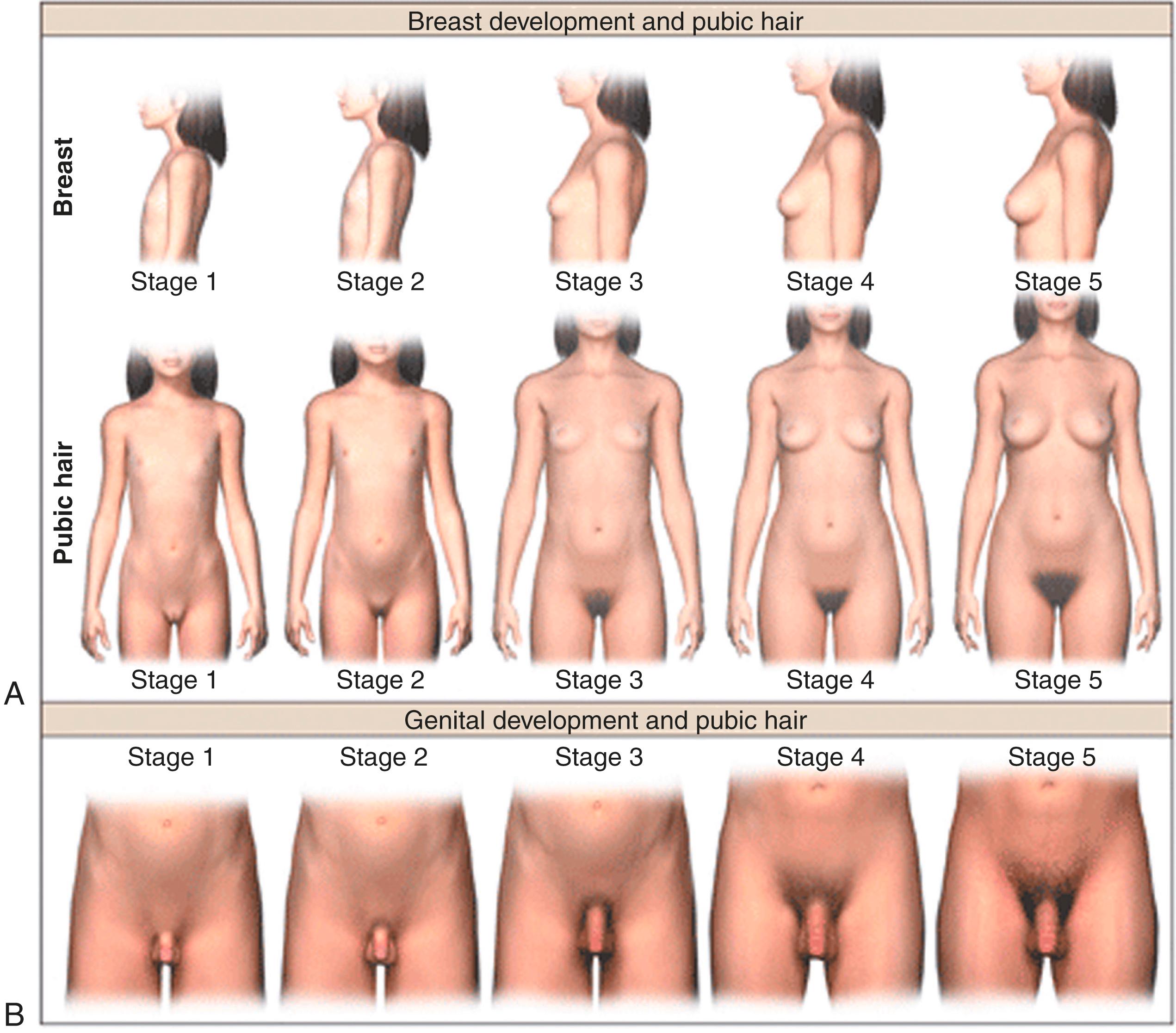
For both sexes, the genital and pubic hair changes that unfold at puberty are classified into five stages: stage 1 is prepubertal and stage 5 is adult ( Fig. 18.1 ; Table 18.1 ). These physical changes reflect the changing hormonal milieu due to gonadarche (as in the case of breast or testicular enlargement) or adrenarche (as in the case of pubic hair development). Although the physical sequelae of gonadarche and adrenarche generally occur concomitantly, discordance between the two processes may also occur in normal development.
| Girls | |||
|---|---|---|---|
| Stage | Breast | Pubic Hair | |
| 1 | Prepubertal | No pigmented hair | |
| 2 | Budding with larger areolae | Small amount of coarse, pigmented hair mostly along labia majora | |
| 3 | Enlargement of breast and areolae | Spread of coarse, pigmented hair over mons pubis | |
| 4 | Secondary mound of areolae | Almost adult pattern | |
| 5 | Mature contour | Adult pattern | |
| Boys | |||
| Stage | Genitalia | Pubic Hair | Testicular Volume |
| 1 | Prepubertal | No pigmented hair | <3 mL |
| 2 | Thinning and darkening of scrotum, increased size of penis | Small amount of coarse, pigmented hair at base of penis | 3–8 mL |
| 3 | Increased diameter of penis | Coarse, pigmented hair extends above penis | 10–15 mL |
| 4 | Increased diameter and length of penis | Almost adult pattern | 15–20 mL |
| 5 | Adult size and shape | Adult pattern | >25 mL |
During puberty, increased ovarian estrogen secretion promotes breast development in girls. The development of breast buds with increased areolar diameter is considered to be stage 2; greater enlargement of the breasts occurs in stage 3, accompanied by increased pigmentation of the areolae and nipples. During stage 4, the areolae are mounded above the breast tissue. Recession of the areola to the general breast contour represents breast stage 5. Palpation of the breast is necessary to better differentiate between the breast tissue and lipomastia. Additional effects of estrogen at this stage of development include cornification of the vaginal mucosa, uterine growth, and morphogenesis of an adult female body habitus.
Menarche follows an anovulatory cycle and generally occurs 2 to 3 years after the onset of breast development. Menstrual cycles during the first year after menarche are typically irregular and anovulatory, with most ranging in duration from 21 to 45 days. By three years postmenarche, over 90% of adolescent females have 10 or more menstrual cycles peryear with an average menstrual interval of 36.5 days. Nevertheless, cycles can remain irregular until the fifth year postmenarche.
Although primordial and preantral follicles predominate during the prepubertal years, small antral follicles can develop during this phase of maturation. These small follicles are gonadotropin-independent. Ovarian volume increases with the onset of puberty, achieves maximum volume soon after (between menarche and age 16 years), and remains stable or decreases slightly thereafter. Ovarian volume assessed by three-dimensional MRI in healthy adolescents and young adults between the ages of 13 and 25 years was 10.8 ± 3.7 mL. Polycystic ovary morphology (PCOM) is commonly detected in healthy adolescent girls and is generally not associated with decreased ovulatory rate, hyperandrogenism, or metabolic abnormalities in this age group. During the early postmenarcheal period, ovarian morphology on transabdominal ultrasound shows multicystic ovaries and increased ovarian volume that differs from ovarian morphology observed in older women. , Healthy adolescents and young adults were recently found to have 15.2 ± 4.0 follicles per ovary, which exceeds the 12 follicles per ovary cutoff of the Rotterdam Criteria for PCOM.
In girls, increased adrenal C19 steroid (androgen) secretion is considered to be responsible for the development of darker hairs along the labia, which is classified as pubic hair stage 2. The hair becomes darker and coarser during pubic hair stage 3, spreading over the pubic symphysis with gradual progression to a full female escutcheon. Apocrine odor may precede or accompany the development of pubic hair. Associated findings include axillary hair, acne, and oiliness of skin and hair.
For boys, increasing testicular and adrenal androgen secretion contributes to the secondary sex characteristics. Genital stage 2 is characterized by an increase in testicular volume and enlargement of the scrotum. At stage 2, the testes are approximately 4 to 8 mL in volume, with the longest axis being approximately 2.5 cm. The volume of the mature human testis is approximately 20 to 30 mL and primarily reflects increased growth of the seminiferous tubule due to Sertoli cell proliferation and differentiation as well as initiation of spermatogenesis. At genital stage 3, further growth of the testes has occurred, and the length and diameter of the penis have increased. At genital stage 4, penile size has increased and the scrotal skin has become darkened. Palpation and use of an orchidometer are preferable to inspection. Male pubic hair stage 2 consists of downy hair at the base of the penis. For pubic hair stage 3, the hair is longer and darker and extends over the junction of the pubic bones. For pubic hair stage 4, the extent of hair has increased but has not yet achieved the adult male escutcheon. Other secondary sexual characteristics in boys include axillary hair, increased size of the larynx, deepening of the voice, increased bone mass, and increased muscle strength. Approximately three years after the appearance of pubic hair, terminal hair appears in androgen-dependent regions on the face and trunk where it may develop for several years thereafter. There is considerable variation in the distribution and density of beard, chest, abdominal, and back hair, presumably reflecting genetic differences. The appearance of spermatozoa in early morning urine specimens (spermaturia) occurs during genital stage 3. Gynecomastia is observed in 50% of boys during pubertal development. This is typically the most prominent in midpuberty when the ratio of circulating concentrations of estradiol to testosterone is relatively high. In most instances, gynecomastia resolves spontaneously by 16 years of age.
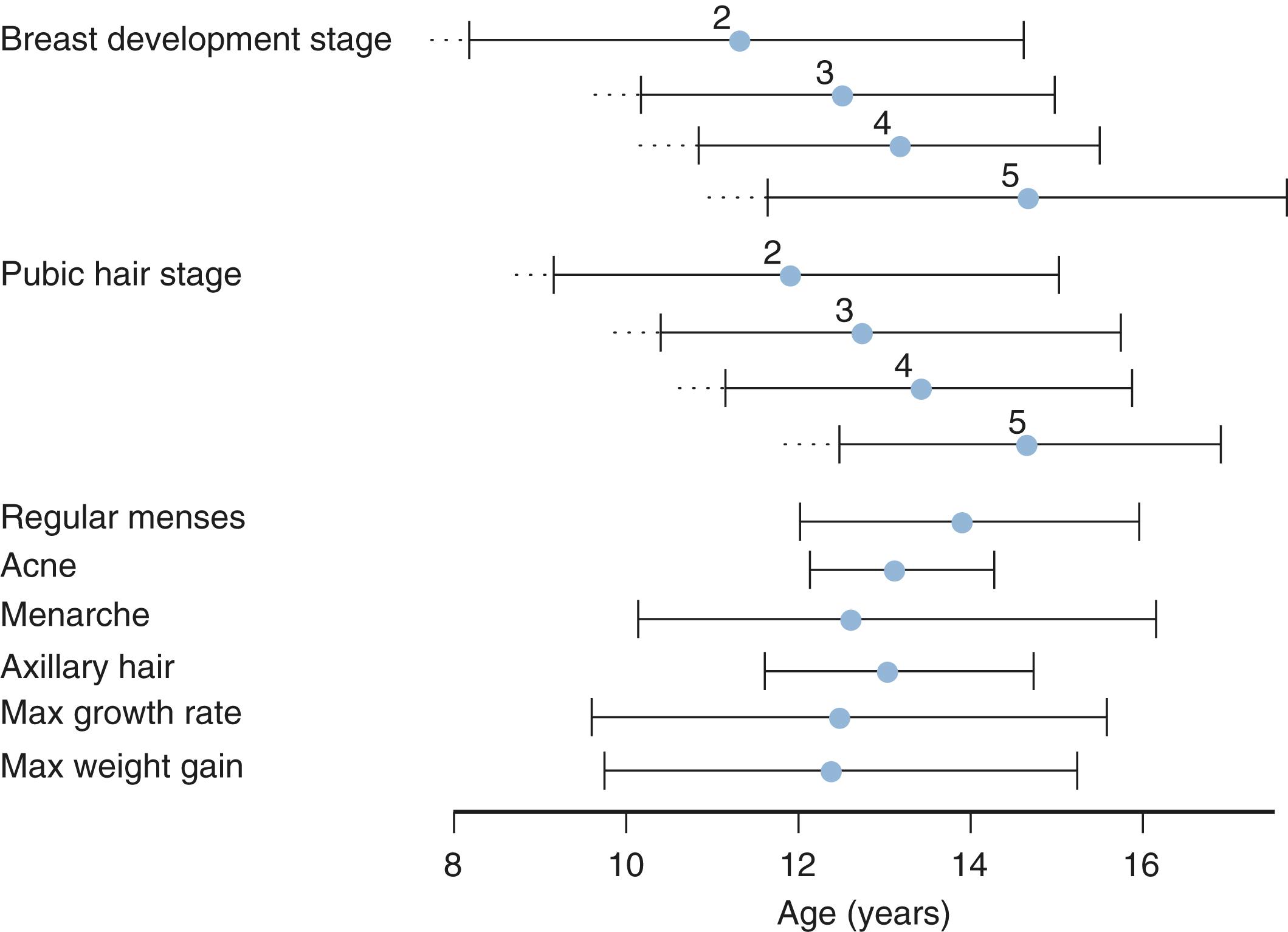
The pubertal growth spurt in girls occurs concurrently with the onset of breast development and reaches its peak in Tanner Stage 3 to 4 prior to menarche. Usually, only 4 to 6 cm of growth occurs after menarche. The pubertal growth spurt in boys, with an average height velocity of 9.5 cm per year, occurs around the genital end of Tanner stage 3 and the beginning of Tanner stage 4. In general, the age at peak height velocity shows an inverse relationship with the magnitude of the growth spurt. , Linear growth is approximately 99% complete for girls at a bone age of 15 years and for boys at a bone age of 17 years.
Breast development in girls and testicular enlargement in boys generally precede pubic hair development. Yet, the tempo for pubic hair development is faster such that synchrony between genital and pubic hair development occurs during the later stages of puberty. Schemata for the temporal development of the secondary sexual characteristic and their relationship to growth velocity are shown for girls and boys in Figs. 18.2 and 18.3 , respectively.
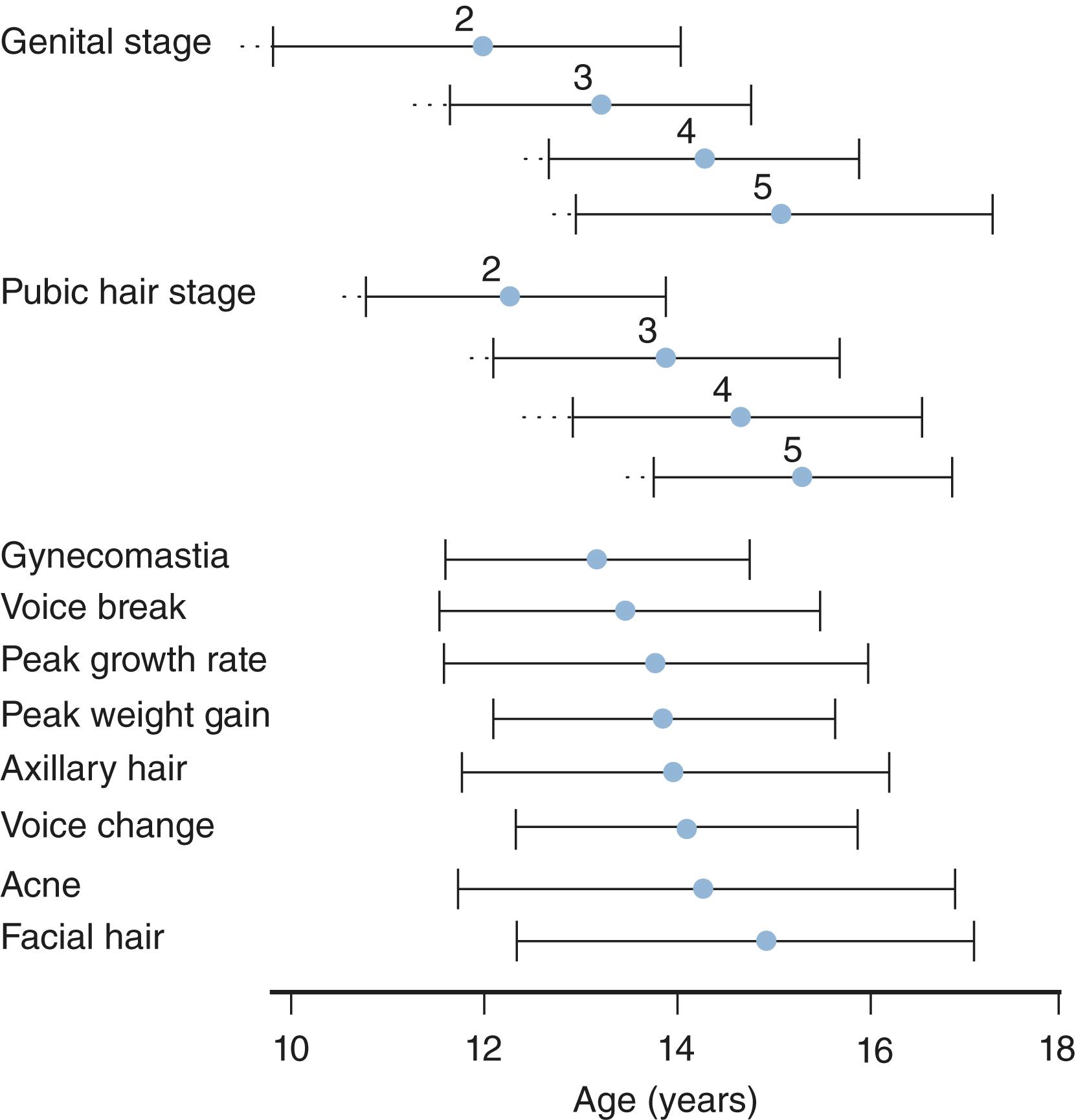
A mixed cross-sectional and longitudinal study of 730 healthy Danish boys showed that voice break and peak height velocity occur during later male puberty. Voice break had a mean age of 13.6 years and was moderately correlated with other male pubertal milestones gonadarche (11.5 years), (testicular enlargement (11.6 years), axillary odor (11.8 years), pubarche (12 years), axillary hair (13.1 years) and peak height velocity (13.7 years)). The authors found that at voice break, testosterone was 10.9 nmol/L, LH was 2.5 mIU/mL and testicular volume was about 12 cc. The longitudinal portion of the study showed that voice break can occur over a broad range of testosterone levels.
Reports of secular changes in the onset of puberty have focused on girls and have typically used age at menarche as the biomarker for puberty. Gluckman and Hanson have suggested that menarche occurred between 7 and 13 years in Paleolithic and Neolithic times. , Based on an analysis of 994 medieval adolescent skeletons (10–25 years) for the pubertal stage, it appears that adolescents likely began puberty around 10 to 12 years, with menarche occurring closer to 15 years in rural areas and 17 years in London. Boys experienced protracted pubertal development during the 10th to 17th centuries. Potential factors that contributed to later puberty during medieval times included poor diet, increased infections, and greater physical exertion. ,
Available historical data indicate that this was followed by a decline in the age of menarche in Europe and North America from the early 19th century (16–17 years of age) to the latter half of the 20th century (13 years of age). This trend has been attributed to the improving socioeconomic conditions during this epoch. Although recent data from North America, several European countries, and other regions of the industrialized world suggest that the trend of earlier menarche has been reduced or halted, breast and pubic hair development are occurring earlier than they would have 50 years ago in both North America and Europe. ,
The biology underlying this continued earlier secular trend in sexual development in girls, which in some populations is loosely associated with a similar trend in growth, is unclear, and may or may not involve an earlier onset of gonadarche or adrenarche. This earlier onset of breast development is not associated with increased gonadotropin or estradiol concentrations, suggesting that it represents a gonadotropin-independent event. , Earlier breast development assessed by palpation was reported in NHW girls, which was likely related to the increased BMI of this group. Analogous studies of boys are limited, but no striking sex differences in secular trends in puberty and growth are apparent. Using both genital staging and the orchidometer, the Copenhagen Puberty Study reported pubertal onset occurring three months earlier in Danish boys and reported that obesity advanced the onset of testicular enlargement ; however, other studies suggest that obesity delays the onset of puberty in boys. More recently, the Copenhagen Puberty Study used two-sample Mendelian randomization (MR) to determine the causal effect of genetically determined BMI on genetically determined pubertal timing. The study used GWAS of 97 BMI-determining variants from the GIANT consortium and GWAS for voice-breaking from 55,871 male research participants in the 23andMe study. They found significant associations between higher BMI and earlier voice break and a linear association between BMI and the timing of male pubertal milestones. In this study, all studied male pubertal events other than pubarche occurred earlier in obese boys compared to nonobese boys.
The age at onset of puberty varies between ethnic groups. Among American girls, mean ages for breast development, pubic hair development, and menarche were 9.5, 9.5, and 12.1, respectively, for NHB girls; 9.8, 10.3, and 12.2, respectively, for MXAM girls; and 10.3, 10.5, and 12.7 years, respectively, for NHW girls. , Data obtained through the cross-sectional Third National Health and Nutrition Examination Survey (NHANES III) between 1988 and 1994 showed that NHB girls enter puberty first, followed by MXAM and NHW girls. , Based on the NHANES III study, luteinizing hormone (LH) and inhibin B concentrations associated with onset of breast development were evaluated. As would be anticipated, LH and inhibin B concentrations increased with pubertal progression. Cut-points for Tanner 2 breast development were LH 1.04 mIU/mL (95% confidence interval [CI]: 0.71–1.37) and inhibin B 17.89 pg/mL (95% CI: 11.63–24.15). The respective median ages at hormonal onset based on LH concentrations were 10.7, 10.6, and 10.1 years for NHW, MXAM, and NHB girls, respectively. Girls with low birthweight and greater postnatal weight gain had relatively earlier onset of puberty based on LH concentrations and a similar pattern regarding pubertal onset was noted based solely on postnatal weight gain.
For boys in NHANES III, median estimated ages for genital and pubic hair stage 2 were 9.3 and 11.1, respectively, for NHB boys; 10.4 and 12.3, respectively, for MXAM boys; and 10.1 and 12.0, respectively, for NHW youths. For genital and pubic hair stage 5, median ages were 14.9 and 15.2, respectively, for NHB boys; 15.8 and 15.7, respectively, for MXAM boys; and 16.0 and 15.6, respectively, for NHW boys. Using the NHANES III data, LH, testosterone, and inhibin B concentrations increased as puberty progressed. Likely reflecting individual and diurnal variation, no single or combination hormone cut-point was found to be predictive of physical pubertal status, either genital or pubic hair stage 2, in this population. ,
Ethnic differences affecting the rate of skeletal maturation have been described in the Generation R study in the Netherlands; using the Greulich and Pyle standards, skeletal age was most advanced in Asians, intermediate in Africans, and least advanced in white Europeans. Skeletal age was more advanced in girls compared to boys in all three ancestral groupings. These ethnic differences in skeletal age persisted after adjustment for age, height, and BMI. Similar findings of advanced skeletal age were found in a longitudinal cohort of American children categorized as African or non-African descent; skeletal age was more advanced in girls. , Normative data in the Greulich and Pyle standard are largely derived from hand and wrist radiographs obtained from a non-Hispanic European white population from the 1930s to the 1960s. Nevertheless, available data suggest that genetic and environmental factors influence rates of skeletal maturation. Hence, clinicians need to be cognizant of these limitations when using the Greulich and Pyle standards to assess skeletal maturation in children from other ethnic/racial backgrounds. ,
The age at puberty reflects interactions between genetic, prenatal, and environmental factors. Twin studies indicate that heredity is responsible for approximately 50% of the variation in age at menarche. , Indeed, pubertal timing of both parents has been demonstrated to influence the timing of pubertal onset of both boys and girls independent of sex. Investigation of genetic variants associated with onset of puberty identified a specific variant, −29G>A, in the promoter region of the follicle-stimulating hormone (FSH) receptor ( FSHR ) gene; breast development occurred 7.4 months later among homozygous carriers of the −29G>A variant compared to the −29GG+GA carriers.
Synthesis of steroids from cholesterol requires expression of specific enzymes, receptors, cofactors, and other proteins in the adrenal cortex and the gonads under the influence of specific trophic hormones, adrenocorticotropin (ACTH), LH, and FSH.
Steroid hormone receptors are ligand-dependent transcription factors comprised of three functional domains: the N-terminal domain serves to modulate function, the DNA-binding domain mediates the binding of the receptor to DNA, and the ligand-binding domain binds to the cognate steroid hormone.
Puberty is characterized by reactivation of the hypothalamic gonadotropin-releasing hormone (GnRH) pulse generator characterized by the increasing amplitude of intermittent bursts of GnRH secretion into the hypophysial portal circulation. These intermittent bursts promote pulsatile LH and FSH secretion by the pituitary gonadotrophs.
Increments in circulating LH and FSH concentrations at the time of puberty drive sex-specific gonadal steroidogenesis, development of secondary sexual features, and gametogenesis.
The KNDy neurons in the infundibular (arcuate in nonprimate species) nucleus in the hypothalamus appear to comprise the major elements of the GnRH pulse generator.
GnRH pulse generation in the KNDy neurons is achieved by reciprocating stimulatory neurokinin B (NKB) and inhibitory (dynorphin) connections within the arcuate nucleus, while the output of the pulse generator is relayed to GnRH fibers projecting to the median eminence by an intermittent kisspeptin signal.
The biosynthetic pathways for gonadal and adrenal steroids are considered together because of their similarities and their importance in understanding the physiology and pathophysiology of puberty ( Fig. 18.4 ; see Chapter 4 ). Synthesis of steroids from cholesterol requires the expression of specific enzymes, receptors, cofactors, and other proteins in the adrenal cortex and the gonads. Steroidogenesis is regulated by specific trophic hormones, ACTH, LH, and FSH.
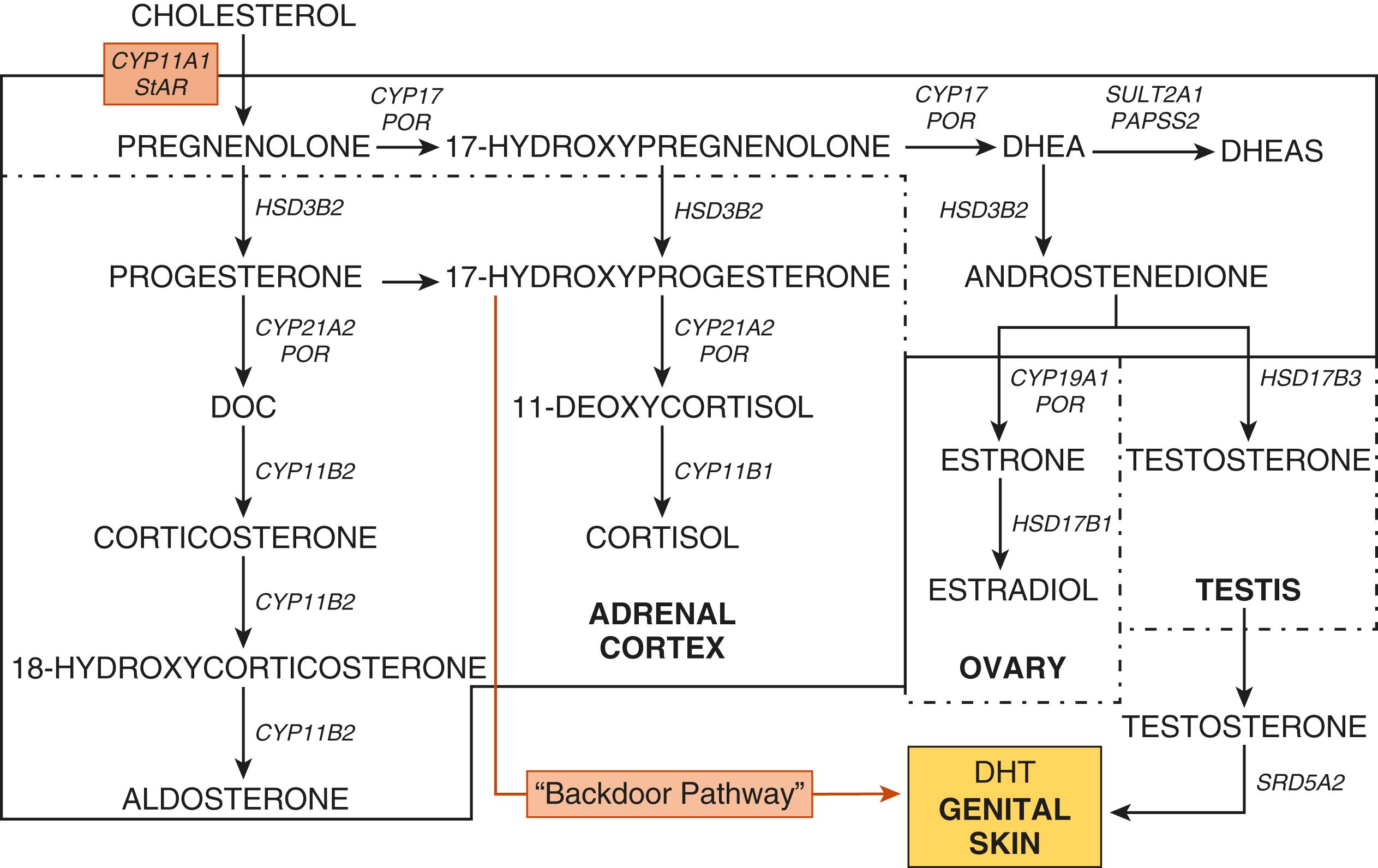
The adrenal cortex consists of three zones, the zona glomerulosa (ZG), zona fasciculata (ZF), and zona reticularis (ZR). The ZG synthesizes aldosterone, a mineralocorticoid, and is primarily regulated by potassium concentrations and renin-angiotensin. The ZF synthesizes cortisol. Steroidogenesis in the ZF is primarily governed by ACTH. The ZR synthesizes C-19 steroids, such as DHEA, DHEAS, androstenedione, androstenediol, and 11β-hydroxyandrostenedione.
Recent studies have confirmed that the human adrenals secrete 11-oxygenated 19-carbon (C 19 ) steroids (11-oxygenated androgens, 11OAs). The 11-OAs include 11β-hydroxyandrostenedione (11OHA4), 11β-hydroxytestosterone (11OHT), 11-ketoandrostenedione (11KA4), 11-ketotestosterone (11KT), and 11-ketodihydrotestosterone (11KDHT). The androgenic activities of 11KT and 11KDHT are comparable to testosterone (T) and dihydrotestosterone (DHT), respectively. In a study of predominantly Caribbean Hispanic prepubertal children with premature adrenarche (n = 13) and controls (n = 11), it was shown that 11OAs were elevated in premature adrenarche compared to controls and a high correlation of T and A4 with 11-ketosterone and 11β-hydroxyandrostenedione. In fact, 11KT values were 4-fold higher than those for T, indicating that 11KT is the major active androgen in the circulation in PA. 11KT was elevated even in children who presented with PA but did not have elevated DHEAS. It was proposed that serum 11OA measurement may be a more accurate way to screen and classify children with PA than DHEAS.
ACTH is a 39 amino acid peptide derived following proteolytic cleavages of proopiomelanocortin (POMC). Its actions are mediated by the ACTH receptor, a seven-transmembrane G protein-coupled receptor encoded by MC2R. This pathway utilizes cyclic adenosine monophosphate (cAMP)-dependent protein kinase A. The acute effects of ACTH include uptake of plasma low-density lipoproteins, stimulation of cholesterol esterase activity, enhanced synthesis and phosphorylation of steroidogenic acute regulatory protein (StAR), cholesterol transfer across the inner mitochondrial membrane, and increased cortisol secretion. The chronic effects of ACTH involve stimulation of transcription and translation of steroidogenic enzyme genes.
In the gonads, LH and FSH modulate steroid biosynthesis. LH promotes ovarian theca cell and testicular Leydig cell steroidogenesis; its actions are mediated by its cognate receptor, LHCGR. Acting through the FSHR, FSH stimulates aromatase expression to promote estrogen biosynthesis in the ovary and Sertoli cell growth in the testis. The LH and FSH receptors are both G protein-coupled receptors and contain leucine-rich repeats in their large ectodomains.
Most enzymes involved in steroidogenesis are cytochrome P450s (CYPs) or hydroxysteroid dehydrogenases (HSDs). The rate-limiting step of steroidogenesis is the transport of cholesterol into mitochondria mediated by StAR. Within the mitochondria, cholesterol desmolase (also known as side-chain cleavage or P450scc) converts cholesterol into pregnenolone. One enzyme, 17α-hydroxylase/17,20-lyase (P450c17), encoded by the CYP17A1 gene, is the qualitative regulator of adrenal and gonadal steroidogenesis. This enzyme mediates 17α-hydroxylation to convert pregnenolone into 17α-hydroxypregnenolone. In the ZR, ovarian theca, and Leydig cells and this same enzyme catalyzes scission of the C17–20 bond to produce DHEA. Although this one protein is capable of two distinct enzymatic reactions, these enzyme activities are differentially regulated. Factors known to modulate 17,20-lyase activity include: (1) the amount of P450 oxidoreductase (POR); (2) the expression of cytochrome b 5 (CYB5A); (3) the phosphorylation of serine/threonine residues on P450c17; and (4) the phosphorylation of noncanonical P450c17 residues. POR is a protein that transfers electrons from nicotinamide adenine dinucleotide phosphate to microsomal cytochrome P450 enzymes, such as P450c17, P450c21, and aromatase (P450aro). CYB5A modulates adrenal androgen secretion by increasing the 17,20-lyase activity of (P450c17).
The Δ 5 -steroids are converted to the Δ 4 -steroids by 3β-hydroxysteroid dehydrogenase type 2 (HSD3B2), the adrenal and gonadal specific isoform. This enzyme converts pregnenolone to progesterone in the ZG, 17-hydroxypregnenolone to 17-hydroxyprogesterone in the ZF, and DHEA to androstenedione in the ZR. In the ZF, 17-hydroxyprogesterone (17-OHP) is converted to 11-deoxycortisol by 21-hydroxylase (P450c21) and, subsequently, to cortisol by 11β-hydroxylase (P450c11β).
The adrenals, ovaries, and testes synthesize sex steroids. The ZR of the adrenal cortex produces DHEA, DHEAS, androstenedione, androstenediol, and 11β-hydroxyandrostenedione. DHEA sulfotransferase (SULT2A1) converts DHEA to DHEAS and this enzyme is also expressed in the liver. Sulfation of steroids by SULT2A1 requires a sulfate donor, 3′-phosphoadenosine-5′-phosphosulfate (PAPS), and the enzyme PAPS synthase. In the ovary, androstenedione is synthesized in the theca cell and diffuses into the granulosa cell, where it is aromatized by aromatase (P450aro) to estrone and converted to estradiol by 17β-hydroxysteroid dehydrogenase type 1 (HSD17B1). Estradiol binds the estrogen receptor. In Leydig cells, androstenedione is converted to testosterone by HSD17B3. In androgen target cells, such as those in the external genitalia and prostate, testosterone is converted to DHT by 5α-reductase type 2. In some androgen-sensitive tissues, such as bone and adipose, testosterone is converted to estradiol by aromatase. Peripheral tissues, including the kidney, adipose, and prostate and genital skin, can convert the adrenal-derived 11-OAS, 11ß-hydroxyandrosterone (11OHA4), and 11ß-hydroxytestosterone (11OHT) into the more potent androgens, 11-ketoandrostenedione (11KA4) and 11-ketosterone (11KT). Androgenic actions are mediated by the androgen receptor. ,
The 17β-hydroxysteroid dehydrogenase enzymes comprise a large family of enzymes involved in steroid biosynthesis and metabolism. The differences in tissue distribution, substrate preferences, subcellular localization, and mechanisms of regulation influence the cellular steroid microenvironment. The type 1 isozyme, 17βHSD1, is expressed in the ovaries, placenta, endometrium, and liver, where it favors conversion of estrone to estradiol. The type 3 isozyme, 17βHSD3, is expressed in the testis, where it preferentially converts androstenedione to testosterone. The type 5 enzyme, 17βHSD5, is an aldo-keto-reductase (AKR) enzyme (AKR1C3) that is expressed in steroidogenic and peripheral tissues; it can convert androstendione to testosterone.
Through investigations of the tammar wallaby and patients with disordered steroidogenesis, the presence of another pathway leading to dihydrotestosterone (DHT) synthesis was elucidated. In this “alternative backdoor pathway,” 17-OHP undergoes 3α- and 5α-reduction followed by 17,20-lyase, 17β-hydroxysteroid dehydrogenase, and 3α-oxidation steps to generate DHT in the absence of the “classic” intermediates, DHEA, androstenedione, and testosterone. In humans, since 17-OHP is not a favorable substrate for the 17,20-lyase reaction, this pathway acquires functional importance in disorders of steroidogenesis associated with increased 17-OHP concentrations, such as congenital adrenal hyperplasia (CAH) due to 21-hydroxylase deficiency and oxidoreductase deficiency.
Once secreted, sex steroids circulate bound to sex hormone-binding globulin (SHBG) and to albumin. The unbound or free hormone is the bioavailable form that diffuses passively into target cells and interacts with nuclear steroid receptors. Steroid hormone receptors are ligand-dependent transcription factors comprised of three functional domains: the N-terminal domain serves to modulate function, the DNA-binding domain mediates binding of the receptor to DNA, and the ligand-binding domain binds to steroid. Steroid receptor activity is modulated by various tissue-specific cofactors; both coactivators and corepressors can influence receptor function.
Steroids also act through nongenomic mechanisms. For example, testosterone can activate phospholipase C, leading to calcium influx into Sertoli cells, and can activate the mitogen-activated protein kinase as well as other intracellular mediators. Sex steroids can be metabolized to inactive forms by a variety of enzymes. Glucuronidation decreases the biological activity of steroid hormones and increases solubility to facilitate renal excretion. This process, catalyzed by UDP-glucuronyltransferase (UGT) enzymes, involves the transfer of glucuronic acid from uridine diphosphoglucuronic acid to steroid hormones. In humans, the UGT2B isoforms show greater specificity for C19 androgens. A second mechanism is sulfoconjugation, in which DHEA sulfotransferase catalyzes conversion of DHEA to DHEAS, and estrogen sulfotransferase converts estrogens to estrone sulfate. The inactive sulfated steroids can be hydrolyzed to active forms by steroid sulfatase.
Gonadarche reflects reactivation of the hypothalamic-pituitary-gonadal (HPG) axis. The increased pituitary secretion of LH and FSH stimulates gonadal steroidogenesis, development of the physical manifestations of puberty, completion of gametogenesis, and maintenance of fertility. LH and FSH are heterodimeric proteins consisting of a common α-subunit and a unique β-subunit. Both LH and FSH are glycosylated peptides. Glycosylation appears to modulate hormone stability, protein folding, cellular trafficking, circulating serum half-life, and receptor signaling. The temporal increments in circulating LH and FSH concentrations at the time of puberty, and their relationships to those of the gonadal steroids, testosterone and estradiol, respectively, at this stage of development, have been well documented in both boys and girls. , The actions of LH and FSH are mediated by their cognate seven-transmembrane domain G protein-coupled receptors: the LH receptor (LHCGR) and FSHR, respectively.
In the female, LH stimulates androgen production by the theca cells of the ovarian follicles and progesterone secretion from luteinized granulosa cells of the corpus luteum. FSH is critical for the process of follicular recruitment and selection. In the granulosa cells of the developing follicle, FSH induces expression of aromatase, which is responsible for the aromatization of the theca cell-derived androgens into estrogens. FSH also induces LHCGR expression in granulosa cells of the dominant follicle, which selectively amplifies the effect of declining FSH concentrations on the dominant follicle. , In the male, LH regulates the secretion of testosterone from Leydig cells.
FSH, together with testosterone, is responsible for initiating and maintaining spermatogenesis. The action of FSH in this regard is indirect and exerted on the somatic Sertoli cell of the seminiferous tubule. While the actions of testosterone are also indirect, several somatic cell types in the testis (Sertoli, Leydig, and peritubular) express AR and are considered to be involved in the control of spermatogenesis.
The pubertal drive to the pituitary-gonadal axis is generated by a diffusely distributed network of hypothalamic neurons expressing GnRH-1, known as the “hypothalamic GnRH pulse generator.” As the name implies, the hypothalamic GnRH pulse generator produces intermittent discharges of GnRH into the hypophysial portal circulation, which is obligatory for gonadotropin synthesis and secretion by the pituitary gonadotrophs. LH and FSH secretion is stimulated by GnRH acting through its receptor, GnRH-R1, located on gonadotropin-secreting cells (gonadotrophs) in the pituitary gland. A unique feature of the human GnRHR, a 7-transmembrane domain G protein-coupled receptor, is its lack of a C-terminal cytoplasmic domain. Other factors studied during puberty include inhibins, activins, antimüllerian hormone (AMH), insulin-like factor-3 (INSL3), and osteocalcin. Activins and inhibins are members of the transforming growth factor-β (TGF-β) superfamily composed of a common α-subunit and two β-subunits (β A and β B ). The activins are dimers consisting of only the β-subunits; activin A is a dimer of β A subunits and activin B is a dimer of β B subunits. The activins are synthesized in the gonadotropes and influence FSH secretion. Mature inhibins are dimers composed of a common α-subunit covalently linked with one of two β-subunits (β A and β B ). The α/β A and α/β B dimers are known as inhibin A and inhibin B, respectively. The gonadal inhibins, like the gonadal steroids, play both an endocrine role in the regulation of gonadotropin secretion and a paracrine role within the gonads. Inhibin B, which is synthesized in part by the Sertoli cell, is the principal inhibin secreted by the testis. Inhibin B concentrations are low in prepubertal boys and increase with the onset of puberty. , The pubertal increase in inhibin B may be attributed to Sertoli cell proliferation and to the initiation of spermatogenesis, both of which reflect the increased gonadotropin drive to the testis at the time of gonadarche.
In girls, circulating levels of inhibin A and B are low or undetectable prior to puberty. Inhibin B begins to rise with the onset of puberty, as does inhibin A in breast stages 3 and 4. Adult levels are attained at approximately 14 to 15 years of age. During the menstrual cycle, inhibin A levels are elevated in the luteal phase, while inhibin B predominates in the circulation of the follicular phase. However, the role of ovarian inhibins in regulating gonadotropin secretion in pubertal and premenopausal women remains to be fully elucidated.
Osteocalcin is secreted by osteoblasts and is a marker of bone formation. Most available data regarding the potential actions of osteocalcin on puberty are derived from mouse studies. Studies of mice have shown that osteocalcin facilitates testicular testosterone secretion and pancreatic β-cell proliferation. The testicular effects of osteocalcin appear to be independent of the HPG axis as the osteocalcin receptor is not expressed in the hypothalamus or pituitary of mice. , Posttranslationally, osteocalcin is carboxylated on three glutamic acid residues. The carboxylated form of the molecule is considered to be biologically inactive, whereas the undercarboxylated form is biologically active. Osteocalcin signals through its cognate receptor, GPRC6A, which is expressed by Leydig cells. Interestingly, increased osteocalcin concentrations are associated with the pubertal rise in testosterone concentrations in boys. Osteocalcin shows sexual dimorphism because it modulates Leydig cell testosterone production, but not ovarian estrogen production. The discovery of a missense mutation in the GPRC6A gene in two men with primary testicular failure, oligospermia, and glucose intolerance encourages speculation that osteocalcin influences testicular function in humans.
Antimüllerian hormone (AMH), also known as müllerian-inhibiting hormone (MIH), is another member of the TGF-β superfamily. AMH signals by binding to a specific type-II receptor (AMHR2); this receptor heterodimerizes with one of several type-I receptors (ALK2, ALK3, and ALK6), leading to recruitment of Smad proteins that are translocated to the nucleus to regulate target gene expression. AMH is secreted by the Sertoli cells of the developing testis and stimulates regression of the müllerian ducts during male fetal development. Postnatally, AMH is secreted by the Sertoli cells of the testis. In boys, AMH concentrations decline at puberty. Although FSH stimulates AMH production, testosterone inhibits Sertoli cell AMH secretion. The decline in AMH concentrations appears to be closely associated with rising inhibin B concentrations at the onset of puberty in boys, the latter presumably reflecting androgen-induced differentiation of the Sertoli cell. Both AMH and inhibin B concentrations are low in boys with bilateral anorchia or complete hypogonadotropic hypogonadism (HH).
Immunohistochemical studies of human ovaries showed no AMH staining in primordial follicles. AMH expression is highest in growing preantral and small antral follicles and disappears in larger follicles (>8 mm). In the ovaries, AMH is primarily secreted by the granulosa cells of the preantral and antral follicles. Among females, AMH concentrations begin to increase in infancy to achieve a plateau during adolescence until age 25 years. Subsequently, AMH concentrations decline and correlate inversely with age. , AMH plays a role as a gatekeeper of follicular development.
Data obtained from studies in rodents have demonstrated the presence of the AMH receptor, AMHRII, in gonadotropes. One report using rats showed that AMH stimulated FSH secretion in immature female rats. Subsets of GnRH neurons express the AMH receptor. In vitro and in vivo studies have demonstrated that AMH increases GnRH-dependent LH pulsatility and secretion. Thus AMH likely influences the migration of GnRH neurons during gestation and contributes to HPG axis function during prepubertal, pubertal, and adult years.
INSL3, a peptide hormone secreted by Leydig cells, plays a major role in directing the transabdominal phase of testicular descent during gestation. INSL3 signaling is mediated by the G protein-coupled relaxin family peptide receptor 2 (RXFP2). After birth, fetal Leydig cells involute followed by quiescence until puberty. Adult-type Leydig cells are derived from a different population of precursors and are dependent on LH stimulation. INSL3 concentrations are low during infancy, rise during puberty, and reflect Leydig cell function during adulthood. Longitudinal data demonstrated that INSL3 concentrations rise with onset of puberty and increasing testicular volume.
INSL3 is also secreted by ovarian theca cells, predominantly by those surrounding medium and large growing follicles. INSL3 and its receptor may play a role in autoregulatory feedback to maintain theca cell androgen production. Despite many variations, INSL3 concentrations tend to rise during late puberty in girls; the variation likely reflects that INSL3 is secreted largely by growing follicles and is a marker of theca cell activity.
In the pubertal and postpubertal individual, the ovaries and testes are governed by feedback control systems. GnRH and the gonadotropins comprise the feed-forward components from hypothalamus to pituitary and from pituitary to gonad, respectively. In turn, steroid and protein hormones from the gonads provide the feedback signals that regulate the secretion of LH and FSH. The feedback actions of these gonadal hormones, which involve both negative and stimulatory (positive) actions, may be exerted directly at the level of the pituitary gonadotrophs to modulate expression of the genes encoding LHβ and FSHβ ( LHB and FSHB , respectively). Feedback may also be exerted indirectly at the level of the hypothalamus to regulate the release of GnRH.
In the male, a negative feedback action of testosterone and inhibin B are the major regulators of LH and FSH secretion, respectively. The action of testosterone is predominantly exerted at the hypothalamic level, while that of inhibin appears to occur directly at the pituitary. The role of aromatization of testosterone to estradiol in mediating the negative feedback action of this androgen on LH secretion continues to be an area of active investigation. The feedback control of LH and FSH throughout the menstrual cycle is complicated and involves both negative and positive feedback actions of ovarian steroids at both the hypothalamic and pituitary levels (see also Chapter 9 ). The maintenance of normal ratios of circulating LH and FSH concentrations is important for gonadal function, particularly for folliculogenesis and ovulation.
In conditions under which pulsatile GnRH release is compromised, such as occurs in anorexia nervosa and during periods of strenuous physical training especially in young women, gonadotropin secretion is attenuated and pubertal development is arrested. Thus the pituitary-gonadal axis in both males and females may be viewed as being a slave to the hypothalamic GnRH pulse generator, and this analogy should be held in mind when considering the mechanisms triggering the onset of gonadarche.
The human hypothalamus contains approximately 2000 diffusely distributed GnRH neurons, many of which send their projections to the median eminence, where they intermittently discharge their peptide into the primary plexus of the hypophysial portal circulation, thereby providing the pituitary gonadotrophs with the pulsatile stimulation essential for maintaining gonadotropin secretion. The mammalian GnRH neuron displays unique characteristics in that the distal portion of the neuron functions as an autonomous regulatory region gathering synaptic input from other neurons; this region lies upstream of the short distal axons responsible for secreting GnRH into the portal circulation. , At the median eminences, these fibers are intertwined, encased by tanycytes (specialized ependymal cells of the third ventricle); they project to multiple blood vessels and receive numerous synaptic inputs. Tanycytes influence GnRH output by modulating access of GnRH neurons to the fenestrated vessels of the median eminence. ,
Compelling evidence indicates the fundamental role of KNDy neurons in the arcuate nucleus (also called the infundibular nucleus) as a major component of the neurobiological mechanism that underlies GnRH pulse generation. , These hypothalamic neurons, named because they coexpress kisspeptin, NKB, and dynorphin, project their axons to the median eminence, where they mingle intimately with GnRH fibers en route to the portal vessels.
GnRH pulse generation is achieved by reciprocal stimulatory (NKB) and inhibitory (dynorphin) connections within the arcuate nucleus; the net output of the pulse generator is relayed to GnRH fibers by an intermittent kisspeptin signal. Kisspeptin, an extremely potent GnRH secretagogue, signals through its cognate receptor, KISS1R, which is expressed in GnRH neurons. Using immunohistochemistry, kisspeptin was detected in the anterior and intermediate lobes of the pituitary in monkeys but was not apparently colocalized with gonadotrophs, somatotrophs, or lactotrophs. , Loss-of-function mutations in the genes encoding for kisspeptin ( KISS1 ), the kisspeptin receptor ( KISS1R ), , NKB ( TAC3 ), or the NKB receptor ( TACR3 ) in humans are associated with hypogonadotropic hypogonadism. ,
Detailed reviews of the neurobiology of the GnRH pulse generator and its applicability to humans are beyond the scope of this chapter but are readily available.
GnRH neurons are derived from a heterogenous stem cell population in the embryonic olfactory placode and migrate to their final destinations in the hypothalamus during embryonic development.
Adhesion and guidance molecules and their cognate receptors influence migration of GnRH neurons, including anosmin-1 (ANOS1), heparin sulfate 6-O-sulfotransferase 1 (HS6ST1), prokineticin 2 (PROK2), prokineticin receptor 2 (PROKR2), semaphorin 3A (SEMA3A), AMH, and others.
A transient reactivation of the HPG axis from 1 through 3 to 6 months of age results in an adult-like endocrine milieu. This time period is labeled as “minipuberty.”
The control system that dictates the up-down-up pattern of GnRH pulse generator activity from early infancy until puberty may be viewed as a conceptual “neurobiological brake.”
The neurobiological brake is “weaker” in girls than in boys.
Pulsatile LH and FSH secretion can modulate all aspects of testicular function and control folliculogenesis and corpus luteum function. A surge of gonadotropin secretion triggered by the positive feedback of the rising estradiol concentrations secreted by the developing dominant follicle is required in females to induce ovulation.
The lack of intact kisspeptin or NKB signaling pathways compromises the output of the GnRH pulse generator, resulting in absence of puberty. This validates the central importance of the KNDy neuron as the GnRH pulse generator.
Available data indicate that the GnRH pulse generator is under the control of an upstream transcriptional gene network.
The precise mechanism(s) that governs the timing of the onset of puberty remains a mystery.
The “somatometer hypothesis” proposes that attainment of a particular state of somatic maturation initiates puberty. Body fat content, leptin, and insulin have been argued to be relevant to this hypothesis.
Another hypothesis invokes a pubertal clock, presumably resident in the CNS.
Using three-dimensional (3D) imaging and transparent human fetal brains, Casoni et al have suggested that approximately 2000 GnRH neurons reside in the hypothalamus and approximately 8000 are widely distributed in other areas of the brain. Meticulously orchestrated development of the GnRH neurons and olfactory neurons in conjunction with precise spatiotemporal expression of multiple factors is essential for normal HPG axis function ( Fig. 18.5 ). Specific mutations in families with disorders of puberty and studies of transgenic mice have established some of the factors involved in GnRH neuron migration. Elements essential to GnRH neuron development, migration, and function include cytoskeletal proteins, adhesion molecules, neurotransmitters, growth factors, receptors, and transcription factors. Some factors function in multiple regions and may have diverging effects depending on the context of the molecular environment. Elucidation of the neurobiology and ontogeny of the GnRH neurons will improve understanding of the pathophysiology of HH and, perhaps, lead to novel therapies.
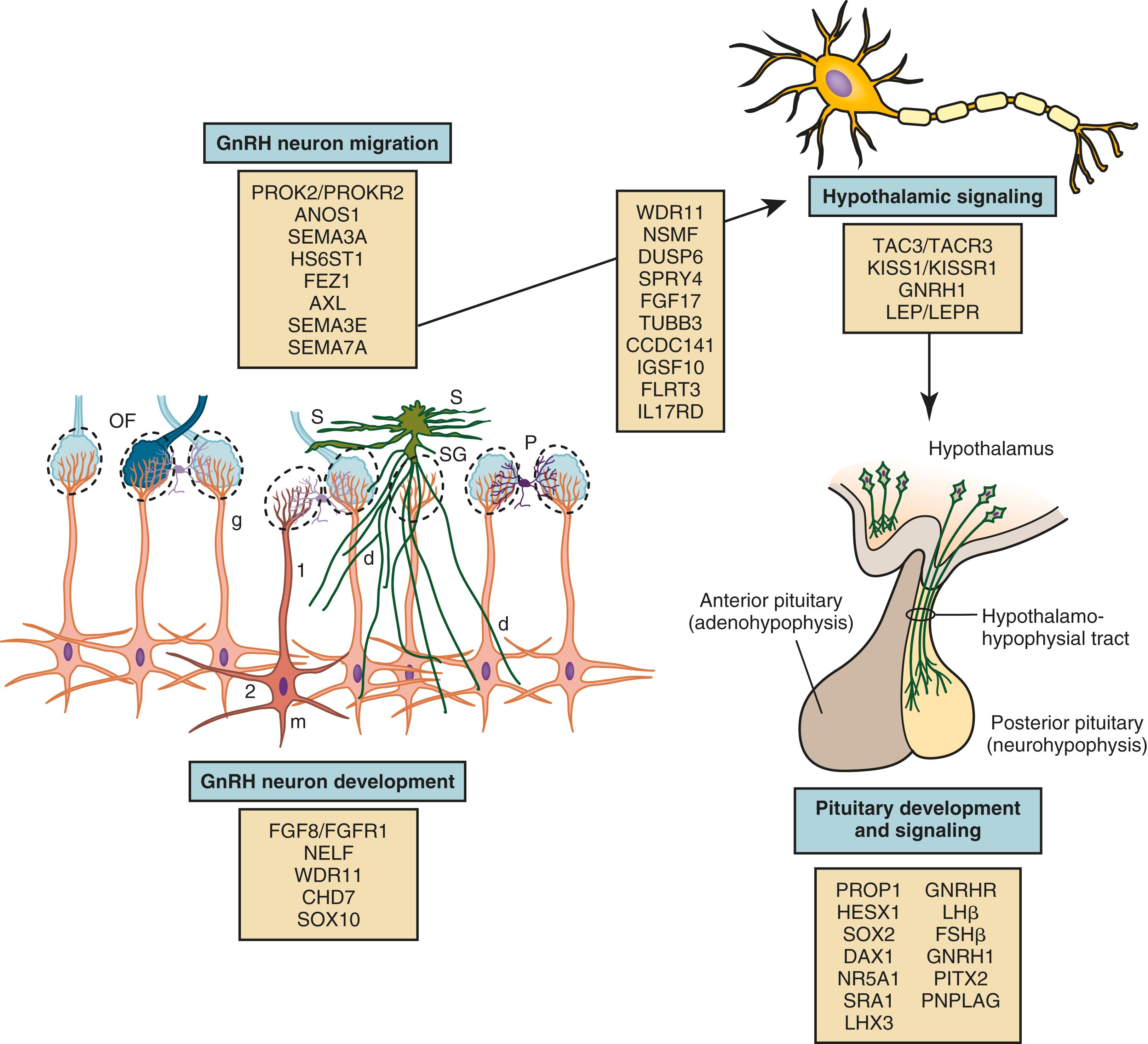
The GnRH neurons are born in the olfactory placode and migrate during early fetal development from the nose through the forebrain to the hypothalamus. The fetal ontogeny of the GnRH neurons can be classified into several stages, each with distinct regulatory mechanisms: (1) differentiation of GnRH neurons; (2) migration with axons of the vomeronasal nerve across the cribriform plate and into the forebrain; (3) localization in the hypothalamus and development of processes to the median eminence; and (4) attainment of final location and functionality.
Most vertebrates have two distinct olfactory systems: the main olfactory system responsible for recognition of volatile odorants and the vomeronasal system responsible for detecting pheromones. During the fifth week of gestation in humans, the olfactory placodes develop as thickenings of the ectoderm on the ventrolateral sides of the head. GnRH neuronal differentiation occurs between 39 and 44 days of gestation. Despite the recognition that GnRH progenitor stem cells are derived from this embryonic olfactory placode, the precise lineage of the GnRH neuron remains unclear.
Between weeks 5 and 6 of human gestation, the migratory mass of neural crest-derived migratory cells and olfactory neurons contains a small number of GnRH neurons. Subsequently, around the sixth week of gestation, the GnRH neurons begin their migration along vomeronasal nerves through the cribriform plate and eventually find their way to the hypothalamus. The GnRH neurons migrate into the brain along two distinct migratory pathways: a ventral pathway directed towards presumptive hypothalamic regions and a dorsal pathway directed toward pallial and subpallial telencephalic regions. These GnRH neurons, accompanied by olfactory ensheathing cells (OECs), travel along with the terminal, vomeronasal, and olfactory nerves into the brain. OECs are glial cells that guide GnRH and olfactory nerves to the forebrain. OECs express several factors important for GnRH migration, such as semaphorin 4D, signaling and neuronal migration factor (NSMF), and stromal-derived growth factor 1 (SDF-1). Available data indicate that SOX10 promotes development of OECs.
Two proteins, chromodomain helicase DNA-binding protein 7 (CHD7) and SOX10, influence neural crest cell development and eventual migration. CHD7 is a large protein that participates in chromatin remodeling and transcription; it interacts with other proteins and may regulate genes involved in neural crest cell guidance. In mice heterozygous for Chd7 mutations, Fgfr1 expression in the olfactory placode, GnRH1 and Otx2 expression in the hypothalamus, and GnRHR expression in the pituitary were decreased, supporting a role for CHD7 upstream of Fgf8 and Fgfr1 in the development and maintenance of GnRH neurons.
Other factors involved in the differentiation of GnRH neurons include fibroblast growth factor-8 (FGF8), fibroblast growth factor receptor-1 (FGFR1), heparan sulfate 6- O -sulfotransferase 1 (HS6ST1), and AMH. FGF8 influences craniofacial development, neuroendocrine cell proliferation, cell fate specification, and cell survival. , In the developing GnRH neuron, FGFR1 is the preferred receptor for FGF8. The FGFR1 is a tyrosine kinase receptor composed of three extracellular immunoglobulin domains, a transmembrane domain, and a cytoplasmic tyrosine kinase domain. Upon ligand binding, FGFR1 and its coreceptor dimerize, leading to autophosphorylation and protein kinase activity. , The extracellular domain of FGFR1 interacts with heparan sulfate proteoaminoglycan, its coreceptor. Heparan sulfates are cell membrane and matrix-associated proteoglycans involved in neural development. These polysaccharides undergo nonrandom modifications of the sugar moieties to facilitate cell-to-cell communication. HS6ST1 introduces a sulfate at the 6- O position within heparan sulfate. This action appears to be necessary for FGFR1 function.
AMH is reported to play roles in both the migration of GnRH neurons during fetal life and the postnatal function of the GnRH neuron. Immunohistochemical studies have identified AMH and AMHR in human fetal GnRH neurons and adult hypothalami. ,
Correct targeting and movement of GnRH neurons depend on multiple cues provided by several factors. These signals may act directly or indirectly through the scaffold of olfactory neurons. Chemokine gradients likely influence GnRH neuronal movement. Such factors include SDF-1 and gamma-aminobutyric acid (GABA). Curiously, SDF-1 and GABA appear to exert divergent effects to accelerate or retard, respectively, neuronal migration. SDF-1 acts through its receptor, CXCR4, via a G protein-activated inward rectifier potassium channel. SDF-1 has been observed in the nasal mesenchyme (NM), whereas its receptor, CXCR4, has been localized in migrating GnRH neurons and olfactory/vomeronasal nerve axons. Further, CXCR4-deficient mice exhibit a loss of GnRH neurons and impaired migration, suggesting the importance of SDF-1/CXCR4 signaling in the development of this system.
CCDC141 encodes a coiled-coil domain containing protein that is expressed in GnRH neurons and olfactory fibers. Knockdown of Ccdc141 did not change olfactory axon outgrowth but was associated with decreased GnRH cell migration out of the nasal pit. In mice, Ccdc141 expression, correlated with migration in nasal regions and decreased when GnRH neurons entered the forebrain, appears to affect cellular motility through its interactions with myosin II.
Additional adhesion and guidance molecules include ANOS1, ephrins, and prokineticin 2 (PROK2). The gene encoding ANOS1 (previously known as KAL1 ) is located at Xp22.3 in the pseudoautosomal region of the X chromosome. Anosmin-1 is an extracellular matrix glycoprotein that contains a whey acidic protein-like protease inhibitor domain and four fibronectin type III domains. It promotes the formation of the lateral olfactory tract and neurite development. Anosmin may also serve as (1) an adhesion molecule to guide migrating GnRH neurons and (2) as a chemoattractant for olfactory axon pathfinding. Also, it may interact with FGFR1. Ephrins are cell surface molecules that play a major role in axon guidance and signal through their cognate membrane tyrosine kinase receptors. PROK2 signals through the prokineticin receptor 2 (PROKR2), a member of the rhodopsin G protein-coupled receptor family. PROK2 and its receptor ( PROKR2 ), a G protein-coupled receptor, appear to play major roles in olfactory bulb neurogenesis and GnRH neuron migration. However, neither protein is expressed in GnRH neurons. Based on the findings in mice with targeted PROK2 mutations, the GnRH neurons appear to be trapped with olfactory neurons with arrested migration just after crossing the cribriform plate.
Semaphorins comprise a large and diverse family of secreted and membrane-associated proteins that influence the navigation of growing axons and play a role in neural network formation. Four class 3 semaphorins, Sema3A, Sema3B, Sema3C, and Sema3F, are expressed around the developing olfactory/vomeronasal region. Semaphorin-3A ( SEMA3A ) is a secreted protein with repulsive effects on primary olfactory axons expressing the coreceptor neuropilin-1 (Nrp1), which may influence the migration of GnRH neurons. Semaphorin 3A is also expressed by OECs. Semaphorin-3E ( SEMA3E ) on the other hand protects maturing GnRH neurons from cell death. Semaphorin 4D is a membrane-bound semaphorin that can also be proteolytically released into the extracellular space in an active form. It can act as a proangiogenic factor through the coupling of its cognate receptor, PlexinB1, with the hepatocyte growth factor (HGF) receptor Met tyrosine kinase (MET). Both semaphorin 4D and PlexinB1 are highly expressed in the developing olfactory placode and the developing NM.
\Semaphorin 7A (Sema 7A) appears to play a role in GnRH neuronal migration. Two mechanisms have been described: it can act as a membrane-bound signaling molecule or, following proteolytic cleavage, as a soluble factor. Sema7A can interact with two different receptors (plexin C1 and β1-integrin). Binding to plexin C1 decreases integrin-mediated cell attachment and spreading and interacting with β1-integrin induces integrin clustering and the activation of MAPK pathways. The phenotype of the mouse model with GnRH neuron-specific β1-integrin conditional KO showed impaired migration of GnRH neurons, delayed pubertal onset, and impaired fertility in female mice. In addition to its role in the development of the GnRH system, Sema7A appears to mediate the plasticity of GnRH neurons and tanycytes in the adult median eminence.
IGSF10, a member of the immunoglobulin superfamily, is also implicated in GnRH neuronal migration. Tissue expression studies using mouse embryos showed that IGSF10 mRNA expression was localized to embryonic NM during the time that GnRH neurons are migrating through the NM. In a zebrafish IGSF10 knockdown model, loss of IGSF10 led to perturbed migration and failed neurite extension of GnRH3 neurons toward the hypothalamus.
HGF, Axl, and Tyro3 maintain GnRH neuronal survival when the neurons are crossing the cribriform plate region. HGF signals through its receptor, cMet, to promote GnRH neuron migration. Axl and Tyro3 are members of the TAM family of tyrosine kinase receptors and contain a fibronectin domain that binds to heparan sulfate proteoglycans. Mice with targeted Axl/Tyro3 mutations show impaired sex hormone-induced gonadotropin surge, resulting in estrous cycle abnormalities. The protein, growth arrest-specific 6 (Gas6) encoded by Gas6, is a ligand that activates Axl and Tyro3. Gas6 is a heparan sulfate proteoglycan-activated ligand with similarities to the FGFs and HGF. The phenotype of Gas6 knockout mice is characterized by early loss of GnRH neurons during embryonic development. Despite a persistent decrease in GnRH neurons and impaired early stages of sexual maturation, these mice eventually manifested normal fertility.
FEZF1 is a zinc-finger gene encoding a transcriptional repressor that is highly and selectively present during embryogenesis in the olfactory epithelium. Fezf1 -deficient mice have impaired axonal projection of pioneer olfactory receptor neurons that cross the cribriform plate and subsequently innervate the olfactory bulb. These mice have smaller olfactory bulbs and an absence of GnRH neurons in the brain. Thus it appears that the FEZF1 product is required for the olfactory receptor neurons, and hence accompanying GnRH neurons, to enter the brain. ,
The roles of microRNAs (miRs) in neuronal development and maturation are becoming apparent. Data obtained using mice with targeted deletion of Distal-less-related 5 ( Dlx5 ) gene demonstrated that specific miRs (-9 and -200 class) influence olfactory and GnRH neuron development. Mice with gonadotrope-specific deletion of Dicer exhibited suppressed gonadotropin β-subunits and infertility. Another microRNA, miR-7a2, is expressed in pituitary gonadotropes. The phenotype associated with the genetic deletion of miR-7a2 in mice includes low gonadotropin concentrations and infertility. miR-7a2 is highly expressed in the pituitary but does not appear to influence GnRH neuron migration. ,
Upon arrival in the hypothalamus, the GnRH neurons extend projections to the median eminence to form a network that can secrete GnRH into the primary plexus of the hypophysial portal circulation. LH and FSH reach detectable levels by the 10th week of gestation in the human pituitary, peak in midgestation, and are higher in female fetuses than male fetuses. Although the hypothalamic control of the fetal pituitary-gonadal axis has not been extensively studied in higher primates, the GnRH pulse generator is clearly driving the gonadotrophs of the fetal pituitary around the 15th week of gestation. Functional activity of this hypothalamic-pituitary system is essential for fetal testicular testosterone synthesis by the Leydig cell and normal male sex development. In contrast to the fetal testis, the ovary at this stage of development is relatively quiescent, and the absence of gonadal feedback signals likely accounts for the higher gonadotropin levels in the female fetus. As gestation progresses, the secretion of estradiol and other steroids by the fetoplacental unit increases dramatically and suppresses gonadotropin secretion from the fetal pituitary by exerting an inhibitory action either directly at the pituitary or indirectly on the hypothalamus to restrain GnRH release.
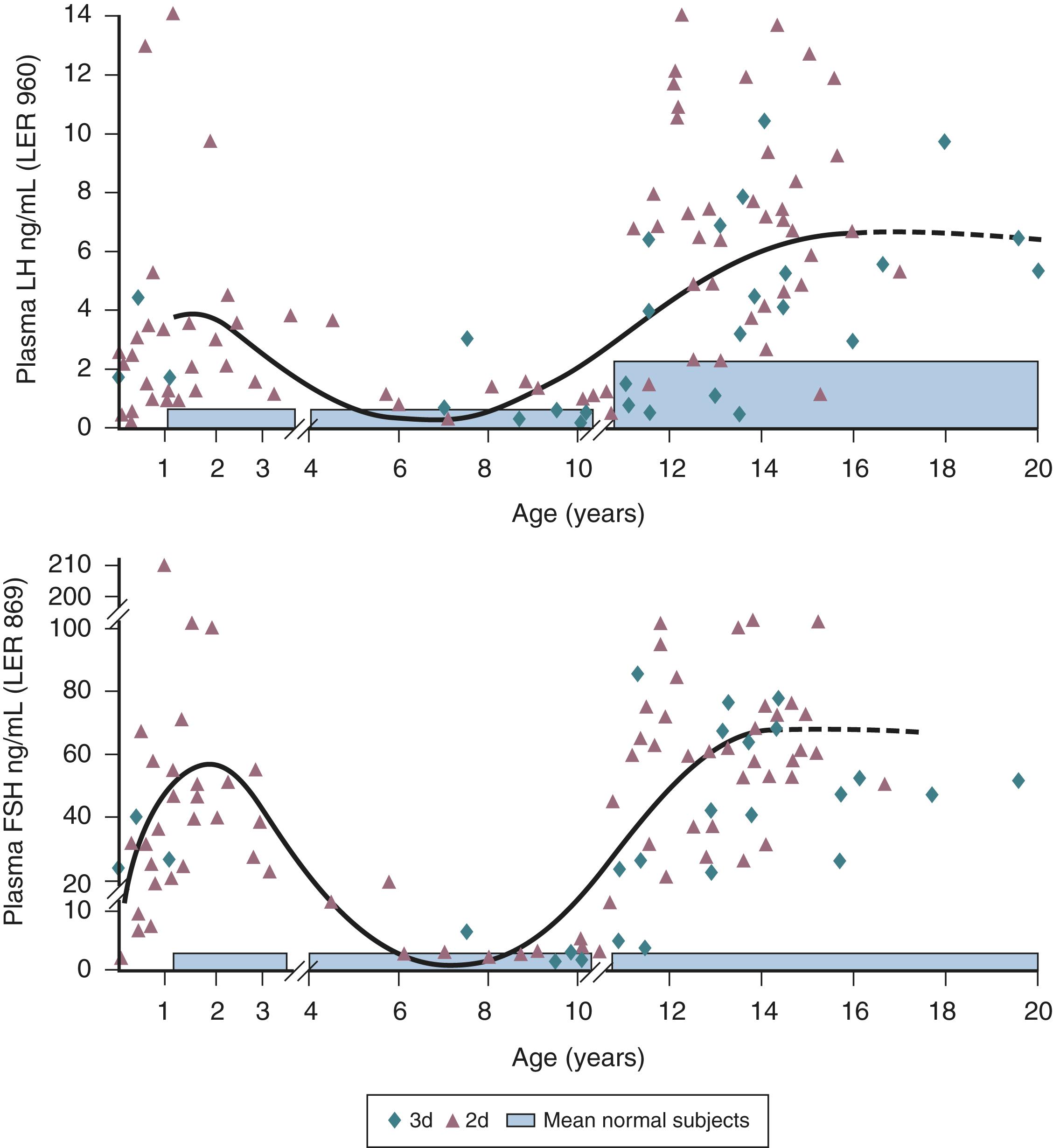
Gonadotropin concentrations are typically low in cord blood samples. However, following birth, GnRH pulse generator activity is robustly expressed, presumably due to the loss of placental steroids. The pituitary gonadotrophs of the infant respond with LH and FSH secretion. , , Moreover, in the infant boy, the Leydig cells of the testis are stimulated so that circulating testosterone levels are similar to those observed in adult men. Peak testosterone concentrations occur at approximately 2 to 3 months of age and typically decline by 6 months of age. Among preterm male infants, LH and testosterone concentrations are higher than among full-term infants; phallic growth was positively correlated with urinary testosterone levels, and testicular growth was positively correlated with urinary FSH levels. Despite the “adult-like” endocrine milieu of increased gonadotropin secretion and elevated testosterone concentrations during the first few months of life, sexual hair does not develop and gametogenesis is not initiated, presumably due to limited AR signaling in the skin and the immature Sertoli cell. Gonadotropin and testosterone secretion during minipuberty promote anchoring of testes in the scrotum, active Sertoli cell proliferation, and expansion of the pool of germ cells. ,
In contrast to the brief peak of LH and testosterone secretion within 24 to 48 hours of birth in infant boys, infant girls show a gradual increase in gonadotropin and estradiol concentrations, with estradiol concentrations typically declining around 6 months of age; LH concentrations decline, whereas FSH concentrations tend to remain higher until 3 to 4 years of age. This transient hypothalamic-pituitary-ovarian axis activity is associated with a transient increase in antral follicles. ,
Utilization of reference ranges for gonadotropins and reproductive hormones during minipuberty can help the management of infants with suspected differences in sex development (DSD) or suspected congenital hypogonadotropic hypogonadism. Although LH and FSH concentrations overlap, LH is higher in boys and FSH is higher in girls. The LH/FSH ratio with a cut-point of 0.32 accurately distinguished boys from girls. AMH concentrations serve as a marker of testicular function and can also separate boys from girls. Girls with Turner syndrome have elevated gonadotropin concentrations. Approximately 25% of men with congenital hypogonadotropic hypogonadism experienced bilateral cryptorchidism. Obtaining LH, FSH, and testosterone concentrations during minipuberty may facilitate early diagnosis of congenital hypogonadotropic hypogonadism. Hence, this brief period of the HPG axis in infancy can be utilized to assess gonadal function in infants with DSD or suspected CHH.
Full hormonal responsivity of the gonad is acquired during childhood. However, the GnRH pulse generator has been brought into check by this stage of development, resulting in the hypogonadotropic state that guarantees continued gonadal quiescence until the prepubertal phase of development is terminated by a resurgence of GnRH pulse generator activity ( Fig. 18.6 ). During childhood and juvenile development, the GnRH neurons, pituitary gonadotrophs, and the cells of the gonads are not limited to the onset of gonadarche. , ,
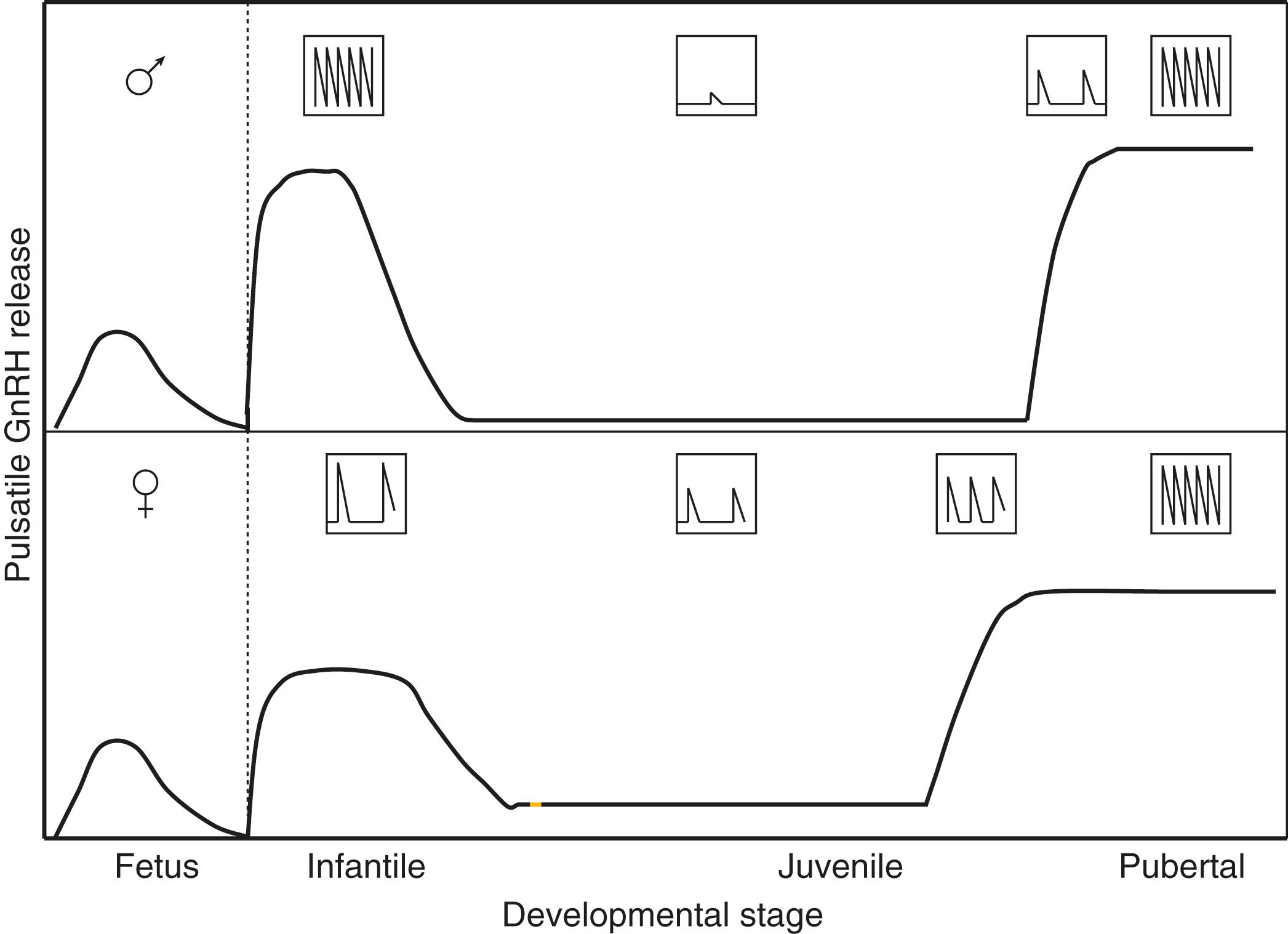
Because GnRH is secreted in only picogram quantities into the hypophysial portal circulation, changes in the concentration of this neuropeptide in the peripheral circulation do not reflect hypothalamic activity. Therefore, studies of the dynamics of the pubertal resurgence of GnRH pulse generator activity in humans and other higher primates have generally utilized the high-fidelity relationship that exists between the frequencies of pulsatile GnRH release and episodic LH secretion. The latter may be tracked with relative ease by measuring moment-to-moment changes in LH concentrations in the peripheral circulation. Although the pubertal increase in hypothalamic GnRH drive to the gonadotroph probably involves both frequency and amplitude modulation of the GnRH pulse generator, the relationship between GnRH and LH pulse amplitude is more complex than the relationship between frequency because amplitude modulation of LH release may not always reflect changes in GnRH pulse amplitude. During the initiation of gonadarche in both boys and girls, LH pulse frequency accelerates and LH pulse amplitude increases in association with amplification of a preexisting sleep-related diurnal pattern in release. This change in neuroendocrine activity may occur before the physical changes of gonadarche are manifest. Particularly in boys, LH pulse frequency appears to decline later in pubertal development, probably due to a negative feedback action of rising testosterone concentrations. A longitudinal study of the agonadal monkey suggests that, as in humans, the pubertal acceleration of pulsatile GnRH release is an early neurobiological event in the initiation of gonadarche, and that it is a rapidly completed process. Thus the slow tempo of the overall progression of puberty probably results from mechanisms downstream from the hypothalamus, particularly at the level of the pituitary.
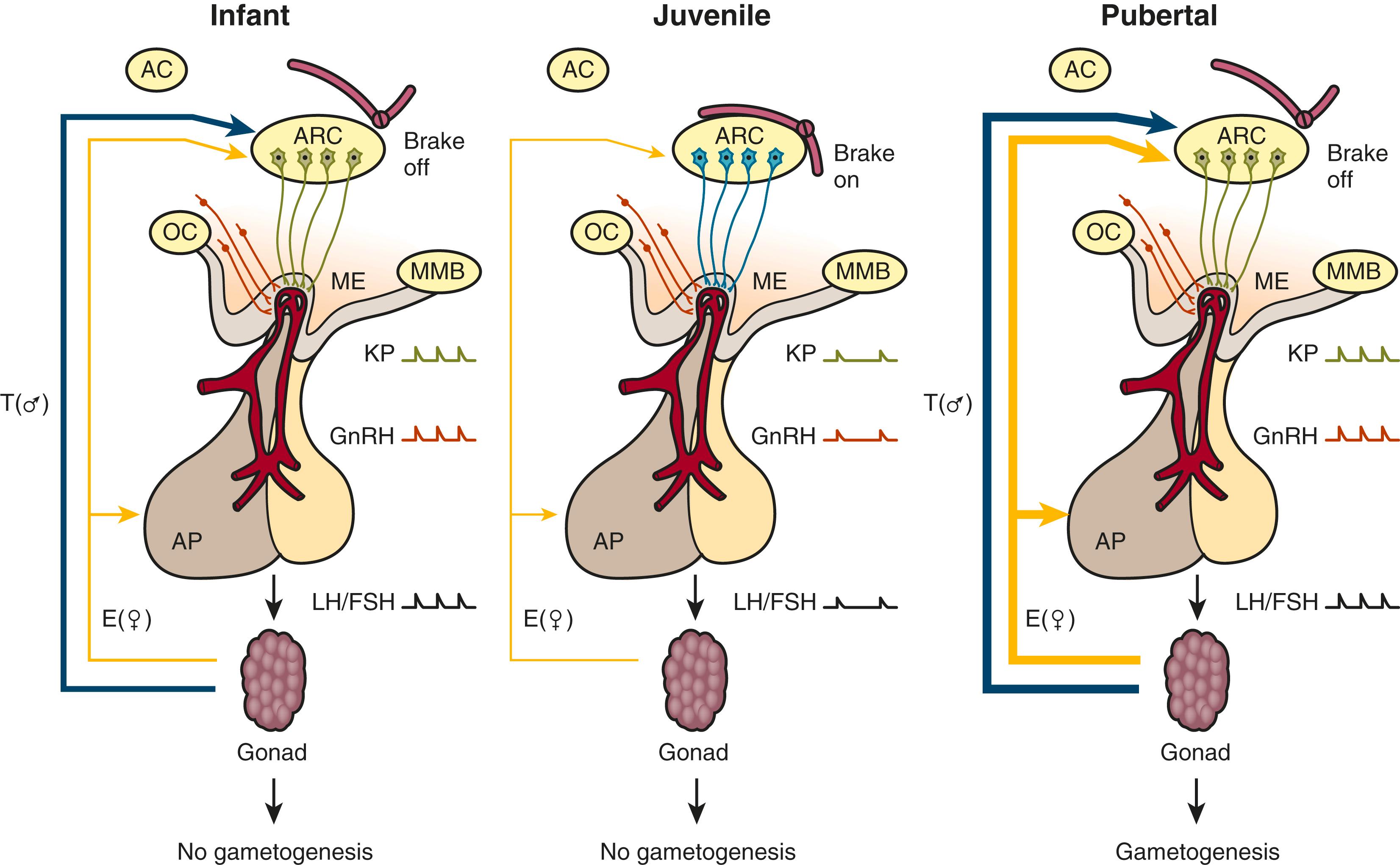
The control system that dictates the up-down-up pattern of GnRH pulse generator activity from birth until puberty may be viewed as a neurobiological “brake” (or central restraint, as it has been previously described in the pediatric literature) that holds GnRH neuronal activity in check during the greater part of prepubertal development. Here it is important to recognize that the notion of a brake is conceptual; that is, the pubertal resurgence in robust GnRH pulsatility could be occasioned either by the removal of an inhibitory input or by the application of a stimulatory signal to the GnRH pulse generator, or a combination of the two. A similar argument may be applied to the earlier transition between infancy and childhood when GnRH pulsatility is markedly diminished. The neurobiological brake on pulsatile GnRH release throughout childhood and juvenile development is imposed in the absence of the ovary or testis. Consequently, the characteristic pattern of gonadotropin secretion observed during postnatal development in humans with robust gonadotropin secretion during infancy and puberty—separated by a prolonged hiatus in LH and FSH secretion—is maintained in the agonadal situation ( Fig. 18.7 ). , ,
Similarly, LH concentrations are generally elevated in male infants with partial androgen insensitivity, which is associated with higher testosterone levels. Yet, infants with complete androgen insensitivity often fail to demonstrate a postnatal rise in LH and testosterone secretion. The latter finding is counterintuitive; an understanding of the molecular basis of this phenomenon may reveal fundamental insights into the ontogeny of GnRH pulse generation.
In agonadal children, the degree of the prepubertal suppression of gonadotropin release is less than that observed in eugonadal individuals. Interestingly, levels are higher in girls than boys in agonadal children circulating gonadotropin, indicating that the intensity of the neurobiological brake imposed on the GnRH pulse generator during prepubertal development is less in females than in males. As a result, the gonadotropin drive to the prepubertal ovary stimulates a low level of estradiol secretion through which negative feedback action on LH and FSH release, amplifying the relatively weaker neurobiological brake restraining gonadotropin secretion in the prepubertal girl. This sex difference in the strength of the neurobiological brake on prepubertal GnRH release is associated with a shorter duration of the brake in girls, which probably accounts for the relatively earlier age of gonadarche in the female. These and other sex differences in the developmental control of the GnRH pulse generator are presumed to result from greater exposure of the fetal male hypothalamus to testosterone.
Transcriptional regulation of pubertal onset is proposed to be controlled by a system of overlapping gene networks organized in a hierarchical manner. , The highest level of control is the transcriptional regulators that direct the expression of subordinate genes leading to pubertal onset. Transcriptional regulators of puberty include the POU-domain gene Oct2 , the homeodomain gene Ttf1/Nkx2.1 , the novel gene Eap1 (Enhanced At Puberty1), and LIN28B . A tumor-related gene (TRG) network has also been identified as transcriptional control of puberty. The TRG network transcriptional regulation of puberty is proposed to function through hubs that are connected to both upper echelon TRG genes ( Oct2, Ttf1, Eap1) and subordinate genes ( KISS1, GPR54, and TSLC1) involved in the regulation of puberty. Kiss1 expression is controlled by both non-TSG transcriptional regulators (TTF1 and Eap1) and TSG transcriptional regulators (CUTL1 and YY1).
Puberty onset is held in check by transcriptional and posttranscriptional repression of genes that are stimulatory to the pubertal process. LIN28A and B block maturation of let-7 MiRNA precursors, whereas excess LIN28B derepresses let-7 miRNA target genes. Other transcriptional repressors include zinc finger (ZNF) genes, POZ-ZF (poxvirus and zinc finger) family of transcriptional regulators, POK (POZ and Krüppel), and PcG (the Polycomb group). The latter three are part of the TSG network.
The discovery in 2003 that loss-of-function mutations in KISS1R in humans were associated with HH and delayed or absent puberty , demonstrated the critical role of kisspeptin in regulating GnRH secretion. Subsequent studies using several different experimental models validated this finding. These data led to the proposal that a major component of the neurobiological brake imposed upon pulsatile GnRH release during the greater part of prepubertal development is due to a hiatus in a stimulatory kisspeptin input to the GnRH neuronal network. This proposal was based on findings in the monkey, that hypothalamic expression of KISS1 and release of kisspeptin in the region of the median eminence increase at the time of the pubertal resurgence in GnRH pulsatility. Additionally, intermittent administration of kisspeptin at hourly intervals during juvenile development elicits a precocious and sustained adult-like pulsatile pattern of GnRH and the pubertal increase in GnRH release may be suppressed by the administration of a KISS1R receptor antagonist directly to the median eminence.
The finding in humans that loss-of-function mutations in the NKB signaling pathway are associated with a phenotype similar to that reported earlier for inactivating mutations in KISS1R, together with the observation that these two neuropeptides are coexpressed in the same neurons (KNDy neurons) in the arcuate nucleus, have led to the concept that these KNDy neurons are responsible for the generation of GnRH pulsatility. Thus kisspeptin expressing KNDy neurons in the arcuate nucleus comprises a critical component of the GnRH pulse generator. In the absence of an intact kisspeptin signaling pathway, the output of the GnRH pulse generator will be abrogated and pulsatile GnRH release will be compromised, resulting in a delay or absence of puberty. Overall, available data suggest that the KNDy neurons in the arcuate nucleus themselves do not govern the timing of puberty; rather, these neurons appear to be subservient to upstream regulatory mechanisms that govern the developmental pattern of pulsatile GnRH release and the onset of puberty ( Fig. 18.8 ).
The nature of the upstream pathways that comprise the neurobiological brake on the GnRH pulse generator during childhood and juvenile development remains poorly understood.
Studies of the female rhesus monkey provide evidence that GABA, the major inhibitory neurotransmitter in the brain, is upregulated during juvenile development, and inhibition of GABA tone in the hypothalamus of the prepubertal monkey leads to precocious menarche and ovulation. Interestingly, infusion of the GABA antagonist bicuculline into the median eminence of prepubertal female monkeys stimulates the release of kisspeptin-54 into this region of the hypothalamus in association with that of GnRH, and the bicuculline-induced GnRH release is blocked by simultaneous infusion of a kisspeptin antagonist. However, it is unclear what reduces GABA inhibition prior to puberty, where the relevant GABAergic neurons are physically located, and how GABA signaling interacts with the GnRH pulse generator.
Other transsynaptic signals implicated in the regulation of the pubertal resurgence of GnRH pulse generator activity include glutamate and neuropeptide Y (NPY). Glutamate is the major excitatory neurotransmitter in the brain and, in contrast to GABA, hypothalamic release of this amino acid is increased at the time of puberty in the female monkey. Also, as discussed earlier in the chapter, repetitive activation of glutamate receptors in the juvenile monkey rapidly leads to the onset of precocious gonadarche. NPY neurons are found in the arcuate nucleus and, in the male rhesus monkey, NPY gene expression in the hypothalamus is inversely related to the up-down-up pattern of GnRH pulse generator activity from birth to puberty. NPY receptors are inhibitory G protein receptors, and their activation leads to hyperpolarization and inhibition of neural activity. However, pharmacological approaches failed to demonstrate that inhibition of NPY signaling in the hypothalamus of the juvenile monkey did not promote GnRH release.
While neuroglia have classically been regarded as subserving only a “supporting role” in the central nervous system (CNS), contemporary views hold that these nonneuronal cells play important functional roles within the brain. Moreover, in the context of the hypothalamus, secretion of TGF-α by astroglia has been postulated to provide the GnRH neuronal network with a stimulatory input at the time of puberty. ,
Attempts to elucidate the neural mechanism dictating the postnatal pattern of GnRH pulse generation have traditionally led investigators to focus on relative isolation on a “favorite” signaling pathway. Using a systems biology approach, global gene discovery has been combined with computational (in silico) biology to identify functional linked networks of hypothalamic genes that are found to be associated with changes in GnRH pulse generator activity. The initial gene discovery approach was conducted without regard to the phenotype of the cells in which the respective genes are expressed and gene networks are operating. Available data indicate that the developmental changes in the transcriptional factors and gene network relevant to GnRH secretion lie upstream of the KISS1 gene and are therefore upstream of GnRH pulse generation. This network of genes serves as a governing hierarchy to orchestrate the resurgence of pubertal GnRH release and, therefore, modulate the timing of puberty. Since such networks of genes are further proposed to function in the absence of signals derived from the periphery, they may conceptually be viewed at a systems level as comprising a pubertal clock (discussed later in the chapter).
Expression of two such transcriptional regulators enhanced at puberty 1 ( EAP1, also known as interferon regulatory factor 2 binding protein-like) and thyroid transcription factor-1 ( TTF-1, also known as NKX2-1 ), increase in the mediobasal hypothalamus of nonhuman primates at puberty. EAP1 is expressed in kisspeptin neurons in the arcuate nucleus of the monkey. Moreover, its expression in the hypothalamus increases at the time of puberty in the female monkey, and the knockdown of EAP1 using a lentivirus approach interrupts menstrual cyclicity in the adult female. Additionally, a single nucleotide polymorphism (SNP) upstream of the EAP1 gene has been associated with irregular menses in the monkey. , Conditional deletion of Ttf1 from terminally differentiated hypothalamic neurons was associated with delayed puberty, decreased Kiss1 expression, and subfertility. Genetic analysis failed to identify germline mutations in either EAP1 or TTF-1 in patients with HH. Nevertheless, both EAP1 and TTF-1 are functionally connected to genes identified by genome-wide association studies (GWAS) to influence age at menarche.
Additional evidence supporting the concept of a gene network to determine the level of function of the GnRH pulse generator have identified a cohort of genes that encode for a group of transcriptional suppressor proteins known as the polycomb group. In the pubertal rat, expression of these genes is downregulated by DNA methylation, leading to a reduction in the silencer proteins. Two of the polycomb group genes are expressed in the arcuate nucleus, and overexpression of one of these genes resulted in a decrease in Kiss1 expression associated with delayed vaginal opening and a disruption of GnRH pulsatility in mediobasal hypothalamic (MBH) explants. A global inhibition of DNA methylation in prepubertal rats resulted in the delay of the vaginal opening, indicating that epigenetic regulation of gene expression may be important in timing puberty. Similarly, data obtained in monkeys indicate that some zinc-finger transcriptional repressors restrain puberty by epigenetically repressing a gene network that operates in the arcuate nucleus and controls puberty by governing pulsatile GnRH release.
Additional support for a zinc-finger motif containing a suppressor gene holding puberty in check has come from human mutation studies. Using whole exome sequencing (WES), inactivating mutations in the gene encoding for makorin RING finger protein 3, MKRN3 , are the most common genetic defects associated with CPP to date. The MKRN3 protein is the first protein to be identified with an inhibitory role in GnRH secretion identified in humans to have reported mutations. It is a maternally imprinted gene on chromosome 15q11.2 in the Prader-Willi critical region. Whereas the maternally inherited allele is methylated in the central nervous system, loss of function mutations are paternally inherited. The makorin family of proteins contains a particular zinc-finger motif that has been associated with ubiquitination, a process that is involved in protein trafficking, which in some cases leads to protein degradation. The relevance of this gene to puberty is further supported by demonstrating declining levels of circulating MKRN3 product prior to pubertal onset in Danish girls and boys. , Expression of this gene decreases immediately before the onset of puberty in the mouse hypothalamic arcuate nucleus. Thus, in mice, makorin-3 appears to contribute to the neurobiological brake on GnRH secretion via neurons producing kisspeptin and neurokinin B. MKRN3 is expressed in KISS1 neurons in the hypothalamic regions critical for puberty initiation and reproduction in rodents and nonhuman primates and associates with KISS1 and TAC3 gene promoters to repress transcriptional activity. In female mice, the prepubertal decline in MKRN3 was shown to be independent of gonadal activation. Together, these studies support the concept that transcriptional repression is a core component of the neuroendocrine circuitry that regulates the timing of puberty.
Recently, genome-wide association studies indicate that several specific loci on the human genome are associated with variations in the age at menarche. , An SNP that was consistently and strongly associated with an earlier age at menarche was found on chromosome 6 near LIN28B, which encodes for a micro-RNA binding protein. The differences in age at menarche in subjects with and without variants in the region of LIN28B are small (1–2 months) compared to the recognized range for the age at menarche in the population at large that may extend from 10 to 16 years of age. A genetic study on girls with early puberty did not find any potentially responsible LIN28B mutations. It is currently unclear the exact causal role of LIN28-Let7, if any, in timing of human puberty. A follow-up meta-analysis study using genome-wide and custom-genotyping arrays in more than 180,000 European women found strong evidence for 123 SNPs at 106 gene loci to be associated with earlier age at menarche. Surprisingly, only a few known puberty genes overlap with the genes associated with age at menarche. These include MKRN3 , LEPR , IGSF1 , and TACR3 , further implicating biological significance of these genes in pubertal development.
Regardless of the components of the neurobiological brake, which dictate the up-down-up pattern of pulsatile GnRH release from birth until puberty, it is to be anticipated that application and withdrawal of the brake will be associated with a corresponding structural remodeling in those hypothalamic neuronal and glial circuits involved. In this regard, the hypothalamus of the postnatal brain retains its capacity for plasticity, as reflected by the expression of polysialic acid-neural cell adhesion molecule (NCAM).
A significant number of boys and girls with delayed puberty do not have a readily recognizable diagnosis, such as an anatomic lesion of the hypothalamus or pituitary, primary gonadal insufficiency, or functional hypogonadotropic hypogonadism due to illness, stress, or negative energy balance. A study of 16 children with delayed puberty showed that unstimulated LH measurements, the presence of overnight LH pulses, and GnRH-stimulated LH secretion did not distinguish between children who would and would not progress through puberty; however, LH response to kisspeptin did distinguish between the two groups. All participants who had a rise in LH of 0.8 mIU/mL or greater in response to kisspeptin progressed through puberty and those with an LH response less than or equal to 0.4 mIU/mL reached age 18 years without developing signs of puberty. The sensitivity and specificity of the kisspeptin stimulation test were both 100%.
The physiological control systems that dictate the timing of puberty have intrigued investigators for decades, but the governance of this fundamental developmental event in humans remains largely a mystery. However, two basic schemata have been proposed. In the first, a cue to reawaken the GnRH pulse generator is provided by the attainment of a particular state of somatic maturation. According to this hypothesis, the brain receives this information by way of a signal in the circulation that is tracked by a somatometer resident within the CNS. In the second schema, a pubertal clock (presumably resident in the CNS) generates the signal.
In the case of the somatometer hypothesis, the attainment of a particular proportion of body fat has long been argued to be necessary for the onset of gonadarche. Interest in this hypothesis was rekindled with the discovery of leptin and its receptor. Leptin, encoded by the leptin ( LEP ) gene, is primarily secreted by adipocytes and regulates feeding behavior and body weight by providing the hypothalamus with information on fat mass and energy status. The leptin receptor (LEPR) is a single transmembrane type I cytokine receptor of the IL-6 receptor family that is encoded by the LEPR gene located at chromosome 1p31.3. Although multiple LEPR isoforms exist, leptin action is primarily mediated by the long form of the LEPR.
Individuals with mutations in leptin signaling, resulting from loss-of-function mutations in either LEP or LEPR, have failed to progress through puberty. The phenotype of LEPR mutations included morbid obesity, abnormal eating behaviors, and lack of spontaneous pubertal development. Over the last 15 years, leptin replacement has been administered to several patients with leptin deficiency due to LEP mutations. When administered at an appropriate age for puberty, leptin promoted pubertal development. Importantly, there has been no evidence of premature puberty in younger children following replacement treatment. Moreover, leptin concentrations among children with GnRH-dependent precocious puberty correlated with BMI and not with pubertal status. These findings provide compelling evidence that the action of leptin, albeit obligatory for the onset of puberty, is nevertheless permissive. This action of leptin may require only low circulating levels of the adipocyte hormone because pubertal development in both male and female subjects with various lipodystrophies has been reported to be normal despite low leptin concentrations.
In girls, it is generally recognized that plasma leptin levels increase progressively through breast stages 1 to 5. In boys, plasma leptin levels also increase during early puberty to reach peak levels between 10 and 12 years of age but subsequently decline as blood levels of testosterone rise into the adult range. The finding that circulating concentrations of the soluble form of the LEPR progressively decrease during childhood until approximately 11 years of age suggests that the increase in bioavailable leptin during early puberty may be greater than that reflected in total leptin concentrations.
Considerable interest is focused on identification of the leptin’s neuronal targets that promote pubertal progression. While it is generally recognized that GnRH neurons do not express LEPR , data obtained in rodents and sheep for the KNDy neurons are inconsistent and the site of action of leptin may be upstream of the GnRH pulse generator. One potential mechanism demonstrated in mice is that leptin induces phosphorylation of neuronal nitric oxide synthase (nNOS) in nNOS-expressing neurons in the preoptic region, which leads to increased LH secretion independent of kisspeptin/KISS1R signaling. Thus NO signaling may play a major role in the crosstalk between leptin and the reproductive axis. ,
Before leaving the subject of leptin and obesity, it should be noted that although overweight girls tend to “mature” earlier, sleep-related increments in LH secretion, and therefore presumable GnRH pulse generator activity, have recently been reported to be blunted in healthy obese premenarcheal girls.
Other somatic factors have been proposed to serve as signals to the somatometer. The hypothesis that such a factor is skeletal in origin is based on the finding that, in children with an accelerated or retarded maturational tempo, menarche and testicular enlargement correlate better with skeletal age than with chronological age. Skeletal age, also known as “bone age,” is a surrogate marker of biological maturation and may be determined by comparing a radiograph of the left hand to gender-specific standards obtained at various chronological ages. Several caveats are relevant when assessing skeletal maturity. The degree of skeletal maturation at various sites, such as hands, elbows, and knees, may differ. Skeletal maturation of the carpal bones, distal radius, and distal ulna often lag behind the metacarpals and phalanges. Nevertheless, the association between bone age and the onset of gonadarche is maintained in disorders of growth. In children with constitutional delay of growth and true isolated growth hormone (GH) deficiency, gonadarche occurs at a late chronological age, but at a normal skeletal age. On the other hand, when skeletal maturation is advanced—as may occur in association with CAH or familial testotoxicosis—secondary GnRH-dependent precocious puberty may develop. Although proteins synthesized in bone enter the vascular compartment, , the ability of osteocalcin and other bone proteins to modulate the activity of the GnRH pulse generator has not been addressed. ,
GH secretion during childhood is relatively stable, but GH release is amplified up to threefold in boys and girls with the initiation of gonadarche and the rise in circulating levels of sex steroids. The pubertal increase in GH, in combination with the increased circulating concentrations of insulin-like growth factor 1 (IGF-I), estrogens, and androgens, contributes to the adolescent growth spurt. The increased secretion of GH at the time of gonadarche is not sustained; by late puberty, GH levels begin to decline. During puberty, height velocity, IGF-1 concentrations, and sex steroid concentrations rise with the synchronization of these changes within individuals. Because the increases in GH and IGF-I appear to be in response to the initiation of gonadarche, particularly to increased gonadal steroid secretion at this time, neither GH nor IGF-I represents compelling candidates for the signal responsible for the resurgence of GnRH release.
At the time of gonadarche, insulin resistance increases. This insulin resistance is greatest among children in Tanner stages 2 and 3 when compared with prepubertal children and adults. Manifestations of insulin resistance appear to be limited to effects on carbohydrate metabolism and are associated with compensatory hyperinsulinemia and normal disposition index. Disposition index, a function of insulin sensitivity and insulin secretion, reflects beta-cell response for a given insulin sensitivity. Using euglycemic-hyperinsulinemic clamp studies in conjunction with an investigation of substrate utilization, one longitudinal study found that puberty was associated with decreased insulin sensitivity, increased insulin secretion, increased total body lipolysis, decreased glucose oxidation, and increased IGF-1 concentrations. In a longitudinal prospective cohort study involving healthy children, insulin sensitivity decreased before the onset of physical features of puberty. In one study, no changes in insulin’s ability to suppress hepatic glucose production were noted during puberty, indicating that decreased insulin sensitivity is limited to peripheral glucose uptake. , Although cross-sectional studies suggest that the magnitude of insulin resistance is influenced by BMI (body weight/height 2 [kg/m 2 ]), gender, and ethnic background, these associations have not been consistently noted in longitudinal studies. IGF-1 concentrations during puberty have been reported to mirror those in insulin sensitivity. Mean 24-hour serum GH and IGF-I concentrations positively correlate with the degree of insulin resistance during puberty, and the pubertal changes in insulin sensitivity may be partially mediated by increased GH and IGF-I concentrations. Yet, while it is generally recognized that increased gonadal steroid levels are responsible for activation of the GH/IGF1 axis at puberty (discussed earlier in the chapter), a causal relationship between testosterone or estradiol and insulin resistance has not been demonstrated.
Treatment of nonobese Catalunyan girls with premature adrenarche and advanced skeletal maturation with the insulin sensitizer metformin was associated with later onset of breast development and menarche. Moreover, in this relatively homogenous ethnic population, metformin treatment for 3 years slowed pubertal tempo among low birth weight girls at risk for early puberty and shorter adult stature. Considered together, the foregoing considerations raise the possibility that decreased insulin sensitivity may represent a component of the cue that times the onset of gonadarche.
Ghrelin is a small peptide, secreted predominantly by the stomach, which circulates in two forms. The active form is acetylated and promotes GH secretion. Ghrelin influences food intake, sleep, body weight, gastrointestinal mobility, and reproduction. , Ghrelin suppresses LH pulsatility in the pituitary. Circulating ghrelin concentrations are higher during fasting and decrease after food intake, indicating that ghrelin signals energy-deficient states and modulates appetite and carbohydrate metabolism. Ghrelin concentrations peak during the first 2 years of life and decrease during puberty. Negative correlations were found between ghrelin concentrations and both age and pubertal stage. The ghrelin receptor, GH secretagogue receptor-1a (GHSR-1a), is expressed in the human hypothalamus, pituitary, testis, and ovaries.
Ghrelin and leptin appear to act as reciprocal regulators of energy homeostasis exerting opposing influences on the HPG axis. Adipokines, hormones secreted by adipocytes, include resistin, adiponectin, leptin, and visfatin. Adiponectin concentrations are inversely related to insulin resistance and have been reported to decrease in pubertal males. Resistin is secreted by adipocytes and peripheral blood mononuclear cells and signals through the Toll-like receptor 4. Although details regarding its physiologic role are unclear, resistin appears to promote insulin resistance. The respective roles, if any, of resistin and visfatin on gonadarche or adrenarche remain to be determined.
The primary signal for adrenarche remains to be elucidated.
Adrenarche appears to occur independently of developmental changes in the HPG axis (gonadarche).
Adrenarche is characterized by increased DHEAS secretion by the ZR associated with increased expression of CYB5A.
Adrenarche is characterized by the development of the ZR in the adrenal cortex and increased DHEA, DHEAS, and androstenedione secretion. DHEA and DHEAS are not bioactive androgens; DHEA is a precursor for more potent sex steroids. Prior to birth, the fetal zone of the adrenal cortex produces large amounts of DHEAS, which serves as the precursor for placental estrogen synthesis. Following involution of the fetal zone after birth, DHEAS concentrations remain low until 6 to 7 years of age, at which time they begin to increase. Rising DHEAS secretion is the earliest hormonal manifestation of adrenarche. Despite no obvious changes in ACTH or cortisol secretion, the failure of children with ACTH receptor mutations to experience adrenarche implicates ACTH in this process.
The onset of adrenarche is associated with increased 17,20-lyase activity and decreased 3β-hydroxysteroid dehydrogenase activity. Available data suggest that, at the time of adrenarche, changes in expression of CYB5A, DHEA sulfotransferase (SULT2A1), and 3β-hydroxysteroid dehydrogenase (HSD3B2) play essential roles in DHEA and DHEAS production. Histologically, increased thickness of the ZR occurs concurrently with the increase in DHEAS concentration. Using more sensitive methodologies such as urinary GCMS, studies indicate that concentrations of DHEA and its metabolites show a continuous rise beginning at 3 to 4 years of age. These data contradict conventional concepts and imply that adrenarche is a gradual process beginning earlier than previously considered. DHEAS concentrations continue to rise and peak between 20 and 25 years of age, followed by progressive decline. Although the findings have been inconsistent, clinical studies suggest that insulin, IGF-I, and GH concentrations influence the timing, onset, and progression of adrenarche. Comparisons of IGF-I concentrations among prepubertal children have shown higher concentrations in African-American children. Whether these ethnic differences contribute to the earlier onset of adrenarche or the increased incidence of premature pubarche in African-American girls is unknown. Clinical observations regarding associations between adrenarche and body size and fatness have been inconsistent; one longitudinal study showed that DHEAS concentrations increased commensurate with the largest increase in BMI, whereas another longitudinal study found no association between DHEAS and weight, BMI, or body surface area.
Despite numerous hypotheses, the primary signal for adrenarche remains to be elucidated. Hypothalamic releasing factors, such as corticotropin-releasing hormone (CRH) and vasopressin (antidiuretic hormone [ADH]), play important roles in governing hypothalamic-pituitary-adrenal axis function. However, in contrast to the role of hypothalamic GnRH in gonadarche, CRH and ADH do not appear to trigger the onset of adrenarche. No specific adrenal androgen-stimulating factors have been isolated from the pituitary.
Moreover, adrenarche occurs independently of developmental changes in the HPG axis. For example, children with gonadal dysgenesis experience normal adrenarche and pubarche, whereas children with primary adrenal insufficiency may have normal gonadarche. Nevertheless, despite the long-established notion that gonadarche and adrenarche are independent events, recent data hint that adrenal androgens may influence pubertal timing. Specifically, using methodology with lower limits of detection, these data show that higher prepubertal urinary androgen excretion correlated with an earlier onset of breast development and penile growth, respectively, and with a shorter duration of pubertal growth spurt.
The temporal correlation between pubertal stages in families indicates genetic influences on the timing of puberty.
GWAS studies identified several loci associated with age at menarche.
Epigenetic factors, methylation status, noncoding regulatory sequences, enhancers, insulators, and other factors modify expression of genes involved in the pubertal process.
The observations that (1) the temporal correlation in somatic maturation and attainment of pubertal stages in monozygotic twins is more robust than that in dizygotic twins ; (2) the age of menarche in mothers and daughters is correlated; (3) pubertal onset in children is influenced by pubertal timing of both parents ; (4) precocious gonadarche in girls may be familial and transmitted in an autosomal-dominant mode ; (5) paternally inherited precocious puberty is associated with MKRN3 mutations ; and (6) the age of menarche varies with racial group, underline the major influence of genetic factors for timing the onset of puberty. However, the nature of the genetic factors that directly dictate the timing of the pubertal resurgence of GnRH release remains to be elucidated. These genetic factors could include a network of clock genes in analogy to circadian timekeeping and/or could affect the balance between stimulating and repressive factors that govern GnRH release.
Despite the accelerating knowledge regarding the genetic causes of disorders of pubertal development, the identity of the specific puberty genes that dictate the timing of the resumption of pulsatile GnRH release (and therefore determine the age of gonadarche) and those that regulate the developmental increase in adrenal androgen secretion that determine the age of pubarche have yet to be identified. The whole genome can be interrogated by linkage and GWAS without a priori hypotheses regarding specific candidate genes. This approach has enabled the examination of millions of loci across the genome. However, these data explain <3% of the variance in age at menarche.
Four GWAS studies identified genomic loci associated with age at menarche. Among more than 100 loci identified in those GWAS, only several loci, LEPR , GNRH1 , TACR3 , and IGSF1, were near genes known to be associated with disorders of puberty ( Fig. 18.9 ). To date, the largest genetic effect on pubertal onset has been associated with FSHR and FSH. However, none of the approximately 50 genes associated with central disorders of puberty timing genes appear to be responsible for triggering the pubertal resumption of pulsatile GnRH secretion.
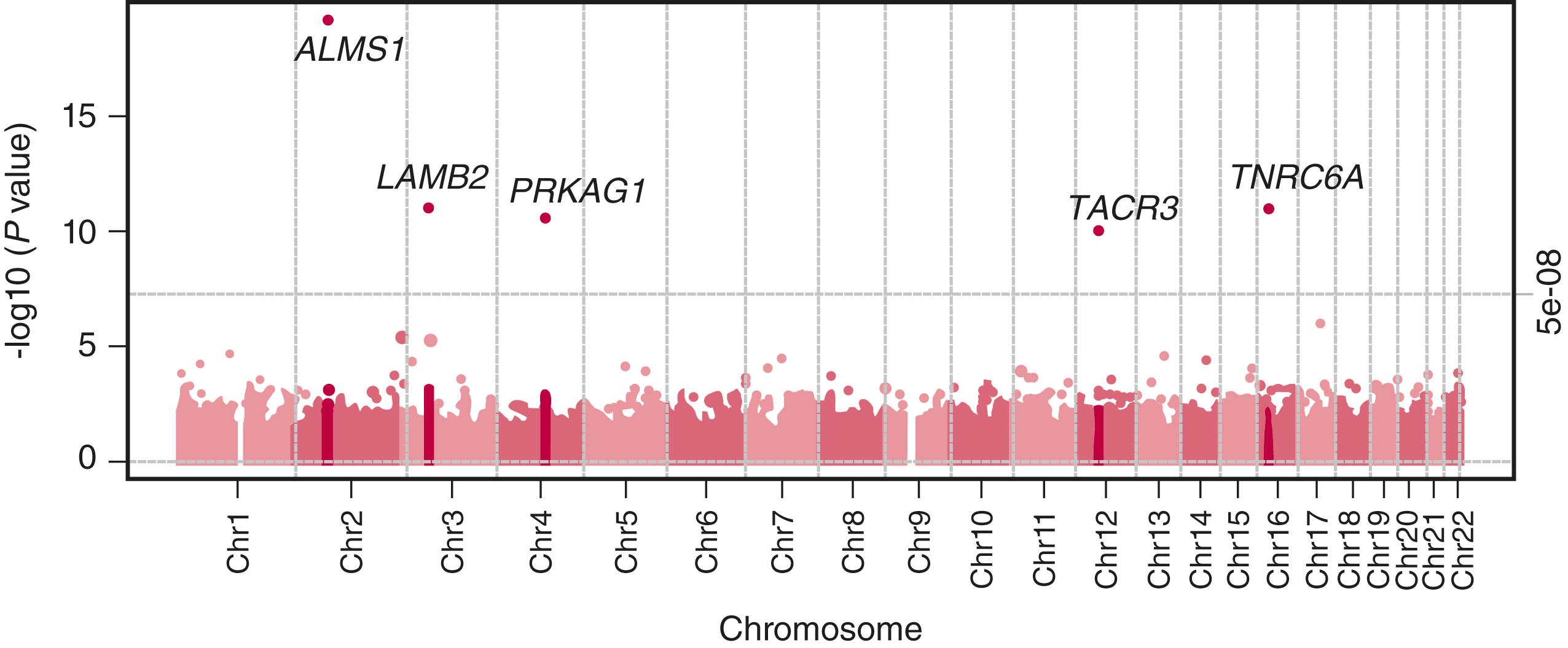
An increasing number of factors have been identified that modulate and regulate gene expression through epigenetic mechanisms. As described above, some growth regulatory genes show parent-of-origin effects due to genomic imprinting. Developmental disorders, such as Prader-Willi syndrome, in which the timing and tempo of puberty is altered (discussed later in this chapter), are associated with abnormal expression of imprinted genes. Maternal nutritional status can influence fetal gene expression through epigenetic mechanisms; animal studies suggest that this effect may be transmitted to subsequent generations. , Environmental factors, such as nutrition, hormone and chemical exposures, and physical elements, can alter gene expression through these epigenetic modifications. These disorders may represent the “tip of the iceberg” for epigenetic influences on the genes that dictate or modulate the timing of puberty.
Genetic information and transcriptional regulation are influenced by DNA (CpG) methylation, chromatin packaging, small RNA or micro-RNA effects, noncoding regulatory sequences, long-range transcriptional regulation (enhancers and insulators), and long noncoding RNA. Interactions between genes and proteins provide another mechanism to modify gene expression. In a longitudinal puberty study involving healthy children, changes in peripheral blood DNA methylation were detected. Several differentially methylated CpGs were found to be associated with changes in testosterone, FSH, AMH, LH, and inhibin B concentrations in boys. One region, situated between Solute Carrier Family 12 Member 9 ( SLC12A9 ) and Thyroid Hormone Receptor Interactor 6, 7q22 ( TRIP6, ) at chromosome 7q22, was coordinately regulated as a function of pubertal development. The function of the protein encoded by SLC12A9 is unclear. Curiously, circulating TRIP6 concentrations were noted to increase during puberty.
Although many genes have been associated with puberty, none have been confirmed to regulate the reactivation of the GnRH pulse generator. Hence, the term “puberty” gene should be restricted to those genes that specifically regulate the timing of either adrenarche or gonadarche. For gonadarche, such genes would determine the age of the pubertal resurgence of pulsatile GnRH release by regulating the timing of the application or withdrawal of the neurobiological brake to the GnRH pulse generator of the juvenile hypothalamus. Puberty genes could time the resurgence of pulsatile GnRH release not only by triggering a hypothalamic signal at puberty but also potentially by determining the duration of the prepubertal brake on pulsatile GnRH release or by timing the “turn off” of the GnRH pulse generator during infancy. In the case of adrenarche, the developmental increase in adrenal androgen secretion would presumably be timed by genes that are responsible for the changes in adrenal steroidogenesis manifested by increased ZR activity.
Adequate energy stores are essential for puberty. The association between extreme energy expenditure, undernutrition, and delayed gonadarche, especially in the female, is well established.
Endocrine disrupting chemicals (EDCs), for example, polychlorinated biphenyl (PCB), polybrominated biphenyl (PBB), bisphenol A (BPA), and similar chemicals have been suspected to be modulators of pubertal timing. EDCs have recently gained considerable attention. However, additional data are needed to establish a direct causative link.
In general, the majority of studies that have examined the influence of various parameters on the onset and tempo of puberty have focused on the process of gonadarche. Considerably less attention has been paid to the timing and progression of adrenarche. Additionally, many factors that influence the onset and tempo of puberty (e.g., nutritional status) also modulate hypothalamic GnRH pulse generator activity in the majority of adults. Therefore, a modulator of puberty is not necessarily a component of the mechanism that initiates this developmental event. Rather, it is more likely that such factors play permissive, albeit in some cases obligatory, roles to allow the process of puberty to unfold once the signal responsible for the resurgence in pulsatile GnRH secretion has been activated.
Adequate energy stores are essential for the accelerated linear growth and achievement of reproductive competence at puberty. Neuropeptides and hormones provide information regarding energy status. The nutritional state modulates the onset of gonadarche in girls, as reflected by the findings that menarche is delayed in malnourished girls, and menarche tends to occur at a particular or “critical” body weight rather than at a set age. Obesity is associated with early breast development and menarche. Rapid weight gain during the first year of life is associated with obesity and early menarche in a prospective UK cohort study. In addition, the timing of adrenarche may also be influenced by nutritional status. In the female rhesus monkey, high-calorie diets were associated with a precocious increase in nipple volume, sex-skin swelling, and menarche in association with increased concentrations of leptin and IGF-1 concentrations, and an increased BMI. , While the impact of undernutrition on gonadarche in boys has received less attention, there is no reason to suspect that the male axis is unaffected in this regard.
Leptin and osteocalcin appear to communicate energy status to several tissues including adipose tissue, bone, CNS, and gonads. In its undercarboxylated (active) form, osteocalcin promotes insulin secretion, improves insulin sensitivity, induces β-cell proliferation, increases adiponectin secretion, and decreases lipolysis. In osteoblasts, insulin signaling increases bone formation, bone resorption, and secretion of active osteocalcin production.
Differences in the timing and tempo of gonadarche unrelated to racial or ethnic background have been reported even among well-nourished girls. Researchers have suggested that diet may account for such variations. Attention has been drawn to subtle relationships between diets high in animal protein and early menarche, and between diets high in vegetable grains and delayed onset or tempo of gonadarche. Moderate to vigorous exercise in the absence of weight restriction appears to have a negligible impact on the timing and tempo of puberty in either sex. However, breast development, menarche, and skeletal maturation can be delayed in girls involved in strenuous physical training. The association between extreme energy expenditure and delayed gonadarche in the female is particularly marked in ballerinas, long-distance runners, and figure skaters, who must maintain their body weight within strict limits. Such girls may experience amenorrhea secondary to HH. The factors responsible for compromising pulsatile GnRH release in pubertal children who exercise vigorously are presumed to be similar to those resulting from undernutrition. The life of the young female dancer or athlete is stressful; therefore stress may also represent a contributing factor underlying exercise-induced delayed gonadarche. Because certain physical and psychological characteristics are necessary for outstanding athletic performances, there may be a significant contribution of self-selection involved in the decision of girls to participate at such an exceptional level. This can be complicated by eating disorders, which are more common in elite artistic athletes such as gymnasts. , In a study of young female gymnasts and their parents, menarche in the mothers was found to be delayed relative to that of mothers of sedentary girls. Therefore, the possibility of a genetic contribution to this phenomenon cannot be totally excluded. The effect of physical training on the timing of pubarche in girls has been less studied, and conclusive data are lacking.
Although the relationship between strenuous physical training and male puberty has received less attention, this developmental process is apparently less susceptible in boys than it is in girls. This may be related, in part, to sex differences in the age at which training is initiated, and to the intensity of the exercise. Nevertheless, in sports like wrestling that require weight control achieved with a combination of strenuous exercise and dietary restriction, impaired testosterone secretion may occur.
Increased attention has been focused on EDCs as modulators of pubertal timing. Most EDCs have estrogenic or antiandrogenic activities. Many occur as mixtures rather than single agents and may have multiple actions. Sexually dimorphic actions can occur. Dioxins, PCB, PBB, BPA, plasticizers (phthalates), pesticides, fungicides (vinclozolin), alcohol, tobacco, and pharmaceutical agents (diethylstilbestrol) are considered to be EDCs. , Phytoestrogens, which can function as selective estrogen receptor modulators (SERMS), are found in soybeans, flaxseed, peanuts, and some vegetables. Most are diphenolic compounds with structural features common to estrogenic steroid agonists and antagonists. EDCs can affect peripheral reproductive systems, such as inducing breast development. These chemicals may also exert neuroendocrine effects to influence hypothalamic and pituitary function. EDCs can act through hormone receptors, can affect enzymatic processes, and may disrupt the complex interactions of endogenous hormones at multiple levels. Available data suggest that EDCs are associated with both early puberty and delayed completion of puberty. The specific consequences of EDCs may depend on age and duration of exposure. Further investigations into EDCs, defining mechanisms of action and consequences, are necessary.
The roles of prenatal influences and environment on postnatal development including puberty are increasingly recognized. , Children with intrauterine growth retardation (IUGR) or born small for gestational age (SGA) have increased risks to develop insulin resistance, hypertension, diabetes mellitus, metabolic syndrome, and coronary artery disease in adulthood. Increased weight gain during the first few years of life exaggerates these risks. Increased adrenal androgen levels and in some cases precocious or exaggerated adrenarche has been reported in both boys and girls born SGA. , Some, but not all, girls with premature adrenarche have an increased risk to develop polycystic ovary syndrome (PCOS) during adolescence. Reports are inconsistent regarding the timing of menarche in girls born SGA compared to girls born at an appropriate size for gestational age. , The timing of puberty in boys born SGA appears to be normal , although subfertility has been reported after they reached adulthood. Perinatal HIV infection is associated with later onset of puberty. , Potential mechanisms responsible for the consequences of abnormal fetal growth are multifactorial and not mutually exclusive. Suggested causes include genetic variations, differential methylation, alterations in microRNA, altered histone acetylation, and other long-range regulatory effectors.
The neuroendocrine consequences of prenatal androgen exposure have been best characterized in sheep. Findings include decreased sensitivity to estradiol and progesterone feedback, decreased content of dynorphin and NKB in KNDy neurons, and altered morphology of KNDy neurons. Whether these data are applicable to humans and whether prenatal androgen exposure alters GnRH pulse generator function is unclear.
Several European countries have reported precocious gonadarche in a relatively dramatic proportion of children—particularly girls—adopted from developing world regions like Asia and South America. , This increased endocrine activity in the hypothalamic-pituitary-ovarian axis prior to the onset of physical manifestations of puberty indicates a central origin to the condition. The cause of the precocity appears to be complex, but improved nutritional and social conditions may contribute to this phenomenon. Precocious gonadarche is also seen in immigrant girls arriving with their parents and lacking evidence of earlier compromised growth and nutrition. Moreover, ethnic background does not appear to influence this trend. A study of immigrant girls in Belgium with premature gonadarche reported an association with previous exposure to organochlorine pesticides. A study in Spain found an increased relative risk for precocious gonadarche in adopted girls (both national and international), but not in immigrant girls. However, this outcome is inconsistent because adopted Chinese girls experienced menarche at ages comparable to nonadopted Chinese girls.
Approximately 90% of cases of CPP in girls are idiopathic in girls versus 50% to 70% of cases in boys.
Hypothalamic hamartomas are a common etiology of CPP.
Paternally inherited heterozygous makorin 3 (MKRN3) mutations are associated with CPP in boys and girls. Circulating makorin-3 concentrations decline immediately prior to puberty among healthy girls and boys.
The nonclassic or milder forms of the virilizing CAHs primarily present with signs of excessive androgen secretion rather than the symptoms of glucocorticoid and mineralocorticoid deficiencies typical of the classical forms.
The protein encoded by the POR gene functions as an electron donor to cytochrome P450 enzymes. Therefore, POR loss-of-function mutations present with a clinical picture showing various combinations of deficiencies of 21-hydroxylase, 17α-hydroxylase, and aromatase enzymes.
Constitutional delay in growth and puberty (CDGP) may be a normal variation of pubertal timing.
Delayed puberty can be due to gonadal failure (hypergonadotropic), neuroendocrine dysfunction (hypogonadotropic), or other endocrine disorders.
In some instances, the underlying pathophysiology may be similar for both constitutional delay and idiopathic HH.
Oligogenic etiology for HH (i.e., more than one HH-associated gene mutation is present in a patient) accounts for 10% to 20% of all cases.
Mutations in genes influencing GnRH neuron development and migration may be associated with HH.
Approximately 10% to 15% of individuals with HH who carry loss-of-function mutations experience a clinical recovery following the initiation of sex steroid replacement therapy.
Primary gonadal failure, disorders of steroidogenesis, or defects in steroid hormone action result in loss of negative feedback inhibition by gonadal steroids at both the hypothalamic and pituitary level leading to hypergonadotropic hypogonadism.
Turner syndrome (TS) is due to deletions or structural rearrangements of the X chromosome and the most common form of gonadal dysgenesis seen in 1:2000 to 1:5000 liveborn females.
In 46,XY gonadal dysgenesis, the degree of testosterone deficiency varies; therefore, the external genital development can range from normal female external genitalia to undervirilization of male external genitalia.
KS (47,XXY karyotype) is associated with failure in both testosterone production and spermatogenesis and may manifest before or after puberty.
Hereditary disorders in adrenal and/or gonadal sex steroid production may lead to ambiguous genitalia or delayed puberty.
Complete androgen insensitivity syndrome (CAIS) is usually associated with mutations in the AR ( NR3C4 ) gene.
To identify the cause of delayed puberty may require reevaluation over time.
The goal of gender-specific sex hormone replacement therapy is to mimic the normal pattern of gonadal secretion to induce secondary sexual characteristics.
Fertility issues should also be frankly discussed.
Fertility preservation should be considered if appropriate.
The terminology used to describe disorders of puberty has evolved as the pathophysiology and molecular etiologies of these disorders have been clarified ( Box 18.1 ; Table 18.2 ). The term “CPP” or GnRH-dependent precocious puberty refers to premature resurgence of GnRH pulse generator activity, which we have labeled as GnRH-dependent precocious gonadarche (CPP). The terms “partial,” “incomplete,” “peripheral,” “pseudo,” and “GnRH-independent precocious puberty” have been used to describe other etiologies of premature sexual development. We refer to these disorders as GnRH-independent precocious puberty. Isosexual refers to the development of sexual characteristics typical for the patient’s gender. Heterosexual refers to the development of sexual characteristics typical of the other gender (e.g., feminizing tumors in males). Delayed puberty has been categorized as being either hypogonadotropic (low gonadotropin concentrations) or hypergonadotropic (elevated gonadotropin concentrations). With expanding knowledge of the functional genomics of the pubertal process and the molecular genetics underlying its pathophysiology, the classification of the etiologies of disorders of puberty will continue to evolve.
Idiopathic: Progressive, nonprogressive
Congenital CNS lesions:
Hypothalamic hamartoma
Septo-optic dysplasia
Arachnoid cysts
Suprasellar cysts
Genetic:
KISS1R activating mutation
KISS1 activating mutation
MKRN3 mutation
Chromosome 14q32 variants
Acquired CNS disorders:
Postinflammatory
Postradiation therapy
Postinfectious
Hydrocephalus
Posttrauma
Tumors (astrocytoma, pineal tumor, optic glioma, craniopharyngioma)
Neurofibromatosis type 1 (optic glioma)
Tuberous sclerosis
Sturge-Weber syndrome
Histiocytosis X
Chronic exposure to androgens:
Congenital adrenal hyperplasia
Familial male-limited precocious puberty
Feminizing disorders:
Estrogen-secreting tumors
Ovarian: granulosa cell, Peutz-Jeghers syndrome, gonadoblastoma/dysgerminoma, carcinoma, cystadenoma, theca cell, lipoid
Adrenal
Feminizing disorders:
Estrogen secretion unrelated to tumors: aromatase mutation
Exposure to exogenous sex steroids or endocrine disruptors
Isolated premature menarche:
Estrogen-secreting cyst
Tumor
McCune-Albright syndrome
Primary hypothyroidism
Premature thelarche:
Variant of normal development
Rubinstein-Taybi syndrome
Kabuki make-up syndrome
Primary hypothyroidism
Virilizing disorders
Premature adrenarche
Congenital adrenal hyperplasias
21-hydroxylase deficiency
3β-hydroxysteroid dehydrogenase deficiency
11β-hydroxylase deficiency
Other disorders affecting steroidogenesis
Oxidoreductase deficiency
Apparent cortisone reductase deficiency
Apparent DHEA sulfotransferase deficiency
Inherited glucocorticoid resistance
Familial male-limited precocious puberty (testotoxicosis)
Androgen-secreting tumors
Adrenal sex steroid-secreting tumors such as adenoma and carcinoma
Ovarian tumors such as arrhenoblastoma
Testicular Leydig cell tumor
hCG-secreting tumors (e.g., hepatoblastoma/hepatoma)
Dysgerminoma
Teratoma
Choriocarcinoma
Cushing syndrome
Cushing disease associated with increased ACTH secretion
Adrenal tumors/disease
Ectopic secretion of CRH or ACTH
Prolonged excessive exogenous steroid use
| Gene | Locus | Phenotype |
|---|---|---|
| CYP21A2 | 6p21 | CAH due to 21-hydroxylase deficiency |
| CYP11B1 | 8q21 | CAH due to 11β-hydroxylase deficiency |
| CYP19A1 | 15q21.1 | Precocious puberty/gynecomastia |
| DLK1 | 14q32.2 | Paternally imprinted precocious puberty |
| GNAS1 | 20q13.2 | McCune-Albright syndrome |
| GRL | 5q31 | Inherited glucocorticoid resistance |
| HSD3B2 | 1p13.1 | CAH due to 3β-hydroxysteroid dehydrogenase deficiency |
| KISS1 | 1q32.1 | Precocious puberty |
| KISS1R | 19p13.3 | Precocious puberty associated with activating mutations |
| LHR | 2p21 | Familial male-limited precocious puberty |
| MKRN3 | 15q11.2 | Paternally imprinted precocious puberty |
| STK11/LKB1 | 19p13.3 | Peutz-Jeghers syndrome |
Although this disorder is labeled GnRH-dependent precocious puberty or puberty (central precocious puberty [CPP]), it represents precocious gonadarche due to either premature resurgence or incomplete suppression of the hypothalamic GnRH pulse generator. It occurs more often in girls than in boys. This sex difference is probably related to the lesser prepubertal suppression of the GnRH pulse generator in girls than in boys. The sequence of pubertal development is typical of a normal puberty including adrenarche in some cases, but it begins at an earlier-than-normal age. Observational data collected from 2008 to 2010 through a stringent Spanish hospital-based registry reported an annual incidence ranging from 0.02 to 1.07 cases per million.
Approximately 90% of cases of CPP in girls are considered to be idiopathic, whereas 50% to 70% of cases in boys are associated with identifiable etiology. The discovery of genes involved in GnRH and gonadotropin secretion has allowed for identification of specific gene mutations in patients with CPP. A heterozygous missense mutation, p.Arg386Pro, in the KISS1R gene was associated with precocious puberty in a girl. This mutation located in the C-terminal tail of the receptor was associated with delayed degradation of the receptor yielding a prolonged duration of action. In addition to this activating mutation in the receptor, a mutation in the KISS1 gene was identified in a young boy who presented at 17 months of age with CPP. This variant, p.Pro74Ser, appears to prolong the half-life due to resistance to degradation. To date, KISS1 and KISS1R mutations are extremely rare in children with CPP.
As described above, paternally inherited MKRN3 mutations are associated with CPP in boys and girls. This gene is mapped to chromosome 15q11.2 in the Prader-Willi Syndrome critical region. The maternal allele is methylated and silenced resulting in expression of the paternal allele. The gene encodes a protein involved in ubiquitination and cell signaling. Several unique mutations have been reported among different ethnic groups. Mutations in this gene appear to be the most common familial etiology of CPP. The median age of pubertal onset is 6.0 years (3.0–7.5) in girls and 8.25 years (5.9–9.0) in boys. No abnormalities of the CNS have been reported. Curiously, despite the physical proximity of MKRN3 to the Prader-Willi syndrome locus, no major signs of Prader-Willi syndrome have been described. A GWAS study looking at the parent of origin effects on puberty identified a signal in the region of MKRN3 gene; the paternal allele of a specific SNP (rs12148769, G>A) affects age at menarche in healthy girls suggesting that variants in this region affect pubertal timing within the normal range. , Circulating makorin-3 concentrations were found to decline prior to puberty among healthy girls and boys ( Figs. 18.10 and 18.11 ). , ,
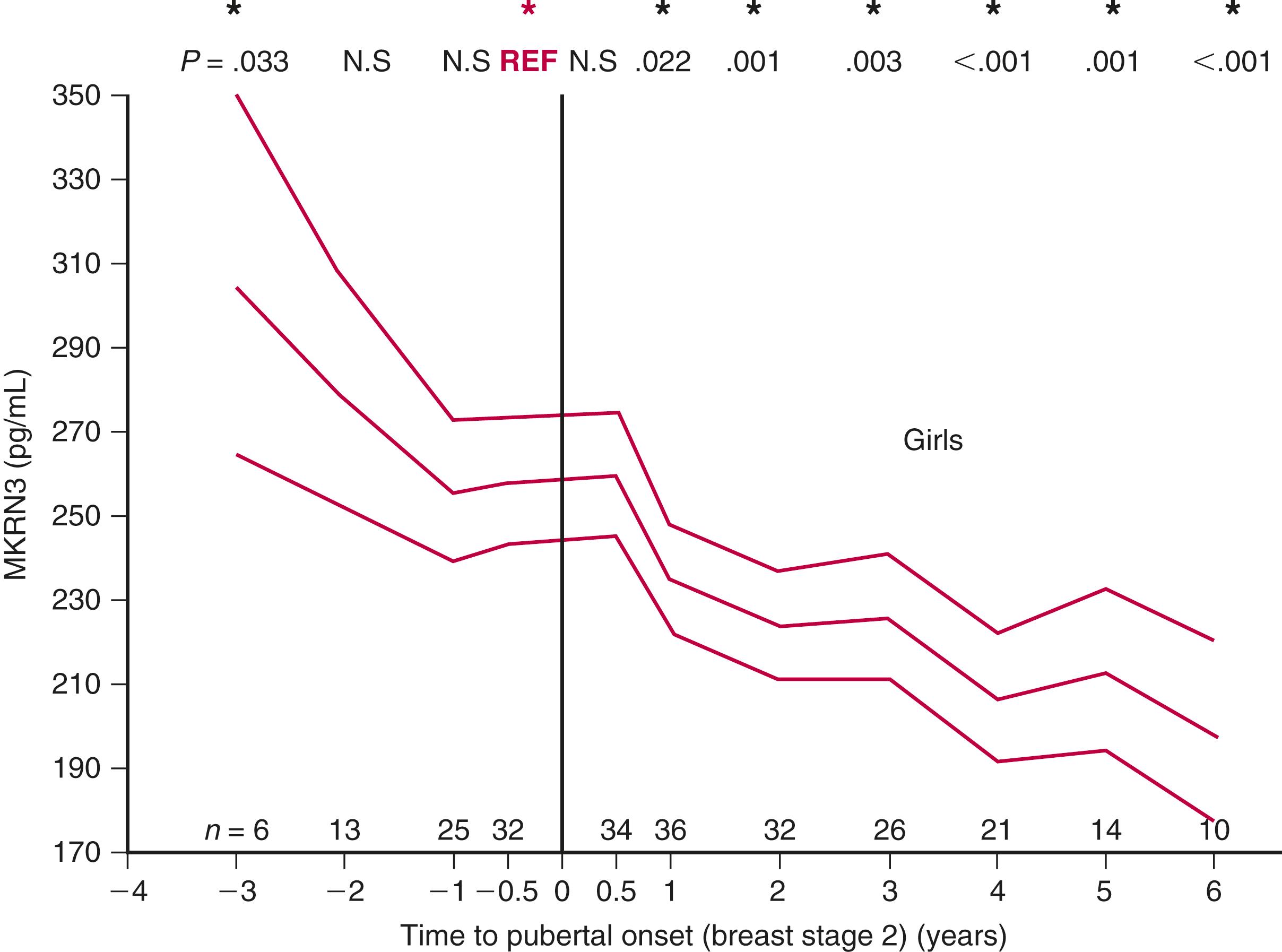
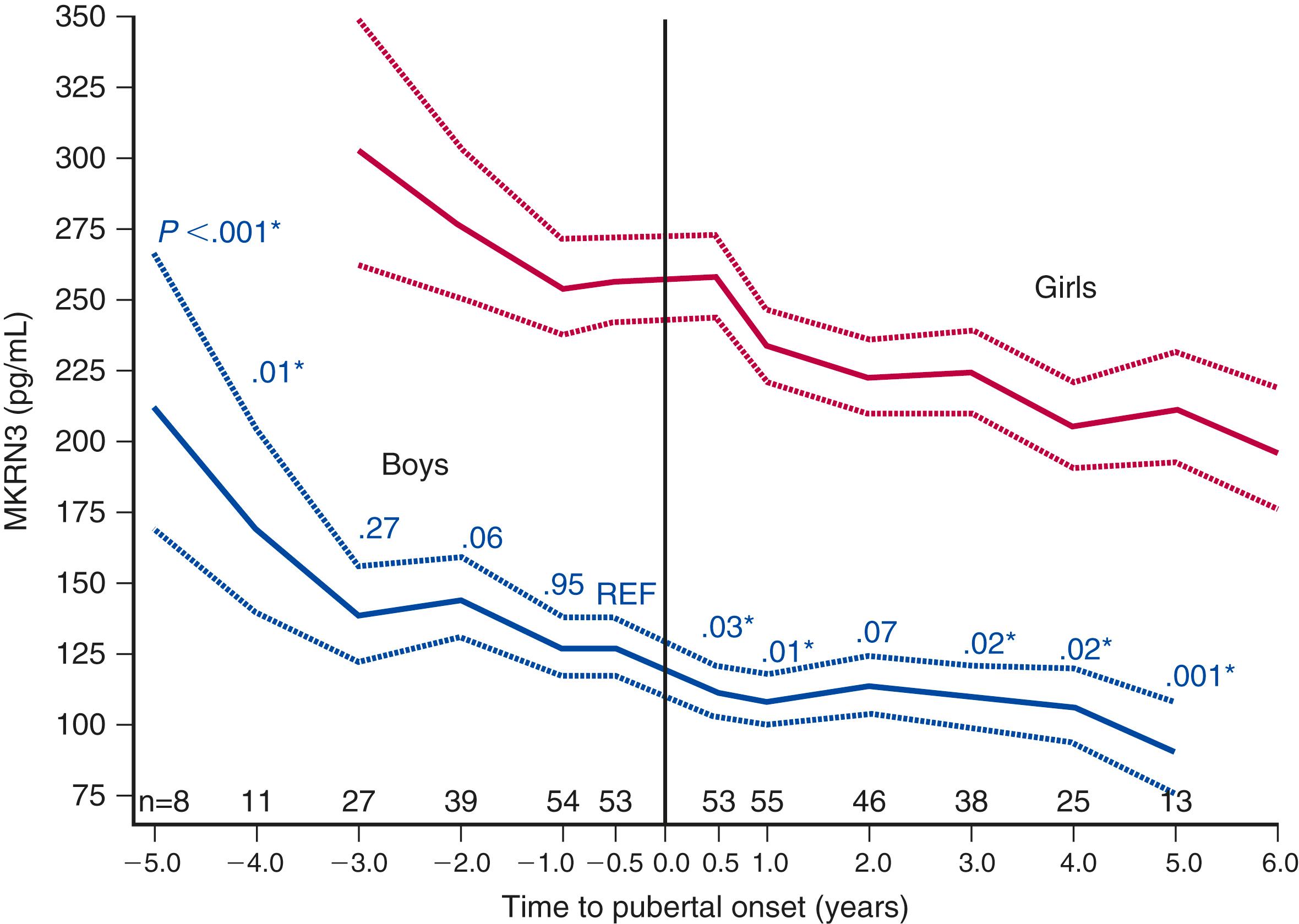
CPP has been described in the Williams-Beuren syndrome, histiocytosis X, and maternal uniparental disomy for chromosome 14. Evaluation of a family with CPP led to identification of a deletion/duplication mutation in the delta-like 1 homologue ( DLK1 ) gene located at chromosome 14q32. The four affected individuals presented with thelarche between the ages of 6.4 to 8.0 years of age. An additional investigation revealed undetectable serum DLK1 concentrations (<0.4 ng/mL) compared to control group (1.9–20 ng/mL). DLK1 is a paternally expressed gene located within the genetic locus associated with Temple and Kagami-Ogata syndromes. Temple syndrome is characterized by IUGR, hypotonia in infancy, CPP, and short stature. Genetic findings in Temple syndrome include maternal uniparental disomy, paternal deletion, or loss of differential methylation at the DLK1/MEG3 region on chromosome 14. Patients with Kagami-Ogata syndrome is associated with prenatal overgrowth, developmental delay, abdominal wall defects, but disorders of pubertal timing do not appear to be a prominent feature. Based on available animal and human data, the opposing directions of regulation of MKRN3 and DLK1 expression prior to puberty may underlie interactions between these factors that modulate the timing of puberty.
Both DLK1 and MKRN3 are maternally imprinted and paternally expressed genes. The role of DLK1 in pubertal timing has been implicated by a GWAS that showed SNPs near paternally inherited DLK1 were associated with a significantly earlier age of menarche. Temple Syndrome occurs with loss of DLK1 expression in conjunction with two other genes paternally inherited genes, RTL1 and DIO2. Polymorphisms in other candidate genes such as LIN28B and the paternally imprinted KCNK9 have also been associated with age of menarche and are candidates of monogenic causes of central precocious puberty. To date, there have been no described patients with central precocious puberty with this mutation. The relatively high frequency of imprinting mutations associated with monogenic causes of central precocious puberty suggests that imprinting may play an important role in the regulation of puberty and that pubertal onset may be associated with specific changes in the regulatory control of epigenetic modification.
Hypothalamic hamartomas, congenital malformations composed of a heterotropic gray matter, neurons, and glial cells usually located on the floor of the third ventricle or attached to the tuber cinereum, are a common etiology of CPP. Two potential mechanisms have been hypothesized for the association between CPP and hypothalamic hamartomas; one is increased GnRH secretion from tissue emancipated from suppression by the prepubertal brake and the other is that factors such as TGF-α provide an ectopic drive to GnRH neurons with a normal distribution in the hypothalamus. The hamartomas can be classified as parahypothalamic , attached or suspended from the floor of the third ventricle, or as intrahypothalamic , in which the mass is enveloped by the hypothalamus and distorts the third ventricle. The lesions do not grow over time, do not metastasize, and do not produce β-human chorionic gonadotropin (β-hCG) and α-fetoprotein. Extreme precocity suggests a hamartoma. Although gelastic or laughing seizures can be associated with precocious puberty due to hypothalamic hamartomas, the majority of patients with hypothalamic hamartomas do not exhibit neurological symptoms. , Among patients in whom hamartomas were removed to treat intractable seizures, hamartomas associated with CPP were more likely to contact the infundibulum or tuber cinereum and were larger than hamartomas not associated with CPP. All hamartomas expressed GnRH, TGF-α, and GnRHR. No differences were detected in the expression of KISS1 and GPR54 between hamartomas associated with CPP and those not associated with CPP. Gene expression profiling of hypothalamic hamartomas associated with precocity may provide clues regarding genes, proteins, and regulatory pathways associated with the timing of puberty.
Hypothalamic hamartomas are generally sporadic but may occur as a feature of several dysmorphic syndrome including Pallister-Hall syndrome and oral-facial-digital syndrome (OFD) types I and VI. Pallister-Hall syndrome is an autosomal-dominant disorder associated with mutations in the GLI3 gene located at chromosome 7p13. Additional features include pituitary anomalies, hypopituitarism, imperforate anus, and polydactyly. In the presence of sonic hedgehog (SHH), the full-length GLI3 translocates to the nucleus, where it functions as a transcriptional activator. When SHH is absent, GLI3 is phosphorylated and cleaved; the cleaved protein translocates to the nucleus to repress specific target genes. The truncated GLI3 protein showed stronger repressor activity than the wild type of protein, suggesting that these mutations are associated with constitutive repression of SHH signaling. Mutations in additional genes in the SHH pathway, especially PRKACA, have been detected in HH associated with epilepsy.
In addition to hypothalamic hamartomas, optic gliomas, suprasellar cysts, arachnoid cysts, previous head trauma, static cerebral encephalopathy, CNS infections, CNS radiation, hydrocephalus, meningomyelocele, and neurodevelopmental disabilities may also be associated with CPP. For hypothalamic-pituitary disorders, the endocrine symptoms of precocious puberty may precede neurological symptoms. Three predictors associated with CNS lesions in girls are: (1) age younger than 6 years; (2) absence of pubic hair; and (3) estradiol concentrations greater than 110 pmol/L. The type of CNS lesion influences the presentation of GnRH-dependent precocious puberty presumably due to differences in the mechanisms inducing puberty and to the hypothalamic-pituitary deficiencies associated with the initial lesion or its treatment.
Optic gliomas are associated with neurofibromatosis type 1 (NF1), an autosomal-dominant disorder diagnosed based on clinical features that include size and number of café-au-lait spots, macrocephaly, and family history of the disorder. The mechanism of action has been presumed to be a mass effect. Pineal tumors can be associated with precocious puberty due to tumor-induced hydrocephalus or germ cell tumors. CNS radiation, dose range 18 to 50 Gray, used to treat intracranial tumors or used prophylactically for malignancies, can induce precocious gonadarche, perhaps as a result of an astroglial response with increased TGFα production. In the situation of CPP, simultaneous GH deficiency (GHD) may be masked by the accelerated growth velocity associated with increased gonadal sex steroid secretion.
Other situations associated with progressive CPP include children with virilizing disorders (such as CAH) and familial male-limited precocious puberty (testotoxicosis) in whom skeletal maturation is usually markedly advanced. In these situations, the precocious gonadarche is considered to be secondary to the virilizing disorder, but the mechanism through which the GnRH pulse generator is prematurely activated is unclear.
The treatment of choice for children with progressive precocious gonadarche is a GnRH receptor agonist (GnRH-Ra). In situations of coexisting GHD, combined treatment with recombinant human GH may be helpful to preserve height potential. GnRH-Ras are modifications of the native GnRH decapeptide, have greater resistance to degradation, and possess an increased affinity for the GnRH-R. Therefore, they are perceived by the pituitary as a continuous GnRH stimulation, which induces downregulation of GnRH-R function and leads to decreased gonadotropin secretion. GnRH-Ras are available as daily injections or depot forms that are currently administered every 28 days. The depot formulation of leuprolide acetate is commonly used in the United States; the recommended dose is 0.3 mg/kg, administered every 28 days. Another GnRH-Ra, histrelin, has been formulated as a hydrogel subdermal implant that is surgically inserted into the inner aspect of the upper arm. Over the course of 1 year, histrelin diffuses from the 50 mg implant. Studies indicate excellent gonadotropin suppression. Additional depot preparations available in the United States, Canada, the UK, and Europe include triptorelin, goserelin, and buserelin.
When the progressive nature of the disorder is equivocal, serial evaluations are necessary to detect a sustained acceleration in tempo of pubertal development, including skeletal maturation (and therefore loss of height potential), before initiating therapy. This strategy is necessary because children with nonprogressive precocious puberty do not benefit from treatment. The major goals of treatment are to prevent further pubertal progression until appropriate for chronological age and to achieve improved adult height.
Clinically, the cessation of pubertal progression is apparent within 3 months of initiating treatment. The signs related to adrenarche neither regress nor are they prevented; they may even progress. In girls, breast size typically decreases but may not completely regress. Vaginal bleeding secondary to estrogen withdrawal, and acne may occur during the first month of treatment. Subsequently, there should be no further vaginal bleeding even if menarche occurred prior to initiation of therapy. The ovaries and uterus decrease in size. In boys, testicular volume decreases. Linear growth velocity and the rate of bone mineral accretion decrease. Longitudinal evaluation of a cohort of children with precocious gonadarche showed increased lumbar bone mineral density and BMI SD scores at the initiation of therapy. After cessation of therapy in children treated with GnRH-Ra, bone mass, bone turnover, and percent body fat were normal, suggesting that peak bone mass would be appropriate. ,
Adequacy of treatment is judged by prepubertal estradiol/testosterone concentrations and prepubertal gonadotropin response to GnRH stimulation. Monitoring to confirm efficacy of therapy includes interim history, physical examination to ascertain height, weight, and stage of pubertal development, and bone age x-rays. In addition, GnRH stimulation tests should be repeated at regular intervals (3–12 months) to document gonadotropin suppression. This monitoring is necessary because bone maturation may progress despite regression of the clinical features associated with gonadarche. Urinary gonadotropin determinations do not provide adequate sensitivity to judge the efficacy of therapy.
The duration of GnRH-Ra therapy should be individualized, with the decision to discontinue therapy based on chronological age, skeletal maturation, projected adult height, and psychosocial readiness for the resumption of puberty. For girls, menstruation usually occurs 9 to 15 months after discontinuation of GnRH-Ra therapy, with earlier onset in those who had experienced menarche prior to treatment. Studies indicate that final height is improved over the initial predicted height at diagnosis but is still less than that based on midparental height. Rapidly progressive pubertal development, advanced skeletal maturation, predicted compromise of adult height, and psychosocial considerations justify treatment. ,
Among the adverse effects of treatment, parents may note increased emotional lability and moodiness just prior to the GnRH-Ra injection. Local or systemic allergic reactions or sterile abscesses may occur but are uncommon. Some children show increased weight gain disproportionate to their linear growth. Intermittent therapy, often due to poor compliance, may have the deleterious effect of increasing gonadotropin and gonadal steroid secretion, leading to progressive skeletal maturation and further compromise of adult height. Since affected children appear older than their chronological age, parents and teachers may have inappropriate expectations regarding psychosocial development and abilities. In a small study, girls treated with GnRHa (median age 10.4 years) showed comparable cognitive performance, behavior, and psychosocial characteristics compared to age-matched control girls with the exception of higher emotional reactivity on one of the two emotional reactivity tasks. However, cognitive and emotional development is normal for chronological age. Thus the guilelessness and naiveté of such children expose them to an increased risk of sexual abuse, with affected girls at risk of becoming pregnant.
The long-term overall positive experience has now accrued for GnRH-Ras. Pregnancies with normal offspring have been observed. Although no major adverse effects on reproductive function have been noted, some girls appear to have an increased risk to develop PCOS. However, similar findings in a cohort containing both treated and untreated girls suggest the possibility of an underlying predisposition to PCOS preceded CPP.
Some children experience a nonprogressive (or slowly progressing) form of precocious gonadarche attributed to premature but intermittent or transient activation of the hypothalamic GnRH pulse generator. Among this latter group of children, basal gonadotropin concentrations and gonadotropin responses to GnRH stimulation may be normal for chronological age but can overlap values observed among children with progressive precocious gonadarche. Because the physical signs of pubertal development do not always correlate with GnRH-stimulated gonadotropin responses, physical findings alone cannot differentiate between progressive and nonprogressive precocious gonadarche. In general, children with this nonprogressive form of precocious gonadarche show no evidence of pubertal responsiveness to GnRH stimulation, no loss of height potential, and do not usually benefit from GnRH-Ra therapy. ,
Precocious pubertal development may occur independently of pulsatile GnRH secretion. In these situations of peripheral precocious puberty, inappropriate gonadal or adrenal steroid secretion or exposure to exogenous steroids induces the physical signs of puberty. In most instances, pubertal development is incomplete and fertility is not attained.
The classical clinical triad of McCune-Albright syndrome (MAS) is precocious pubertal development, café-au-lait spots, and bony fibrous dysplasia. The café-au-lait lesions are generally large, do not cross the midline, and have irregular “coast of Maine” margins, but exceptions to the classic features can occur. Precocious pubertal development is not observed in all cases and appears to be more common among girls than boys. This disorder is due to constitutive activation of the G s α protein coupled to membrane-bound glycoprotein hormone receptors and is associated with autonomous function of the endocrine glands. The syndrome is due to postzygotic somatic cell mutations in the GNAS1 gene; missense mutations Arg201His and Arg201Cys are among the most common. Partial or atypical forms are increasingly recognized; among 113 children with 1 to 3 features typical of MAS, a PCR-based mutation analysis protocol identified a missense mutation at codon 201 in 90% when an affected tissue (e.g., ovarian, bone, or adrenal tissue) was analyzed. These gain-of-function mutations in the G s α gene lead to constitutive activation of gonadotropin receptors, and subsequent increased autonomous ovarian estrogen and testicular testosterone secretion in affected girls and boys, respectively.
Vaginal bleeding, with or without breast development, is the typical presentation in prepubertal girls. The vaginal bleeding represents estrogen withdrawal following the resolution of an estrogen-secreting cyst. Estrogen concentrations may be elevated and pelvic ultrasound may confirm a unilateral ovarian cyst. The frequency of recurrent episodes of ovarian cysts and vaginal bleeding is variable. Because the precocious pubertal development is GnRH-independent, GnRH-Ra treatments are ineffective. Treatment with aromatase inhibitors, estrogen receptor modulators, and medroxyprogesterone acetate has been tried. , Aromatase inhibitors have been tried; anastrozole had limited efficacy, whereas letrozole showed sustained beneficial effects on skeletal maturation, growth velocity, and predicted adult height. A partial estrogen antagonist, tamoxifen, has been helpful. Preliminary results using fulvestrant, a pure estrogen blocker, have been promising to decrease vaginal bleeding and slow bone maturation. , One outcome study showed variable gonadal function in affected adult women, with some having regular menses and fertility while others had persistent autonomous gonadal function associated with irregular menses and infertility.
Among boys with MAS, precocious puberty is less common. In a cohort of 54 males, 11 (21%) presented with precocious puberty. Gonadal involvement occurred in approximately 80%. Testicular embryonal carcinoma is rare. Macroorchidism may be noted on examination. Ultrasound features can include macroorchidism, hyperechoic lesions, hypoechoic lesions, and testicular microlithiasis. Leydig cell hyperplasia can occur in postpubertal men but is rare in boys. AR blockers, aromatase inhibitors, and blockers of sex steroid synthesis have been used to treat precocious puberty. A conservative approach to MAS-associated testicular lesions has been endorsed with emphasis on serial imaging and testicular preservation. Combined treatment with bicalutamide and anastrozole in one boy was well tolerated and associated with normalization of growth velocity and prevention of pubertal progression.
The typical bone lesion is polyostotic fibrous dysplasia. Lesions tend to be asymmetric and can affect any bone, including the skull. Skeletal disease is generally apparent by 10 years of age. The cystic bone lesions can lead to pathological fractures and deformities. Pseudoarthrosis can occur. With involvement of the skull, hyperproliferation of the preosteoblastic cells results in impingement into cranial foramina, leading to compression of cranial nerves. Blindness, deafness, facial asymmetry, or ptosis can result. Rather than being associated with a single abnormality of bone, the specific histopathology and clinical consequences vary depending on anatomical location: axial or appendicular skeleton, cranial bones, or gnathic bones. Total body bone scintigraphy can be performed to identify and determine the extent of bone lesions before they are visible on x-rays. Hypermetabolic bone disease is indicated by increased serum osteocalcin and alkaline phosphatase concentrations, as well as increased urinary hydroxyproline concentrations. Studies suggest that bisphosphonate therapy may benefit the bone disease with reduced fracture rate, decreased bone pain, and radiological evidence of long bone healing. ,
Hypophosphatemic rickets can occur secondary to increased production of the phosphaturic hormone, FGF23, by the bone lesions. There is a positive relationship between disease activity and FGF23 concentrations. Renal tubulopathy with some phosphate wasting may be evident. However, significant hypophosphatemia is uncommon. Treatment of hypophosphatemia includes phosphate and vitamin D. Other endocrine manifestations include nodular thyroid hyperplasia with hyperthyroidism, multiple pituitary adenomas associated with gigantism, acromegaly or hyperprolactinemia, and parathyroid adenoma or hyperplasia with hyperparathyroidism. Cushing syndrome is extremely rare and almost always occurs during infancy.
Estrogen-secreting tumors are a rare cause of premature or abnormal pubertal development. Types of tumors include granulosa cell, gonadal stromal cell, ovarian sex cord stromal, and theca cell tumors. , The majority of juvenile granulosa cell tumors are large, can be palpated on bimanual examination, and are limited to the ovary at the time of diagnosis. Rapid pubertal progression is common. Estrogen concentrations may be very elevated, gonadotropin concentrations are suppressed, and circulating tumor markers such as α-fetoprotein, inhibin, CA-125, or hCG may be detected. Somatic mutations involving AKT1 have been identified as a major event in the pathogenesis of juvenile granulosa cell tumors; 60% (i.e., 10/16) of the ovarian samples carried in-frame duplications that activated AKT1. The primary treatment is surgical excision with staging that includes peritoneal cytolog.
Rarely, gonadoblastomas in streak gonads, lipoid tumors, cystadenomas, and ovarian carcinomas can secrete estrogens, androgens, or both. Elevated serum inhibin and AMH concentrations and the finding of AMH immunoreactivity in the tumor indicate that the tumor cells are of granulosa or Sertoli cell origin. Expression of FOXL2 , SOX9 , and other specific genes can be helpful to discriminate between gonadoblastomas and intratubular germ cell neoplasias; FOXL2 expression suggests that a gonadoblastoma has a granulosa cell component. If originally positive, these markers may be useful to recognize recurrence.
Sex cord tumors with annular tubules are common in patients with Peutz-Jeghers syndrome (PJS), an autosomal-dominant disorder characterized by mucocutaneous pigmentation and gastrointestinal polyposis. PJS is associated with mutations in the STK11/LKB1 gene on chromosome 19p13.3, which encodes a serine-threonine kinase. The endocrine tumors, which may be multifocal and bilateral, can differentiate into granulosa cells or large cell-calcifying Sertoli tumors with the potential to secrete estrogen. Thus affected females may present with precocious puberty and affected males may present with gynecomastia. Although usually benign, granulosa or Sertoli cell tumors can undergo malignant changes. Affected individuals have an increased risk for colon, stomach, small intestinal, breast, and pancreatic cancers. Aromatase inhibitors were associated with decreased gynecomastia in a small series of boys with large cell calcifying Sertoli cell tumors.
Sertoli cell tumors have been found in association with both PJS and the Carney Complex (CNC). CNC is often associated with mutations in the protein kinase, c-AMP dependent, regulatory, type 1, alpha ( PRKAR1A ) gene, which encodes the regulatory subunit type 1 of protein kinase A. , Among boys, adrenal, testicular, or hepatocellular tumors can express aromatase, leading to secretion of estradiol and estrone. In instances where gynecomastia is excessive, prolonged, and apparent at a time other than midpuberty (around stage 3), further evaluation may be warranted. The evaluation should include testosterone, estradiol, hCG, LH, FSH, thyroid-stimulating hormone (TSH), and DHEAS measurements. In addition to tumors, the differential diagnosis of gynecomastia includes Klinefelter syndrome, impaired testosterone biosynthesis, androgen insensitivity, and hyperprolactinemia.
In one family, autosomal-dominant familial gynecomastia was due to aberrant transcription of the aromatase gene; the affected girl developed GnRH-independent precocious pubertal development. Autosomal-dominant aromatase excess syndrome is characterized by high systemic estrogen levels, short stature, prepubertal gynecomastia, testicular failure in males, and premature breast development, macromastia, and uterine pathology in females. Small chromosomal arrangements in the promoter region of the aromatase gene appear to be associated with increased promoter activity, resulting in increased aromatase activity. Novel cryptic deletions and duplications were identified; phenotypic severity in this gain of function disorder is primarily determined by the tissue expression pattern of CYP19A.
Isolated premature menarche refers to vaginal bleeding at an inappropriately early age in the absence of other signs of puberty. The duration of bleeding is usually limited to a few days. The most common endocrine etiology is a spontaneous resolution of an estrogen-secreting ovarian cyst. Often, the ultrasound shows no abnormality because the cyst has resolved by the time the study is obtained. Most instances are self-limited, remit spontaneously, and are associated with normal pubertal development. Typically, isolated menarche due to an estrogen-secreting cyst is a sporadic event that usually occurs once. However, such cysts and episodes of vaginal bleeding may recur. The bloody vaginal discharge noted in female infants during the first week of life is a physiological event secondary to estrogen withdrawal.
Tumors or trauma usually do not cause cyclic bleeding. The other major differential diagnoses of isolated vaginal bleeding include sexual abuse, vaginal foreign bodies, vaginal infection, MAS, and primary hypothyroidism. Neoplasms such as rhabdomyosarcoma and sclerosing stromal tumors can present with isolated vaginal bleeding.
Premature thelarche is defined as isolated breast development without other signs of pubertal maturation. Typically, the parents or pediatrician note breast development, either unilateral or bilateral, between 9 and 18 months of age. No significant nipple development or pigmentation occurs, and the vaginal mucosa remains pink and shiny.
Breast ultrasound can help distinguish breast tissue from cysts, fibroadenomas, neurofibromas, or other less common lesions but is usually not needed. Pelvic ultrasound may show a bilateral increase in the number of ovarian follicular cysts. Using a recombinant cell bioassay with increased sensitivity, estradiol concentrations are higher among girls with premature thelarche than in healthy controls. However, estradiol concentrations in such patients are still low and remain below the assay detection limit for most radioimmunoassays. FSH concentrations may be increased for chronological age, but LH concentrations and LH responses to GnRH stimulation are prepubertal. No acceleration in linear growth velocity or skeletal maturation occurs, and the breast development usually regresses spontaneously over time. Although premature thelarche prior to 2 years of age is often transient, closer observation is indicated if growth velocity is accelerated or if the basal LH concentration is greater than 0.3 IU/L. In most instances, onset of puberty, adult height, and adult reproductive function are normal. , Premature thelarche can usually be considered a normal variant. Nevertheless, longitudinal evaluation is helpful to ensure the nonprogressive nature of this disorder.
Premature thelarche has been described in Rubinstein-Taybi syndrome (RTS), an autosomal-dominant disorder characterized by short stature, psychosocial retardation, a characteristic that fades, broad thumbs and halluces, and increased risk for neoplasia. RTS has been associated with mutations in CREB binding protein ( CREBBP ) and E1A binding protein p300 (EP300) genes. Premature thelarche and precious puberty have been reported in girls with the Kabuki make-up syndrome, which is characterized by a peculiar facies with eyes reminiscent of Kabuki actors, mental retardation, and decreased growth velocity. Kabuki syndrome has been associated with mutations in the KMT2D and KDM6A genes.
Girls with primary hypothyroidism can, on rare occasions, present with breast development or isolated vaginal bleeding. This constellation of clinical features was first described by Van Wyk and Grumbach in 1960. On ultrasound, enlarged multicystic ovaries may be noted. Additional features may include delayed bone age, ascites, and pleural and pericardial effusions. This is the only etiology for precocious puberty associated with delayed bone age. Thyroid hormone replacement therapy is associated with regression and resolution of the cysts; surgical treatment is not indicated. The mechanism underlying the ovarian stimulation is unclear. One possibility is that the excessively elevated TSH concentrations cross-react with the FSHR to promote estrogen secretion. Another explanation is that increased FSH secretion observed in the hypothyroid state is responsible.
Hypothyroid boys may show increased testicular volume (macroorchidism) for age. Interestingly, after thyroxine replacement to a cohort of such boys and the subsequent attainment of stage 5, testicular volume was found to be considerably greater than that in controls. The macroorchidism associated with prepubertal hypothyroidism is probably the result of an expanded population of undifferentiated Sertoli cells resulting from increased FSH signaling in response to elevated concentrations of either FSH or TSH.
It should be noted that at the hypothalamic-pituitary level, hypothyroidism leads to a delay in the pubertal resurgence of LH secretion, which presumably accounts for the delayed puberty that is generally associated with chronic hypothyroidism (discussed later in the chapter).
Exposure to exogenous estrogenic steroids or estrogen receptor agonists can induce pubertal development. Information regarding possible environmental exposures from tea tree oil, lavender oil, or personal care products should be elicited because use of these substances has been associated with thelarche and gynecomastia. Potential sources of estrogenic steroids include oral contraceptives, creams, shampoos, and various lotions. In addition, phytoestrogens found in a variety of foods, and phthalate esters present in plastics are environmental endocrine disruptors with estrogen agonist activity. Epidemics of premature breast development reported in Puerto Rico and Italy have been attributed to increased exposure to estrogenic steroids, phthalates, phytoestrogens, or estrogenic mycotoxins. Several case reports have described precocious puberty and virilization in children accidentally exposed to the testosterone gels.
Mycotoxins are naturally occurring substances that can be found as environmental contaminants in cereals, corn, and nuts. Certain drugs, such as marijuana, isoniazid, spironolactone, ketoconazole, and cimetidine, can induce gynecomastia by a variety of mechanisms. It has been suggested that prenatal exposure to endocrine disruptors may influence fetal programming of the endocrine system and, therefore, may influence the timing and tempo of puberty. Potential mechanisms of action include binding to nuclear hormone receptors, influencing cofactor recruitment, altering steroidogenesis, and epigenetic modifications.
Premature pubarche is defined as the development of pubic hair, axillary hair, and apocrine body odor prior to 8 years of age in girls and age 9.5 years of age in boys. The differential diagnosis includes premature adrenarche, CAH, and androgen-secreting tumors.
While the age of adrenarche varies considerably among ethnic groups, it is generally considered premature when it occurs prior to age 8 in girls and age 9.5 in boys. As in normal adrenarche, pubic hair, axillary hair, adult type apocrine odor, and acne may develop, whereas skeletal maturation may be appropriate for chronological age or slightly advanced. Clitoromegaly and marked phallic enlargement are unusual findings in patients with premature adrenarche. Gonadarche usually occurs at an appropriate chronological age and subsequent pubertal development proceeds normally. More girls tend to be referred for evaluation than boys. Despite BMI comparable with controls, one recent study reported that girls with premature pubarche appear to have increased total and central fat mass. In one study of Spanish and Italian girls, mean age at menarche for girls with documented premature pubarche was 6 months earlier than healthy controls.
Typically androgen concentrations are elevated for chronological age but within normal limits for the stage of pubic hair development. However, a subset of girls with premature adrenarche show persistent hyperandrogenism upon gonadarche. , Insulin resistance, hyperinsulinemia, and dyslipidemia have been described in these girls. These girls may develop chronic anovulation, hirsutism, insulin resistance, hyperinsulinemia, severe acne, and an increased LH:FSH ratio. These features are suggestive of incipient PCOS. Subsequently, adolescent girls with PCOS have an increased risk to develop impaired glucose tolerance and type 2 diabetes mellitus. In some populations, the frequency of heterozygosity for mutations in the 21-hydroxylase ( CYP21A2 ) gene is higher among children with premature pubarche and adolescent girls with incipient PCOS.
Premature pubarche due to premature adrenarche is a diagnosis of exclusion. The majority of children with premature adrenarche require no pharmacological intervention. However, at this time, the ability to predict outcome and risk for PCOS is imperfect. Because lifestyle interventions involving food choices and exercise programs decrease the progression from impaired glucose tolerance to diabetes mellitus among adults, it seems prudent to counsel children with premature pubarche to adopt healthy lifestyles.
Rett syndrome is a rare neurodevelopmental disorder, which is characterized by loss of hand skills, loss of acquired spoken language, gait abnormalities, and stereotypic hand movements. This disorder is associated with heterozygous mutations in the Methyl-CpG-binding protein 2 (MECP2) gene, which is mapped to the X chromosome. Affected girls enter puberty early but they experience delayed menarche. Based on data generated through the multicenter RTT Natural History study, median duration of puberty, from thelarche to menarche, was reported to be 3.9 years.
The virilizing CAHs are a group of autosomal-recessive disorders in which cortisol synthesis is impaired due to decreased 21-hydroxylase, HSD3B2, or 11β-hydroxylase activity (see also Chapter 4 ). The specific pattern of circulating steroid hormone concentrations reflects which steroid enzyme is involved. Approximately 90% to 95% of cases are due to 21-hydroxylase deficiency, which is due to mutations in the CYP21A2 gene. Mutations in HSD3B2 and 11β-hydroxylase ( CYP11B1 ) genes account for the remaining 5% to 10% of patients with virilizing CAH.
In all cases, decreased cortisol concentrations lead to loss of negative feedback inhibition, increased ACTH secretion, and increased adrenal androgen biosynthesis. The clinical spectrum for these disorders ranges from complete loss-of-function mutations, which typically present during infancy with genital ambiguity, to partial loss-of-function mutations, which may present in childhood, adolescence, or adulthood. Here, discussion is limited to nonclassic CAH (NCAH), where the major symptoms are secondary to hyperandrogenism rather than to adrenal cortical insufficiency, as in the classical forms of this disease.
During childhood, boys or girls with NCAH can present with premature pubic hair, adult-type apocrine odor, increased growth velocity, and tall stature. In contrast to premature adrenarche, clitoromegaly or phallic enlargement and advanced skeletal maturation are more common. In female patients, the symptoms of NCAH are similar to those of PCOS. Males with NCAH are often not identified unless sisters with NCAH have been diagnosed.
The reported incidence of NCAH is approximately 1 in 1000 patients, whereas the reported incidence of the more severe forms is approximately 1 in 14,000. In 21-hydroxylase deficiency, decreased 21-hydroxylase activity leads to increased concentrations of 17-OHP, 17-OH-hydroxypregnenolone, DHEA, androstenedione, and testosterone. The gene coding for 21-hydroxylase, CYP21A2 , maps to chromosome 6p21. A highly homologous nonfunctional pseudogene, CYP21A1P , is located in close proximity to the functional gene. The majority of mutations associated with 21-hydroxylase deficiency represent gene conversion events in which the functional gene has acquired deleterious nucleotide sequences from the pseudogene. Over 200 mutations have been reported to date, but approximately 10 mutations account for the majority of affected alleles. Most affected individuals are compound heterozygotes and carry different CYP21A2 mutations on each allele. For individuals with nonclassical CAH, the missense mutation, V281L, accounts for at least one of the CYP21A2 alleles for most patients with nonclassical CAH. Phenotype–genotype correlations are fairly consistent, with the phenotype usually representing the least severe mutation.
For children with symptoms suggestive of NCAH, early morning basal 17-OHP values have been suggested as an effective screening test. Armengaud et al reported 100% sensitivity and 99% specificity with a threshold value of 200 ng/dL (6 nmol/L) to diagnose NCAH in children with premature pubarche. Defective 21-hydroxylase activity also promotes accumulation of other steroid hormone intermediates, such as 21-deoxycortisol and 16α-hydroxyprogesterone, 11-ketoandrostenedione, and 11-ketotestosterone. Although random 17-OHP concentrations can be diagnostic (especially in the more severe forms), ACTH-stimulation tests may be necessary to confirm the diagnosis of milder forms. Despite the increased availability of CYP21A2 genotype analyses, the complexity of this locus precludes routine use of molecular diagnostics. Techniques that genotype each allele or inclusion of parental DNA samples to segregate the alleles are helpful because a single CYP21A2 allele can carry multiple mutations. Two mutations on the same allele can act synergistically to impair enzyme activity to a greater extent than would be anticipated for each mutation individually.
In this form of virilizing CAH, decreased activity of the adrenal and gonadal specific form of HSD3B2 leads to increased concentrations of 17α-hydroxypregnenolone and DHEA. This disorder is due to mutations in the gene coding for HSD3B2 . Patients with classical HSD3B2 deficiency have been found to have mutations in the HSD3B2 gene. However, nonclassical CAH due to HSD3B2 mutations is extremely rare. Consequently, the correlation of molecular genotype data with hormonal responses has led to the adoption of more stringent criteria for the diagnosis of 3 β -HSD deficiency. In general, ACTH-stimulated 17-OH-hydroxypregnenolone and DHEA responses are elevated.
The clinical features associated with 11β-hydroxylase deficiency are similar to 21-hydroxylase deficiency. Patients with this type of virilizing adrenal hyperplasia may develop hypertension attributed to increased deoxycorticosterone secretion. This disorder is due to mutations in the CYP11B1 gene located on chromosome 8. Nonclassical CAH due to CYP11B1 mutations is extremely rare. The incidence of 11β-hydroxylase deficiency has been estimated to be 1:100,000 among Caucasians, but the incidence among Israeli Jews of Moroccan origin is reported to be higher. Elevated basal and ACTH-stimulated 11-deoxycortisol concentrations are typically found in this form of CAH. Serum 17-OHP, androstenedione, and testosterone concentrations may be mildly elevated. Plasma renin activity concentrations are low or suppressed.
This autosomal-recessive (AR) disorder of steroidogenesis is characterized by a steroid profile suggesting combined 17α-hydroxylase and 21-hydroxylase deficiencies. The more severe phenotype is characterized by ambiguous genitalia, adrenal insufficiency, and skeletal anomalies and is known as the Antley-Bixler syndrome. The skeletal abnormalities can include craniosynostosis, midface hypoplasia, choanal atresia, low-set ears, pear-shaped nose, arachnodactyly, clinodactyly, and radiohumeral synostosis. This disorder is due to mutations in the POR gene located at chromosome 7q11.2. , The protein encoded by the POR gene functions as an electron donor to cytochrome P450 enzymes. POR loss-of-function mutations influence the activities of 21-hydroxylase, 17α-hydroxylase, and aromatase enzymes.
Prenatal virilization of female fetuses occurs, but there is minimal postnatal virilization. Maternal virilization during pregnancy may occur and has been attributed to aromatase deficiency. Curiously, some patients, both male and female, present with delayed puberty; and ovarian cysts can occur in adolescent females. One adolescent girl presented with breast development, primary amenorrhea, bony anomalies, and cystic ovaries. Basal and ACTH-stimulated steroid profiles are variable because oxidoreductase deficiency affects multiple steroidogenic enzymes; serum 17-OHP concentrations tend to be elevated. Urinary analysis of steroid excretion using gas chromatography and mass spectroscopy (GC/MS) can provide hormonal confirmation of the diagnosis. Cortisol deficiency may occur; affected individuals may benefit from glucocorticoid replacement therapy. , Mutations in the FGFR2 gene are associated with similar skeletal anomalies but have normal steroidogenesis and normal external genitalia.
Cortisol is the active glucocorticoid secreted by the adrenal ZF. Concentrations of cortisol and its inactive metabolite, cortisone, are modulated by the enzymes, 11β-hydroxysteroid dehydrogenase type 1 (HSD11B1) and type 2 (HSD11B2). Apparent cortisone reductase deficiency (ACRD) is due to hexose-6-phosphate dehydrogenase (H6PD) deficiency and has been associated with premature adrenarche. This disorder is due to loss-of-function mutations in the H6PD gene, which interferes with HSD11B1 oxo-reductase activity and prevents local conversion of cortisone to cortisol. The net result is accelerated peripheral clearance of cortisol resulting in decreased negative feedback inhibition of the hypothalamic, pituitary, and adrenal axis, increased ACTH secretion, and increased ACTH-mediated adrenal androgen secretion. DHEAS, androstenedione, and testosterone concentrations are also increased. Urinary steroid metabolite profiling by combined gas chromatography and mass spectrometry may be helpful in the differential diagnosis of ACRD.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here