Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Preterm neonates are exposed to frequent painful and agitating stimuli during intensive care. Pain and agitation have a negative impact on long-term outcome.
Laboratory tests and invasive procedures should be used judiciously in neonates.
A systematic scoring system should be used prior to, during, and after acute painful procedures.
Neonatal intensive care units should develop an algorithm for the treatment of pain, including standardized nonpharmacologic and pharmacologic interventions.
Nonpharmacologic interventions such as facilitated tucking should be consistently used prior to mild to moderately painful procedures.
Sucrose may be used to mitigate behavioral responses to minor painful procedures; clinicians can safely use the lowest effective doses ≤10 times every 24 hours.
Rapidly acting opioids with short durations of action (fentanyl or remifentanil) should be administered before moderately painful procedures, including nonemergency intubations. Benzodiazepines provide anxiolysis but may cause hypotension in preterm neonates.
Continuous multimodal analgesia and sedation (such as morphine/fentanyl and dexmedetomidine) should be used to promote ventilator synchrony and minimize oxygen consumption in ventilated term neonates. These agents may be avoided for short durations of mechanical ventilation.
Currently, no pharmacologic therapy has shown benefit in preterm neonates requiring prolonged, invasive mechanical ventilation. Low-dose morphine may be used selectively on the basis of clinical judgment. In the setting of insufficient sedation from low-dose morphine infusion, dexmedetomidine infusion may be considered, titrated carefully to effect while monitoring closely for adverse reactions.
Optimal and compassionate neonatal care demands the prevention and treatment of acute pain and chronic agitation. The traditional definition of pain relies on self-report, presenting challenges in nonverbal populations including neonates. Late into the 20th century, preterm neonates underwent major surgical procedures without perioperative or postoperative analgesia. However, extensive research has elucidated the developmental physiology of nociception, documenting completion of ascending pathways connecting peripheral sensory neurons to the thalamus between 20 and 24 weeks of gestation, which is at or below lower limits of viability. Additionally, clinical research clearly documents the unique susceptibility of preterm neonates to adverse metabolic, behavioral, and clinical effects from acute painful procedures in the absence of appropriate analgesia. In fact, pain has emerged as an important modifiable risk factor for altered brain maturation and neurodevelopmental disability after preterm birth. ,
Despite these data, sedation and analgesia are still not used consistently in painful procedures in neonatal care. , More than 40 acute-pain assessment tools exist, combining vital signs and behavioral responses in an attempt to quantify the subjective experience of the neonate. Research continues regarding more objective measures of pain and modalities to assess chronic pain and agitation in neonates. Nonpharmacologic comfort measures represent the standard of care for procedural pain. The provision of analgesia prior to moderate and major procedures ranging from endotracheal intubation to invasive surgery is now increasingly accepted, but further work is needed. Specifically, the selection of the optimal cocktail of analgesic, sedative, and/or anesthetic agents for specific clinical situations represents an ongoing challenge for clinicians. Finally, the optimal approach to neonates requiring invasive mechanical ventilation requires careful consideration of clinical status and available evidence.
Nociception, derived from the Latin nocere , or to harm or hurt, defines the transmission of noxious stimuli from peripheral sensory neurons to the brain, triggering physiologic and behavioral responses. Contrary to historical misconceptions, ascending pathways mediating nociception connect peripheral sensory neurons to the thalamus before neonatal viability whereas descending inhibitory pathways do not mature until far later in gestation. Peripheral sensory neurons first develop in the perioral area of the human fetus during the seventh week of gestation. The face, palms, and soles of the feet are populated by the 11th week of gestation, followed by the trunk, arms, and legs in the 15th week, with all cutaneous and mucous surfaces populated by the 20th week of development. Importantly, complete myelination of these neurons is not necessary, with conduction of nociceptive signals occurring via unmyelinated C fibers and thinly myelinated A delta fibers ( Fig. 4.1 ). In fact, incomplete myelination slows conduction velocity, but neonates have shorter interneuron length. On balance, preterm neonates have lower flexor reflex thresholds compared with older patients.
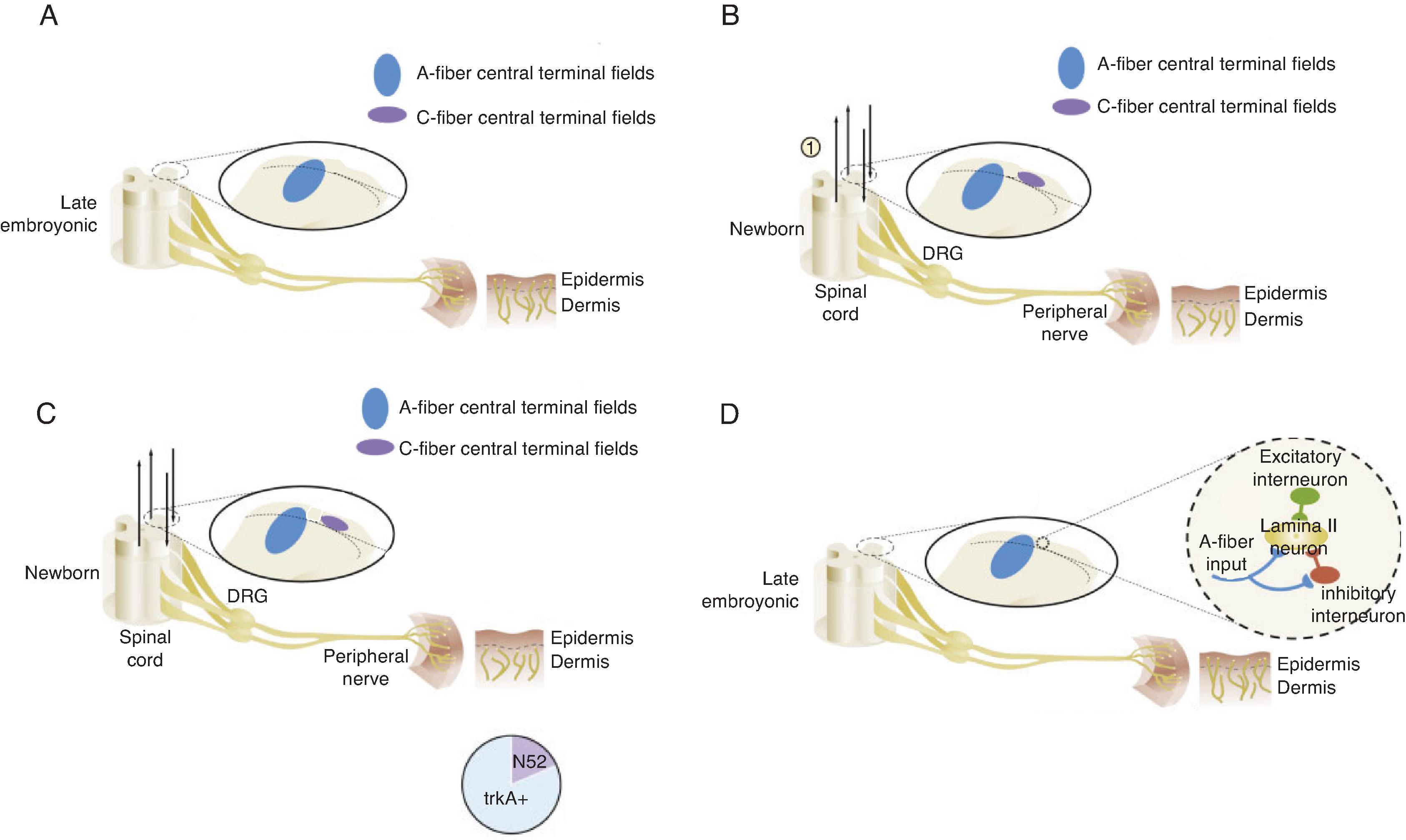
Peripheral sensory neurons synapse with the dorsal root ganglion of the spinal cord via dorsal horn interneurons on formation. However, laminar arrangement and synaptic interconnection (along with a significant degree of myelination) does not begin until 30 weeks of gestation, partially explaining the inability of neonates to attenuate noxious stimuli. Within the spinal cord, glutamate and tachykinin stimulate N-methyl-D-aspartate (NMDA) and tachykinin receptors, amplifying sensory inputs. The NMDA receptor fields in the dorsal horn are larger than adult fields through the newborn period. Additionally, γ-aminobutyric acid (GABA), a primary inhibitor of the action of glutamate in adults, induces neuronal depolarization in neonates due to overexpression of the sodium-potassium-chloride cotransporter NKCC1 and resultant elevation in baseline intracellular chloride concentrations.
The thalamus relays sensory input from the spinal cord to the cerebral cortex. Lateral spinothalamic connections appear in the fetus at the time of initial peripheral sensory neuron formation. Thalamocortical connections occur by 24 weeks of gestation, completing the link between peripheral sensory neurons and the cerebral cortex, which develops neurons itself beginning at 8 weeks of gestation with arborization by 20 weeks. Unfortunately, descending inhibitory pathways originating in the brainstem do not connect to the dorsal horn until late in the third trimester of human gestation. This immaturity appears to have both anatomic and chemical origins due to delayed interneuron maturation in the substantia gelatinosa and delayed expression of noradrenaline, dopamine, serotonin, and other neurotransmitters. More specifically, descending projections appear late in fetal life but do not begin to express neurotransmitters vital to the inhibitory functionality of the pathway until at least 42 weeks postmenstrual age, with full functionality delayed until 48 weeks.
These unique aspects of nociception in immature neonates contribute to the significant consequences of untreated pain. Preterm neonates experience prolonged hyperalgesia and allodynia after tissue damage, leading to chronic periods of nociception and stress. Early tissue damage results in persistent dendritic sprouting in peripheral sensory nerve terminals and within the dorsal root ganglion, resulting in hyperinnervation of injured areas and their spinal projections that persist in adulthood. The rapidly maturing fetal and neonatal central nervous system is also susceptible to long-term epigenetic alteration from noxious stimuli. Repeated painful procedures enhance perceptual sensitivity and lower flexor reflex thresholds into childhood. , Finally, repetitive pain in infancy appears to impact brain morphology in clinically important ways, a subject discussed in more detail in the Long-Term Outcomes section of this chapter.
Neonates demonstrate a complex behavioral, physiologic, and hormonal response to painful stimuli, further reinforcing the necessity of optimal approaches to prevention and treatment ( Table 4.1 ). Behavioral responses have historically been divided into four general categories: simple motor responses, facial expressions, crying, and complex behavioral responses. Simple motor responses include flexion and adduction of extremities in response to a focal painful stimulus. , Neonates produce similar facial responses to acute pain compared with older patients, including brow bulge, eye squeeze, nasolabial furrow, and open mouth. Crying is the primary method of neonatal communication; however, crying owed to pain, hunger, or fear can be distinguished by both the subjective evaluation of trained observers and by spectrographic analysis. Common complex behavioral responses to acute pain in the neonate include alterations of the sleep-wake cycles, decreased attentiveness, and increased irritability compared with neonates who undergo a similar painful procedure after local anesthesia.
| Physiologic | Hormonal | Behavioral |
|---|---|---|
| ↓Oxygen saturation ↕Heart rate ↕Blood pressure ↕Intracranial pressure |
↑Cortisol ↑Epinephrine ↑Norepinephrine ↑β-Endorphins ↑Insulin ↑Glucagon ↑Aldosterone ↑Growth hormone |
Simple motor Flexion and adduction Facial expressions Brow bulge Eye squeeze Nasolabial furrow Open mouth Crying Complex Altered sleep–wake cycle ↓Attentiveness ↑Irritability |
Physiologically, neonates respond to painful stimuli with decreased oxygen saturations, tachycardia with increased variability in heart rate and blood pressure, and consequent fluctuations in intracranial pressure, increasing the risk for intraventricular hemorrhage (IVH) in preterm neonates. Hormonally, neonates demonstrate increases in plasma cortisol, catecholamines (both epinephrine and norepinephrine), β-endorphins, insulin, glucagon, aldosterone, and growth hormone in response to acute noxious stimuli. Chronic stress also suppresses the immune system in the already susceptible neonatal host. Importantly, both physiologic and hormonal responses to acute pain can be attenuated with appropriate analgesia or anesthesia. ,
Clinical pain scoring systems are used to assess procedural pain, acute distress associated with handling, or postoperative pain. Neonatal pain assessment tools generally include physiologic (e.g., heart rate, blood pressure, respiration rate, oxygen saturation, and skin color), behavioral (e.g., crying, facial expression, and bodily reactions), and contextual (e.g., gestational age and behavioral state) components. The five most commonly used neonatal assessment tools include the Neonatal Facial Coding System–Revised, Premature Infant Pain Profile–Revised, , Neonatal Pain, Agitation and Sedation Scale (N-PASS), , Neonatal Infant Pain Scale, and Bernese Pain Scale for Neonates ( Table 4.2 ). The interrater reliability of these five scales is very high (intraclass coefficients >0.96) for venipuncture in late preterm and term infants, and all the tools discriminate between a painful and stressful stimulus. Behavioral items (specifically body tenseness, restlessness, and extremity tone) more consistently indicate pain compared with physiologic measures. It is important for individual neonatal units to choose one tool and rigorously train caregivers to consistently assess before, during, and after painful procedures. Of these tools, only the N-PASS has been evaluated to assess chronic pain and agitation during mechanical ventilation. The COMFORTneo tool was also developed and validated for this purpose. These tools may be used for regularly scheduled assessments such as those during routine care of neonates receiving mechanical ventilation.
| Gestational Age, Weeks | Tool | Score | |
|---|---|---|---|
| ACUTE PAIN | |||
| Neonatal Facial Coding System–Revised | 25–40 | Physiologic: none Behavioral: brow bulge, eye squeeze, nasolabial furrow, horizontal mouth stretch, taut tongue Contextual: none |
0–5 |
| Premature Infant Pain Profile–Revised | 25–40 | Physiologic: heart rate, oxygen saturation Behavioral: brow bulge, eye squeeze, nasolabial furrow Contextual: gestational age, behavioral state (active or quiet, awake or asleep) |
0–18 |
| Neonatal Infant Pain Scale | 26–47 | Physiologic: breathing pattern Behavioral: facial expression, cry, arm tone, leg tone Contextual: behavioral state |
0–7 |
| Bernese Pain Scale for Neonates | 27–41 | Physiologic: respiratory pattern, heart rate, oxygen saturation, skin color Behavioral: duration of cry, time to calm, brow bulge with eye squeeze, posture Contextual: behavioral state |
0–27 |
| ACUTE PAIN OR PROLONGED PAIN OR AGITATION | |||
| Neonatal Pain, Agitation and Sedation Scale | 23–40 | Physiologic: vital sign changes (choice of heart rate, respiratory rate, blood pressure, oxygen saturation) Behavioral: crying or irritability, facial expression, extremities tone Contextual: gestational age, behavioral state |
7–13 for preterm; 10–10 for term |
| PROLONGED PAIN OR AGITATION | |||
| COMFORTneo | 25–43 | Physiologic: none Behavioral: calmness or agitation, respiratory response to mechanical ventilation or crying, body movement, facial tension, body and muscle tone Contextual: alertness |
6–30 |
Objective markers of neonatal pain have been targeted by investigators for decades. Electroencephalography, near-infrared spectroscopy (detecting increases in oxygenated hemoglobin in the contralateral somatosensory cortex), skin conductance (reflecting autonomic function), and salivary cortisol have all been documented to discriminate noxious painful stimuli from touch. In term neonates, near-infrared spectroscopy, heart rate, and oxygen saturation appear to capture an acute response to pain, whereas skin conductance and salivary cortisol represent a more prolonged stressful response. These objective measures moderately correlate with behavioral assessment ( r = 0.20–0.42). Further research is required to determine the optimal measure or combination of measures of acute and chronic pain and stress in infancy.
Optimal treatment of pain and agitation in neonates requires a multimodal approach, including both nonpharmacologic and pharmacologic strategies. Nonpharmacologic therapies are underused in clinical practice. Nonpharmacologic strategies with documented efficacy before acute mildly to moderately painful procedures such as needle sticks include nonnutritive sucking, breast milk, skin-to-skin contact, kangaroo care, and facilitated tucking. Facilitated tucking improves both pain reactivity (immediately after the painful stimulus) and immediate regulation (at least 30 seconds after the painful stimulus) in preterm neonates, whereas nonnutritive sucking positively impacts both domains in term neonates, emphasizing the importance of tailoring nonpharmacologic therapy bundles based on neonatal maturity. Although the optimal bundle of nonpharmacologic interventions remains undefined, individual neonatal units should select the most feasible interventions with supporting evidence and consistently use those interventions prior to all mildly to moderately painful procedures.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here