Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Large bowel screening programs have been shown to substantially reduce the incidence of colorectal carcinoma by identifying and removing premalignant polyps. Increased use of colonoscopy has led to an increase in the number of polyps encountered in daily pathology practice. Broadly speaking, the term polyp refers to any form of lesion that projects above the surrounding colonic mucosa. The vast majority of colorectal polyps are adenomatous, serrated, inflammatory, or hamartomatous. In addition, polyps may develop from mesenchymal proliferations, benign or malignant hematolymphoid tissue, metastatic tumors, and a wide variety of non-neoplastic substances, such as air. The relative proportions of these various types of polyps depends on the type of population undergoing endoscopy (e.g., age, associated risk factors such as inflammatory bowel disease [IBD] or polyposis syndrome) and the method of investigation (e.g., sigmoidoscopy vs. colonoscopy), but serrated and adenomatous polyps are by far the most common polyps in general.
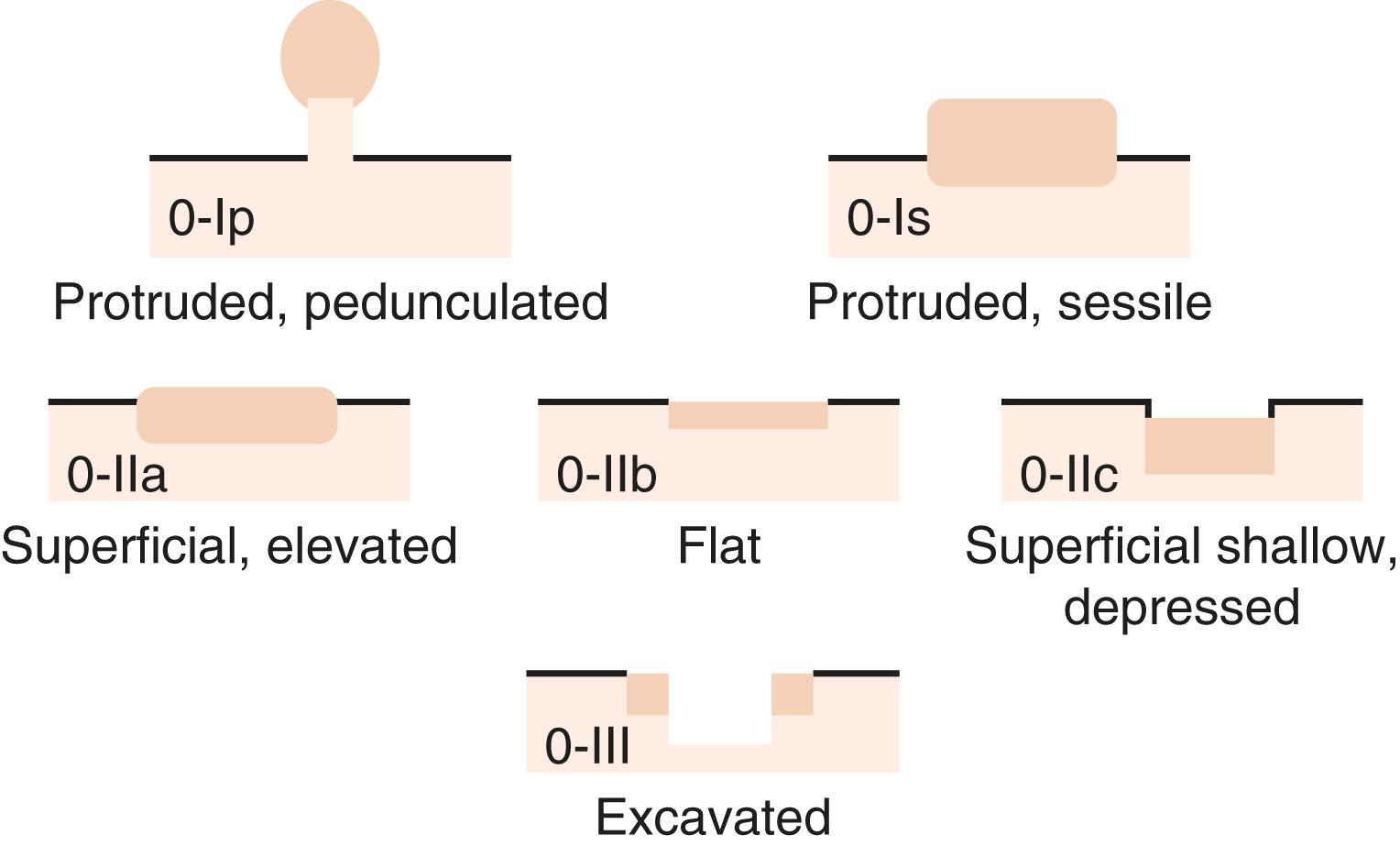
In 2002 an international group of endoscopists, surgeons, and pathologists gathered in Paris, France, to revise and modify a prior Japanese classification of superficial neoplastic lesions. This resulted in the Paris Endoscopic Classification for Superficial Neoplastic Lesions of the colon, and it applies to all polyps and early carcinomas that do not invade deeper than the submucosa ( Fig. 22.1 ). This classification scheme is useful because each subtype of lesion carries a different risk of invasive carcinoma, and it also helps determine the type of endoscopic resection that must be performed.
Subsequently, endoscopists have used narrow-band imaging to classify colorectal lesions as either serrated/hyperplastic, adenomatous, or invasive carcinoma based on the color, the quality of the blood vessels, and the surface mucosal pattern. The Narrow-Band Imaging International Colorectal Endoscopic (NICE) classification system has been shown to have a high degree of sensitivity for predicting the presence of submucosal carcinoma, but it has limited utility in distinguishing adenomatous from serrated polyps in routine clinical practice. The endoscopic pit pattern and granularity have also been used to classify polyps and help predict the risk of submucosal invasive carcinoma. Using these various endoscopic features and classification systems, gastroenterologists can determine whether the polyp in question requires an advanced endoscopic technique for removal or it can simply be removed by piecemeal polypectomy.
Adenomas are common lesions. They are almost always asymptomatic. However, some patients may develop either overt, or occult, rectal bleeding, and large polyps may lead to iron-deficiency anemia. The clinical importance of adenomas is almost entirely related to their well-established premalignant nature. In general, the prevalence of adenomas increases dramatically with age. By the fifth decade of life, approximately 12% of individuals have adenomas, of which approximately 25% are considered high-risk lesions (see later). After 50 years of age, the prevalence rate of adenomas continues to increase to approximately 50% of the population in high-risk Western countries such as the United States. The likelihood that adenomas will develop is strongly influenced by family history and by a variety of nutritional factors. ,
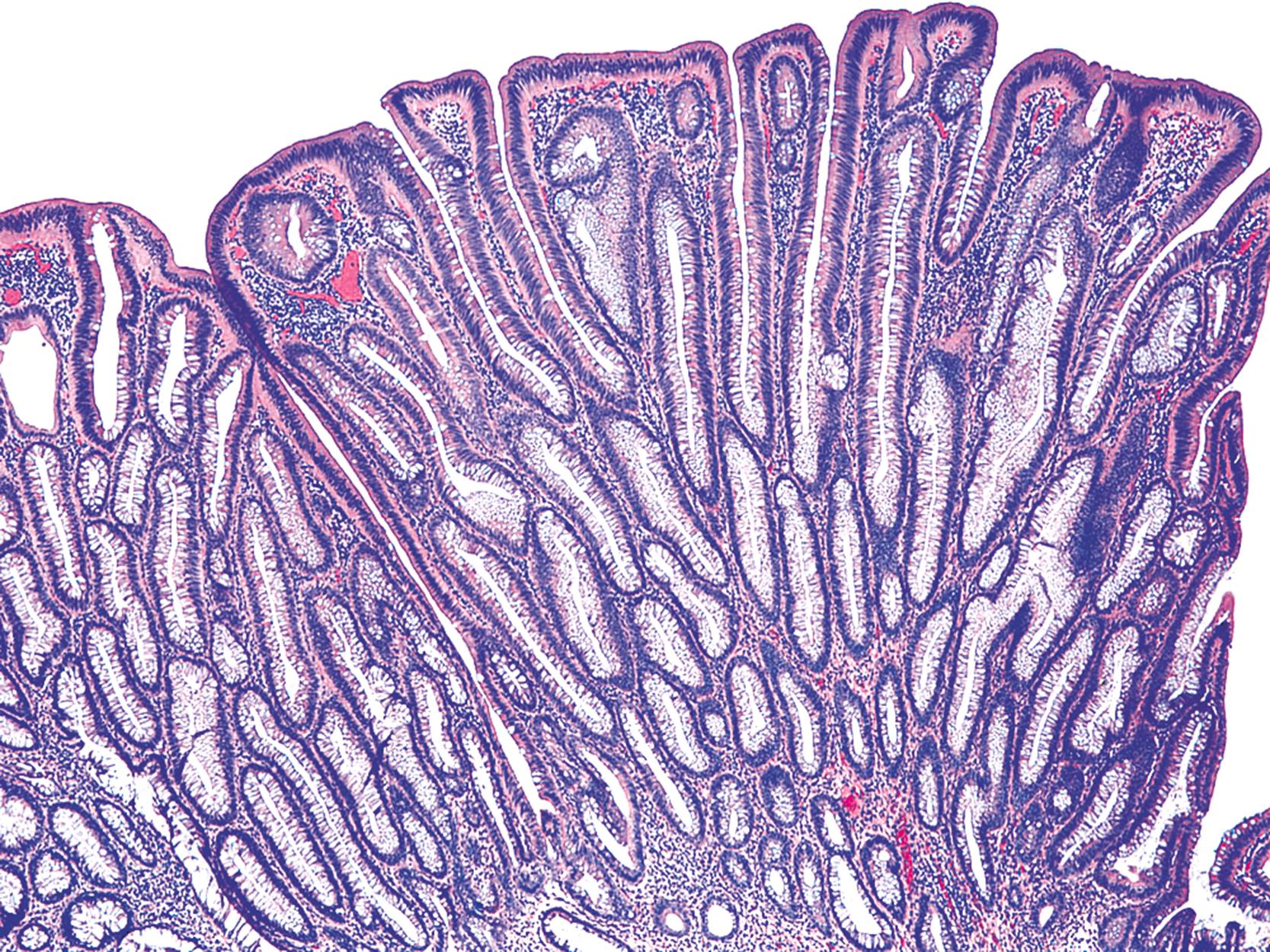
Adenomas are defined as in situ dysplastic clonal proliferations of epithelium ( Fig. 22.2 ). Microscopically, adenomas are categorized, architecturally, as either tubular, tubulovillous, or villous ( Fig. 22.3 ). However, precise histological criteria for each of these three categories vary widely. One reasonable rule of thumb is that villous adenomas should contain at least 75% villi, whereas pure tubular lesions should contain less than 25% villi. Thus tubulovillous lesions are those that contain between 25% and 75% villous epithelium. The degree of villous differentiation has been shown to increase with increasing size of the adenoma. Villous lesions are considered “advanced” for the purpose of clinical management (see later discussion).
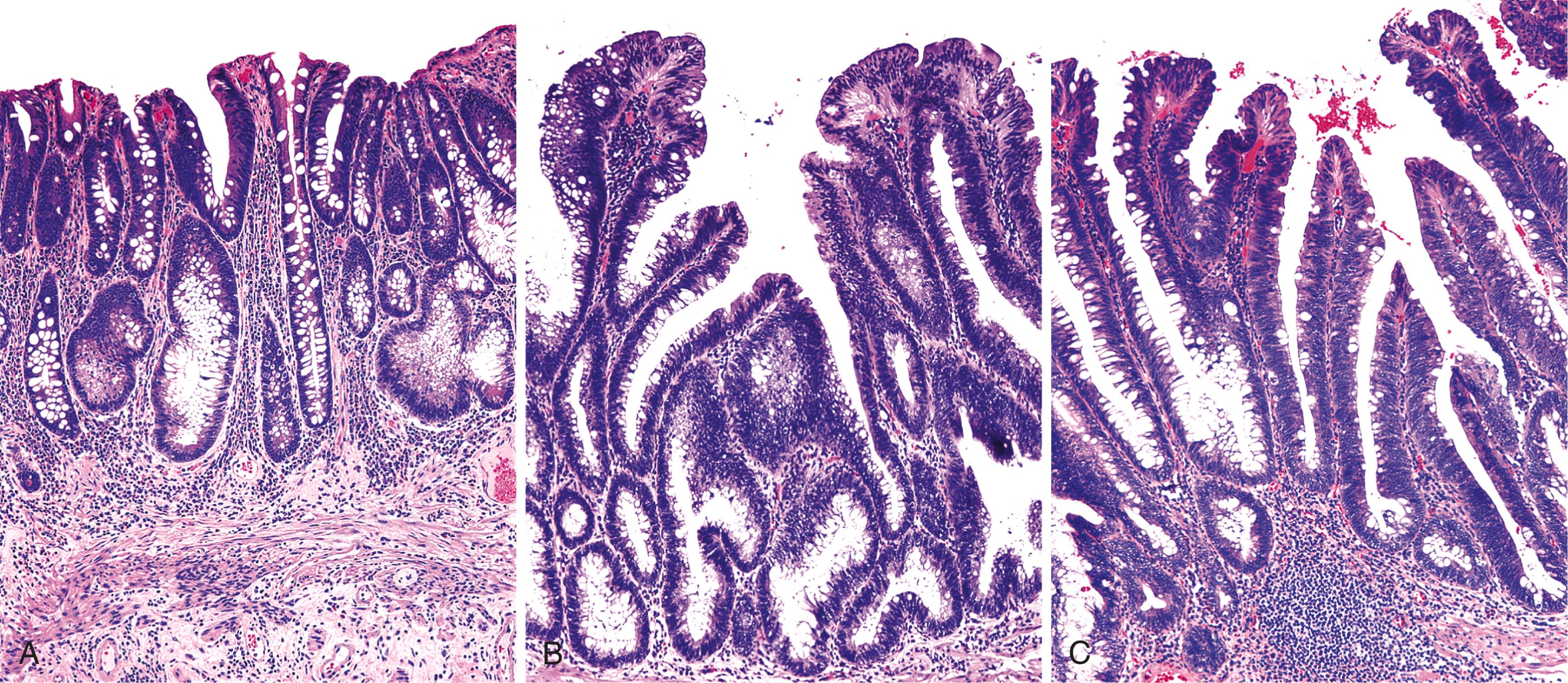
The lamina propria of adenomas may contain a variable amount of lymphocytes, plasma cells, neutrophils, and eosinophils. Some adenomas, particularly those with high-grade dysplasia, may be ulcerated. Paneth cell or neuroendocrine cell metaplasia is a common finding and may be marked in some cases. Rarely, one may see squamous metaplasia (or squamous morules) in adenomas. Some adenomas, particularly those that are pedunculated, may contain dilated and ruptured crypts with mucin extravasation into the lamina propria. Often, these cases are associated with epithelial displacement into the submucosa. Desmoplasia is not normally seen in benign colonic adenomas. When present, it should raise a very strong suspicion of a submucosal invasive adenocarcinoma.
Very rarely, adenomas are associated with a minute proliferation of well-differentiated neuroendocrine cells either adjacent to, or underlying, dysplastic crypts in either the mucosa or submucosa. These lesions have been referred to as composite adenoma-microcarcinoid tumors. The neuroendocrine component consists of small clusters and nests of uniform cells with round nuclei and eosinophilic cytoplasm ( Fig. 22.4 ). The vast majority of studies indicate that composite adenoma-microcarcinoid have a benign clinical course, even when the neuroendocrine cells extend into the submucosa; however, care must be taken to exclude a high-grade neuroendocrine carcinoma arising from an adenoma.
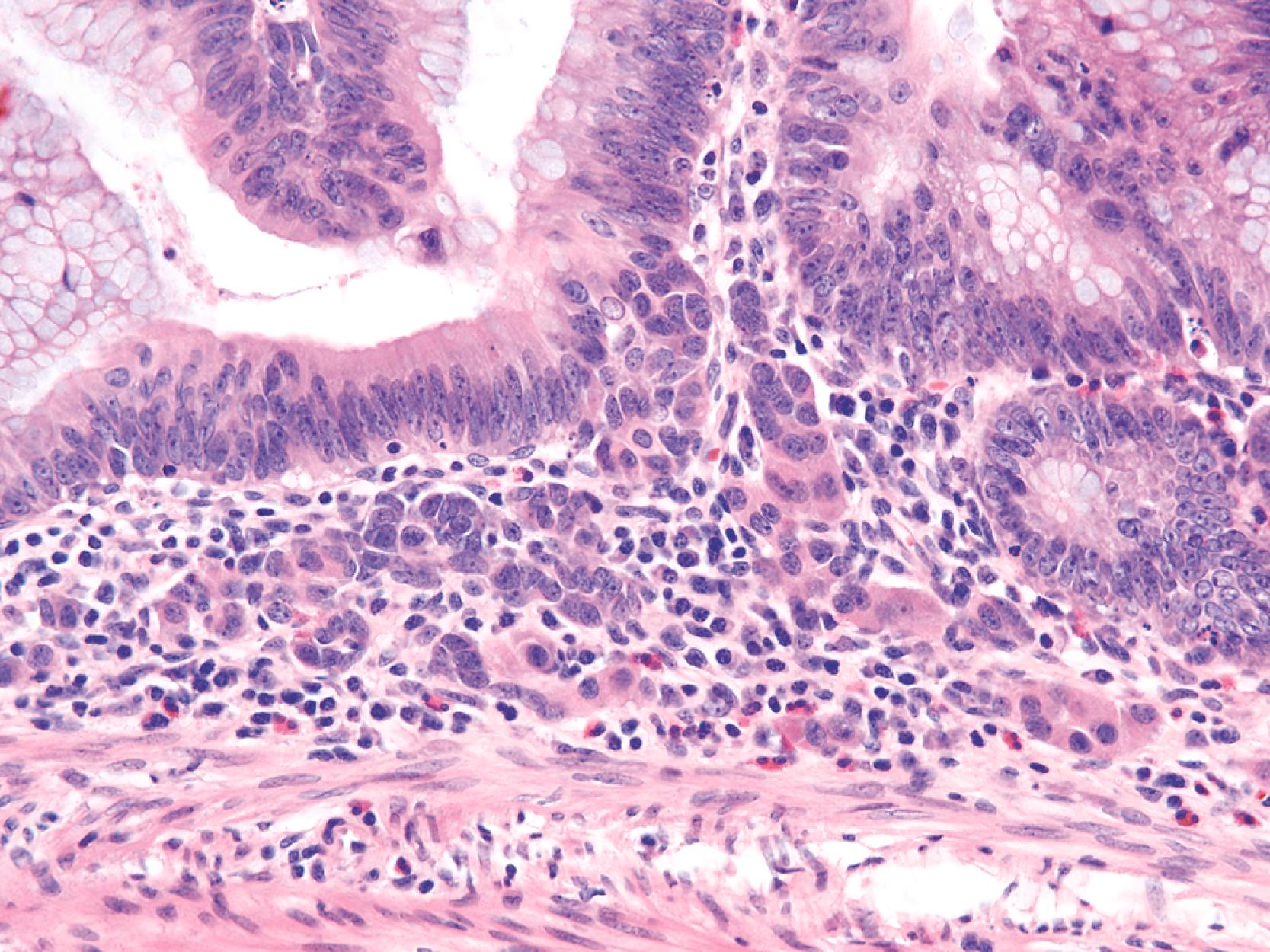
Microscopically, by definition, all adenomas contain at least low-grade dysplasia. Dysplasia in adenomas is generally classified as either low grade or high grade based on a combination of cytological and architectural features. This classification system is favored because (1) a decrease in the degree of interobserver variability has been documented regarding interpretation of dysplasia by pathologists; (2) improved clinical pathological relevance is seen with regard to surveillance and treatment options; and (3) the term carcinoma in situ is often misinterpreted by clinicians as indicative of malignant behavior, which may lead to an unnecessary colonic resection.
Low-grade dysplasia is defined by the presence of architecturally noncomplex crypts containing nuclei that are pseudostratified, or partially stratified, such that the cell nuclei reach only the lower half of the cell cytoplasm. Mitotic activity may be brisk, but atypical mitoses, significant loss of polarity, and pleomorphism are minimal, if present at all. Apoptosis is usually readily identifiable throughout the adenomatous epithelium. The crypts are arranged in a parallel configuration without significant back-to-back configuration, cribriforming, or complex budding.
High-grade dysplasia is defined by marked pseudostratification, or stratification, of neoplastic nuclei that extend toward the luminal half of the cells and usually contain significant pleomorphism, increased mitotic activity, atypical mitoses, and marked loss of polarity. Architectural changes such as back-to-back gland configuration and cribriforming may also be observed. With progression of neoplasia, glands lose their orderly configuration and become more irregular and complex. In addition, neoplastic nuclei become more “open” in appearance and may contain prominent nucleoli ( Fig. 22.5 ). The N:C ratio of the cells increases, and loss of polarity becomes marked.
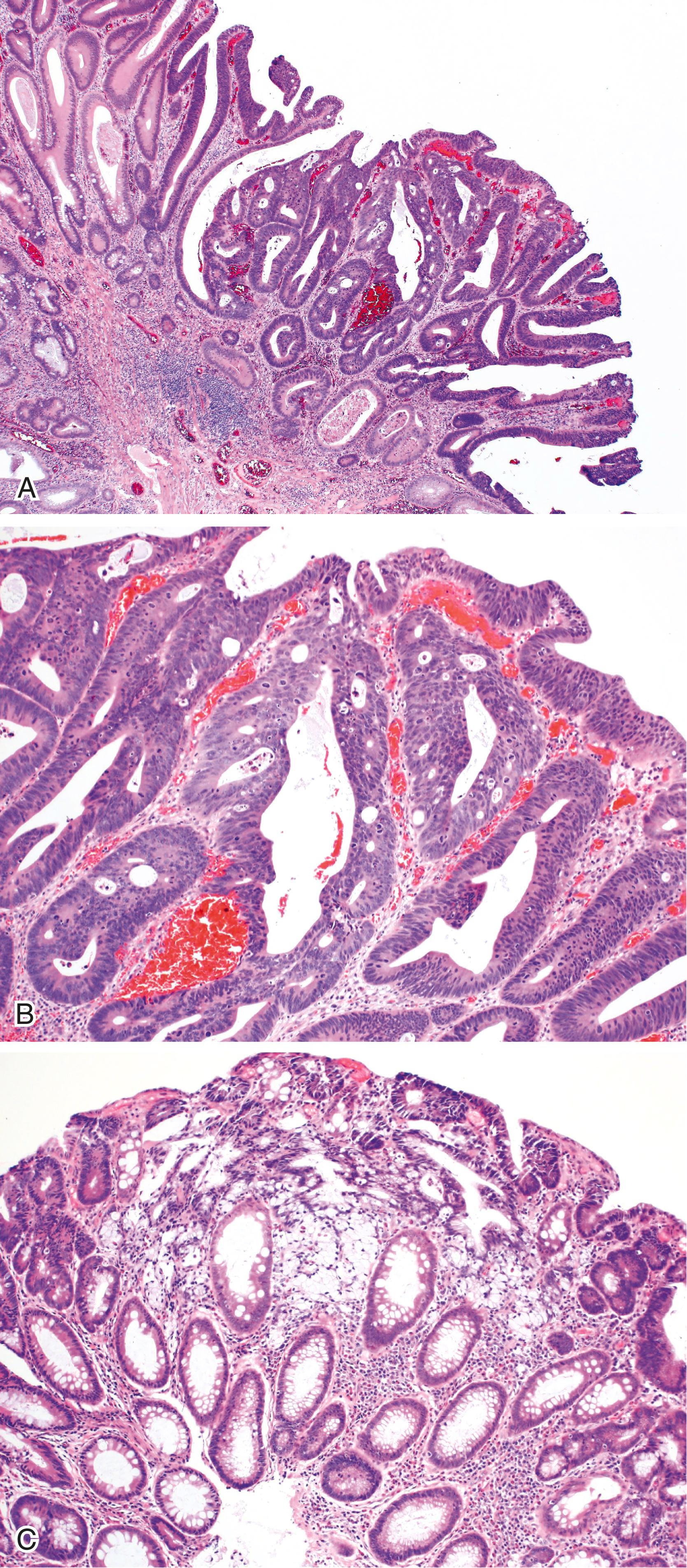
The pathological criteria and clinical importance of intramucosal adenocarcinoma in adenomatous polyps is a controversial topic. Intramucosal adenocarcinoma is defined as invasion into the lamina propria, including the muscularis mucosae, but not through the muscularis mucosae into the submucosa (see Fig. 22.5 ). Although this is a well-recognized diagnosis in other sites of the gastrointestinal (GI) tract, such as the stomach and esophagus, many studies have shown that invasion of the lamina propria of the colon has virtually no increased risk of lymph node metastasis provided that the polyp has been completely excised. This has led some authorities to conclude that the term intramucosal adenocarcinoma should not be used in the setting of colonic adenomatous polyps, given the lack of clinical significance. However, other experts argue that the term intramucosal adenocarcinoma should be used in any setting in which there is pathological evidence of invasion into the lamina propria, but that it is incumbent on the pathologist, gastroenterologist, and surgeon to understand the biological potential of this phenomenon, and that polypectomy is sufficient to treat these lesions. Ultimately, the goal is to avoid unnecessary surgery for patients with intramucosal adenocarcinoma. However, if one feels compelled to use the term adenoma with intramucosal adenocarcinoma, the diagnostic report should always include a statement regarding the absence of submucosally invasive cancer, the status of the resection margin, and the overall adequacy of resection and need for further therapy, which in most cases is best accomplished with endoscopic polypectomy.
Screening colonoscopy has played a major role in the steady decline in the incidence of colorectal carcinoma in the United States. The National Polyp Study, , a large study initiated in 1990, has demonstrated that removal of adenomas by endoscopic polypectomy significantly decreases both the incidence of colorectal adenocarcinoma and colorectal cancer–specific mortality. Patients with colon cancer identified on screening colonoscopy not only have lower-stage disease at presentation, but also have more favorable outcomes independent of their staging. The best predictor for the presence of malignancy within an adenoma at the time of excision is the polyp size. Among adenomas larger than 2 cm, there is a 10% to 20% risk of carcinoma in the polyp at the time of removal. Adenomas measuring between 1 and 2 cm have a 5% risk of harboring cancer, and those smaller than 1 cm have a much lower risk of adenocarcinoma (<1%).
The degree of dysplasia also represents an independent risk factor for malignancy in adenomas, regardless of polyp size. Most adenomas harbor only low-grade dysplasia. However, larger lesions have a greater likelihood of harboring high-grade dysplasia. , , Nevertheless, high-grade dysplasia within a colorectal adenoma does not increase the risk of carcinoma elsewhere in the patient’s colon. Because of the time involved for an adenoma to acquire sufficient molecular changes for it to become invasive, most remain benign. In general, adenomas are slow-growing lesions. The lifetime prevalence rate of adenomas is approximately 50%, whereas the lifetime prevalence of colorectal adenocarcinoma is approximately 6%. Therefore it is evident that only a small minority of polyps ultimately develop adenocarcinoma. Actual longitudinal follow-up data have been difficult to obtain because adenomas are typically removed at the time of endoscopic identification. One cohort of 35 patients, all of whom had an adenoma smaller than 5 mm in maximum diameter, was followed up for 2 years. In 50% of the patients, an increase in size of the adenoma occurred; in the remainder, the lesion remained unchanged or even regressed slightly.
The appropriate treatment for all colorectal adenomas, regardless of their size, architectural type, or degree of dysplasia, is complete endoscopic removal with negative margins ( Box 22.1 ). ,
Average risk (no first-degree relative with colon cancer)
Colonoscopy at 50 years of age:
If no adenoma or carcinoma, repeat in 10 years
If 1 to 2 tubular adenomas <10 mm, repeat in 7 to 10 years
If 3 to 4 tubular adenomas <10 mm, repeat in 3 to 5 years
If 5 to 10 tubular adenomas <10 mm, repeat in 3 years
If adenoma ≥10 mm, repeat in 3 years
If adenoma with high-grade dysplasia, tubulovillous or villous architecture, repeat in 3 years
If >10 adenomas on single examination, repeat in 1 year (consider genetic testing for these patients and for those with >10 cumulative adenomas)
If piecemeal resection of and adenoma ≥20 mm, repeat in 6 months
Moderate risk (first-degree relative with colon cancer before 60 years of age or ≥2 first-degree relatives with colon cancer)
Colonoscopy at 40 years of age or 10 years before age of occurrence in relative, whichever comes first: follow guidelines as above
High risk (familial adenomatous polyposis [FAP], Lynch syndrome, serrated polyposis)
FAP: Colonoscopy every 1 to 2 years beginning at 10 to 12 years of age
Lynch syndrome: Colonoscopy every 1 to 2 years beginning at 20 to 25 years of age
Serrated polyposis: Colonoscopy every 1 year after diagnosis
Colorectal adenocarcinoma prevention screening recommendations are based on the distribution of adenomas throughout the colon, the age of the patient, and the patient’s family history (see Box 22.1 ). Although most adenomas occur distal to the splenic flexure, up to 40% occur in the proximal colon. At least one-fourth of patients have adenomas that are present only proximal to the splenic flexure. For this reason, full colonoscopy is the preferred method for screening in the United States over all other modalities. Yearly fecal immunochemical testing (FIT) is suitable for those who decline colonoscopy. Second-tier tests include CT-colonography every 5 years, FIT-fecal-DNA test every 3 years, and flexible sigmoidoscopy every 5 to 10 years. The current consensus regarding screening guidelines supports the use of colonoscopy starting at 50 years of age, although there have been recent recommendations to reduce the age to 45. Persons with a family history of colorectal carcinoma in a first-degree relative diagnosed before 60 years of age should undergo colonoscopy every 5 years beginning at 40 years of age, or 10 years before the age at which the patient’s relative was diagnosed with an adenomatous polyp. , African Americans should undergo initial screening at 45 years of age. Colonoscopy should be repeated every 1 to 10 years, depending on the findings at the initial screening, and the patient’s family history. See Chapter 2 for details of screening and surveillance.
Aside from the diagnosis of an “adenoma,” pathologists should also report the architectural pattern (tubular, tubulovillous, or villous), particularly when villous; the presence or absence of high-grade dysplasia; and the status of the polypectomy margins whenever possible. Small, “diminutive” tubular adenomas are usually excised in total, and these are usually removed in such a manner that it is often not possible to evaluate the margins of the specimen. This is normally acceptable from a clinical point of view because the endoscopist is keenly aware of the method used to excise the polyp, and as a result, the possibility of a lack of ability of the pathologist to evaluate margins when removed as such. Similarly, margins cannot normally be evaluated in large adenomas that are removed by piecemeal polypectomy. In this situation, the endoscopist would normally know whether the polyp was removed in total (and as such, determination of the margins by the pathologist is not necessary) and also whether a certain fragment of polyp represented the stalk of the polyp where the deep margin resides. In this case, the pathologist may issue a comment regarding the status of the piece of tissue that was designated as the polyp stalk. For polyps that are removed in piecemeal, it would be expected to find multiple pieces of tissue with cautery effect at the edges of tissue fragments, either with or without dysplasia. These do not normally represent the final margins of the polyp, however. In situations in which the margin cannot be evaluated, it is usually helpful to indicate this in the diagnostic report. In contrast, if a polyp is removed in one piece, then the status of the resection margins should be evaluated and commented on in the pathology report. If possible, the number of each polyp type should be provided; however, this is often difficult when multiple polyps are placed in the same jar. The endoscopic size of the polyp is used for postpolypectomy surveillance guidelines; however, if the endoscopic size is markedly different from the histological size, it is helpful to comment on this finding as well.
A key issue is risk stratification of patients based on the likelihood of developing advanced neoplasia in the future. Patients are divided into those with low risk (defined as 1 to 2 tubular adenomas <10 mm in size), intermediate risk (3 to 4 tubular adenomas <10 mm), and high risk defined as an advanced adenoma (adenoma with villous features, high-grade dysplasia, or adenocarcinoma, size ≥10 mm, or five or more adenomas) (see Box 22.1 ). Advanced adenomas require more aggressive colonoscopic surveillance given the increased risk for further metachronous adenomas and adenocarcinoma. Unfortunately, the degree of interobserver variability is high for determination of the presence and degree of villous architecture and the presence or absence of high-grade dysplasia, even among expert GI pathologists. However, the vast majority of villous adenomas, or adenomas with high-grade dysplasia, are ≥10 mm in size. Thus these polyps are considered “advanced” regardless of the presence or absence of these high-risk features, therefore the lack of interobserver agreement for villosity and/or high-grade dysplasia would not affect surveillance. Importantly, the finding of advanced morphological features should not be considered an indication for colectomy because advanced lesions have no potential for metastasis unless they harbor an invasive cancer.
Foci of displaced or “misplaced” epithelium or mucin extravasation within the submucosa is a rather common finding in adenomas, particularly in polyps that have a long or pedunculated stalk, and those from the left colon versus the right colon. This may lead to a misdiagnosis of invasive adenocarcinoma. This pathological reaction occurs in 2% to 4% of adenomatous polyps; it tends to occur more commonly in the left colon and particularly in the sigmoid colon, where the intraluminal pressures are higher and peristalsis is more vigorous. It occurs in patients of all ages and is slightly more common among males. Displaced epithelium in adenomas is believed to occur secondary to peristalsis-induced twisting and torsion of the polyp stalk, which leads to vascular compromise, inflammation, breakdown of the muscularis mucosae, and eventually herniation of adenomatous epithelium into the submucosa. Thus this reaction occurs much more commonly in polyps that have a long stalk, which are more susceptible to twisting and torsion. Rarely, adenomatous glands can herniate into a submucosal lymphoid aggregate. This results in formation of a “lymphoglandular complex.” This phenomenon tends to occur in regions of the mucosa in which the muscularis mucosae is incomplete or shows breaches in its integrity and sites in which blood vessels enter the mucosa from the submucosa. Such adenomas tend to be sessile in structure, in contrast with the more common pedunculated polyp that more often contains epithelial displacement resulting from tissue injury.
The intramucosal portion of an adenoma that contains displaced epithelium may have features that range from low- to high-grade dysplasia, and it may be tubular, tubulovillous, or villous. More often than not, foci of displaced epithelium show a similar degree of dysplasia to that in the intramucosal portion of the polyp. The epithelium in the submucosa is usually well-lobulated and composed of well-circumscribed aggregates of crypts in a pattern reminiscent of the mucosal portion of the polyp. Displaced epithelium is usually surrounded by a distinct rim of lamina propria, is often associated with hemorrhage or hemosiderin deposition in the stroma, shows no evidence of desmoplasia, and lacks the cytological and architectural features of invasive carcinoma ( Fig. 22.6 ). Mucin pools, when present in association with displaced epithelium, are usually either acellular or lined by dysplastic epithelium of a grade similar to that in the surface mucosal portion of the polyp. Furthermore, mucin pools are typically smooth and regular, and they are usually associated with extravasated mucin associated with ruptured crypts in the surface of the polyp. In contrast, the presence of irregular, small, jagged mucin pools dissecting through the submucosal stroma or the presence of mucin pools associated with cytologically malignant cells floating in the pools of mucin are features suggestive of adenocarcinoma.
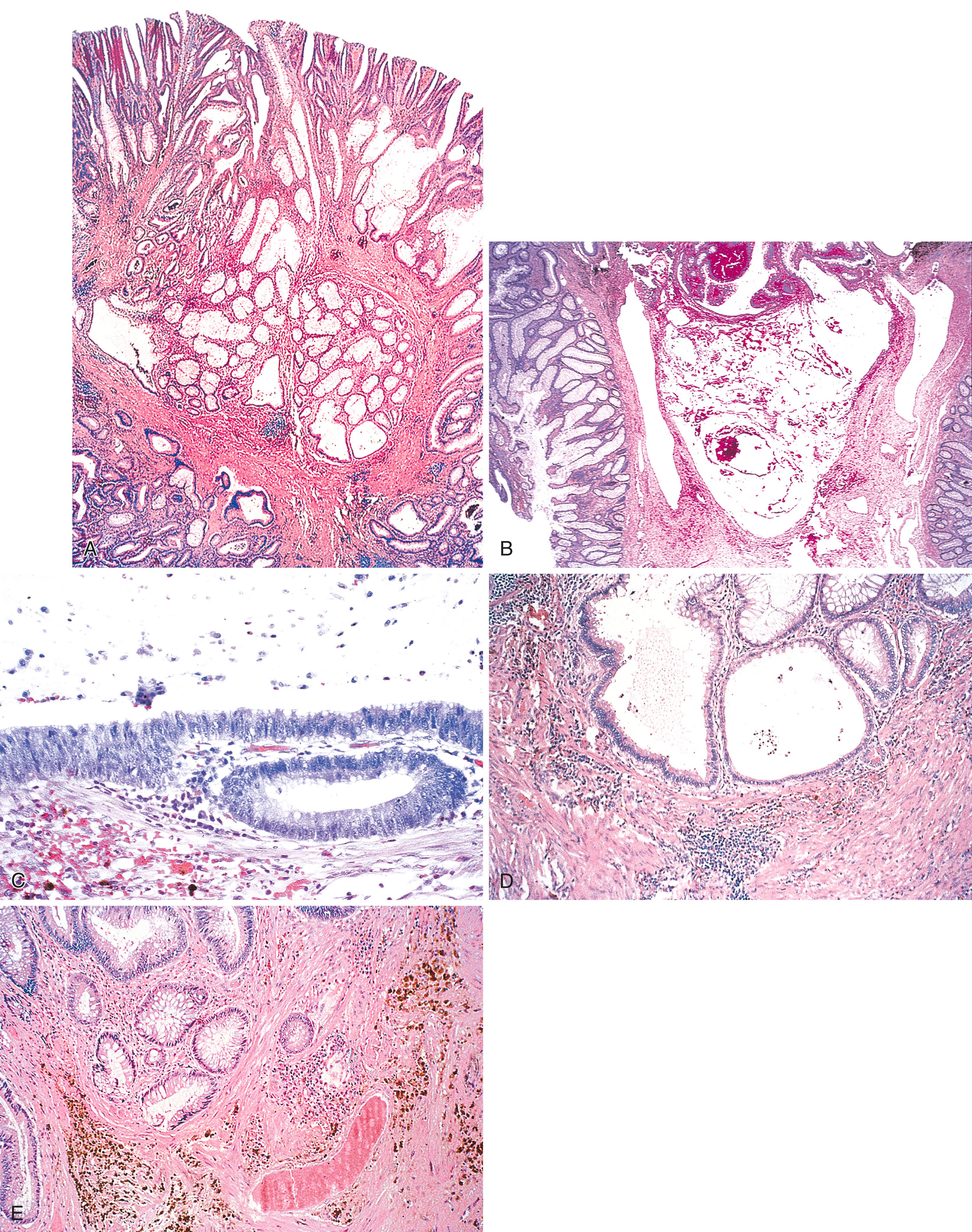
Table 22.1 outlines features of adenomas with displaced epithelium from invasive adenocarcinoma. Features suggestive of adenocarcinoma include (1) epithelium that contains a greater degree of cytological and architectural atypia than that of the intramucosal portion of the adenoma; (2) architectural complexity, such as the presence of irregular, tortuous, and jagged crypts; (3) back-to-back and cribriform crypt formation; (4) a nonlobular configuration of crypts; (5) single or small clusters of cells without a surrounding rim of lamina propria; (6) a desmoplastic reaction; and (7) the absence of hemorrhage or hemosiderin deposition in the stroma ( Fig. 22.7 ). Although displaced benign crypts usually demonstrate a communication of the intramucosal portion of the polyp on deeper sectioning, this feature may also occur in cases with adenocarcinoma, but it is more common in the former compared with the latter.
| Feature | Displaced Epithelium | Invasive Adenocarcinoma |
|---|---|---|
| Pedunculated shape | Usually present | Present or absent |
| Architecture | Round, smooth, lobular arrangement of crypts | Irregular, variably sized tortuous crypts, single cells, small clusters of cells |
| Crypts | Noncomplex | Complex, cribriform, budding |
| Mucin pools | Round, smooth, lined by dysplastic epithelium at periphery | Irregular, floating cells may be present |
| Hemorrhage/hemosiderin | Usually present | Usually absent |
| Desmoplasia | Absent | Usually present |
| Lamina propria around crypts | Usually present | Absent |
| Communication to surface | Often present | Present or absent |
| Degree of dysplasia | Similar to polyp surface | Carcinoma-like |

Adenomas with epithelial displacement should be excised in total, with confirmation that the mucosal and deep cauterized margins of the polyp are negative, similar to adenomas without epithelial displacement. For management purposes, pools of mucin associated with a rim of dysplastic epithelium at the deep resection margin should be considered “positive” for adenomatous epithelium. The significance of the presence of acellular mucin pools at the cauterized deep resection margin is unclear. As such, this should probably warrant close endoscopic follow-up. Cases in which a definite distinction from invasive adenocarcinoma cannot be made with complete certainty should probably be managed individually according to other clinical health factors (see Malignant Polyps). If invasion cannot be ruled out with certainty, but there are no unfavorable histological features, then polypectomy would be considered adequate treatment regardless. In almost all cases in which uncertainty regarding the presence of a small focus of invasive carcinoma exists, complete polypectomy will usually be sufficient because “unfavorable” histological features are unlikely to be present. However, in cases in which invasion cannot be ruled out with certainty, but the foci of uncertainty involve a deep or lateral resection margin, then surgical resection should be considered if the clinical circumstances support surgery.
Flat adenomas are defined as noninvasive dysplastic lesions without an intraluminal polypoid component (Paris 0-IIa, IIb, and IIc). They may be slightly raised at the edges, but typically they have a central depression. Some authorities define these as adenomas with a height (thickness) less than two times that of adjacent normal mucosa. Flat adenomas are associated with molecular abnormalities somewhat different from conventional polypoid adenomas. , The molecular phenotype is thought to be of a more aggressive nature than that of typical adenomas by some authorities, but this is highly controversial. Some reports cite an increased prevalence rate of high-grade dysplasia and a higher rate of progression to adenocarcinoma than conventional adenomas. , However, one large follow-up study of 474 flat adenomas from the National Polyp Study cohort found neither a higher rate of high-grade dysplasia nor an increased risk of advanced adenomas on follow-up, compared with either conventional sessile or pedunculated adenomatous polyps.
Of practical clinical importance, these lesions may also be more difficult to identify endoscopically. An accurate assessment of the prevalence rate and natural history of these lesions may depend on more extensive studies that utilize chromoendoscopy or high-resolution endoscopy, or both. Lynch et al. described two families with flat adenomatous polyposis. The lesions in these patients were predominantly right-sided, but they did not have typical features of familial adenomatous polyposis (FAP) or Lynch syndrome. Subsequent studies showed that patients with so-called hereditary flat adenoma syndrome actually have attenuated FAP (see later discussion). , A flat adenoma may result in the development of flat, or depressed, invasive adenocarcinoma without evidence of residual overlying or adjacent adenomatous epithelium. However, this phenomenon may also occur in large cancers that, upon growth, obliterate the overlying adenomatous epithelium.
FAP is an autosomal dominant condition caused by either inheritance of a mutated APC gene (located on chromosome 5q) or a new germline mutation in the same gene (up to one-third of cases). Adenomas then develop from loss of the second APC allele in colonic epithelial cells. APC is a tumor suppressor gene, and its absence allows for additional mutations in other genetic loci, such as in the KRAS and TP53 genes (see Chapter 23, Chapter 27 ). FAP patients develop large numbers of colorectal adenomas during late childhood, adolescence, and early adulthood. FAP has been traditionally defined by the presence of more than 100 adenomatous polyps in the colon, although many patients have several hundred or even thousands of polyps. Adenocarcinoma develops in most patients by their mid-30s, but it can occur as early as 17 years of age. FAP accounts for approximately 1% of all colon cancers.
The frequency of FAP is from 1 in 8000 to 1 in 14,000 in the general population, with equal sex representation worldwide. FAP patients may express a variety of extraintestinal phenotypes (see later discussion), but all include the presence of adenomatous polyps of the GI tract. They are predisposed to a high rate of adenocarcinoma. Upper GI tract adenomas and adenocarcinomas may also develop in FAP patients at a high frequency; they are particularly prominent in the first and second portions of the duodenum and in the periampullary region. Periampullary adenomas and adenocarcinomas have recently become the most common causes of morbidity and mortality in FAP patients who have undergone prophylactic colectomy. The prevalence of noncolonic adenomas in FAP ranges from 9% to 50% in the stomach and from 50% to 100% in the duodenum. Much less commonly, patients with FAP develop thyroid carcinoma, brain tumors (discussed later), pancreatic carcinoma, and hepatoblastoma.
Cyclooxygenase-2 (COX-2) inhibitors have received considerable attention lately as a method of decreasing polyp burden in FAP patients or reducing or eliminating rectal adenomas in patients who have had a subtotal colectomy. COX-2 inhibitors may also decrease the extent of duodenal polyposis in FAP patients. , Gardner’s syndrome, Turcot’s syndrome, and attenuated FAP are considered subtypes of FAP; these are discussed in the following sections ( Table 22.2 ).
| Syndrome | Colorectal Polyps | Extracolonic Lesions | Genetics | Risk of Malignancy |
|---|---|---|---|---|
| Familial adenomatous polyposis (FAP) | Adenomatous polyps (100s to 1000s) | Duodenal/periampullary adenomas, gastric fundic gland polyps, congenital hypertrophy of the retinal pigment epithelium, osteomas, dental abnormalities, desmoid tumors, brain tumors | Autosomal dominant APC mutations |
100% risk of colorectal carcinoma (mean age 35-40 years) 3%-5% risk of duodenal/periampullary carcinoma |
| Attenuated FAP | <100 adenomatous polyps | Similar to conventional FAP | Autosomal dominant APC mutations |
80% risk of colorectal carcinoma (mean age 50 years) |
| MUTYH -associated polyposis | Oligopolyposis with adenomas and serrated polyps | Uncertain | Autosomal recessive MUTYH mutations |
80% risk of colorectal carcinoma |
| Polymerase proofreading–associated polyposis | >10 adenomas | Uncertain | Autosomal dominant POLE and POLD1 mutations | 40%-60% risk of colorectal carcinoma |
| NTHL1 polyposis | >10 adenomas | Sebaceous skin lesions | Autosomal recessive NTHL1 mutations |
High risk of colorectal carcinoma |
| MSH3 -associated polyposis | >10 adenomas | Duodenal adenomas, gastric cancer, early-onset astrocytoma | Autosomal recessive MSH3 mutations | High risk of colorectal carcinoma |
| AXIN2 -associated polyposis | >10 adenomas | Ectodermal dysplasia | Autosomal dominant AXIN2 mutations |
High risk of colorectal carcinoma |
For most patients, the treatment of choice is screening during adolescence followed by a postadolescent prophylactic colectomy.
Desmoid fibromatosis affects 15% of patients with FAP and has been referred to as Gardner’s syndrome, although use of this term is no longer recommended by the World Health Organization (WHO). , , Desmoid tumors in these patients occur most commonly in the small bowel mesentery and abdominal wall. Surgical incisions can also increase the incidence of desmoid fibromatosis. Osteomas (particularly in the mandible, skull, and long bones), epidermoid cysts, dental abnormalities, and congenital hypertrophy of the retinal pigmented epithelium (CHRPE) can also occur. Overall, CHRPE affects 70% to 80% of FAP patients.
Central nervous system tumors can occur in association with both FAP and Lynch syndrome. This is referred to as Turcot’s syndrome, although this term is also no longer recommended by the WHO as many patients have been found to have constitutional mismatch repair deficiency. , , Although medulloblastomas are the most common primary central nervous system tumor in FAP patients, astrocytomas and ependymomas can also occur. In contrast, the most common primary central nervous system tumor in patients with Lynch syndrome and constitutional mismatch repair deficiency is glioblastoma.
The term attenuated FAP refers to a hereditary colon cancer syndrome with fewer than 100 colonic polyps (usually <30). , Many such patients, on closer endoscopic examination or with the assistance of methylene blue staining, are found to harbor more than 100 polyps, so these patients may, in fact, have conventional FAP. When patients are seen with a suspicious family history, or mild polyposis, or both, the diagnosis can be further investigated by assaying for mutations in the APC gene. In attenuated FAP, as opposed to classic FAP, APC mutations tend to occur at the most 5′ or 3′ aspect of the gene. , Once the mutations are discovered, these patients require close endoscopic screening for removal of polyps and prevention of adenocarcinoma development. In addition to being fewer in number, adenomas and adenocarcinomas develop at a later stage in life compared with classic FAP; the lifetime risk of colorectal carcinoma in attenuated FAP is approximately 80%. These adenomas, and adenocarcinomas, are otherwise of unremarkable morphology, although many of the adenomas in some patients with attenuated FAP are “flat” (see earlier discussion). Upper GI lesions and extraintestinal manifestations develop at a rate similar to that observed in classic FAP.
There is an alternative genetic mechanism for the attenuated FAP phenotype, one that is inherited in an autosomal recessive fashion (unlike the mutations that cause FAP). , In 2002, Al-Tassan et al. studied a family whose members harbored multiple colorectal adenomas and carcinomas, but lacked APC mutations. They discovered that affected patients carried biallelic mutations in the mutY DNA glycosylase gene (MUTYH) located on the short arm of chromosome 1, which encodes a base excision repair enzyme responsible for preventing mutations after oxidative DNA damage. Mutations in MUTYH results in G→T conversions throughout the genome. Other groups have detected homozygous or compound heterozygous MUTYH mutations in 15% to 40% of patients who have between 10 and 100 adenomas. , The highest frequency of MUTYH -associated polyposis is seen in patients with more than 30 adenomatous polyps, but who have no family history of polyposis. Patients with MUTYH -associated polyposis can also have multiple serrated polyps, suggesting some overlap with serrated polyposis. Similar to FAP and attenuated FAP, duodenal adenomas and adenocarcinomas occur in these patients. Sebaceous tumors have also been described in affected patients.
Two specific missense mutations (p.Y179C and p.G396D) account for most MUTYH -associated polyposis in northern Europeans. Other mutations are more common in southern Europeans (p.E480del), Pakistanis (p.Y104∗), and Indians (p.E480∗). One large population-based study of 2239 cases by Farrington et al. found a 93-fold increased risk of colorectal cancer compared with wild-type controls. The lifetime risk of colorectal carcinoma in affected patients is 80%, and the risk of duodenal carcinoma is 4%. The risk of cancer in heterozygous carriers of an MUTYH mutation remains unknown but is probably slightly increased. , ,
Recently, germline mutations in the exonuclease domain of POLE and POLD1 have been associated with a clinical phenotype that overlaps with FAP and attenuated FAP. , Similar to APC mutations, mutations in POLE and POLD1 are dominantly inherited. Patients present with oligo adenomatous polyposis and early-onset colorectal and endometrial cancer. The risk of colorectal carcinoma appears higher for POLD1 mutation carriers compared with POLE carriers; however, carriers of both mutations warrant colonoscopic surveillance. The cancers that develop in these patients are characterized by a hypermutated phenotype, and they are amenable to immunotherapy.
NTHL1 is a protein involved in base excision repair similar to MUTYH. Mutations in NTHL1 result in C→T transitions and predispose to adenomatous polyposis that presents in adulthood, generally by 50 years of age. , Endometrial carcinoma and sebaceous skin tumors are also seen, which results in overlap with both MUTYH -associated polyposis and Lynch syndrome. Other more rare polyposis syndromes include MSH3 -associated polyposis and AXIN2 -associated polyposis. MSH3 encodes the MMR gene that is involved in repairing di-, tri-, tetra-, and pentanucleotide repeats. Biallelic mutations in MSH3 result in multiple adenomas and colorectal carcinoma. Duodenal adenomas, gastric cancer, and early-onset astrocytomas have also been reported. AXIN2 regulates β-catenin degradation and functions in the same pathway as APC. Mutations in this gene result in colonic polyps, colorectal cancer, gastric polyps, and ectodermal dysplasia and is inherited in an autosomal dominant manner.
Lynch syndrome is an autosomal dominant condition caused by inherited defects in one of the DNA mismatch repair genes (MLH1, MSH2, MSH6, PMS2) or EPCAM , that lead to microsatellite instability and a rapid accumulation of somatic mutations in genes that control pathways of tumor progression (see Chapter 23, Chapter 27 ). Most mutations occur in the MLH1 and MSH 2 genes and are truncating. Lynch syndrome is the most common form of hereditary colon cancer, accounting for approximately 3% of all colon cancers (see Chapter 23, Chapter 27 ); universal screening of all colorectal carcinomas is recommended by numerous GI societies. The disorder is characterized by the development of colon cancer, often at an early age, and with predominance of right-sided cancers. Lynch syndrome patients do not usually develop an excess of polyps. However, rarely they can present with multiple adenomas. When adenomas do occur, they carry a significant risk of malignant degeneration. However, individuals with constitutional mismatch repair deficiency caused by biallelic mutations in MMR genes develop adenomas at a very young age. Constitutional mismatch repair deficiency also predisposes to a wide variety of other tumors including leukemia, lymphoma, and brain tumors.
The risk of colon cancer in Lynch syndrome varies widely according to the pathogenic MMR gene mutation. The risk is highest in those with MSH2 mutations (∼95%) and lowest in those with PMS2 mutations (∼10%). , Immunohistochemical staining of adenomas in patients with Lynch syndrome demonstrates loss of expression of the inherited faulty mismatch repair protein in 50% to 79% of adenomas and is influenced by the adenoma size and the presence of high-grade dysplasia. Defective DNA mismatch repair functionality suggests that the progression from adenoma to carcinoma could be quite rapid in these patients. It has also been suggested that Lynch syndrome is associated with a higher proportion of flat adenomas. Patients with Lynch syndrome also have a ∼15% lifetime risk of small-intestinal carcinoma; the duodenum and the jejunum are the most common sites. ,
The histological features of colorectal carcinomas in Lynch syndrome are distinctive, similar to those of sporadic microsatellite instability-high (MSI-H) carcinomas (see also Chapter 23, Chapter 27 ). Sixty percent occur in the proximal colon. Compared with microsatellite stable colorectal carcinomas, MSI-H carcinomas more commonly have a mucinous or signet ring cell component, a microglandular or medullary growth pattern, and an expansile (“pushing”) margin. These tumors are often poorly differentiated and characteristically show prominent tumor-infiltrating lymphocytes. A peritumoral lymphoid or Crohn’s-like response is often seen at the leading edges of the tumors.
Recognized by their bland cytological features and classic serrated architecture, serrated polyps were historically considered non-neoplastic and without malignant potential. However, it is now well-recognized that some types of serrated polyps may, in fact, progress to adenocarcinoma. , Recognition of the potential neoplastic nature of these lesions has led to identification and characterization of the serrated pathway of carcinogenesis, which is discussed further in Chapter 27 . Box 22.2 describes the classification of serrated polyps. The serrated neoplasia pathway describes a morphological progression that begins in nondysplastic serrated polyps and terminates with the development of carcinomas that have morphological, biological, and clinical characteristics that differ from carcinomas that develop in the conventional adenoma-carcinoma pathway.
Nondysplastic serrated polyps
Normal architecture, normal basilar crypt proliferation
Microvesicular hyperplastic polyp
Goblet cell hyperplastic polyp
Abnormal architecture, abnormal proliferation along the length of the crypts
Sessile serrated polyp (or sessile serrated lesion)
Dysplastic serrated polyps
Sessile serrated polyp (or sessile serrated lesion) with dysplasia
Traditional serrated adenoma
Serrated adenoma unclassified
Unclassifiable serrated polyps (with or without dysplasia)
Table 22.3 outlines the histological and molecular features of serrated colorectal polyps. Two principle morphological subtypes of “hyperplastic polyps” are wellrecognized. These are termed microvesicular polyps and goblet cell hyperplastic polyps . From a clinical point of view, it is not yet considered mandatory or even recommended to distinguish these two variants in diagnostic reports. Rather, it is considered clinically important to recognize and report goblet cell hyperplastic polyps, which can have very subtle serrated features, to distinguish them from normal mucosa and from other types of serrated polyps. They can be difficult to distinguish from normal epithelium if one does not look carefully at the crypts for evidence of numerous goblet cells and a sawtooth surface contour. The microvesicular variant of hyperplastic polyp is the most common. A third type of hyperplastic polyp (mucin-poor hyperplastic polyp) was initially described by Torlakovic et al. However, this lesion should not necessarily be regarded as a distinct entity because little is known of its biological nature, and most authorities believe that it likely represents a mucin-depleted variant of a microvesicular hyperplastic polyp, perhaps as a result of tissue injury.
| Histological Features | Molecular Features | ||||||
|---|---|---|---|---|---|---|---|
| Type | Crypt Architecture | Proliferation Zone | Cytological Features | Mucin Type | BRAF Mutation | KRAS Mutation | CpG Island Methylation |
| Microvesicular hyperplastic polyp | Funnel-shaped crypts with serrations limited to upper two-thirds | Located uniformly in the basal portion of crypts | Small basally located nuclei, no dysplasia | Mixed micro-vesicular and goblet cell | 70%-80% | 0% | + |
| Goblet cell hyperplastic polyp | Elongated crypts that resemble enlarged normal crypts; little to no serrations | Located uniformly in the basal portion of crypts | Small basally located nuclei, no dysplasia | Goblet cell only | 0% | 50% | − |
| Sessile serrated lesion | Horizontal growth along the muscularis mucosae, dilation (often asymmetric) of the crypt base (basal third of the crypt), and/or serrations extending into the crypt base | Proliferation may be abnormally located away from the crypt base, variable from crypt to crypt | Small basally located nuclei with occasional larger nuclei with inconspicuous nucleoli, no dysplasia | Mixed micro-vesicular and goblet cell | >90% | 0%-5% | ++ |
| Sessile serrated lesion with dysplasia | As for sessile serrated lesion | As for sessile serrated lesion with more proliferation in dysplastic component | Varied morphological appearance to dysplastic component | Varied type | >90% | 0% | +++ |
| Traditional serrated adenoma | Slitlike serrations, often ectopic crypt foci | Present within ectopic crypt foci and crypt base | Elongated pencillate nuclei with nuclear stratification and cytoplasmic eosinophilia; may develop overt (conventional or serrated) dysplasia | Occasional scattered goblet cells; rare goblet cell variant has been described | 20%-40% | 50%-70% | BRAF mutated ++ KRAS mutated + |
| Serrated adenoma–unclassified | Varied | Varied | Unequivocal dysplasia must be present | Varied | Uncertain | Uncertain | Uncertain |
Torlakovic and Snover first recognized an unusual type of serrated polyp that occurred mainly in patients with serrated polyposis. In 2003, Torlakovic et al. provided the first description of serrated polyps in patients without any known polyposis syndrome. Since 2003, this polyp has been termed sessile serrated adenoma, sessile serrated polyp, sessile serrated adenoma/polyp, and, more recently, sessile serrated lesion (SSL) by the most recent WHO publication. The original argument for including the term adenoma in its description was to emphasize clinically that these lesions are in fact valid precursors of colorectal carcinoma. However, over the years, use of the term adenoma has resulted in much confusion among clinicians because they have the tendency to associate adenomas with dysplastic epithelium, which of course most SSLs do not contain. In fact, these polyps have significant differences in their natural history and molecular phenotype compared with conventional adenomas. If dysplasia is present within an SSL, the WHO recommends the term SSL with dysplasia. Thus the term cytological dysplasia has been dropped in the current edition of the WHO classification of serrated polyps.
Traditional serrated adenoma (TSA) remains the recommended diagnostic term to be used for “serrated adenoma” of the type reported by Longacre and Fenoglio-Preiser in 1990. We have only recently begun to appreciate the wide diversity of molecular and morphological alterations that can occur in these particular lesions (see later). Finally, over the past several years, new and unusual types of dysplastic serrated polyps that do necessarily fulfill the precise diagnostic criteria of a typical TSA or an SSL with dysplasia have been increasingly described. In this setting, the WHO recommends use of the generic term serrated adenoma unclassified for unequivocally dysplastic serrated lesions that do not fit neatly into one of the above two discrete diagnostic categories. Another acceptable term for polyps that do not fit into a specific category is serrated lesion with dysplasia, unclassified, although this is not specifically endorsed by the WHO. Regardless, both terms convey the essential feature, which is the presence of a serrated polyp of some kind with dysplasia. Ultimately, knowledge of the presence or absence of dysplasia is important for clinicians to be aware of with regard to assurance of completeness of resection and determination of frequency of future surveillance.
Elucidation of the molecular alterations in serrated polyps has been critical in our understanding the neoplastic progression in the serrated pathway of carcinogenesis ( Fig. 22.8 ). The molecular hallmarks of the serrated pathway include activating mutations involving a component of the RAS-RAF-MAP-kinase signaling pathway as well as methylation of CpG DNA islands. Both microvesicular hyperplastic polyps and SSL commonly demonstrate activating mutations in BRAF. , CpG island DNA methylation is uncommon in hyperplastic polyps but occurs in up to 50% of SSLs. , Given the histological and molecular overlap between hyperplastic polyps and SSLs, it is likely that SSLs arise from microvesicular hyperplastic polyps, although this still remains a matter of debate. In contrast with SSLs and microvesicular hyperplastic polyps, goblet cell hyperplastic polyps often demonstrate KRAS mutations.

The development of dysplasia in SSLs is frequently accompanied by methylation-induced silencing of the MLH1 gene. Silencing of MLH1 results in microsatellite instability and accumulation of mutations in a wide variety of genes, including CDKN2A and TP53 . Activation of the WNT pathway also occurs, but this often occurs late in neoplastic progression, in contrast with the conventional adenoma-carcinoma pathway, where aberrant WNT signaling happens early. ,
TSAs have more complex molecular alterations. They are molecularly heterogeneous. Most TSAs also harbor alterations in the RAS-RAF-MAP-kinase signaling pathway. Approximately 50% have a BRAF mutation, and 30% have a KRAS mutation. More extensive CpG island methylation is seen in BRAF -mutated TSAs. In addition, many of the BRAF- mutated TSAs arise in association with SSL-like and/or microvesicular hyperplastic changes in the surrounding mucosa. The origins of the KRAS -mutated cases are less clear, but they also may arise in association with a precursor lesion with features of a goblet cell hyperplastic polyp. , Additional mutations in TSAs include frequent RSPO fusion transcripts in KRAS -mutated TSAs and frequent RNF43 mutations in BRAF -mutated TSAs. , These molecular alterations tend to occur at the transition from precursor polyp to TSA. The development of high-grade dysplasia and invasive carcinoma in TSAs is often accompanied by loss of p16 expression and mutations in TP53 . In contrast with SSLs with dysplasia, the progression from TSA to invasive carcinoma is never accompanied by MLH1 inactivation.
Hyperplastic polyps are considered serrated polyps with normal architecture and normal hierarchical proliferative characteristics. They are small, innocuous lesions that may be found throughout the colon of adults, but they are especially common in the rectum. Up to 90% occur in the left colon. Their prevalence rate increases with age. Up to 35% of asymptomatic individuals older than 50 years of age demonstrate hyperplastic polyps. Specific lifestyle and dietary factors commonly associated with conventional adenomas are also associated with hyperplastic polyps. These include cigarette smoking, alcohol consumption, low folate intake, and obesity. , These lesions are typically asymptomatic and are often identified as an incidental finding during endoscopic examination of the colon. Endoscopically, hyperplastic polyps are small, sessile, smooth bumps or nodules that appear pale and often flatten on insufflation of air.
Microvesicular hyperplastic polyps are usually small or diminutive sessile polyps. Endoscopically, they can often have a stellate pit pattern, as described by Kudo et al. Histologically, they are composed of serrated epithelium with funnel-shaped, evenly spaced crypts and with proliferative zones confined to the crypt bases ( Fig. 22.9 ). Nuclear atypia is minimal, but some polyps show a mild degree of nuclear stratification at the base of the polyp, which should not be confused with a conventional adenoma in poorly oriented tissue sections. Dystrophic goblet cells and cells with round vesicular nuclei and prominent nucleoli are rare; if present, they are usually confined to the lower portions of the crypts. Most notably, these lesions show distinct evidence of surface maturation and an absence of cells with eosinophilic cytoplasm characteristic of SSLs and TSAs (see later discussion). The cells lining the crypts consist mainly of goblet cells and cells with abundant fine apical vacuoles containing microvesicular mucin (see Fig. 22.9 ). The nuclei in the cells in the surface epithelium are basally located and quite small in size. Importantly, the luminal epithelial serrations are usually limited to the upper one-half to two-thirds of the crypt in contrast with SSL (see later). When cut in cross section, the serrated crypts of these polyps have a uniform stellate appearance.

Some polyps also show thickening, and even extension, of the muscularis mucosae into the lamina propria. Occasionally, these prolapse-type changes can be quite striking and result in distortion of the serrated crypts, mimicking an SSL. In addition, larger-sized microvesicular hyperplastic polyps (>0.5 cm) may be slightly distorted and even show mild, often symmetric crypt dilation. In this instance, care should be taken to distinguish these changes from an SSL (see latter discussion). Occasional reports of inverted hyperplastic polyps exist in the literature. However, it is now apparent that an inverted growth phenomenon, wherein serrated crypts are present within the submucosa, occurs almost exclusively in SSLs. , It is similarly unclear how these lesions differ, if at all, from SSL with epithelial displacement.
Goblet cell hyperplastic polyps are the second most common type of hyperplastic polyp. They are typically small (<0.5 cm) sessile lesions and are commonly found in the left colon, but they can certainly be present in the right colon as well. Goblet cell hyperplastic polyps are often overlooked because their morphological alterations are usually quite subtle. These polyps show elongated crypts that are rich in goblet cells without microvesicular mucin ( Fig. 22.10 ). They are normally sessile lesions but demonstrate a far lesser degree of luminal serration than microvesicular hyperplastic polyps. The luminal section is usually limited only to the surface and uppermost portions of the crypts. The crypts may be mildly elongated as well. Nuclear atypia, stratification, and mitoses are not seen in this type of hyperplastic polyp. In these polyps, the crypts are taller and wider than normal. Occasional branching or tortuosity may occur, but this is rare.

Until recently, small hyperplastic polyps were not believed to require definitive treatment, although they are typically removed in the process of endoscopy with biopsy. However, the biological characteristics and natural history of hyperplastic polyps, particularly the microvesicular type, are now considered controversial. Large lesions, particularly those located in the right colon, should be removed in total. , Small proximal hyperplastic polyps may carry the same risk of synchronous advanced neoplasia as similarly sized SSLs, although the risk of metachronous neoplasia is unclear. Further studies are needed to determine the natural history and malignant potential of hyperplastic polyps and each of the different subtypes. There is a strong correlation between the occurrence of hyperplastic polyps and conventional adenomas in similar populations worldwide. Although some studies suggest that distally located hyperplastic polyps predict an increased risk of proximal adenomas or even advanced adenomas, this is controversial. Thus at present, the finding of a distal hyperplastic polyp at sigmoidoscopy is not considered to be an absolute indication for full colonoscopy. Furthermore, surveillance for detection of metachronous hyperplastic polyps, or adenomas, after removal of a hyperplastic polyp is not currently recommended.
In contrast with hyperplastic polyps, SSLs are usually larger in size (>5 mm) and occur more often in the proximal colon (75%). , The prevalence of SSLs varies according to endoscopist experience and has been found in up to 20% of individuals undergoing screening colonoscopy by skilled endoscopists. SSLs are associated with female sex, smoking, high body mass index, and alcohol intake. , SSLs can be difficult to identify at endoscopy. However, awareness and specific training can improve detection. They often appear as ill-defined, sessile, pale lesions with an irregular shape and a “cloudlike” surface ( Fig. 22.11 ). , These polyps are often covered by adherent mucin, and a rim of bubbles or fecal debris can sometimes collect at the periphery of these lesions. They have a stellate configuration (Kudo pit pattern II) and appear as superficially elevated lesions (Paris classification 0-IIa). , , Incomplete resection is common in SSLs given the lack of a sharp demarcation between the SSL and adjacent normal colonic mucosa.

Morphologically, SSLs show distinctive features, characterized more specifically by the presence of asymmetric crypt dilation, crypt irregularity (horizontally shaped crypts), prominent and often exaggerated lower (and upper) crypt serration, mitoses in the upper levels of the crypts, vesicular nuclei in the upper crypts, reduced amounts of lamina propria between crypts, hypermucinous epithelium, and, occasionally, an inverted (epithelial displacement) growth pattern ( Fig. 22.12 ; see Table 22.3 ). Rows of cells with eosinophilic (“pink”) cytoplasm are not uncommon. These may represent “senescent” cells or possibly early dysplastic cells, but this topic is controversial. The basal portions of the crypts are usually branched and may appear flask- or boot-shaped (similar to an inverted T ), indicative of horizontal rather than vertical growth. An expert panel of pathologists suggested that the presence of one or more unequivocally architecturally distorted crypts of this kind situated at the base of the lesion is enough evidence to establish a diagnosis of an SSL rather than a hyperplastic polyp in the appropriate clinical and endoscopic setting. This definition has been adopted in the current WHO classification. Mature goblet cells, or mucinous cells, show an irregular distribution by being uncharacteristically located at the bases of the crypts. Some polyps also contain perineurial-like proliferations in the lamina propria.

SSLs may show a variable degree of nuclear atypia. At the low end of the spectrum, lesions show little or no stratification, a low mitotic rate, and clear evidence of surface maturation. SSLs may occasionally show an increased degree of nuclear stratification, consisting of cells with open vesicular chromatin and small nucleoli as well as increased eosinophilia of the cytoplasm. These features approach those of a traditional serrated adenoma, but the cytological changes are often focal and present only in the surface epithelium, not along the entire length of the crypts. This focal eosinophilic change should not be considered indicative of a TSA or an SSL with dysplasia.
It is important to emphasize that the size, location, number, and endoscopic appearance are not part of the pathological diagnosis of a hyperplastic polyp or an SSL. Using these parameters can hamper future efforts to refine the risk profile of patients with serrated polyps, especially in scenarios such as small proximal hyperplastic polyps or large distal hyperplastic polyps. Only a proper morphological classification of serrated polyps will allow for refinement of the risk of metachronous neoplasia in these patients. Results from such analyses can then be used to modify the diagnostic criteria and include other features for the diagnosis if necessary. From a practical perspective, it is extremely rare to encounter a microvesicular hyperplastic polyp >9 mm, and such polyps should be considered advanced serrated polyps for the purpose of subsequent surveillance intervals. The main dilemma occurs when encountering small or diminutive proximal serrated polyps that morphologically are best classified as a microvesicular hyperplastic polyp (see Fig. 22.8 ). Furthermore, goblet cell hyperplastic polyps are not infrequently seen in the proximal colon. Despite the lack of diagnostic features, there has been a tendency to consider all proximal serrated polyps as SSLs because of the presumption that hyperplastic polyps do not exist in the proximal colon. Such a diagnostic strategy will lead to an increased frequency of colonoscopy for these patients and an increased burden on surveillance programs. In rare cases of polyps with marginal histological features, size and location can be used to tip the diagnosis one way or another.
With histological and molecular progression (often characterized by loss of MLH1 immunostaining and microsatellite instability), SSLs may acquire morphological evidence of dysplasia (see Table 22.3 ). , , SSL with dysplasia represents the most “advanced” type of serrated polyp, and these lesions can progress fairly rapidly to invasive carcinoma (see later discussion). These lesions have been historically referred to as mixed hyperplastic/adenomatous polyps. However, it is now well recognized that these polyps do not occur as a result of a coincidental growth of hyperplastic and adenomatous elements, but as a result of dysplastic change within an SSL. Morphologically, these lesions usually show discrete areas of a typical SSL with dysplasia ( Fig. 22.13 ). Occasionally, background SSL-type crypts may not be apparent in polyps that contain a large dysplastic component, and thus have “overgrown” their SSL precursors. In such cases, the presence of any convincing nondysplastic serrated crypts is considered sufficient to diagnose an SSL with dysplasia.

The morphological heterogeneity of dysplasia seen in SSLs has only recently been described. Two general types occur: serrated dysplasia and intestinal-type dysplasia . However, until more follow-up and natural history data are obtained, it is not necessary to grade or morphologically subclassify the type of dysplasia for clinical purposes. Rather, it is more prudent to recognize the various types of dysplasia seen in these polyps to ensure an accurate diagnosis and for future studies. Most commonly, dysplasia is intestinal and characterized by the presence of elongated, cigar-shaped hyperchromatic nuclei that resemble low-grade dysplasia in conventional adenomas. Architecturally there is often increased complexity characterized by crypt branching, crypt elongation, and a villous and/or cribriform architecture. The dysplasia frequently involves the full thickness of the mucosa and is usually associated with loss of MLH1 expression, although frequencies vary. , Serrated dysplasia is less common. SSL with serrated dysplasia shows architectural complexity and tightly packed glands composed of epithelial cells with prominent cytoplasmic eosinophilia, luminal serration, enlarged nuclei, and prominent nucleoli (see later; see Fig. 22.13 ). MLH1 is normally preserved in this type of dysplasia. Polyps composed of a mixture of these two types of dysplasia is also common. Rarely, SSLs can show very subtle cytological and architectural abnormalities, often with glandular crowding, mild nuclear enlargement, a slight change in mucin content, and a slight change in serrations termed minimal deviation dysplasia. This is also often associated with loss of MLH1. However, because little is known regarding the biology and natural history of such lesions, more study is needed before this becomes a diagnostic entity. Occasionally dysplasia closely resembles that of a conventional adenoma with typical basophilic cytoplasm, penicillate nuclei, and straight crypts without significant serrations and architectural complexity. The dysplasia is often limited to the surface of the polyp. MLH1 is preserved in this pattern. Recent studies have suggested that such lesions may represent a collision between a conventional adenoma and an SSL given the differences in BRAF mutations between the dysplastic component and adjacent SSL.
The natural history and risk of progression to malignancy of SSLs are poorly understood but are under intense investigation. The mean age of patients with SSLs with dysplasia is similar to SSLs that harbor invasive carcinoma, suggesting that once dysplasia occurs in an SSL, progression to invasive adenocarcinoma occurs fairly rapidly. It is commonly believed that loss of DNA repair ability and subsequent microsatellite instability are mechanisms that drive rapid neoplastic progression. Although SSLs that harbor dysplasia progress rapidly to carcinoma, the acquisition of dysplasia in SSLs takes years to occur. , , In one study of 55 patients with SSL who were monitored for a mean of 7 years, colorectal cancer or high-grade dysplasia developed in 15% of patients. In other studies, patients with SSL were found to be at increased risk for synchronous advanced neoplasia and interval adenomas during surveillance. One rational proposal for the management of SSL is based on the fact that most traditional hyperplastic polyps (goblet cell or microvesicular type) are unlikely to progress to carcinoma, and that SSLs without dysplasia are probably slow, but progressive, lesions. Table 22.4 provides a summary of the recommendations from the U.S. Multi-Society Task Force on Colorectal Cancer, British Society of Gastroenterology, and Cancer Council Australia.
| Organization | Serrated Polyps Only | Serrated Polyps and Adenomas |
|---|---|---|
| US Multi-Society Task Force on Colorectal Cancer | 10 years for:
5-10 years for:
3-5 years for:
3 years for:
6 months for:
|
No specific recommendations because of low quality of evidence (see Box 22.1 for adenoma surveillance recommendations) |
| British Society of Gastroenterology | 3 years:
|
3 years:
|
| Cancer Council of Australia | 10 years:
5 years for:
3 years for:
1 year for:
|
Clinically significant serrated polyps and synchronous conventional adenomas 5 years for:
3 years for:
1 year for:
Synchronous high-risk conventional adenoma (tubulovillous or villous adenoma, with or without HGD and with or without size ≥10 mm)
1 year for:
|
For SSLs without dysplasia, complete endoscopic removal is recommended. , However, if this is not possible, then repeat endoscopy with biopsies should be performed within 1 year to evaluate for signs of dysplasia, and this should be followed by continued surveillance at shorter intervals until the lesion is completely removed. Surgical excision for large SSLs without dysplasia, particularly those that are recurrent, is also a reasonable alternative, although this is becoming less common with the increased use of advanced endoscopic techniques. In contrast, complete excision is considered mandatory for SSLs with dysplasia based on the likelihood that these lesions have undergone hypermethylation and acquired microsatellite instability and therefore are prone to carcinoma progression. Complete excision should be accomplished by endoscopy or surgical resection. If removed by piecemeal polypectomy, patients usually undergo repeat endoscopy within 6 months to 1 year to ensure that the lesion has been removed in its entirety and that there has been no progression.
Due to heterogeneity in the morphology, molecular characteristics, and frequency of association with non-neoplastic serrated or “hyperplastic” precursor polyps, the clinical and epidemiological features of these polyps are poorly understood. Nevertheless, TSAs are relatively uncommon, representing less than 1% to 2% of all colonic polyps in most studies. In one Japanese study, TSAs accounted for only 1.8% of more than 10,000 colonic polyps. Endoscopically, TSAs manifest variable characteristics, but they are more often pedunculated than sessile. In one study, 63% of TSAs were pedunculated, 29% were sessile, and 8% were described as flat or carpetlike. They may occasionally be rather large and filiform in contour. In one study, “filiform” TSAs were exclusively found in the rectum.
Overall, TSAs are more common in females and occur more commonly in the left colon, particularly the sigmoid colon and rectum. Right-sided TSAs, however, do occur, and these may be flat rather than polypoid. The mean age at diagnosis is 60 to 65 years of age, which is typically older than for patients with hyperplastic polyps or SSLs. As mentioned previously, TSAs are molecularly heterogenous: there are BRAF -mutated and KRAS -mutated types. BRAF -mutated TSAs more commonly are associated with a precursor hyperplastic polyp or SSL and occur in the right colon in a higher percentage of cases than the KRAS -mutated type. It is based on these observations that some authorities have suggested that these lesions, in particular, may reflect development of TSA-like dysplasia in an SSL, but this topic remains highly controversial. Regardless of the terminology used for these lesions, the treatment ultimately is the same because both are considered, generically, serrated lesions with dysplasia. In fact, it is not currently recommended to diagnose a “TSA arising in an SSL” as diagnostic of an “SSL with TSA-like dysplasia.” SSLs with dysplasia are considered advanced lesions that often demonstrate microsatellite instability and CpG island DNA hypermethylation, and they can rapidly progress to carcinoma. In contrast, TSAs typically progress more slowly, have distinctive morphology, and do not normally develop microsatellite instability. Thus future studies are needed to elucidate the true relationship and potential biological continuum between SSLs and TSAs.
The origin of KRAS -mutated TSAs is less clear, although goblet cell hyperplastic polyp-like changes can, occasionally, be found at the periphery of these lesions. Of course, this suggests a possible progression pathway of goblet cell hyperplastic polyp to TSA, but further research is needed in this regard.
Most TSAs are composed of epithelial cells with abundant and intensely eosinophilic cytoplasm and bland oval-shaped palisaded nuclei, usually without significant mitotic activity ( Fig. 22.14 ; see Table 22.3 ). , , The second and probably most typical feature of TSA is the presence of distinctive slitlike serrations, similar to the narrow slits present in normal small-intestinal epithelium. , , , , The association of both the traditional serrated adenoma cytology and slitlike serrations makes the diagnosis of traditional serrated adenoma straightforward. A third characteristic feature is the presence of ectopic crypt foci. Ectopic crypt foci are small buds of epithelial cells resembling the bases of normal crypts that are not anchored to the muscularis mucosae, but instead are situated perpendicular to the villous projections of the polyp. Ectopic crypt foci are not specific for TSAs. They occur in other polyps, such as conventional adenomas, and as such may simply represent small zones of proliferation in polyps with a villiform growth pattern. , Interestingly, flat TSA often lack ectopic crypt foci. Some TSAs with typical slitlike serration and at least focal ectopic crypt foci may show a predominance of goblet cells (mucin-rich variant) rather than the characteristic eosinophilic cells with abundant cytoplasm. ,

Architecturally, TSAs are often protuberant and villous, but they can grow as flat, superficially elevated lesions as well. Filiform traditional serrated adenomas show elongated, finger-like villous projections and often show inflammation, ulceration, and dilated lymphatics within the lamina propria. Up to 50% of TSAs reveal an adjacent area of nondysplastic serrated precursor, either a hyperplastic polyp or SSL. Distal TSAs may show an adjacent “shoulder” area of flat growth at the edge of the protuberant polyp. In a recent study, small (<10 mm) polyps resembling these shoulder areas were shown to be early forms of TSA that can be recognized by subtle superficial serrations and cytoplasmic eosinophilia. ,
Despite being a polyp with malignant potential, most TSAs do not reveal the classic cytological characteristics of “dysplasia” because they lack significant nuclear hyperchromasia and mitotic activity outside of the areas of ectopic crypt foci. It is controversial among experts whether the “baseline atypia” of TSAs represents a very low grade of dysplasia (because molecular aberrations are common in these lesions) versus truly non-dysplastic “senescent epithelium” (based on the absence of significant cytological atypia). Regardless of the term used to describe the cytological features of TSAs without traditional high-grade dysplasia, TSAs are nonetheless considered premalignant polyps because most, if not all, will eventually progress through various stages of overt (conventional) dysplasia to cancer if left untreated. Overt (conventional) dysplasia does develop in TSAs with progression. The dysplasia in TSAs can be either intestinal type, resembling dysplasia seen in conventional adenomas, or serrated type, characterized by tightly packed serrated glands composed of epithelial cells with prominent cytoplasmic eosinophilia, enlarged nuclei, and prominent nucleoli (see Fig. 22.14 ). , Both high-grade intestinal and serrated dysplasia should be reported when present because this indicates a more advanced stage of the lesion. However, it is also unclear whether “advanced” TSAs, as such, portend any increased risk of further synchronous or metachronous neoplasia or risk of cancer after excision compared with conventional TSAs without high-grade dysplasia. Furthermore, it is similarly unclear whether the risk of progression is affected by the type (intestinal vs. serrated) or extent of dysplasia in the polyp.
The natural history of TSA and the risk of progression to malignancy are poorly understood given its infrequent occurrence as an isolated polyp. , Most TSAs occur synchronously with other polyps. An additional complication is the occasional lack of use of consistent terminology, as mentioned earlier. For instance, the terms TSA with associated SSL versus SSL with TSA-like dysplasia when the lesion occurs on a background of SSL or a hyperplastic polyp. Thus evaluation or progression has been challenging. Nevertheless, progression to high-grade dysplasia was reported in 37% of TSAs in one study. In this study, 11% contained intramucosal adenocarcinoma. In another study of filiform TSAs by Yantiss et al., 22% showed high-grade dysplasia, and another 6% showed invasive adenocarcinoma. Some studies have suggested that the rate of malignant transformation in TSAs is similar to that of conventional adenomas. , It is likely that the risk of progression is related to the size and location of the lesion. Large TSAs in the proximal colon may progress at a more rapid rate than those in the left colon.
Prior studies suggesting a high risk of concurrent colorectal carcinoma and metachronous adenomas in patients with a baseline TSA were limited by a small number of patients. , More recently, in a relatively large study of Korean patients with TSAs, the risk of metachronous serrated polyps, conventional adenomas, and high-risk adenomas was higher than a control population with only baseline conventional adenomas. A recent population-based study from Denmark also showed that patients with TSA were nearly five times more likely to develop colorectal carcinoma on follow-up compared with those without TSA. The treatment of TSAs is complete endoscopic removal. The surveillance interval is similar to that for patients with conventional adenomas (i.e., every 3 to 5 years).
Recently, unusual types of dysplastic serrated polyps have been identified that do not fit neatly into a discrete diagnostic category, such as a TSA or SSL with dysplasia. In fact, some tubular, tubulovillous, or villous adenomas may show areas of architectural serration, but these polyps can be distinguished from TSAs most easily by the characteristics of the cytological features of the nuclei ( Fig. 22.15 ). In one study, re-review of 180 distal villous and tubulovillous adenomas resulted in reclassification of 20 polyps as TSA. , However, some of these so-called serrated tubulovillous and villous adenomas also demonstrated molecular features intermediate between a TSA and conventional tubulovillous adenoma. It is unclear why some conventional adenomas acquire a serrated growth pattern, but this may be related to the acquisition of KRAS mutations, which has been proposed to add a “serrated” molecular signature to traditional adenomas, and provides evidence of a “fusion” molecular pathway of carcinogenesis in some cases. Some of these polyps may represent TSAs overgrown by conventional intestinal-type dysplasia. Recently, a superficial serrated adenoma has been described that consists predominantly of straight adenomatous glands with only superficial serrations. These polyps lack eosinophilic cytoplasm, slitlike serrations, and ectopic crypt foci of TSA. They have been demonstrated to harbor KRAS mutations and RPSO fusions similar to TSAs. Some of these likely represent early forms of TSA but lack the features needed to a definitive diagnosis. Finally, some SSLs with small areas of superficial eosinophilic cells can be confusing to classify in some cases. These SSLs have been regarded as having either low-grade serrated dysplasia, features of TSA, or enteric metaplasia by various authors. These changes can be differentiated from TSA arising from an SSL caused by involvement of the deeper portions of the serrated crypts in TSA compared with SSLs with focal eosinophilic change. However, given the uncertainties in the classification of these unusual serrated lesions, the term serrated adenoma, unclassified is proposed by the recent WHO.

Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here