Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The diagnosis of infectious diseases ranges from straightforward clinical diagnoses to those that are possible only with the use of advanced molecular methods. Between these two extremes are the many infectious diseases for which an accurate and timely diagnosis requires the combined use of microbiologic cultures, histopathology/cytopathology, and molecular methods. It is this broad group of infectious diseases where the histopathologist and medical microbiologist, either alone or together, play a central role in diagnosis.
As with all clinical or pathologic evaluations, pathologists need all available clinical, radiographic, and laboratory findings in order to make accurate and timely diagnoses. This is particularly important in infectious disease pathology: many infectious diseases are restricted geographically, present with distinctive (or at least highly suggestive) clinical signs and symptoms, can present as localized or disseminated disease, may be associated with environmental or zoonotic exposure, and, because many are contagious, can present as part of an outbreak or be linked to transmission from another host. The immune status of the host also is important, as some infectious manifestations are seen primarily in the setting of immune compromise. Therefore, the amount of information necessary for the diagnosis and treatment of infectious diseases can be substantially greater than that needed for many noninfectious diseases. It is imperative that clinicians provide this information to pathologists, and that pathologists make an active attempt to obtain it when it is not initially received.
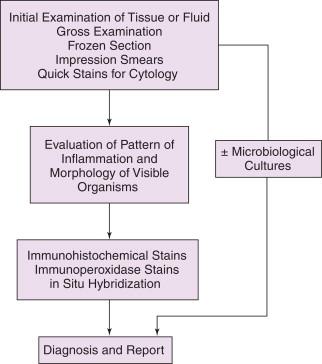
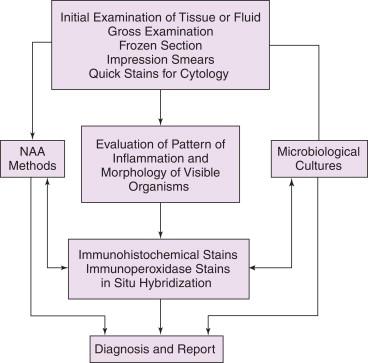
The histopathologic and cytopathologic diagnoses of infectious diseases are progressive and sequential processes that move from the general to the specific based on results from a combination of diagnostic methods. The traditional approach is shown graphically in Figure 1-1 , where the starting point is gross examination of specimens, frozen sections, aspirate smears, or hematoxylin and eosin (H&E)–stained permanent sections. In this approach, the evaluation is limited to the sequential use of histologic methods with or without the accompanying use of microbiologic cultures. This is a cost-effective and adequate approach for many common infectious diseases. For more challenging cases, an integrated approach as shown in Figure 1-2 is more appropriate. In such an approach, several methods are used simultaneously and in parallel to obtain the most accurate and timely diagnosis, including correlating histopathologic findings with the results of clinical signs and symptoms, other laboratory test results, radiographic findings, and, when appropriate, consultation with infectious disease pathology specialists. Ideally, the result of this process is an integrated, composite report that incorporates all of these findings into a summary and interpretation.
The inflammatory response to pathogenic microorganisms is sufficiently consistent and predictable to usually allow pathologists to identify that an infection is present. The type of inflammatory response—as well as its distribution in tissues, fluids, and organs—also may allow the pathologist to categorize the infection as one likely caused by bacteria, fungi, mycobacteria, or parasites ( Table 1-1 ). Some histopathologic clues, when combined with other findings in tissue, help narrow the differential diagnosis. One example is the Splendor-Hoeppli phenomenon, which is a radial aggregate of eosinophilic material surrounding a nidus of infection. The protein was once believed to be composed of aggregates of antigen-antibody complexes, but it is now known to be composed of major basic protein. When it is identified in tissue sections it suggests that an infection is caused by Sporothrix, some other fungi, Schistosoma eggs, and others. Although it is not a specific finding, it does help the pathologist narrow the differential diagnosis. Thus, although infections in tissues may show considerable overlap in their histopathologic characteristics, the type and distribution of inflammation—and often a combination of otherwise nonspecific findings—provides sufficient information for pathologists to order additional studies in a logical and sequential manner.
| Pattern of Inflammation | Likely Pathogens |
|---|---|
| Acute Inflammation | Bacterial Early stages of mycobacterial, fungal, and parasitic infections |
| Granulomatous inflammation | Mycobacterial, fungal, rare bacterial, parasitic |
| Endothelial damage, focal acute hemorrhage | Viral (e.g., viral hemorrhagic fevers) Rickettsial Pseudomonas aeruginosa |
| Focal necrosis | Viral Bacterial |
| Ulceration | Bacterial Mycobacterial Fungal Viral Parasitic |
Most common bacterial pathogens elicit the acute inflammatory immune response, a typical example being acute bronchopneumonia caused by Streptococcus pneumoniae . The acute inflammatory response consists primarily of an infiltrate of segmented neutrophils, although in more severe or prolonged cases the infiltrate can also contain neutrophil precursors. The surrounding tissue is often edematous, shows vascular congestion with margination of neutrophils along the vascular endothelium, and may show focal necrosis with severe inflammation. As the segmented neutrophils lyse, they release cellular debris that can mimic the appearance of extracellular bacterial cocci, but in general the cellular debris varies more in size and shape than do cocci. If a tissue Gram stain is performed, histopathologists should remember to look for both free bacteria and intracellular bacteria. The location of bacteria is determined in part by the nature of the infection (e.g., intracellular gram-positive cocci in pneumococcal pneumonia) and also by the degree of cellular lysis, severity of infection, and stage of infection.
A small number of pathogenic bacteria are associated with inflammatory responses other than typical acute inflammation. Rickettsia and other pathogens associated with endothelial infections typically do not cause acute inflammation but rather cause vascular leakage with edema of affected tissues. Vascular thromboses and vascular necrosis may also occur, depending on which rickettsial pathogen is the cause of the infection. Affected vessels may show a surrounding cuff of mononuclear inflammatory cells, but in general these bacterial infections are not associated with an infiltrate of neutrophils. Similarly, Pseudomonas aeruginosa pneumonia is characterized by damage to the walls of blood vessels with the subsequent development of acute hemorrhage with or without associated acute inflammation. Bacterial infections that are caused by toxin-producing bacteria, such as Clostridium species, may show only extensive tissue necrosis with minimal or no inflammation. Rapidly progressive infections caused by Streptococcus pyogenes in skin and subcutaneous soft tissues may progress so rapidly that extensive tissue necrosis occurs before acute inflammation can develop. One of the less intuitive inflammatory responses is that elicited by Brucella spp., Yersinia pestis, Francisella tularensis, Bartonella henselae (the causative agent of cat-scratch disease), and the Chlamydia trachomatis strains that cause lymphogranuloma venereum. Unlike most other bacterial pathogens, these bacteria are associated with the formation of stellate necrotizing granulomas in infected tissues.
Some bacterial infections are associated with patterns of infection that are distinctive but may not be recognized due to their relative infrequency in routine clinical practice. The foamy macrophages observed with Whipple disease in gastrointestinal biopsies is one example, as is the similar tissue reaction observed in the spleen with infections caused by Mycobacterium avium complex in patients with severe immunosuppression caused by HIV infection. Infections caused by Rhodococcus are typically associated with malakoplakia. The finding of xanthogranulomatous inflammation, while nonspecific, strongly suggests a chronic bacterial infection rather than an ongoing fungal or mycobacterial infection.
The histopathologic changes associated with bacterial infections are discussed in detail in 12, 13, 15, 16, 17, 18, 19 .
Fungal infections are typically associated with granulomatous inflammation, which may be necrotizing or non-necrotizing depending on the stage of the infection. Early stages of fungal infections are typically associated with acute inflammation, with the evolution of granulomatous inflammation occurring as cell-mediated immunity develops. As occurs with mycobacterial infections, the early stages of fungal infections are rarely seen: by the time the patient is symptomatic, or a biopsy is necessary, the infection almost always has progressed to the stage at which granulomatous inflammation has developed. One notable exception occurs with cutaneous fungal infections, where mixed acute and granulomatous inflammation occurs. In addition, many granulomas in cutaneous fungal infections have a necrotic center, but rather than be filled with necrotic debris the centers of the granulomas are filled with neutrophils. These granulomas do not have a specific name, being referred to as pyogenic granulomas or mixed inflammation, among other terms. When identified in skin and subcutaneous tissues, they are highly suggestive of a cutaneous fungal infection such as phaeohyphomycosis or chromoblastomycosis. As with mycobacteria and other causes of granulomas, necrosis of the center of the granulomas is more typical of infections that have persisted for some time. It is important for the diagnostician to remember that granulomatous inflammation varies considerably in its histopathologic appearance.
One distinctive tissue reaction to fungal infection occurs with the diverse group of fungi that cause what is variably termed zygomycosis, phycomycosis, or mucormycosis . Because the causative fungi all belong to the class Zygomycetes, perhaps the best term is zygomycosis . Infection with any of these agents, which are indistinguishable in tissue sections, results in invasion of arterial walls with subsequent vascular occlusion and thrombosis. Similar findings occur when members of the hyaline hyphomycetes (such as Aspergillus ) invade pulmonary arteries. Necrosis of infected tissues is the result of these fungal infections, with minimal acute inflammation in the early stages of infection.
More comprehensive descriptions of the histopathologic changes associated with fungal infections are presented in 23, 24, 25, 26 .
As with fungi, mycobacterial infections are classically associated with granulomatous inflammation. As with bacteria and fungi, however, mycobacteria elicit a spectrum of inflammatory reactions depending on the type of mycobacterium, the site of infection, and the stage of infection. Although the immediate tissue response to mycobacterial infection is that of acute inflammation, this is rarely (if ever) seen in diagnostic specimens: by the time clinical signs and symptoms develop, the inflammatory response has evolved to the stage of granulomatous inflammation. Early infections show non-necrotizing granulomas; if infections persist, the granulomas will begin to show necrosis of the central portions, which eventually become fully necrotic, eventually assuming the classic finding of caseation. At this stage of the infection, a rim of viable tissue, consisting mostly of epithelioid histiocytes, surrounds the central area of necrosis. From a practical standpoint, the most important feature for the histopathologist is that residual mycobacteria are limited to the interface between the necrotic and viable tissue; mycobacteria are almost never found in either the necrotic or the viable granulomatous tissue.
A number of unique patterns of inflammation with mycobacterial infections have also been described. Buruli ulcer, a tropical infection caused by Mycobacterium ulcerans, is manifested by the development of chronic skin ulcers that show acute and chronic inflammation at the leading edge of ulcers, with minimal granulomatous inflammation along the edge of ulcers. Tuberculoid leprosy shows a granulomatous inflammation, particularly in a perineural pattern, whereas lepromatous leprosy shows aggregates of lipid-laden macrophages that may be filled with Mycobacterium leprae bacilli. The absence of granulomas in lepromatous leprosy reflects a lack of an effective T-cell immune response. Mycobacterium avium complex infections in patients with profound immunosuppression also are characterized by foamy macrophages distended by innumerable bacilli, and again the absence of granulomas reflects a lack of an effective T-cell immune response. Mycobacterium marinum causes small subcutaneous granulomas in the skin (usually of the extremities) where the body temperature is sufficiently low to support the growth of the bacterium.
The histopathologic changes associated with mycobacterial infections are discussed in detail in 20, 21, 22 .
Excluding viral cytopathic effects, most viral infections are not associated with characteristic inflammatory or other tissue reactions in tissues or organs, although the distribution of changes within tissue or organs may suggest certain viral infections. For example, changes limited to the liver would suggest certain viral infections but not others due to the organ- and cell-specific tropism for many viruses. Other examples include the pattern(s) of acute hemorrhage associated with viral hemorrhagic fevers, changes associated with progressive multifocal leukoencephalopathy caused by JC virus infection, and viral myocarditis. That is not to say that a histopathologic diagnosis of viral infections is not possible, rather that the pattern of inflammation per se is not as characteristic as that of many bacterial, fungal, or mycobacterial infections.
It is important for pathologists to remember that many viral infections can be accompanied by secondary bacterial infections. Classic examples include bacterial superinfection during and following viral pneumonias and bacterial infections following ulcers caused by viruses (e.g., esophageal ulcers caused by the Herpes simplex virus or Cytomegalovirus ). Depending on the timing of any biopsy or collection of fluid, the acute inflammatory infiltrate associated with the secondary bacterial infection can easily obscure the underlying viral infection.
The histopathologic changes associated with viral infections are discussed in detail in 2, 3, 4, 5, 6, 7, 8, 9 .
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here