Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Diabetic nephropathy is the major cause of end-stage renal disease (ESRD) in the industrialized world. Although the incidence of nephropathy due to type 1 diabetes may not be increasing, type 2 diabetes mellitus, considered 30 years ago a rather benign condition invariably associated with the “normal” aging process, is now the most common single cause of chronic kidney disease in the United States, Japan, and Europe. The associated health care costs are massive and not only is diabetic nephropathy a major burden on the quality of life, it also predicts an ominous prognosis despite advances in the medical management of renal and cardiovascular diseases. For instance, the diabetic patient with proteinuria has a two- to fourfold increased risk of morbidity and mortality from cardiovascular diseases. Even with chronic dialysis, the cardiac death rate of diabetic patients is ~50% higher than nondiabetic patients.
Diabetic nephropathy is the major cause of end-stage renal disease (ESRD) in the industrialized world. Although the incidence of nephropathy due to type 1 diabetes may not be increasing, type 2 diabetes mellitus, considered 30 years ago a rather benign condition invariably associated with the “normal” aging process, is now the most common single cause of chronic kidney disease in the United States, Japan, and Europe. The associated health care costs are massive and not only is diabetic nephropathy a major burden on the quality of life, it also predicts an ominous prognosis despite advances in the medical management of renal and cardiovascular diseases. For instance, the diabetic patient with proteinuria has a two- to fourfold increased risk of morbidity and mortality from cardiovascular diseases. Even with chronic dialysis, the cardiac death rate of diabetic patients is ~50% higher than nondiabetic patients.
The pathophysiology of diabetic nephropathy is complex. Renal injury was initially thought to be caused by hemodynamic alterations (renal hyperperfusion, intraglomerular hypertension, and glomerular hyperfiltration), but there is now clear evidence that these changes are only one aspect of a complex series of metabolic and biochemical alterations caused by disturbed glucose homeostasis. Hyperglycemia is a necessary prerequisite but genetic susceptibility is also crucial for the development of diabetic nephropathy. Several lines of evidence, including familial aggregation, suggest the existence of genes where allelic variation contributes to risk of diabetic nephropathy. Metabolic changes and hemodynamic stress can induce the release of vasoactive peptides, cytokines, and growth factors that can trigger a host of autocrine and/or paracrine effects that eventually mediate the effects of hyperglycemia and nonenzymatically glycated proteins on the functional alterations (hyperperfusion, hyperfiltration) as well as the structural changes (early hypertrophy, excess extracellular matrix deposition, podocyte abnormalities) of diabetic nephropathy. This chapter will provide a comprehensive review of our current understanding of the pathophysiology of diabetic nephropathy.
There are approximately 200 million people with diabetes mellitus worldwide, and this number is projected to increase to 366 million by 2030. In the United States, it is estimated that 18.8 million people (or ~6% of the population) have diabetes (ADA statistics webpage found at http://www.diabetes.org/diabetes-basics/diabetes-statistics/ ), but a significant portion of those with type 2 diabetes have not yet been clinically identified. Up to one third of patients with type 1 diabetes eventually develop nephropathy after ~20 years of diabetes. Among those with type 2 diabetes, the risk of nephropathy is less clear and varies with ethnicity. However, some European studies have suggested that the risk of ESRD in type 2 diabetic patients is almost as high as in type 1 diabetic patients. African Americans, Native Americans, Asians, and Hispanics are more prone to developing both type 2 diabetes and diabetic nephropathy than non-Hispanic whites. The reasons for this difference remain unknown but genetic factors may be implicated. There has been a secular trend of decreasing prevalence of nephropathy in type 1 diabetic patients, presumably because of progressively better management of glycemia and hypertension. Although in type 2 diabetic patients, aggressive early, multi-factorial intervention reduces the frequency of cardiovascular and renal endpoints, there was little evidence of a decreasing incidence of nephropathy in patients with type 2 diabetes. As of 2005, however, the incidence of diabetic nephropathy leading to end-stage renal disease seems to be leveling off, but whether that positive trend continues will require additional years of data to confirm (USRDS 2011). The increasing prevalence of type 2 diabetes in the aging population and the better survival of diabetic patients with nephropathy had been the major reasons for the rising numbers of type 2 diabetics with ESRD, but incident rates have recently stabilized or are falling in older populations and among whites. The sobering news is that diabetes-related ESRD is increasing among younger minority patients (USRDS 2011). Importantly, the risk of dying from cardiovascular disease is greater than the chance that one may live long enough to develop ESRD. An increasing proportion of diabetic patients can have acute but irreversible renal failure, superimposed on chronic kidney disease, usually after cardiac or septic complications. Administration of radiocontrast media agents or nonsteroidal anti-inflammatory agents may sometimes precipitate renal failure that may not resolve.
According to the US Renal Data System ( www.usrds.org , 2004; incidence data from 2002) diabetic nephropathy was the primary diagnosis in 45% of incident patients starting renal replacement therapy (i.e., 148 of 326 patients per million). This was an increase of 221% compared with 1990, (updated for USRDS 2011, incidence data from 2009, diabetic kidney disease accounted for 43% of incident patients, i.e., 154 of 354.8 patients per million). The number of type 2 diabetic patients with ESRD in the United States in 2000 far exceeded that of type 1 (38% and 6% of the total ESRD dialysis population, respectively). This is due to the much higher prevalence of type 2 diabetes in the overall diabetic population (90–95%) as compared with type 1 diabetes (5–10%). Survival of diabetic patients on dialysis is considerably worse than that for nondiabetic patients. Five-year mortality is between 40% and 80% in diabetic patients with ESRD in European countries and the United States. The main causes of death in hemodialyzed diabetic patients are primarily cardiovascular events but infection also plays a significant role. Survival rates in dialyzed diabetic patients are much better in Eastern countries such as Japan, presumably related to the lower rate of cardiovascular death in the background population of Japan.
The risk of nephropathy is strongly determined by genetic factors and only approximately 30% of patients with type 1 or type 2 diabetes will ultimately develop nephropathy. Genetic factors may directly influence the development of diabetic nephropathy and/or be clustered with genes influencing cardiovascular diseases. One support for genetic transmission is an experimental study showing that bone marrow–derived mesangial cell progenitors transmit diabetic nephropathy from donors with type 2 diabetes ( db/db mice) to naive, normoglycemic recipients. There is ongoing research through genomic screening and candidate gene approaches to better identify genetic loci for diabetic nephropathy susceptibility. Although some potential genes have been identified, linkage was only present in defined ethnic subpopulations and not in the majority of patients. An incomplete list of previously implicated genes is shown in Table 78.1 . The major problem with such studies is that diabetic nephropathy is a complex disease trait, meaning that several genes are likely involved and the inheritance patterns do not follow simple Mendelian rules. For example, a potential association between polymorphisms in candidate genes and the development and progression of nephropathy has been widely studied. These case-control studies are often problematic and clear guidelines for such polymorphism studies have been provided. The complexity of this genetic linkage analysis is exemplified by studies with controversial results investigating the insertion or deletion polymorphism of the angiotensin-converting enzyme (ACE) gene. Discrepancies could be explained by genetic heterogeneity and by small effects in limited cases in most studies (often <200 cases). The current opinion about the role of ACE gene polymorphism is that it may be associated with progression of disease in certain ethnic populations, but it is not a predictor of the development of diabetic nephropathy.
| Gene | Gene Variant |
|---|---|
| Promoter of RAGE | 63-bp deletion (decreased risk) |
| Histocompatibility antigen | DR3/4 |
| Angiotensin-converting enzyme | D/I |
| Angiotensinogen | M235T |
| Aldose reductase | Z+2 alleles |
| Transforming growth factor-β1 | Leu10Pro, Arg25Pro |
| Apolipoprotein E | e2 allele |
| Paraoxonase 1 | T107C, Leu54Met |
| Interleukin-1γ | T105C |
| Atrial natriuretic peptide | C708T |
| Glucose transporter 1 | Xba1/HacIII |
| Mannose-binding lectin | YA/YA, XA/YA |
The introduction of Genome-Wide Association Studies (GWAS) is projected to bring new insights into the genetics of diabetic nephropathy. After scanning the entire genome, this method tries to identify single nucleotide polymorphisms (SNPs) associated with a particular disease, and thus can pinpoint areas of the genome involved in renal disease for example. So far, 2 GWAS have been conducted on patient populations with diabetic nephropathy. Pezzolesi et al. identified two loci that are significantly associated with renal disease in patients with type 1 diabetes: the FRMD3 (4.1 protein ezrin, radixin, moesin [FERM] domain containing 3) and CARS (cysteinyl-tRNA synthetase). These findings were replicated using samples from the Diabetes Control and Complications Trial (DCCT)/Epidemiology of Diabetes Interventions and Complications (EDIC) study. On the other hand, using a Japanese cohort, Maeda et al. suggest that ELMO1 (engulfment and cell motility 1 gene) and SLC12A3 (solute carrier family 12, member 3) may be associated with type 2 diabetic nephropathy. This was later confirmed by studies on African-American cohorts with type 2 diabetes as well as data from the GoKinD collection which includes Caucasians with type 1 diabetes. Because it casts a wide net, GWAS should increase our understanding of the pathology of diabetic nephropathy, and help identify novel therapeutic targets. This powerful method also promises to give clinicians and researchers a better understanding of the differences in disease progression and therapeutic responsiveness, paving the way towards a personalized medical practice.
More recently, the tools of genome-wide association studies have been applied to diabetic nephropathy. This method allows for a comprehensive genetic survey of the entire genome for chromosomal regions that are linked with a specific trait, in this case diabetic nephropathy. A genome scan for diabetic nephropathy in African-Americans identified susceptibility loci on chromosomes 3q, 7p, and 18q. Another scan in Pima Indians, a native American tribe with an alarmingly high incidence of early-onset type 2 diabetes, also identified linkage to diabetic nephropathy on chromosome 7. This powerful method, or its derivative technologies, may in the future more clearly identify the genetic risk for developing diabetic nephropathy. However, the genetic findings still need to be sussed out for their functional significance and explained in the larger pathophysiological context.
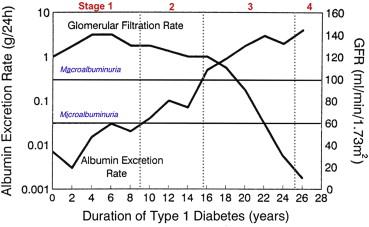
Five clinical stages characterize the progression of diabetic nephropathy ( Fig. 78.1 , Table 78.2 ). These stages are classified on the basis of the values of the glomerular filtration rate (GFR), urinary albumin excretion (UAE), and systemic blood pressure. The discrete structural lesions in the renal parenchyma and vasculature (discussed in subsequent sections) generally become more severe with advancing clinical stages, but the diagnosis of diabetic nephropathy is often made on clinical grounds without the need for renal biopsy except in atypical presentations. The stages are best delineated in the setting of type 1 diabetes because the patient is often young, typically does not have essential hypertension that can cause renal injury, and the onset of diabetes is much more easily pinpointed. In these patients, sustained albuminuria due to diabetic nephropathy rarely develops before the first 10 years of diabetes. On the other hand, if a patient has diabetes for longer than 25 years and has not developed proteinuria, the future risk of developing nephropathy is only about 1% per year.
| Stage | GFR | UAE | Blood Pressure | Years After Diagnosis |
|---|---|---|---|---|
| 1. Hyperfiltration | Supernormal | <30 mg/d | Normal | 0 |
| 2. Microalbuminuria | High normal–normal | 30–300 mg/d | Rising | 5–15 |
| 3. Overt proteinuria | Normal-decreasing | >300 mg/d | Elevated | 10–20 |
| 4. Progressive nephropathy | Decreasing | Increasing | Elevated | 15–25 |
| 5. ESRD | <15 ml/min | Massive | Elevated | 20–30 |
It should be noted that transient albuminuria can be first detected in human type 1 diabetes when the patient has uncontrolled diabetes or co-existent infections. This feature is not reflective of established diabetic nephropathy because the patient typically reverts to the normoalbuminuric state when the blood glucose is better controlled or an infection is cleared.
The natural history for patients with type 2 diabetes is not as easily characterized as in type 1 patients, because 5% to 20% of these patients have some degree of albuminuria at the time of recognition of diabetes. Unlike type 1 patients, patients with type 2 diabetes commonly have hypertension at presentation and the renal disease in these patients can be attributed to diabetes only in approximately 75% of the cases. A renal biopsy may sometimes be indicated to reach the correct diagnosis. Moreover, due to increased cardiovascular mortality, many type 2 diabetic patients die before they ever progress to ESRD. Nevertheless, longitudinal observations in the Pima Indians have revealed that the course of diabetic nephropathy in type 2 diabetes is very similar to that of type 1 diabetes. Thus, although it may be debated that diabetic nephropathies due to either type 1 or type 2 diabetes are specific entities, there is convincing evidence that basic pathophysiological mechanisms that eventually lead to nephropathy are essentially similar in types 1 and 2 diabetes. However, in type 2 diabetes cases, other noxious factors, being or not related to diabetes mellitus itself, such as hypertension, obesity, dyslipidemia, and macrovascular ischemic renal disease could additionally injure the kidney resulting in complex patterns of nephropathy. The pathophysiological changes prior to the development of type 2 diabetes have been classified as the metabolic syndrome (see subsequent sections).
The earliest renal manifestations in type 1 diabetes are nephromegaly and glomerular hypertrophy, which are accompanied by afferent arteriolar vasodilation, renal hyperperfusion (although some find that renal blood flow is reduced ), and glomerular hyperfiltration ( Fig. 78.1 ). Microscopically, there is thickening of the glomerular and tubular basement membranes. All these early functional and structural manifestations appear even if the patient is not destined to develop overt diabetic nephropathy. The UAE in this stage is normal (<30 mg/d or <20 µg/min) ( Fig. 78.1 ) but occasionally a transient increase in UAE (“transient microalbuminuria”) is present secondary to poor glycemic control or infection. Typically the blood pressure is below the hypertensive range (<140/90 mm Hg). The GFR is increased by 20 to 40% above normal values, with higher levels being frequently achieved when glycemic control is poor. Some studies suggest that patients with more renal hypertrophy or higher degrees of hyperfiltration (GFR >150 ml/min) are at increased risk for the future development of overt nephropathy. In patients with type 2 diabetes, this early stage is not readily discernible owing to the increased age but there may be modest degrees of nephromegaly and hyperfiltration.
Incipient or latent nephropathy is defined by the appearance of microalbuminuria (UAE of 30–300 mg/day or 20–200 µg/min). Without intervention, UAE increases at the rate of 10% to 20% per year and is almost always accompanied by a steady rise in blood pressure. Hypertension (>140/90 mm Hg) is typically diagnosed one to two years after the appearance of microalbuminuria in type 1 diabetes. Microalbuminuria rarely develops before five years of disease duration (median, 10 years) in type 1 diabetes, but in type 2 diabetes microalbuminuria may be present at the time of diagnosis of hyperglycemia in up to 20% of patients with as many as 40% of patients having elevated blood pressure as well. This may be due to the fact that many of these patients have had impaired glucose tolerance for years before actually being diagnosed with diabetes, or it may reflect another disease causing increased UAE as part of the metabolic syndrome, most notably essential hypertension. Other possible causes of increased UAE in this population include renovascular hypertension, morbid obesity, and sleep apnea. Even during this clinically “silent” phase of the disease, there may be significant mesangial matrix expansion or diffuse glomerulosclerosis, further thickening of the glomerular and tubular basement membranes, and some degree of podocyte loss. The GFR may remain elevated or may decrease to within the “normal” range (100–120 ml/min) ( Fig. 78.1 ). Longitudinal studies have shown that the patient with microalbuminuria is at greater risk for the development of overt proteinuria (UAE >300 mg/d) compared with patients with normoalbuminuria. For example, type 1 diabetic patients with persistent microalbuminuria have approximately an 80% chance of developing established nephropathy within the ensuing five to seven years. In contrast, the percentage of patients with type 2 diabetes who progress to overt proteinuria is significantly lower, in part because of excess mortality due to cardiovascular events. Thus, the predictive value of microalbuminuria in patients with type 2 diabetes is much less clear than in type 1 patients.
This stage is characterized by the development of overt proteinuria (total protein excretion >500 mg/day) or macroalbuminuria (UAE >300 mg/d) ( Fig. 78.1 ). In type 1 patients this occurs after an average of 15 years of diabetes. Hypertension is almost always present, and the worse the blood pressure control is, the more rapidly the GFR declines ( Fig. 78.4 ). If a renal biopsy were to be performed, the glomeruli would typically demonstrate diffuse glomerulosclerosis and/or nodular glomerulosclerosis, further podocyte loss with focal areas of foot process effacement, arteriolar hyalinosis in both the afferent and efferent arterioles, and variable degrees of tubulointerstitial fibrosis. Structure–function correlations indicate a highly significant inverse correlation between declining GFR and mesangial expansion. The progressive expansion of the glomerular mesangium causes a reduction in the glomerular filtering surface area. Nephron loss due to tubulointerstitial fibrosis is another major cause of the reduction in GFR. In the untreated patient, the GFR falls at a rate of about 1 ml/min/month, but this rate of fall can vary significantly from patient to patient. As the GFR falls from previously supernormal levels, the serum creatinine may remain in the normal range or be slightly elevated and is therefore not a reliable indicator of the magnitude of disease progression.
After approximately five years of overt nephropathy, untreated patients progress to advanced nephropathy, as characterized by nephrotic-range proteinuria (>3.5 g/d), worsened hypertension that becomes difficult to control, and a progressive decline in GFR ( Fig. 78.1 ). In fact, diabetic nephropathy is the most common cause of the nephrotic syndrome in the adult population. Parenchymal and vascular lesions become more severe. The rate of decline in GFR is steady over a period of months but is variable from patient to patient and depends on the degree of elevation of blood pressure as well as the amount of UAE.
The final stage is progressive renal failure reaching ESRD, with the GFR declining to 15 ml/min or lower. The patient may soon need renal replacement therapy to control uremic symptoms or excessive volume expansion. The average time for progression to ESRD from the time of diagnosis of diabetes is about 20 to 25 years, with a more rapid course developing in patients with uncontrolled hypertension and/or heavy proteinuria. Many patients, especially those with type 2 diabetes, never reach ESRD because of the severely increased risk of cardiovascular mortality in this population.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here