Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Despite the ever-evolving field of pediatric surgery, it remains the last bastion of true general surgery. Pediatric surgical conditions span prenatal (fetal) and neonatal to adolescent and young adult age groups. Pediatric surgeons must assess and manage a wide spectrum of surgical conditions with vastly different pathophysiology. Pediatric surgery covers a wide spectrum of organ systems, ranging from head and neck to thoracic and the gastrointestinal (GI) tract, and conditions such as congenital anomalies, solid tumors, and pediatric trauma. This chapter provides an overview of common and unique pediatric surgical conditions, their pathophysiology, and treatment strategies.
The unique newborn physiology stems in part from the smaller size of patients and the cellular and functional immaturity of their organ systems. Newborns are at risk for cold stress, and an ideal thermal environment must be maintained to reduce oxygen consumption and metabolic demands. Isolates in nurseries and warm operating rooms serve to maintain thermogenesis. The major risk factors for hypothermia in infants are their relatively large body surface area to body weight and subcutaneous tissue and greater insensible fluid losses. Infants also respond to cooler ambient temperature by nonshivering thermogenesis, whereby increases in metabolism and oxygen consumption occur. The use of overhead radiant heaters is also common in the operating rooms, where an ideal ambient temperature (∼23°C) is kept in order to maintain a neonate’s core body temperature between 36°C and 37.5°C.
During fetal circulation, arterial blood from the placenta bypasses the fetal lungs through the patent foramen ovale and ductus arteriosus. After birth, the foramen ovale closes with the newborn’s first breath, then a precipitous drop in pulmonary vascular resistance occurs, thereby increasing pulmonary blood flow. Decreased blood flow, along with a higher oxygen content, promotes spontaneous closure of the ductus arteriosus. Nonsteroidal antiinflammatory drugs (e.g., indomethacin) can promote closure of a patent ductus arteriosus, particularly in premature infants. If persistently open, patent ductus arteriosus closure is achieved via open minithoracotomy or a catheter-based coil procedure. An increase in pulmonary vascular resistance can be caused by hypoxemia and acidosis, resulting in persistent pulmonary hypertension (PPHN) and a right-to-left shunt. Nitric oxide gas, a potent inducer of vascular smooth muscle relaxation, is effective against PPHN.
The infant heart has a limited capacity to increase the stroke volume; therefore, cardiac output is largely heart rate-dependent. As such, heart rate change is a sensitive indicator of intravascular volume status. Capillary refill is a sensitive indicator of adequate tissue perfusion. A prolonged capillary refill (>2 seconds) may represent substantial shunting of blood from the peripheral tissues to the central organs, which may occur with cardiogenic or hypovolemic shock.
The respiratory rate for a normal newborn ranges from 40 to 60 breaths/min, with a tidal volume of 6 to 10 mL/kg. Nasal flaring, grunting, intercostal and substernal retractions, and cyanosis are symptoms and signs of respiratory distress. Infants are obligate nasal and diaphragmatic breathers, and therefore any condition that obstructs the nasal passages (e.g., nasogastric tube) or interferes with diaphragmatic function may severely compromise respiratory status. In addition, the newborn airway is quite small, with an average tracheal diameter of 2.5 to 4 mm, and it can easily be plugged with airway secretions.
At birth, the lungs are considered functionally immature and continue to develop new terminal bronchioles and alveoli until 6 to 8 years of age. The neonatal lung has fewer type II pneumocytes, which produce surfactant, a lipoprotein mixture of phospholipid, protein, and neutral fats. Surfactant regulates alveolar surface tension, thereby increasing functional residual capacity. Lecithin, the most predominant phospholipid, can be measured in amniotic fluid, and the lecithin-to-sphingomyelin ratio is used to determine fetal lung maturity. Hence, premature infants are at greater risk for alveolar collapse, hyaline membrane formation, and barotrauma from mechanical ventilatory support. Exogenous surfactant therapy has had a major impact on the management of premature infants. This has resulted in improved survival and decreased incidence of bronchopulmonary dysplasia, a condition characterized by oxygen dependence, radiologic abnormality, and chronic respiratory symptoms beyond the first 28 days of life.
Infants have lower levels of immunoglobulins (IgA, IgG, and IgM) and of the C3b complement at birth and therefore are at higher risk for systemic infection. Sepsis work-up for neonates has largely remained the same with surveillance cultures, including cerebrospinal fluid, and complete blood count as well as C-reactive protein levels. Infants are also at risk for potential septic sources arising from invasive monitoring lines and interventions such as prolonged endotracheal intubation, umbilical vascular catheters, and bladder catheterization. An empirical antibiotic regimen is often started based on subtle clinical suspicions (e.g., decreased tolerance of enteral feeding, temperature instability, reduced capillary refill, tachypnea, irritability). Antibiotic therapy is targeted at common bacterial pathogens, such as Group B beta-hemolytic streptococcus, methicillin-resistant Staphylococcus aureus, and Escherichia coli .
Fluid and electrolyte balance must be carefully assessed in pediatric surgical patients, especially in smaller neonates with a narrow margin for error. Due to higher insensible water losses through a thin immature skin barrier, fluid requirements for premature infants can be substantial. Insensible water losses are directly related to gestational age, which range from 45 to 60 mL/kg/day for premature infants weighing less than 1500 g to 30 to 35 mL/kg/day for term infants. Radiant heat warmers, phototherapy for hyperbilirubinemia, and respiratory distress can result in additional fluid losses. During the first 3 to 5 days of life, physiologic water loss can total up to 10% of the body weight. Fluid requirements are calculated according to body weight. During the first few days of life, the fluid recommendations are on the conservative side; however, infants require 100 to 130 mL/kg/day for maintenance fluids by the fourth day of life. Surgical conditions such as gastroschisis and necrotizing enterocolitis (NEC) demand a significantly higher volume. Urine output and osmolarity are good indicators of adequate tissue perfusion. The ideal minimum urine output in a newborn is 1 to 2 mL/kg/hour. Infants can respond to prerenal azotemia by concentrating urine only up to approximately 700 mOsm/kg. The daily requirements for sodium and potassium are 2 to 4 and 1 to 2 mEq/kg, respectively. These requirements are usually met with 5% dextrose in 0.45% normal saline with 20 mEq/L of potassium at the calculated maintenance rate. Fluid losses from gastric drainage, ostomy output, or diarrhea should also be carefully assessed and replaced with an appropriate solution. Gastric losses should be replaced in equal volumes with 0.45% normal saline with 20 mEq/L of potassium. Diarrheal, pancreatic, and biliary losses are replaced with isotonic lactated Ringer solution. Hypovolemia due to acute hemorrhage should be corrected with prompt transfusion of blood products at a bolus of 10 to 20 mL/kg of packed red blood cells, plasma, or 5% albumin.
Energy requirements vary substantially from infancy to childhood and also under different clinical conditions. Appropriate weight gain remains the most reliable crude indicator of adequate caloric intake. Total daily caloric requirements and the weight curve plateau with age. Nearly 50% of the energy in term infants younger than 2 weeks and 60% of energy in premature infants weighing less than 1200 g is consumed for growth. A general guideline for the enteral calorie requirement of infants is 120 calories/kg/day to achieve an ideal weight gain of ∼1% of body weight per day. Breast milk and standard infant formulas contain 20 calories/ounce. Formulas with higher calorie density are available for those who are unable to consume sufficient volumes to meet their calorie requirements or those with strict fluid restriction. Breast milk is the ideal form of enteral nutrition. However, hypoallergenic, lactose-free to amino-acid-based formulas are available to meet the specific needs of infants with particular GI tract conditions. In general, infants with a stressed gut are given continuous enteral feedings and then transitioned to gastric bolus feedings. Toleration of enteral feeding is carefully monitored by assessing for abdominal girth, gastric residuals, and stool output.
Carbohydrates are stored mainly as glycogen in the liver and muscles. Because newborn liver and muscle mass is disproportionately smaller than that of adults, infants are susceptible to hypoglycemia with risks of seizure and neurologic impairment. The minimum glucose infusion rate for neonates is 4 to 6 mg/kg/min. This rate must be calculated daily while the infant is receiving parenteral nutrition. For total parenteral nutrition (TPN), the glucose infusion rate is increased daily in increments of 1 to 2 mg/kg/min to a maximum value of 10 to 12 mg/kg/min. Hyperglycemia from a less than ideal glucose infusion rate should be avoided because it can lead to rapid hyperosmolarity and dehydration. Hyperglycemia could also reflect an underlying sepsis and therefore, should be investigated.
The average protein intake is ∼15% of total daily calories and ranges from 2 to 3.5 g/kg/day in infants. By 12 years of age, this protein requirement is reduced in half and approaches the adult requirement (1 g/kg/day) by 18 years of age. The provision of greater amounts of protein relative to nonprotein (carbohydrate plus fat) calories will result in rising blood urea nitrogen levels, resulting in a nonprotein calorie-to-protein calorie ratio (when expressed in grams of nitrogen) greater than 150:1. For infants receiving parenteral nutrition, protein administration usually starts at 0.5 g/kg/day and advances in daily increments of 0.5 g/kg/day to a target goal of approximately 3.5 g/kg/day.
Fat is a major source of nonprotein calories. Linoleic acid, an 18-carbon chain with two double bonds, is considered an essential fatty acid, and its deficiency results in dryness, rash, and desquamation of skin. In pediatric patients, fat is provided as a major source of calories to prevent the development of essential fatty acid deficiency. The lipid requirements for growth are significant, and fat is a robust calorie source. Similar to protein, fat infusions are started at 0.5 g/kg/day and advanced up to 2.5 to 3.5 g/kg/day. In infants with unconjugated hyperbilirubinemia, fat is administered with caution because fatty acids may displace bilirubin from albumin. The free unconjugated bilirubin may then cross the blood-brain barrier and can lead to kernicterus, resulting in mental retardation.
TPN is reserved for infants for whom adequate daily enteral nutrition cannot be achieved. Infants only have energy reserve to withstand periods of starvation as few as 2 to 3 days. Thus, an infant’s need for parenteral nutrition should be addressed promptly. The total TPN infusion rate is kept at a steady-state to meet daily fluid requirements, and the concentration of nutrients is gradually increased daily until goals are met. Infants with surgical conditions often become cholestatic, typically caused by prolonged TPN support, however, other causes should be ruled out. Serum bile acid levels are usually elevated first, then direct bilirubin concentration, followed by liver enzyme levels. The ideal treatment for TPN-associated cholestasis is restoring enteral feeding. The use of omega-3 fat emulsion (Omegaven) has been critical in preventing TPN-induced cholestasis. A medium-chain triglyceride–containing formula is used, and if an infant is receiving total enteral nutrition, fat-soluble vitamins should be supplemented.
Dermoid and epidermoid cysts are slow-growing benign lesions that typically occur in the scalp and the skull. These cysts usually arise from part of the dermal or epidermal tissues, forming a small cyst filled with normal skin components. Dermoid cysts may contain hair, teeth, and skin glands. Epidermoid cysts typically have only epidermal tissue and keratin debris. They commonly occur on the forehead, lateral corner of the eyebrow, anterior fontanelle or in the postauricular space. They are generally asymptomatic but may increase in size over time and can often become osteolytic. Most scalp lesions require only the clinical exam for diagnosis and subsequent surgical excision. However, imaging studies (e.g., ultrasound) can be critical to identify midline lesions such as a communicating cephalocele.
Enlarged lymph nodes presenting as small, mobile, discrete clusters in the anterior cervical triangle are one of the most common conditions referred for a pediatric surgical evaluation, biopsy, or possible excision. They usually occur along the sternocleidomastoid muscle border, often in clusters. The exact cause is unknown but presumed to be multifocal. A detailed history and physical examination are sufficient to determine whether surgery is indicated. The use of ultrasound has become widely prevalent and can, at times, identify those nodes with central necrosis requiring surgical intervention. Occasionally, lymph nodes presenting as fixed, nontender, progressively enlarging nodes in the supraclavicular region should raise a suspicion for a serious underlying etiology. Constitutional symptoms such as night sweats and weight loss should prompt a thorough investigation with chest radiography, which could detect mediastinal adenopathy.
Acute, bilateral cervical lymphadenitis from respiratory viral infections (e.g., adenovirus, influenza virus, respiratory syncytial virus) require observation alone. S. aureus and Group A streptococcus are responsible for the majority of acute pyogenic lymphadenitis.
Cat-scratch disease is a self-limited infectious condition characterized by painful regional lymphadenopathy. A gram-negative bacillus, Bartonella henselae, is responsible for the majority of cases. A history of exposure to cats is helpful but not always present. Indirect immunofluorescent antibody testing has only moderate specificity. Polymerase chain reaction assay of a lymph node biopsy specimen is a more useful study for diagnosis. Cat-scratch disease is usually self-limited. A less common infectious cause of cervical lymphadenitis is nontuberculous mycobacterial infection. The nodes are fluctuant, with a violaceous appearance of the overlying skin. The diagnosis is made by positive cultures for nontuberculous acid-fast bacilli along with a tuberculin skin test. Surgical excision is usually indicated because most nontuberculous mycobacteria are resistant to conventional chemotherapy.
Cystic hygroma is a multiloculated cyst lined by endothelial cells occurring as a result of lymphatic malformation. Most involve the lymphatic jugular sacs and present in the posterior neck region. Other common sites are the axillary, mediastinal, inguinal, and retroperitoneal regions, and approximately 50% of these cystic lesions are present at birth. Cystic hygromas are soft cystic masses that can distort the surrounding structure, including the airway. A large cystic mass of the neck in the fetus can pose a significant threat to airway at birth. Prenatal ultrasound and fetal magnetic resonance imaging (MRI) studies can better demonstrate the extent of disease along with its mass effect on the airway. If present, a careful coordination of surgical intervention, ex-utero intrapartum treatment (EXIT) procedure, at the time of delivery can be life-saving, though this is more likely with solid tumors such as teratomas. Cystic hygromas are prone to infection and hemorrhage into the mass. MRI is useful in outlining the extent of lymphatic channels. Complete excision with tedious isolation and ligation of lymphatic branches is the surgical goal. Aggressive blunt and electrocautery dissection, as well as radical resection, must be avoided, as this can lead to recurrence or infection due to incomplete control of lymphatics. When a safe, complete surgical excision is not feasible, injection with sclerosing agents, such as bleomycin, doxycycline, or OK-432 derived from Streptococcus pyogenes , should be considered as an effective nonsurgical option.
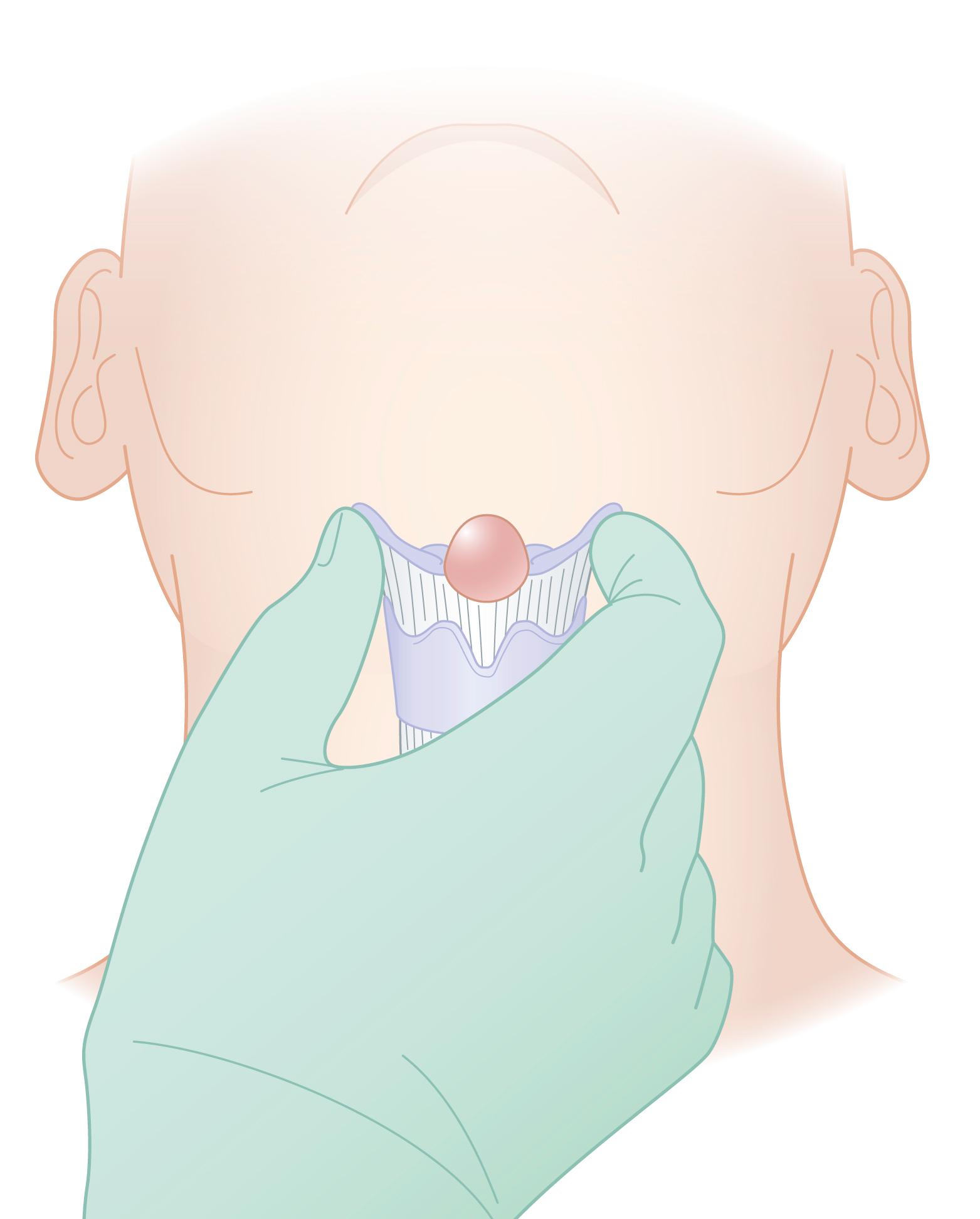
An upper-midline cystic neck lesion in toddlers is a thyroglossal duct cyst until proven otherwise ( Fig. 67.1 ). It originates at the base of the tongue at the foramen cecum and descends through the central portion of the hyoid bone. Although thyroglossal duct cysts may occur anywhere from the base of the tongue to the thyroid gland, most are found at, or just below, the hyoid bone. A thyroid diverticulum develops as a median endodermal thickening at the foramen cecum in the embryonic stage of development. The thyroid diverticulum descends in the neck and remains attached to the base of the tongue by the thyroglossal duct. Also, as the thyroid gland descends to its normal pretracheal position, the ventral cartilage of the second and third branchial arches forms the hyoid bone. Hence, the intimate anatomic relationship of the thyroglossal duct remnant exists with the central portion of the hyoid bone. The thyroglossal duct normally regresses by the time the thyroid gland reaches its final position. When the elements of the duct persist despite complete thyroid descent, a thyroglossal duct cyst may develop. Failure of normal caudal migration of the thyroid gland results in a lingual thyroid in which no other thyroid tissue is present in the neck. Ultrasound or radionuclide imaging may be useful to identify the ectopic thyroid gland in the neck. The Sistrunk procedure, first described in 1928, is the gold standard operation for thyroglossal duct cysts. It involves complete excision of the cyst in continuity with its tract, the central portion of the hyoid bone, and the tract interior to the hyoid bone extending to the base of the tongue. Failure of complete resection results in cyst recurrence in as high as 40% to 50% of cases.
Branchial cleft remnants present as lateral neck masses. Embryologically, the structures of the head and neck are derived from six pairs of branchial arches, their intervening clefts, and pouches. Congenital cysts, sinuses, or fistulas result from failure of these structures to regress, persisting in aberrant locations. The location of these remnants generally dictates their embryologic origin and guides the subsequent operative approach. Failure to understand the embryology may result in incomplete resection or injury to adjacent structures. Branchial lesions can manifest as sinuses, fistulas, or cartilaginous rests in infants. The clinical presentation ranges from a continuous mucoid drainage, a fistula or sinus, or an infected cystic mass. Branchial remnants may also be palpable as cartilaginous lumps or cords corresponding with a fistulous track. Dermal pits or skin tags may also be present. First branchial anomalies are typically located in the front or back of the ear, or in the upper neck near the mandible. Fistulas typically course through the parotid gland, deep or through branches of the facial nerve, and end in the external auditory canal. The second branchial cleft anomalies are the most common type. The external ostium of these remnants is located along the anterior border of the sternocleidomastoid muscle, usually in the vicinity of the upper half to lower third of the muscle. A tortuous and long course of the fistula tract can be found, which requires stepladder counter-incisions in order to excise the fistula track completely. Typically, the fistula penetrates the platysma, ascends along the carotid sheath to the level of the hyoid bone, and turns medially to extend between the carotid artery bifurcations. The fistula courses adjacent to the hypoglossal and glossopharyngeal nerves, behind the posterior belly of the digastric and stylohyoid muscles to end in the tonsillar fossa or other nasopharyngeal spaces. Third branchial cleft remnants usually do not have associated sinuses or fistulas and are located in the suprasternal notch or clavicular region and can descend into the mediastinum. They present more commonly as cysts in toddlers and older children. These most often contain cartilage and present clinically as a firm mass or subcutaneous abscess.
Extracorporeal life support (ECLS) is a cardiopulmonary bypass delivering temporary life support for the critically-ill patient with acute respiratory and/or cardiac failure. ECLS achieves sufficient gas exchange, with carbon dioxide removal and oxygenation of blood to maintain normal circulatory support. Since its first reported neonatal experience in 1976, ECLS has not only become a standard therapeutic option for cardiopulmonary failure that is refractory to maximal medical therapy, but it also has become widely used for a variety of clinical applications, such as extracorporeal cardiopulmonary resuscitation (eCPR). Although the exact role of eCPR in neonatal and pediatric resuscitation remains controversial and yet to be determined, the use of eCPR has become a common practice in many hospitals around the globe. There are over 800 centers around the world contributing registry data to the Extracorporeal Life Support Organization database (ELSO registry data; July 2019).
The major indications for neonatal extracorporeal membrane oxygenation (ECMO) include meconium aspiration, respiratory distress syndrome, PPHN, sepsis, and congenital diaphragmatic hernia (CDH). Meconium aspiration is the most common application for neonatal ECMO with the highest survival rate (>90%). Indications for neonatal ECMO vary among institutions. In general, ECMO is indicated when an infant’s overall cardiopulmonary function deteriorates to a point of ∼80% predicted mortality. Two guidelines have been used as predictors for survival without ECMO: the alveolar-arterial difference in the partial pressure of oxygen (P ao 2 − Pa o 2 [also known as AaD o 2 ]) and the oxygen index. AaD o 2 greater than 610 for longer than 8 to 12 hours and AaD o 2 greater than 620 for 6 hours, associated with extensive barotrauma and severe hypotension requiring inotropic support, are considered to be criteria for ECMO. The oxygen index is calculated as the fraction of inspired oxygen (usually 1.0) × mean airway pressure × 100 divided by Pa o 2 . An 80% mortality is observed with an oxygen index greater than 40. Contraindications are severe prematurity due to a high risk of intracranial bleeding, weight less than 2 kg, presence of an intracranial bleed (grade II intraventricular hemorrhage), and a nonreversible pulmonary disease such as congenital alveolar dysplasia. Additional exclusion criteria include the presence of cyanotic congenital heart disease or major genetic defects that preclude survival, as well as intractable coagulopathy.
The right internal jugular vein and common carotid artery are used for venoarterial cannulations because of vessel sizes to accommodate cannulas and collateral circulation. The ECLS circuit is composed of a silicone rubber bladder that collapses when venous return is diminished, roller pump, membrane oxygenator, heat exchanger, tubing, and connectors. The basic principle of ECLS is that desaturated mixed venous blood from the right atrium drains through the venous cannula to the bladder and is pumped to the membrane oxygenator, where carbon dioxide is removed and oxygen is added. The oxygenated blood then passes through the heat exchanger and is returned to the patient through the arterial cannula. Systemic anticoagulation to prevent clotting of the ECLS circuit puts patients at risk for bleeding complications. Indicators of lung recovery include an increasing Pa o 2 , improved lung compliance, and clearing of the chest radiograph. As the pulmonary function recovers, the patient is trialed off bypass by clamping the cannulas. If tolerated, the patient is taken off ECLS on moderate conventional ventilatory settings. Venovenous bypass using a double-lumen single cannula placed via the right internal jugular vein has the advantage of avoiding carotid arterial cannulation. Often, perfusion of well-oxygenated blood through venovenous ECMO restores hemodynamic stability. In older pediatric, as well as adult, patients, venous cannulas can be placed via both the internal jugular and femoral veins to achieve venovenous ECMO circulation.
Bleeding is the most common complication of ECLS, and it can occur anywhere from catheter sites and surgical sites such as the thoracic cavity to intracranial bleeds. Gestational age is the most significant predictor of intracranial hemorrhage on ECLS, and premature infants less than 34 weeks of gestational age are at highest risk. Other complications include seizures, neurologic impairment, real failure requiring hemofiltration or hemodialysis, hypertension, infection, and mechanical malfunction of membrane oxygenator, pump, and heat exchanger, as well as the cannulas themselves.
The overall incidence of CDH is 1 in 2000 to 5000 live births. CDH is diagnosed prenatally in most cases and occurs more commonly on the left side (84%); bilateral defects occur but are rare (2%). A hernia sac is present 10% to 15% of the time and must be excised at the time of repair. Despite multimodality treatment strategies, such as fetal tracheal occlusion, ECMO, inhaled nitric oxide, and permissive hypercapnia, the overall survival rate remains at 70% to 90%. True survival data for CDH is somewhat distorted by the fact that infants with more severe CDH are often stillborn, and therefore not captured by the registry. The lung area to head circumference ratio (LHR) is a sonographic predictor of prognosis and is determined by taking the product of the longest two perpendicular linear measurements of the lung contralateral to CDH and is divided by head circumference. An LHR of less than 1 and an abnormal liver position at 24 weeks of gestation are strong predictors of unfavorable outcomes. Fetal MRI is also used at many centers to determine fetal lung volume, as well as ratio of lung to the spinal fluid signal intensity.
In the embryo, the pleuroperitoneal cavities become separated by the developing membrane during 8 to 10 weeks of gestation. When the pleuroperitoneal canal persists, it leads to a posterolateral CDH defect. The posterolateral location of this hernia is known as Bochdalek hernia; it is distinguished from a CDH of the anteromedial location known as Morgagni hernia. Abdominal contents herniate into the thoracic cavity through the diaphragmatic defect, compressing the ipsilateral developing lung. These lungs have smaller bronchi, with less bronchial branching and less alveolar surface area. The ipsilateral lung is affected more severely, however, both lungs are affected by pulmonary hypoplasia. In addition, the pulmonary vasculature is significantly affected by the increased thickness of arteriolar smooth muscle. Arteriolar vasculature is also extremely sensitive to local and systemic vasoactive factors. Hence, the severity of pulmonary hypoplasia and pulmonary hypertension significantly affect the overall morbidity and mortality in CDH infants.
The diagnosis of CDH is frequently made prenatally, as early as 15 weeks of gestation, during routine ultrasound evaluation. Infants who have a late onset of CDH (beyond 25 weeks of gestation) have a better overall survival. Herniation of the stomach and liver, polyhydramnios, and associated anomalies have been associated with poor outcomes. The delivery of a fetus with CDH should be planned at a hospital capable of providing advanced neonatal care, including ECMO.
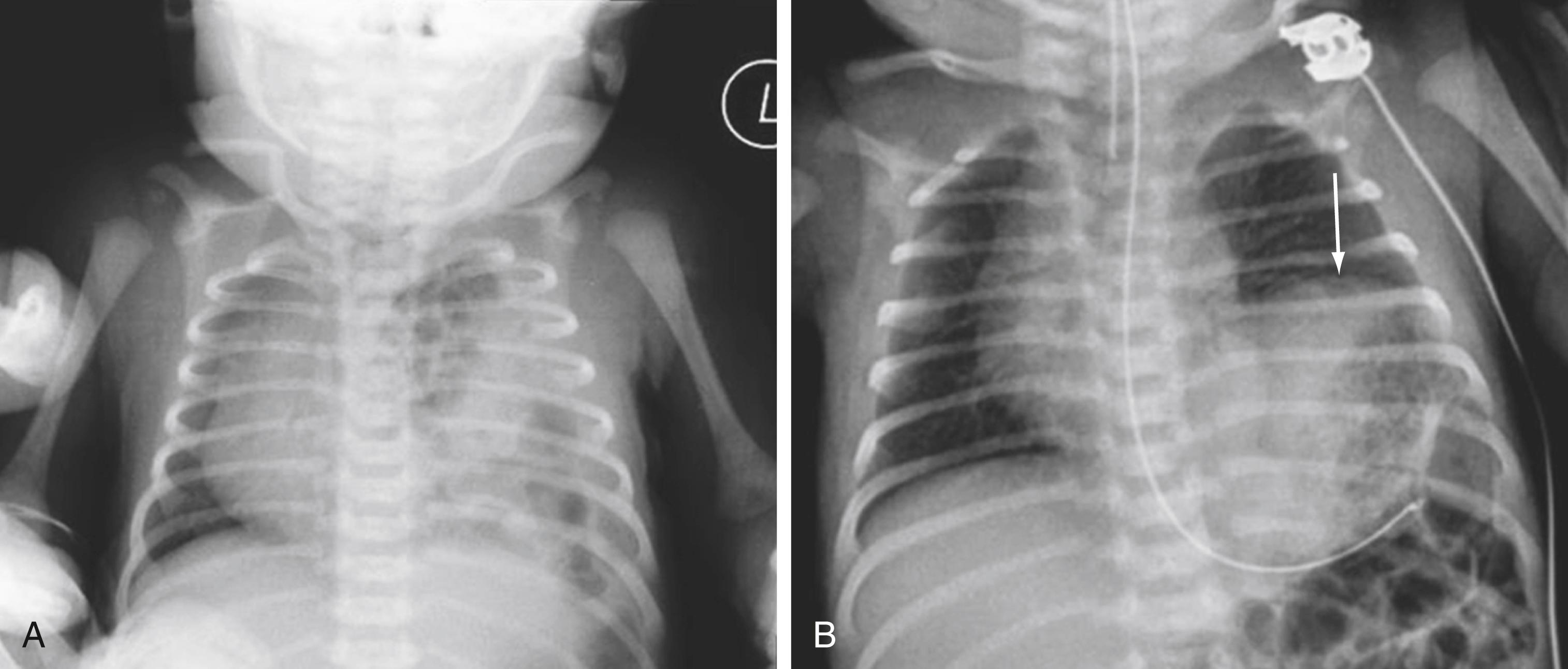
Most infants with CDH experience respiratory distress immediately after birth. The initial symptoms and signs may include grunting respiration, chest retractions, dyspnea, and cyanosis with a scaphoid abdomen. Decreased breath sounds, along with bowel sounds, may be auscultated over the chest with CDH. The shifting of heart sounds to the right (for left-sided CDH) is common. A significant preductal and postductal pulse oximetry differential indicates right-to-left shunting due to PPHN. The chest radiograph demonstrates multiple bowel loops in the thoracic cavity along with mediastinal shift ( Fig. 67.2A ). The differential diagnosis includes congenital cystic adenomatoid malformation, bronchogenic cyst, diaphragmatic eventration ( Fig. 67.2B ), and cystic teratoma. Typically, the infant does well for several hours after delivery during the “honeymoon period” and then begins to demonstrate worsening respiratory function. Therapeutic interventions are aimed at stabilizing and treating PPHN. In 10% to 20% of cases, CDH is diagnosed beyond the first 24 hours, when infants experience symptoms of feeding difficulties, respiratory distress, and pneumonia. In Morgagni hernias, the diagnosis is often delayed until childhood because most infants are asymptomatic.
Open fetal surgery for CDH was first attempted in the late 1980s with initial success in fetuses without liver herniation. However, in fetuses with liver herniation and subsequent kinking of the umbilical vein reduction of the liver resulted in fetal demise; therefore, the trial was abandoned. Subsequently, fetal endoscopic tracheal occlusion (FETO) showed encouraging results in promoting lung growth by preventing the egress of pulmonary fluid and increasing intrabronchial pressure. However, a randomized controlled trial failed to show improved survival. A multicenter randomized trial, Tracheal Occlusion to Accelerate Lung Growth (TOTAL), which had been led by several European centers, is now enrolling patients in the United States. The evolution of fetal surgery and its clinical applications are discussed in Chapter 73 .
The postnatal management of CDH is directed toward stabilization of the cardiorespiratory status while minimizing iatrogenic injury from therapeutic interventions. Immediate securing of the airway with endotracheal intubation is critical; however, excessive mean airway pressure ventilation can lead to barotrauma along with compromised venous blood return to the heart. An orogastric tube is placed to prevent gastric distention, which may worsen the lung compression, mediastinal shift, and ability to ventilate. The major emphasis on gentle ventilatory management with permissive hypercapnia has resulted in improved survival for CDH infants. Inhaled nitric oxide is used widely for its pulmonary vasodilatory effect. The use of tolazoline, a nonselective α-adrenergic blocking agent, as a pulmonary vasodilator has not produced clinically significant results. Sildenafil, a phosphodiesterase-5 inhibitor, works by inducing pulmonary vascular smooth muscle relaxation and has been used in many centers with variable results. A retrospective cohort study showed an increasing trend in the use of a variety of vasodilators for CDH patients.
Infants with CDH not requiring ECMO can safely undergo operative repair soon after birth. Laparoscopic repair of CDH has gained popularity in recent years, but its overall benefit and effects on long-term outcome remain uncertain. The ideal timing of CDH repair on ECMO remains quite controversial; some advocate early operative repair on ECMO, whereas others recommend repair at the time of weaning from ECMO or even after decannulation. There are no indisputable prospective data on the ideal timing of CDH repair for those requiring ECMO support. A recent report suggested that CDH repair after ECMO therapy is associated with improved survival compared to repair while on ECMO. The preferred open approach for a posterolateral CDH is through a subcostal abdominal incision. The viscera are reduced into the abdominal cavity, and the posterolateral defect in the diaphragm is approximated with interrupted nonabsorbable sutures. The hernia defect is often quite large, with only a small leaflet of diaphragmatic tissue present anteromedially. Although primary repair of the defect is ideal, closure with excessive tension must be avoided in order to prevent hernia recurrence. Some advocate for the use of pledgeted sutures. A number of reconstructive techniques and materials are available for the repair of large hernia defects. The surgical technique of abdominal or thoracic muscle flaps may be considered, but a prosthetic material, such as a Gore-Tex patch, is most widely used. The advantages of a prosthetic patch are shorter operative time and a tension-free repair. Some advocate use of regenerative extracellular matrix biomaterials as an ideal biodegradable patch (e.g., Surgisis [Cook Medical Bloomington, IN] and AlloDerm [LifeCell, Branchburg, NJ]) to repair diaphragmatic hernia defects. In general, the chest tube is spared and postoperative radiographs show immediate mediastinal shift toward the center. The chest cavity quickly fills with serous drainage, which later gets absorbed as fluid status returns to baseline. During abdominal cavity closure, it can often be difficult to accommodate the reduced viscera from the thoracic cavity. A temporary abdominal silo may be considered, however allowing an incisional hernia with skin-only closure until the definitive fascia closure can be performed is an alternative surgical option. When CDH is repaired on ECMO, postoperative bleeding is a common major complication and therefore, meticulous hemostasis must be achieved.
Long-term outcomes in infants with CDH vary. Some can develop a chronic condition due to PPHN and respiratory dysfunction. Moreover, infants who received aggressive and prolonged care in the neonatal intensive care unit have a high incidence of developmental delay, seizures, and hearing loss. Other morbidities for CDH survivors include chronic lung disease, scoliosis, growth retardation, pectus excavatum deformities, as well as gastroesophageal reflux (GER) disease and foregut dysmotility.
Abnormal elevation of the hemidiaphragm can significantly affect respiratory function. Eventration of the diaphragm can be congenital or acquired. Congenital eventration can occur due to birth trauma (Erb palsy) or due to an anatomic abnormality of the diaphragm. Erb palsy is a paralysis of the arm caused by injury to the brachial plexus, comprising the ventral rami of spinal nerves C5 to C8. These injuries typically result from shoulder dystocia during a difficult birth. Erb palsy commonly includes ipsilateral diaphragmatic paralysis due to traction injury of the phrenic nerves and upper portion of the brachial plexus. Acquired eventrations are usually secondary to iatrogenic phrenic nerve injury during open cardiac surgery. Elevation of the diaphragm is seen on chest radiographs ( Fig. 67.2B ); however, it can easily be misdiagnosed as CDH. The diagnosis is confirmed by dynamic visualization of the diaphragm using either fluoroscopy or ultrasound of the chest. Absent or paradoxical movement of the diaphragm on inspiration is diagnostic of eventration. When symptoms progress, resulting in respiratory distress or inability to wean from ventilatory support, repair is indicated. The surgical treatment of diaphragm event ration is an open or laparoscopic diaphragm plication in which the diaphragm is folded taut using multiple interrupted nonabsorbable sutures.
Bronchopulmonary malformations are congenital abnormalities of the airway and include bronchogenic cysts, intralobar sequestrations (ILSs) and extralobar sequestrations (ELSs), congenital pulmonary airway malformations (CPAMs), and congenital lobar emphysema (CLE). In the perinatal period, these lung lesions can result in pleural effusions, polyhydramnios, hydrops, and pulmonary hypoplasia with subsequent respiratory distress and even airway obstruction. If severe enough, fetal demise can occur. The majority of these lesions are diagnosed prenatally with ultrasound. Fetal surgery has been pursued when fetal viability is at risk. After birth, congenital lung lesions are often asymptomatic and some may even spontaneously regress. However, there is a concern that some lesions may lead to recurrent pulmonary infections and exhibit long-term malignant potential.
The bronchogenic cyst wall consists of fibroelastic tissue, smooth muscle, and cartilage, whereas the cyst itself is lined with respiratory tract ciliated columnar epithelial cells. It can also contain mucus-producing cuboidal cells, which contribute to enlargement of the cyst with mucus. These cysts can occur anywhere along the tracheobronchial tree, but are usually found near the carina and right hilum, and represent the most common mediastinal cysts. These cysts can enlarge resulting in compression of the airway or other vital structures. Infants are particularly at risk because of their narrow, easily compressible trachea and bronchus. Cysts can also cause dysphagia, pneumothorax, cough, and hemoptysis or become infected, especially when presenting later in life. The diagnosis is often suspected on a routine chest radiograph and is confirmed by computed tomography (CT), which demonstrates a spherical nonenhancing, mucus-filled cystic mass, although an air-fluid level can be seen if the cyst communicates with the airway. Cysts within the pulmonary parenchyma typically communicate with a bronchus, whereas those in the mediastinum usually do not. Bronchogenic cysts are resected regardless of symptoms ( Fig. 67.3A ). Rare cases of malignant transformation have been reported.
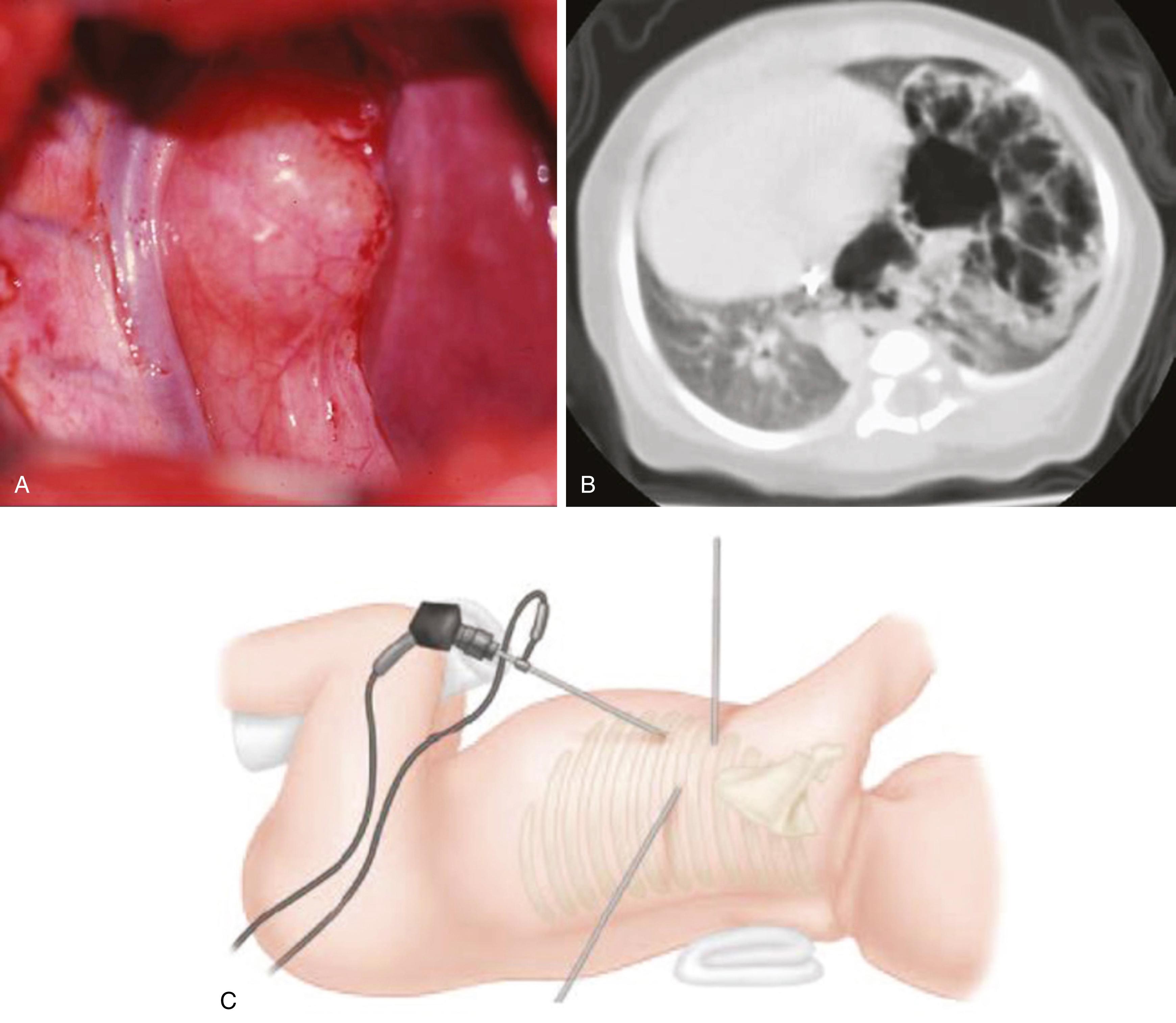
CPAMs have been described as hamartomatous lesions in which a multicystic mass replaces normal lung tissue. They are connected to the tracheobronchial tree, and the blood supply is pulmonary. CPAMs can undergo malignant transformation, and rhabdomyosarcoma has been reported in older children. They are classified on the basis of their appearance on imaging, and confirmation is made by pathologic examination. According to the Stocker classification, type I lesions account for almost 75% of all cases and consist of a small number of large, 2- to 10-cm cysts that can compress normal lung parenchyma. Type II lesions have numerous cysts, usually measuring less than 1 cm in diameter. Type III lesions are rare and appear to be only a few millimeters in diameter; however, they are associated with mediastinal shift, hydrops, and a poor prognosis. Prenatal fetal MRI can be used to distinguish CPAM from other thoracic anomalies. If fetal distress occurs, options include fetal thoracotomy and thoracoamniotic shunting (if the fetus is <32 weeks), but this is extremely rare. Most fetuses with a prenatally diagnosed CPAM experience partial regression in the third trimester and can be treated with expectant management. A single course of betamethasone has been shown to be effective in promoting spontaneous regression or size reduction and resolving hydrops. Although they are usually unilateral and unilobar, they can present in the immediate perinatal period with life-threatening respiratory distress. However, the majority of CPAMs are asymptomatic in infancy. Unrecognized CPAMs can present with chronic cough or recurrent pneumonia at a later time. A chest radiograph is usually diagnostic, revealing a cystic thoracic mass, with or without air-fluid levels; however, ultrasound and CT studies are now routinely obtained ( Fig. 67.3B ). In general, the involved lobe should be resected given the risk of infection and malignant transformation (e.g., pleuropulmonary blastoma). There is a significant variability among surgeons as to the ideal timing of the operation for asymptomatic CPAM, but most surgeons advocate for elective resection at 3 to 6 months of age.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here