Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
In the cerebrovascular literature, perfusion imaging refers to an all-encompassing term of various methods to measure hemodynamically derived functional parameters in the brain. In radiology, the three most common parameters that are assessed and utilized clinically are cerebral blood volume (CBV), cerebral blood flow (CBF), and mean transit time (MTT). CBV refers to the amount (volume) of blood within a given mass of brain tissue at a given time and is measured in mL/100 g. CBF refers to the flow of blood through an area of the brain and is usually measured in mL/100 g of brain tissue per minute. MTT refers to the time it takes for blood to flow between the arterial inflow and venous outflow in the brain, that is, the time that blood spends in the cerebral capillary circulation and is often measured in seconds. The general relationship between these three parameters is depicted by the central volume theorem:
A parameter that is being increasing used recently along with MTT is Tmax, or the time to maximum tissue residue function after deconvolution of the tissue contrast agent concentration. Evaluation of these various perfusion parameters can give us an understanding of the physiologic state of the brain and cerebral vasculature in various disorders and help in patient management. Cerebral perfusion can be measured by a variety of methods including magnetic resonance imaging (MRI), computed tomography (CT), and nuclear medicine techniques. These techniques differ in terms of image acquisition duration, spatial resolution, the types of endogenous or exogenous tracers used, and their ability or accuracy in measuring different perfusion parameters. The clinical applications of perfusion imaging include investigation of cerebrovascular disease, brain tumors, and the effect of other diseases on cerebral perfusion.
Perfusion MRI can be performed via the injection of dynamic gadolinium-based contrast material or without contrast material injection by tagging intravascular protons using MR labeling schemes (i.e., arterial spin labeling [ASL]).
Dynamic susceptibility contrast (DSC) perfusion MRI is based on susceptibility effects using T2-weighted or, more commonly, T2*-weighted images and is the most commonly studied and clinically used perfusion technique in assessment of brain masses and stroke. This method is based on the principle that the signal change that occurs during passage of a high-concentration bolus of gadolinium contrast material in the vessels causes a difference in susceptibility between the vessels that contain contrast material and brain tissue, and this signal change can be converted to a relaxation rate change proportional to the fraction of blood volume within each voxel. Data generally are acquired during first-pass perfusion over approximately 1 minute, during which time a high-concentration bolus of gadolinium chelate is rapidly injected intravenously. Time-signal intensity curves drawn during this acquisition will show a rapid decline in signal as the contrast enters the brain, with return of the signal to near baseline after the first pass ( Fig. 28.1 ). These measurements are used to construct a relative CBV map, and the time signal intensity curve can be used to derive other perfusion parameters by various mathematical methods.
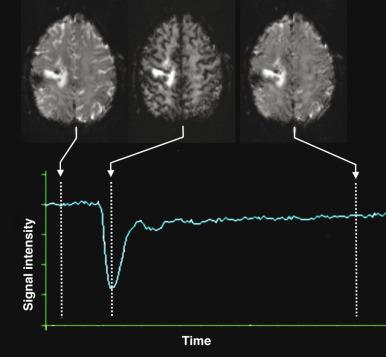
To provide adequate temporal resolution during dynamic contrast administration, most centers currently use gradient echo planar imaging sequences to perform clinical DSC perfusion MRI. This technique is very sensitive to structures that cause magnetic field inhomogeneity such as blood, calcium, bone, metals, or air interfaces such as the skull base. The use of DSC imaging entails multiple technical considerations. Accuracy of CBV maps can vary substantially depending on the acquisition and postprocessing methods used. It is important to take into consideration the type of acquisition used in applying research results for characterization of lesions, because the results and thresholds may vary. Derivation of the perfusion parameters also depends on the mathematical models used by the processing software, and caution must be exercised in comparing results from different calculation methods. Sometimes a small prebolus dose of contrast material is given before the actual DSC acquisition to correct for leakage and more accurate calculation of the perfusion parameters.
DSC perfusion applications are used predominantly to assess cerebrovascular disease and in brain tumor evaluation ( e-Fig. 28.2 ). DSC perfusion can assess the ischemic penumbra in persons who have had an acute stroke and aid in their imaging triage and management. DSC perfusion has been used in differentiation of brain tumors from other mass-like lesions in the brain, preoperative grading of brain tumors (higher grades generally have higher CBV), differentiation of primary high grade from metastatic tumors, assessment of treatment response, and as an adjunct in differentiation of radiation necrosis from a recurrent tumor.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here