Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
For decades, the specific dermatoses of pregnancy represented a confusing group of overlapping entities, understood largely through anecdotes and case reports. However, more recent reviews have simplified and condensed the list of pregnancy-specific dermatoses ( Table 27.1 ) . For example, impetigo herpetiformis is now generally recognized as pustular psoriasis, perhaps induced by the relative hypocalcemia of pregnancy (see Ch. 8 ). Also, prurigo annularis, first reported in 1941 (without histology or laboratory information), and linear IgM disease of pregnancy (described in 1988) have not been subsequently reported, yet are frequently perpetuated in reviews.
| CLASSIFICATION OF THE DERMATOSES OF PREGNANCY | |
|---|---|
| Classification | Synonym(s) |
| Pemphigoid gestationis * | Herpes gestationis † Gestational pemphigoid |
| Polymorphic eruption of pregnancy (PEP) | Pruritic urticarial papules and plaques of pregnancy (PUPPP) † Toxic erythema of pregnancy Late-onset prurigo of pregnancy Toxemic rash of pregnancy |
| Intrahepatic cholestasis of pregnancy (ICP) | Cholestasis of pregnancy † Obstetric cholestasis Cholestatic jaundice of pregnancy Pruritus/prurigo gravidarum |
| Atopic eruption of pregnancy (AEP) | Prurigo of pregnancy * , † Prurigo gestationis (Besnier) Early-onset prurigo of pregnancy (Nurse) Papular dermatitis of pregnancy (Spangler) Pruritic folliculitis of pregnancy * Linear IgM disease of pregnancy Eczema in pregnancy |
Much of the confusion can be dispelled by reviewing the original articles: Besnier (1904) first used the term “prurigo gestationis” to include all patients with pregnancy-related dermatoses, other than those with pemphigoid gestationis. Costello (1941) subsequently referred to these patients as “prurigo gestationis of Besnier” and estimated an incidence of 2% of all pregnancies. Bourne (1962) characterized a subset of patients with intensely pruritic papules or urticarial plaques that tended to appear during the later part of the third trimester, coining the term “toxaemic rash of pregnancy”. Initial lesions typically developed within the abdominal striae of short women who experienced excessive weight gain during pregnancy. However, he offered no histopathologic or laboratory details.
Spangler et al. (also 1962) reported on a series of women with intensely pruritic, widely scattered, excoriated papules. Initial onset was during the second or third trimester and all patients suffered recurrences during subsequent gestations. The hallmarks of Spangler's cases were biochemical: elevated urinary human chorionic gonadotropin (HCG), decreased plasma hydrocortisone, and decreased serum half-life of hydrocortisone. Liver function tests and histopathology were not reported and immunofluorescence (IF) was not yet available. Most notably, Spangler's papular dermatitis was associated with high fetal wastage, a finding now thoroughly discredited.
Nurse (1968) cited all of the above literature but ignored it, dividing patients with (non-pemphigoid gestationis) pregnancy eruptions into “early” and “late” forms. The late papular/urticarial form clearly overlapped with Bourne's “toxaemic rash of pregnancy”, and has since been described as “pruritic urticarial papules and plaques of pregnancy” (PUPPP), “toxic erythema of pregnancy”, and, most recently, “polymorphic eruption of pregnancy” (PEP). Nurse's “early-onset” patients and Spangler's “papular dermatitis” patients were undoubtedly drawn from the same clinical spectrum, and subsequently these entities were reclassified as forms of “prurigo of pregnancy” (see Table 27.1 ).
Pruritic folliculitis of pregnancy was added to the list of pregnancy-specific diseases in 1981, yet all six of the original patients were thought to have papular dermatitis clinically. They were classified as having a different entity based solely upon shared histologic features, that is, sterile folliculitis. In spite of their clinical similarity to the patients reported by Spangler, none had appropriate biochemical investigations. In 1998, an adaptation of the Holmes and Black classification scheme (1983) was published and listed pruritic folliculitis as a variant of prurigo of pregnancy (see Table 27.1 ).
Introduction of the term “atopic eruption of pregnancy (AEP)” represents the most recent revision to the classification of dermatoses of pregnancy . Based upon an analysis of over 500 pregnant women with pruritus , significant overlap, both clinical and histologic, was noted amongst those patients with the diagnoses of prurigo of pregnancy, pruritic folliculitis of pregnancy, and eczema in pregnancy (~50% of the patients in this series) . AEP was thought to be a useful diagnostic term that encompassed these three disorders and reminded the clinician that the eczema was more likely to be of new onset as opposed to a flare of previously diagnosed atopic dermatitis.
Because of the lack of primary lesions, intrahepatic cholestasis of pregnancy (ICP) had long been omitted from the list of pregnancy dermatoses. However, failure to appreciate ICP in a pregnant woman with widespread excoriations or even prurigo nodularis surely, in retrospect, accounts for some of the confusion in terminology. Furthermore, ICP is the most important diagnosis to exclude in a pregnant patient with pruritus, as it is associated with significant fetal risk (see below).
▪ Gestational pemphigoid ▪ Herpes gestationis
Rare, pruritic, vesiculobullous eruption that develops during late pregnancy or the immediate postpartum period
Linear C3 deposition along the basement membrane zone (BMZ) by direct IF
IgG1 autoantibodies are directed against a transmembrane hemidesmosomal protein (BP180; BPAG2; collagen XVII)
Increased risk of prematurity and small-for-gestational age neonates; the risk correlates with disease severity
Commonly recurs in subsequent pregnancies
Pemphigoid gestationis is a rare, self-limited, autoimmune bullous disease. It is the most clearly characterized dermatosis of pregnancy and the only one that may also affect the skin of the newborn.
Milton first coined the term “herpes gestationis” in 1872 and Bulkley (1874) canonized the term “as embodying the clinical characters of the eruption and signifying at the same time the sex and state of the body in which it appears”. Little additional insight was gained until IF techniques revealed complement deposition along the BMZ in 1973, a feature now accepted as essential for the diagnosis of pemphigoid gestationis.
The incidence of pemphigoid gestationis has been estimated at 1 : 1700–1 : 50 000 pregnancies , correlating with the prevalence of HLA-DR3 and -DR4 in different populations. While occurring primarily during pregnancy and the immediate postpartum period, pemphigoid gestationis has rarely developed in association with trophoblastic tumors (hydatidiform mole, choriocarcinoma). Interestingly, no case of a pemphigoid-like disease has been reported in men with choriocarcinoma, who have a biochemically similar yet entirely syngenic tumor; of note, the nuclear genome in placental tissue (and hence choriocarcinoma in women) is primarily paternal in origin. Patients with a history of pemphigoid gestationis appear to be at increased risk for the development of Graves disease .
Historically, pemphigoid gestationis was thought to be caused by an anti-BMZ “serum factor” (the “herpes gestationis [HG] factor”) that induces C3 deposition along the dermal–epidermal junction. This factor is now known to be complement-fixing autoantibodies of the IgG1 subclass directed against a 180 kDa transmembrane hemidesmosomal protein (BP180; BPAG2; collagen XVII). As in patients with bullous pemphigoid (BP), it is the non-collagenous (NC) segment closest to the plasma membrane of the basal keratinocyte, NC16A, that constitutes the immunodominant region of BP180 (see Fig. 31.9 ). Circulating antibodies are almost exclusively directed against this domain, as demonstrated by ELISA and immunoblot studies of maternal or neonatal sera.
What initiates the production of autoantibody remains enigmatic. Because the antibodies also bind to amniotic basement membrane (a structure derived from fetal ectoderm and antigenically similar to skin), attention has focused on immunogenetics and potential cross-reactivity between placental tissue and skin. Immunogenetic studies have revealed a marked increase in HLA antigens DR3 or DR4, and, curiously, nearly 50% of patients have the simultaneous presence of both. There is essentially a 100% incidence of anti-HLA antibodies in patients with a history of pemphigoid gestationis . Since the only source of disparate HLA antigens is typically the placenta (which is primarily of paternal origin), the universal finding of anti-HLA antibodies implies a high frequency of immunologic insult during gestation. Women with pemphigoid gestationis also have increased expression of MHC class II antigens (DR, DP, DQ) within the villous stroma of their chorionic villi. It has therefore been proposed that pemphigoid gestationis is a disease, initiated by the aberrant expression of MHC class II antigens (of paternal haplotype), that serves to initiate an allogeneic response to placental BMZ, which then cross-reacts with skin .
Pemphigoid gestationis may develop during any trimester as well as immediately postpartum, but classically it presents during late pregnancy. There is an abrupt onset of cutaneous lesions on the trunk, in particular the abdomen and often within or immediately adjacent to the umbilicus ( Fig. 27.1 ). Rapid progression to a generalized pemphigoid-like eruption then occurs, with pruritic urticarial papules and plaques, followed by clustered (herpetiform) vesicles or tense bullae on an erythematous base. The eruption may involve the entire body, sparing only the mucous membranes. While the clinical presentation and course may vary considerably, spontaneous improvement during late gestation is common. This is followed, however, by a flare at the time of delivery in 75% of patients. Such flares may be dramatic, with an explosive onset of blistering within hours.
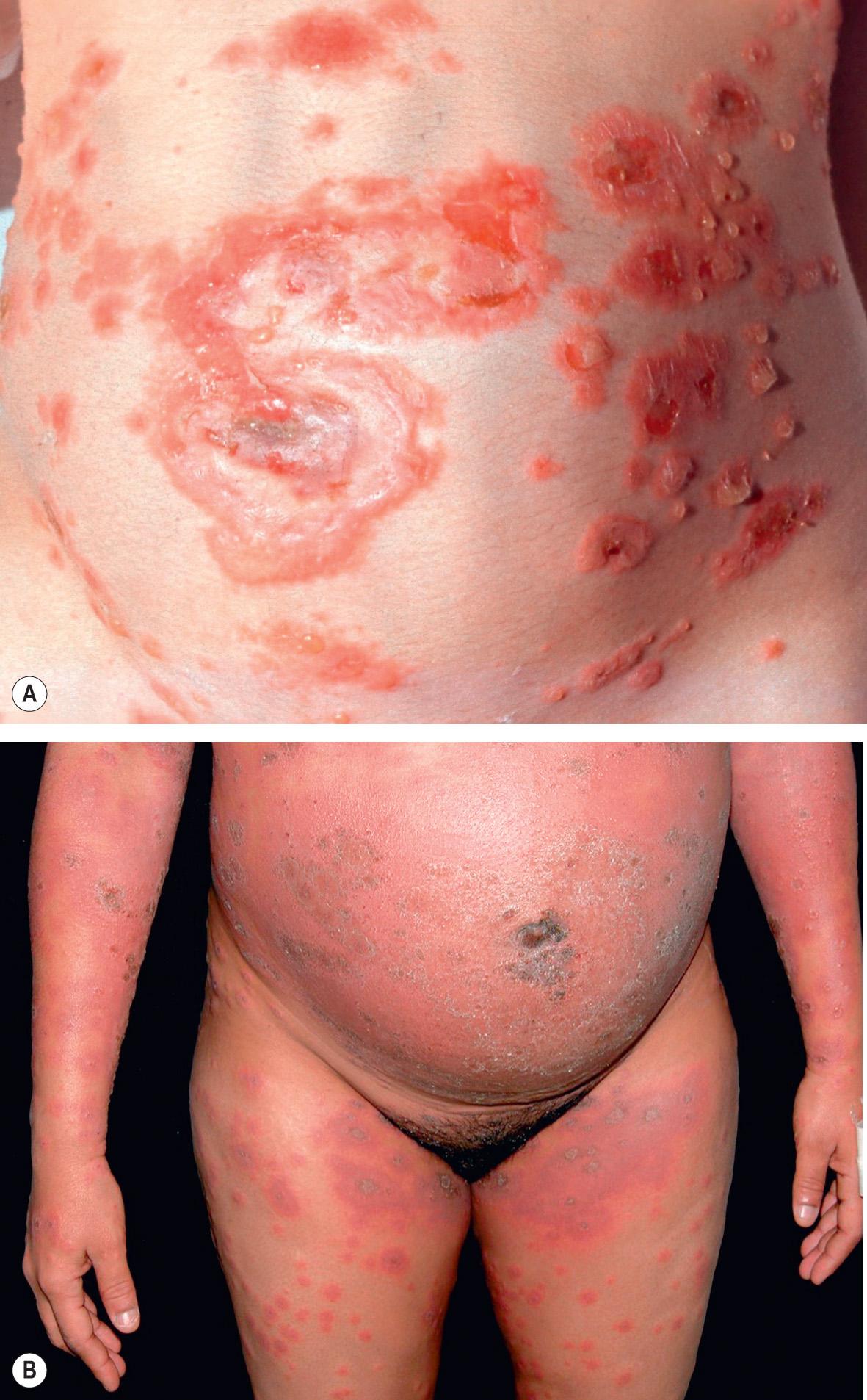
Most disease activity spontaneously remits during the weeks to months following delivery, but there are isolated reports of a protracted course postpartum. Flares and/or recurrences in association with menstruation are common, and in 25–50% of patients, they may also be induced by oral contraceptives ( Table 27.2 ). Pemphigoid gestationis may not develop during the patient's first pregnancy, but, once established, it is quite likely to recur in subsequent pregnancies, usually with an earlier onset and more severe course. “Skipped” pregnancies have been observed in 5–8% of women.
| DERMATOSES OF PREGNANCY – FETAL RISK, INVOLVEMENT OF NEWBORN SKIN, AND RISK OF RECURRENCE | |||
|---|---|---|---|
| Dermatosis | Fetal risk | Newborn skin involvement | Risk of recurrence |
| Pemphigoid gestationis | Increased risk of prematurity and small-for-gestational age neonates; risk correlates with disease severity | Mild and transient lesions of pemphigoid gestationis in up to 10% | Commonly recurs (“skipped” pregnancies in only 5–8% of women); recurrences induced by oral contraceptives in 25–50% |
| Polymorphic eruption of pregnancy | None | None | Usually does not recur |
| Intrahepatic cholestasis of pregnancy | Increased risk of premature labor (20–60%), intrapartal fetal distress (20–30%), and stillbirths (1–2%) | None | Recurrence in 45–70% of subsequent pregnancies; may be triggered by oral contraceptives |
| Atopic eruption of pregnancy | None | None | Commonly recurs due to atopic diathesis |
Approximately 10% of newborns develop mild skin involvement due to passive transfer of maternal antibodies and this resolves spontaneously within days to weeks (see Ch. 34 ). There seems to be an increased risk of prematurity and small-for-gestational age neonates, presumably due to chronic placental insufficiency. Recently, it was shown that this risk correlates with disease severity, i.e. occurrence of blistering and early onset, and not with the use of systemic corticosteroids .
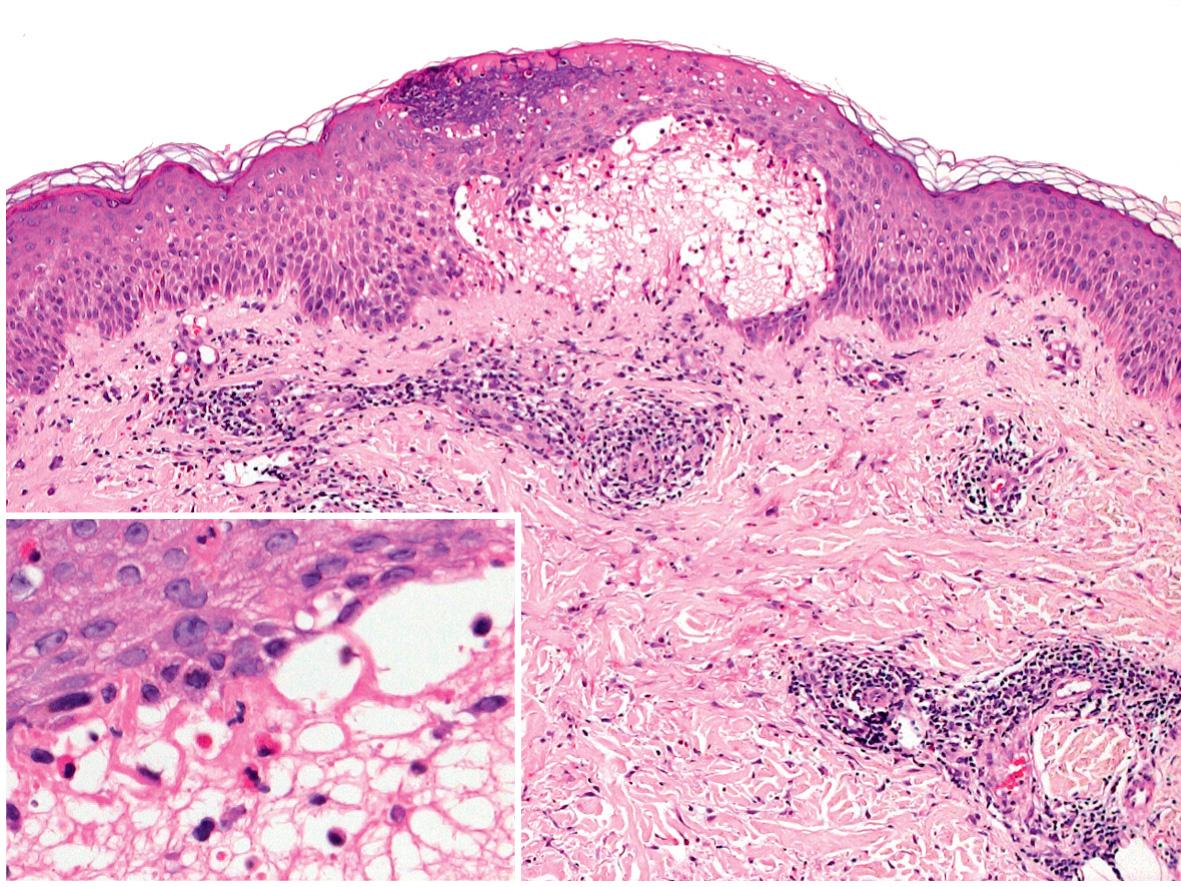
The classic histologic finding of a subepidermal vesicle is seen in the minority of patients. Instead, a nonspecific mixed cellular infiltrate containing a variable number of eosinophils is more common. The presence of eosinophils is the most constant histologic feature of pemphigoid gestationis ( Fig. 27.2 ).
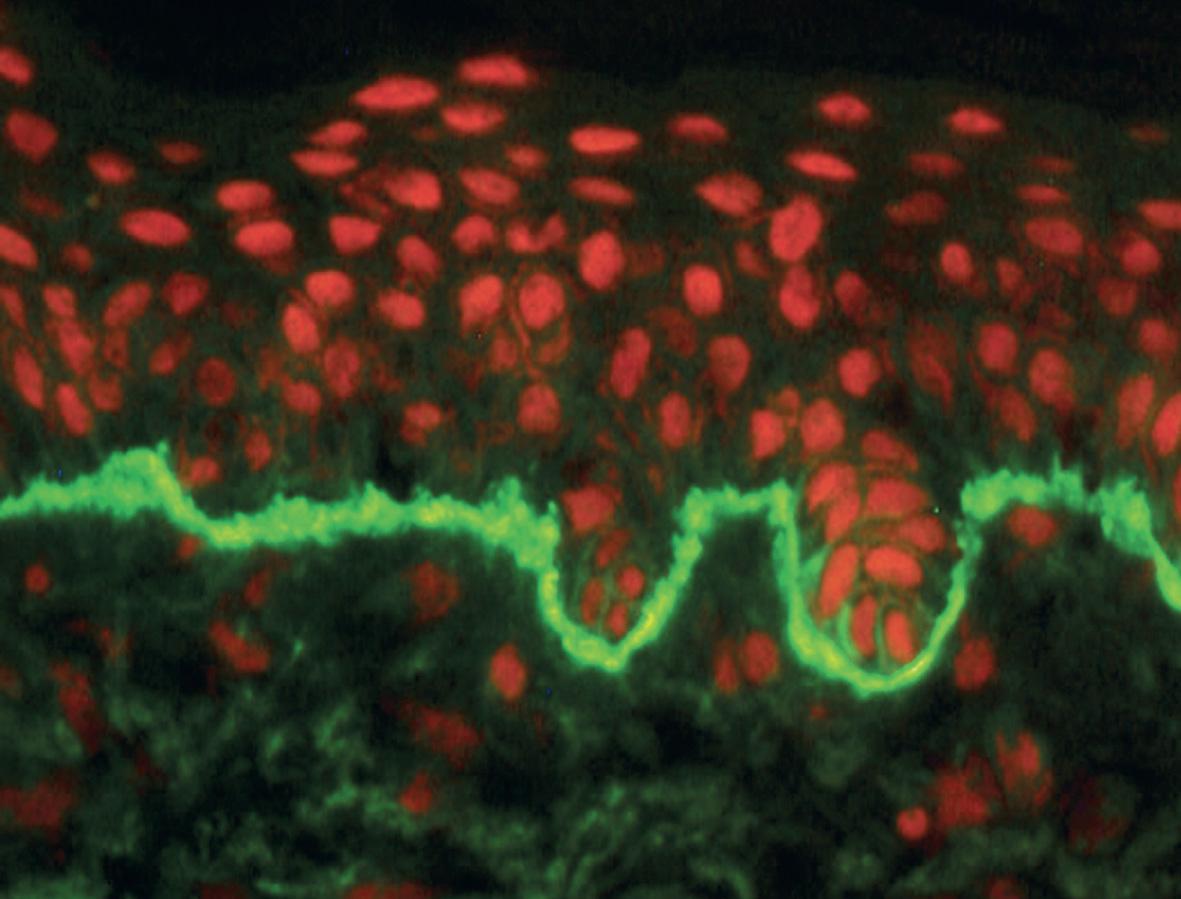
The essential component for the diagnosis of pemphigoid gestationis is a linear deposition of C3 along the BMZ of perilesional skin by direct IF microscopy ( Fig. 27.3 ). This is observed in 100% of patients, and linear IgG deposition is seen in 30% of patients. When salt-split skin specimens are employed for conventional indirect IF, deposition of IgG along the bottom of the epidermal fragment is observed in 30% of patients. However, complement-added indirect IF reveals the circulating anti-BMZ IgG1 autoantibodies in essentially all patients. Determination of antibody titers via BP180-NC16A ELISA may be helpful in following disease activity and monitoring therapy.
An increased incidence of anti-thyroid antibodies has been documented, but clinically apparent thyroid dysfunction is uncommon . Routine laboratory investigations are normal.
The most frequent considerations in the differential diagnosis are PEP and drug eruptions. PEP is a particularly challenging exclusion, given the difficulty of distinguishing PEP and urticarial lesions of pemphigoid gestationis. IF and, more recently, the BP180-NC16A ELISA are key to the differentiation and are especially relevant in helping the patient plan for future pregnancies.
The primary goal in treating this self-limited disease is to relieve pruritus and suppress blister formation. In mild cases, the use of potent topical corticosteroids combined with emollients and systemic antihistamines may be adequate. However, systemic corticosteroids remain the cornerstone of therapy ( Table 27.3 ). Most patients respond to 0.5 mg/kg of prednisolone daily; the dose is tapered as soon as blister formation is suppressed. The common flare associated with delivery usually requires a temporary increase in dosage. Those rare patients with refractory disease may benefit from plasmapheresis during pregnancy. Persistent disease after delivery is uncommon and is treated like BP.
| SPECIAL CONSIDERATIONS FOR CORTICOSTEROID AND ANTIHISTAMINE USE DURING PREGNANCY | |
|---|---|
| Corticosteroids | |
| Topical |
|
| Systemic |
|
| Antihistamines | |
| Systemic |
|
Anecdotal alternatives to corticosteroids (dapsone, doxycycline or minocycline ± nicotinamide, pyridoxine, cyclosporine) or adjuvants (methotrexate, cyclophosphamide, gold, IVIg) have been tried. None of these medications, with the possible exception of cyclosporine, are safe prior to term and thus should be avoided.
▪ Pruritic urticarial papules and plaques of pregnancy (PUPPP) ▪ Bourne's “toxaemic rash of pregnancy” ▪ Nurse's “late-onset prurigo” of pregnancy ▪ Toxic erythema of pregnancy
Urticarial papules and plaques that usually first appear within striae distensae during the latter portion of the third trimester or immediately postpartum
Development of polymorphous features (vesicles, erythema, target, and eczematous lesions) with disease progression
Most frequent in primiparous women
Nonspecific histologic features, negative IF, and normal routine laboratory evaluation
No maternal or fetal risks; usually does not recur
Polymorphic eruption of pregnancy (PEP), formerly known as PUPPP, is a common gestational dermatosis. It is characterized by a typical clinical presentation, normal laboratory tests, and negative IF or ELISA.
The term PUPPP, introduced by Lawley et al. in 1979, focuses on the initial clinical findings in this disorder but overlooks its later polymorphous features. In order to encompass the entire clinical spectrum, the term “polymorphic eruption of pregnancy” has been introduced and is now generally accepted.
The incidence is ~1 in 160 deliveries . It is seen predominantly in primiparous women and tends not to recur in subsequent pregnancies. There is neither an autoimmune diathesis nor an association with a specific HLA type.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here