Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Biomarker development has profoundly affected basic understanding of carcinogenesis and expanded means of intervention in human cancers. Biomarkers have been applied with variable success in three broad areas that correspond to phases of tumor development and progression: (1) early detection, (2) diagnosis, and (3) prediction of clinical outcome (prognosis) and response to targeted treatment.
For early detection, biomarkers go through five stages of development: (1) identification of promising directions; (2) validation of a clinical assay; (3) demonstration that the biomarker can detect disease before it is clinically relevant; (4) evaluation of the biomarker during prospective screening; and (5) quantification of the effect of screening on reducing the burden of disease in a population.
Early detection using quite different approaches has changed the natural history of cervical, colorectal, and lung cancer. Progress depends on improving our understanding of the biology of site-specific carcinogenesis and using intervention strategies informed by basic biology.
Cancer diagnosis still relies on an evaluation of tumor tissue histologic findings, a complex process associated with interindividual variation; however, new approaches to automated quantitative histologic evaluation and immunohistochemical analyses are under active development.
New molecular diagnostic approaches that use assessment of quantitative messenger RNA signatures, fluorescence in situ hybridization, quantitation of microRNAs, and genetic sequencing can now be accomplished with accurate use of formalin-fixed paraffin-embedded tissues.
Molecular testing can provide guidance in prioritizing therapies for patients most likely to benefit and can identify patients who have been proven not to benefit from a targeted therapy.
Molecular testing can also provide clinicians with a rationale to guide specific patients toward specific clinical trials.
As new molecular techniques have been applied to cancer diagnosis and treatment, the necessity of developing standardized processing approaches for solid tumor specimens has been appreciated and has helped to change tissue collection and specimen-handling routines.
Since the time of Virchow more than 150 years ago, diagnosis and treatment of cancer have depended on the appearances of tumor cells under the microscope and the diverse and often subtle morphologic differences that distinguish benign from malignant cells. Careful study of cellular and later of molecular features of both hematologic and solid tumors led to the idea that cancer results from a series of genetic and phenotypic changes in progenitor cells that occur over a period of many years. In epithelial tumors, the final histologic changes that define transformation of premalignant lesions into malignancy are the invasion of stromal tissues and the elicitation of a stromal response including angiogenesis and metastasis to lymph nodes and distant sites. The clinical implications of this multistep process are best illustrated in the cervix, colon, and lung, where early intervention has resulted in reduced mortality from cancer. Once invasive carcinoma has occurred, testing becomes more urgent and intervention more aggressive. Recently, the great diversity of genetic changes that drive invasive cancers and the broad range of coverage needed to identify them have come into focus. This chapter reviews concepts of solid tumor carcinogenesis and biomarker development for early detection and distinguishes early detection testing from biomarker applications in diagnosis, prognosis, and treatment of established solid tumors. Hematologic biomarkers are described elsewhere in this book.
Remarkable strides have been made in reducing morbidity and mortality through early intervention in several different kinds of solid tumors. The triggers for intervention have until now been largely based on cytologic evaluation of exfoliated cells or advances in imaging technology. To date, few molecular markers have been sufficiently sensitive, specific, or accessible to be useful in population-based studies for early detection or prevention.
There are several reasons for this. Sensitive biomarkers for risk assessment and early detection must be inexpensive, noninvasive, and easily applied to be useful for population-based screening. However, such biomarkers are rarely sufficiently specific for diagnosis, prognosis, or response to treatment. A formal process to guide the development of early detection biomarkers has been defined by Pepe and colleagues. In this construct, biomarkers must pass through five phases of development that correspond to the strength of evidence that the biomarker will be a useful population screening tool. These phases of development are analogous to the structure for therapeutic drug development that has been used for many years. Pepe's phases are listed in Table 15.1 and are the framework for the discussion of biomarker development in individual organs later in this chapter.
| Phase | Category | Descriptor |
|---|---|---|
| 1 | Preclinical/Exploratory | Promising directions identified |
| 2 | Clinical Assay and Validation | Clinical assay detects established disease |
| 3 | Retrospective/Longitudinal | Biomarker detects disease early, before it becomes clinically evident and a screen-positive rule is defined |
| 4 | Prospective Screening | Extent and characteristics of disease detected by the test and the false referral rate are identified |
| 5 | Cancer Control | Impact of screening on reducing the burden of disease on the population quantified |
As will be seen, biomarker development for early detection has been uneven across the various organs sites, and successes and challenges faced for three exemplary organs are placed in context below. These three organs were chosen because they exemplify organ sites where early detection trials have demonstrated reduction in disease morbidity and mortality but where success of early detection biomarker development has been variable. The organ site for which biomarker development is most advanced is the cervix, wherein a clear viral cause has been identified and where the constituents of that virus can be used for risk assessment, early detection, diagnosis, triggering colposcopic intervention, and vaccination, achieving Pepe's stage 5 status of changing clinical practice as described later.
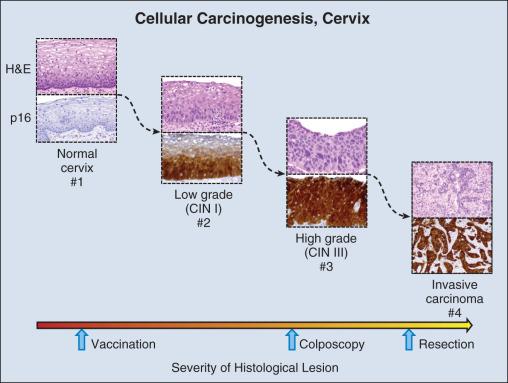
Invasive cervical carcinoma is one of the few tumors that results from well-defined premalignant lesions that can be identified and effectively treated, reducing morbidity and mortality from a tumor that is the third most common in women worldwide. More than 60 years ago, it was recognized that cellular abnormalities in cervical epithelium detectable in exfoliated cells could predict the presence of carcinoma. By 1988, premalignant cervical cytology had been codified as the Bethesda system for reporting cervical and vaginal cytologic diagnoses. Population-based light microscopic screening for atypical cells in cervical smears with surgical removal of detected abnormal epithelium in Great Britain is estimated to have reduced the prevalence of invasive squamous carcinoma by 80% after 1988.
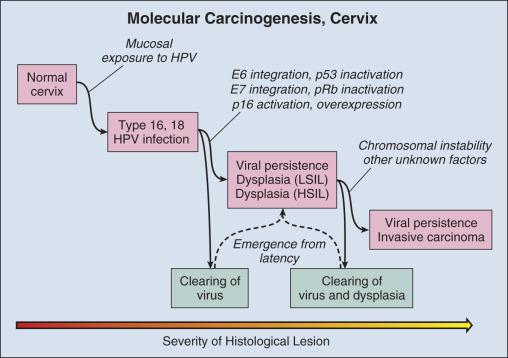
Delineation of molecular carcinogenesis in the cervix ( Figs. 15.1 and 15.2 ) has provided alternative methods for detection, risk assessment, and diagnosis. Nearly all cervical carcinomas and their precursor lesions are caused by human papillomavirus (HPV). HPV is a DNA tumor virus shown by Peyton Rous to produce squamous tumors in rabbits in 1934. It was not until 1974 that zur Hausen recognized the association between HPV and cervical cancer. Persistent infection with high-risk strains of HPV, most commonly types 16 and 18, is an essential step in cervical carcinogenesis (reviewed in zur Hausen ). The virus encodes three proteins that stimulate cell proliferation and survival, E5, E6, and E7 (reviewed in zur Hausen ). The viral proteins E6 and E7 interfere with p53 and Rb, respectively resulting in resistance to apoptosis and chromosomal instability. The HPV genome may be integrated into the host cell genome, accounting for long-term persistence of the virus. Integration of E6 and E7 genes is crucial to progression of the infected cell to a malignant state, whereas other viral genes may be lost.
Detection of HPV DNA in clinical samples can be accomplished by a number of methods, with the dominant techniques being based on PCR or hybridization with signal amplification (reviewed in Leal and Gulley ). These include the Hybrid Capture 2 method (HC2, Qiagen), COBAS (Roche Molecular Diagnostics), and Aptima HPV (HOLOGIC) assays, which have similar sensitivities and specificities ; the latter two systems provide genotyping information in addition to simple detection information.
Understanding the molecular oncogenesis of cervical squamous carcinoma has provided a standardized biomarker of risk. More than 95% of HPV infections clear without further consequences. However, persistent infection for months to years with HPV types 16 or 18 carries a 40% risk of squamous intraepithelial lesions (SIL) ; untreated high-grade SILs have been estimated to result in invasive carcinoma in approximately 30% of individuals over 5 to 10 years after diagnosis. Persistent HPV infection thus indicates high risk for eventual invasive carcinoma and represents an additional biomarker that could be useful in the prevention of invasive carcinoma.
Testing for HPV DNA has been found to be more sensitive than cytologic screening for detecting high-grade SIL but somewhat less specific. In view of the number of infections that are self-limited, relying on a single HPV test may result in greater numbers of negative colposcopy examination results. To address this problem, cotesting strategies have been evaluated whereby both cytologic evaluation and HPV testing are performed on initial examination. Negative results have a high negative predictive value and the rescreening interval can be extended from 1 to 5 years, reducing overall screening expense. Individuals with a positive viral DNA test result but negative cytologic findings have been rescreened at 1 year and examined with colposcopy if results are positive. Whether this or other strategies will be universally adopted for cervical cancer risk management remains to be determined. What is clear is that HPV testing provides a means for improving sensitivity for detection of early cervical lesions, further reducing mortality from invasive cervical carcinoma while potentially reducing screening expense.
Finally, HPV not only is a biomarker for risk assessment and colposcopic intervention, but also is an immunogen that has been successfully incorporated into bivalent (types 16 and 18; Cervarix), quadrivalent (types 6, 11, 16, and 18; Gardasil), and nonavalent (types 16, 18, 31, 33, 45, 52, 58, 6; and 11; Gardasil 9) vaccines to prevent infection and carcinogenesis (reviewed by Stanley and Villa ) in both female and male patients. In-depth discussion of HPV vaccines for tumor prevention is beyond the scope of this chapter. However, the many facets of HPV expression in cervical carcinoma and the emerging role of HPV in other tumors including vulvar and vaginal condylomata, certain skin cancers, and head and neck carcinoma illustrate how a valuable biomarker that is integral to oncogenesis can be used to improve the accuracy of estimating tumor risk and the specificity of treatment. Unfortunately, such clearly defined and easily accessible biomarkers are rare. Rather, the more usual case is described here for colon and lung, wherein potential biomarkers are defined but have not found a role in early detection, diagnosis, prevention, or treatment.
Like cervical carcinoma, colonic adenocarcinoma can be effectively prevented by extirpation of premalignant epithelium, which in colon is represented by polyps. Several trials have indicated that removal of polyps through colonoscopy reduces the frequency of colorectal carcinoma (CRC), and consensus opinion is that endoscopic polypectomy has a significant impact on the incidence of CRC. Data have also suggested that colonoscopic screening reduces CRC mortality by more than 50% and that the less extensive sigmoidoscopy reduces mortality from distal CRC by a nearly equal percentage (50%). The latter sigmoidoscopic study was a large randomized population-based screening trial. Colonoscopic screening for colon polyps beginning at age 50 has become a standard of care.
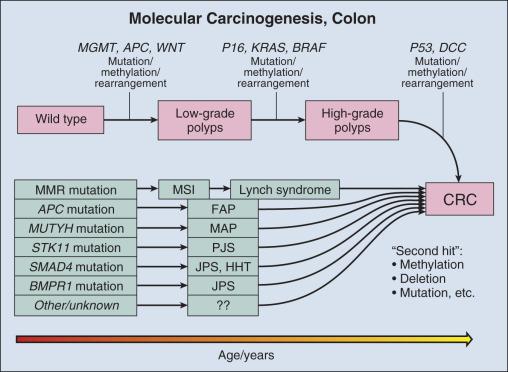
Molecular biomarkers to date have not played a direct role in reduction of morbidity and mortality in CRC. However, molecular studies have facilitated understanding of colon carcinogenesis ( Figs. 15.3 and 15.4 ) and provided a rationale for colonoscopic surveillance and polypectomy. In the mid-20th century an association between between benign polyps and invaive carcinoma was suspected, but at that time there was no direct evidence that polyps undergo transition to CRC. Suspicions were finally confirmed in the 1980s, when the clonal nature of both sporadic polyps and CRC was documented. Activating mutations in the KRAS oncogene and inactivating mutations in several tumor suppressor genes (TSGs) were described. The same mutations were demonstrated in both polyps and carcinoma, and the number of mutations was found to increase with histologic grade of the adenoma. This suggested a progression to invasive carcinoma through the accumulation of genetic and epigenetic changes. These changes provide a proliferative and survival advantage to mutant cells, which continue to proliferate and mutate until a malignant phenotype is reached. The combined data from these studies led to the proposal of a multistep model of colon carcinogenesis, which continues as the standard model.
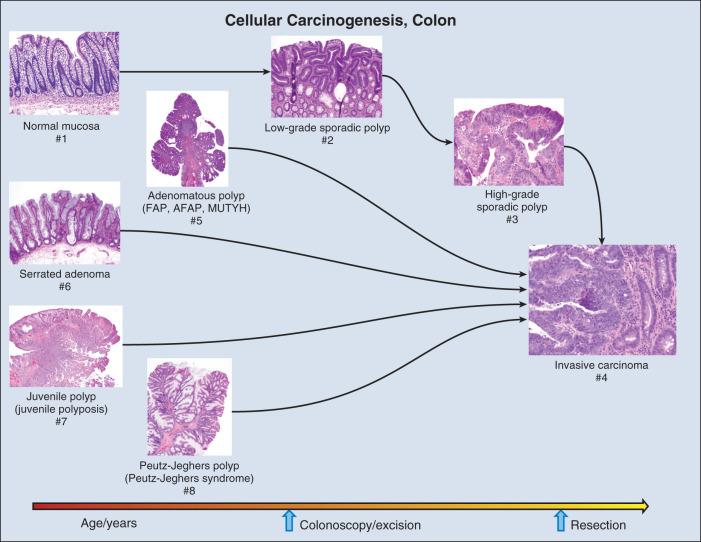
Further insight into colon carcinogenesis has been provided by studies of hereditary forms of colon cancer and the molecular changes that underlie them. A detailed discussion of hereditary colon cancer is provided elsewhere in this book and is beyond the scope of this chapter. However, a brief description of the major forms of hereditary tumors will provide insight into colon carcinogenesis and perspective on the strengths and limitations of the role of genetic data in early detection of this prevalent tumor type.
Several forms of hereditary colon cancer are recognized (reviewed by Jasperson and colleagues ) and are listed in Table 15.2 .
| Syndrome (Abbreviation, Synonym) | Gene(s) | Molecular Phenotype | Noncolonic Organs Affected |
|---|---|---|---|
| Sporadic adenomatous polyp | Unknown | Chromosomal instability (aneuploidy) | None known |
| Familial adenomatous polyposis (FAP) | APC | Chromosomal instability (aneuploidy) | Duodenum, stomach, pancreas, thyroid, liver, central nervous system |
| Attenuated familial adenomatous polyposis (AFAP) | APC | Chromosomal instability (aneuploidy) | Duodenum, thyroid, liver, central nervous system |
| Hereditary nonpolyposis colon cancer (HNPCC, Lynch syndrome) | hMLH1, hMSH2, hMSH6, hPMS2 | MSI | Endometrium, stomach, ovary, biliary and urinary tracts, small bowel, central nervous system |
| MUTYH-linked adenomatous polyposis (MAP) | MUTYH | Chromosomal instability (aneuploidy) | Duodenum |
| Peutz-Jeghers syndrome (PJS) | STK1 (70%) | Unknown | Breast, pancreas, stomach, ovary, lung, small bowel, uterus, testis |
| Juvenile polyposis syndrome | SMAD4, BMPR1A | Unknown | Blood vessels (hereditary hemorrhagic telangiectasia) |
| Hyperplasic polyposis | SMAD4, BMPR1A (40%), PTEN | Unknown | Stomach, pancreas, small bowel |
The first hereditary colon carcinoma to be described, familial adenomatous polyposis (FAP), is characterized in its classic form by the occurrence of hundreds to thousands of polyps in the colon of affected individuals with the formation of invasive carcinomas at an early age. Polyps may also sometimes be found in the upper gastrointestinal tract. As long ago as 1882, siblings were described with numerous and recurring polyps, and by the mid-20th century FAP had been established as a hereditary autosomal dominant disorder. A gene mutation associated with FAP was identified on chromosome 5q by linkage analysis and positional cloning studies. The gene, adenomatous polyposis coli (APC) , is now known to encode a tumor suppressor that modulates Wnt signaling through binding and stabilization of a complex of proteins that includes GSK3b, β-catenin, and axin family members. Mutations in APC truncate the protein at its carboxyl terminus and disrupt the protein signaling complex. This results in deregulation of cell cycle control, cell migration, differentiation, and apoptosis (reviewed by Goss and Groden ). In addition to being responsible for FAP, APC mutation is present in approximately 60% of sporadic colon adenocarcinomas and an approximately equal number of sporadic polyps, suggesting that APC mutation is an early event in sporadic carcinogenesis as well as in hereditary disease. An attenuated form of FAP with smaller numbers of tumors (<100) has been described. This condition is referred to as attenuated familial adenomatous polyposis (AFAP) and may be driven by the mutations in APC in the 3′ and 5′ ends rather the central portion of the gene that is affected in classic FAP.
The most common form of hereditary colon cancer is hereditary nonpolyposis colorectal cancer (HNPCC). HNPCC is a familial syndrome that differs from FAP by involving smaller numbers of polyps in comparison to FAP. The earliest description of this disorder can again be traced back to the early 20th century, but the hereditary character and syndromic features of the disease were firmly established by the diligent kindred studies of Lynch and colleagues, after whom the syndrome is named (Lynch syndrome). In brief, the syndrome consists of CRC occurring in close relatives, often younger than age 50, without adenomatous polyposis. HNPCC is also associated with an increased frequency of tumors in the endometrium, stomach, ovary, hepatobiliary tract, upper urinary tract, small bowel, and central nervous system (CNS). In contrast to FAP polyps, which are frequently found on the left side of the colon, individuals with HNPCC frequently have flat lesions on the right side of the colon with a distinct histologic appearance referred to as serrated adenoma (see Fig. 15.3 ).
The molecular genetic underpinnings of HNPCC were deciphered beginning with the linkage to microsatellite markers on chromosome 2p. The genetic basis of the disease was established when it was found that patients with the syndrome harbored mutations in the mismatch repair (MMR) genes hMLH1, hMSH2, hMSH6, and hPHS2, with mutations in hMLH1 and hMSH2 accounting for over 90% of HNPCC cases. The germline mutations are heterozygous, and colon carcinogenesis results from silencing of the nonmutant allele by methylation or by acquired allelic loss.
The phenotypic consequence of gene silencing is loss of MMR protein and the resulting inability to repair DNA replication errors, ultimately leading to accumulation of mutations contributing to carcinogenesis. At a clinical level, protein loss can be demonstrated by immunochemistry and optimally requires four separate stains. The inability of affected cells to repair DNA replication errors results in insertions or deletions in short tandem repeats (microsatellites) that exist throughout the genome and is referred to as microsatellite instability (MSI). Currently, MSI is documented by testing of a standard set of five microsatellite markers recommended by a consensus panel. Results are scored on the basis of the number of unstable loci and are classified as microsatellite instability–high (MSI-H), microsatellite instability–low (MSI-L), or microsatellite instability–stable (MSI-S). More than 90% of HNPCC tumors are MSI-H, but approximately 15% of all sporadic tumors are also MSI-H.
A third hereditary variant of colon carcinoma is MUTYH-linked polyposis (MAP), the newest polyposis syndrome, first described in 2002. This is a disorder similar to AFAP and is characterized by adenomatous polyps that may number in the hundreds but not in the thousands, usually ranging from 10 to 1000. Polyps may also occur in extracolonic sites, usually the duodenum. Adenomas tend to occur in the proximal colon. MUTYH is a glycosylase and part of the base excision repair (BER) pathway. Oxidative damage to DNA frequently results in the modification of guanine (G) to 8-oxoG. 8-oxoG can pair with adenine (A) during DNA replication, a mismatch that is recognized by MUTYH, which removes the mismatched A and restores the correct base, cytosine (C). The damaged G (8-oxoG) is then replaced with an intact G. Mutation of MUTYH at specific hot spots hinders recognition of the 8-oxoG:A mispairing, resulting in G:C to A:T transversion at sites of oxidative damage. The mutational hot spots are found in exon 7, resulting in amino acid substitution Y179C, and in exon 13, resulting in substitution G496D. Germline mutation at both hot spots (biallelic mutation) occurs in 0.3% of patients with CRC. Biallelic mutation confers 28-fold relative risk for CRC compared with controls. Monoallelic mutation is more common, with a frequency of approximately 2% in CRC, but also occurs at approximately the same frequency in control populations. Most studies suggest that monoallelic mutation confers little or no increased risk of CRC. Among control patients without polyposis, biallelic mutation is rare.
The fourth hereditary form of colon cancer are the hamartomatous polyposes, a category that includes Peutz-Jeghers syndrome (PJS) and juvenile polyposis syndrome. First described in the 1890s, PJS was named for the work of Peutz and Jeghers in 1954 by Bruwer. PJS is an autosomal dominant condition characterized by multiple histologically distinct polyps (see Fig. 15.3 ) occurring in the large and small bowel in association with melanin pigmentation of the lips, mouth, nose, anus, and extremities. The condition is associated with a high risk for cancer in not only the large and small intestine but also in the esophagus, stomach, pancreas, breast, uterus, ovary, and lung. Gene mapping has identified a locus on chromosome 19.3p13 that encodes a serine/threonine kinase, STK11 (LKB1), a gene that is mutated in more than 90% of affected individuals. A variety of mutations including deletions, truncations, and missense point mutations may occur over nearly the full coding region of the gene. STK11 protein is multifunctional, playing a role in cell cycle arrest, wnt signaling, and the TSC pathway, with effects on mammalian target of rapamycin (mTOR) signaling. Mutation, often associated with loss of heterozygosity (LOH), results in loss of function, and the gene is thus considered to be a TSG. The overall frequency of PJS is estimated at 0.5 to 2.0 per 100,000 live births.
Juvenile polyps are characteristically solitary peculated lesions that contain tortuous, often cystically dilated crypts, stromal granulation tissue, and inflammation and denudation of the surface epithelium. These lesions frequently occur in children, and there is no evidence that solitary juvenile polyps pose an increased risk for carcinoma. However, multiple juvenile polyps may occur in young individuals and their family members, and the presence of more than five juvenile polyps or a single juvenile polyp in an individual with a family member with juvenile polyposis now defines the condition juvenile polyposis. This form of polyposis carries a lifetime risk of colon carcinoma of 39% and a relative risk of 34. Fifty percent to 60% of affected individuals carry germline mutations in SMAD4 or BMPR1A. Nearly all patients with SMAD4 mutations also have hereditary hemorrhagic telangiectasia (overlap syndrome). Both SMAD4 and BMPR1A are involved in the transforming growth factor–β (TGF-β)/bone morphogenetic protein (BMP) pathway.
Finally, a variety of polyposis syndromes with potential genetic linkage have been described and are best exemplified by hyperplastic polyposis syndrome (serrated polyposis syndrome). This condition consists of the occurrence of multiple sessile polyps variously referred to as hyperplastic polyps or serrated adenomas, frequently on the right side of the colon and numbering from 5 to more than 100. Although familial associations of the syndrome and linkage to high risk of colon carcinoma have been reported, the genetic basis is complex and associated with mutations in several driver genes including BRAF and KRAS. No single genetic mechanism has been linked to the syndrome, and the true frequency is not established. To date, the number of reported cases is small.
Although the number of highly penetrant mutations that predispose to colon carcinoma is large, the proportion of colon carcinomas that can be accounted for by these mutations is approximately 5% and has been overestimated in the past. Markers of hereditary predisposition and molecular progression in sporadic tumors that have been delineated to date have not been easily converted into an early detection test suitable for broad population-based applications for several reasons. First, the types of accessible specimens that can be used for testing for early detection consist primarily of blood and fecal DNA. Although DNA from invasive tumors can be detected in the blood by PCR-based mutation testing, it is not as useful at earlier polyp stages. In addition, collection and processing of fecal specimens for a biomarker that is expected to be present at low level and heavily contaminated by nonneoplastic cells is a logistic challenge. Second, some of the genetic abnormalities identified in colon carcinogenesis do not readily lend themselves to screening assays. Mutations in APC, for example, consist mainly of truncations and deletions that may occur over a wide region of the gene, so designing a sensitive clinical assay has been difficult. Finally, wide availability of colonoscopy services that combine polyp detection and excision reduces the cost and improves the attractiveness of direct and systematic colonoscopic population-based surveillance.
Biomarkers that have been most successful for colon cancer screening are fecal tests such as the guaiac fecal occult blood test (FOBT), the fecal immunochemical test (FIT), and the stool DNA (sDNA) test. The greatest experience has been with the FOBT. This test is based on a color change whereby α-guaiaconic acid extracted from the wood of tropical Guaiacum tree is rapidly oxidized by hydrogen peroxide to a blue quinone in the presence of heme derived from blood. There is wide variability in results as a consequence of the level of compliance with collection methods, type or brand of kit, frequency of testing, user interpretation, and other factors. Despite a lack of standardization, FOBT screening with follow-up colonoscopy is reported to reduce both incidence and mortality from colon carcinoma. The former is attributed to polyp removal. FIT is a second test for occult blood and is reported to be more sensitive and specific than the guaiac assays. It has been shown to have a cancer detection rate similar to colonoscopy but has a lower sensitivity for polyps.
Occult blood tests measure host response (hypervascularity and bleeding) to tumor. The sDNA test, however, aims to measure a direct product of tumor cells. A recent version of the sDNA test measures multiple markers in stool using an array-based platform and for this reason has been referred to as “next generation,” although this technology does not employ deep sequencing methods and should not be confused with “next-generation sequencing.” The biomarkers that are incorporated into the platform include four methylation probes, as well as mutated KRAS and hemoglobin. Patterns of gene methylation have been identified in colon tumors that have been characterized as CpG island methylation (CIMP) high and CIMP low (reviewed by Kim and colleagues ). When applied to a preliminary cohort of 678 subjects with polyps or CRC, the combination test including methylation markers, KRAS mutation, and occult blood had a sensitivity of 85% for carcinoma and 54% for polyps.
Despite the availability of these fecal-based tests, the target in early colon cancer intervention appears to be shifting from detection of invasive carcinoma to prevention by detection and thorough clearing of adenomatous polyps from the colon. Defining the biology of colon carcinogenesis and confirming that polyps are the precursor lesions along with improvements in colonoscopic methods have led to reduction in mortality from colon cancer. Colonoscopy enables direct identification of potential precursor lesions and at the same time provides a means for ablating them. This is strikingly evident in the new sigmoidoscopy findings indicating that whereas sigmoidoscopy reduces mortality from visible distal colon tumors, mortality from proximal lesions is unaffected. It seems likely that colonoscopy will be the main vehicle for early detection and intervention in colon cancer for the foreseeable future.
For several decades lung carcinoma has had the highest mortality of all tumors, accounting for more than twice as many deaths as the next most lethal tumor. Initial large-scale efforts during the 1970s and 1980s to reduce mortality by early detection using chest radiography and/or sputum cytology were not successful. Chemoprevention using β-carotene and/or α-tocopherol dietary supplementation also failed. However, early detection efforts have begun to yield some success in reducing mortality even in this problematic tumor. Low–radiation dose computed tomography (LDCT) has a higher clinical sensitivity for detecting smaller tumors than chest radiography. Early studies in smokers documented a 2.7% prevalence of malignant disease detected by LDCT versus 0.7% in the same patients screened by chest radiography. In a multicenter study of more than 30,000 smokers, LDCT revealed lung cancer in 1.3% at baseline and an additional 0.3% at annual follow-up examinations, for an overall total detection rate of approximately 1.5%. Although this study was not randomized and had no unscreened control arm, it showed a survival advantage in comparison to historical controls. Finally, a large randomized trial of computed tomography (CT) versus radiography conducted annually in three rounds found that LDCT enabled detection of carcinoma in 2.4% of the screened high-risk population and resulted in a 20% reduction in lung cancer mortality that was not observed in the chest radiograph control group. The weight of evidence supporting improved long-term mortality rates in smokers has prompted the publication of multisociety consensus guidelines recommending annual LDCT screening for smokers and former smokers aged 55 to 74 years with access to appropriate follow-up. This study has raised awareness of possible benefits of early detection of lung cancer but has been criticized for possible overdiagnosis, a question that is currently unresolved.
What is clear is that lung carcinogenesis is a multistep process and that premalignant lesions exist in both central and peripheral airways ( Figs. 15.5 and 15.6 ) before invasive carcinoma occurs.
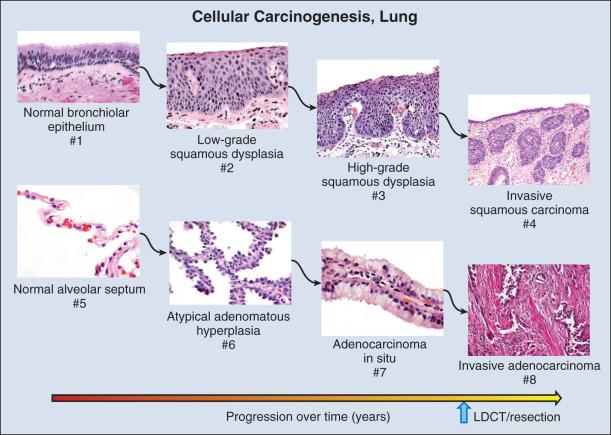
The predominant premalignant lesion of the central airways is squamous dysplasia (also referred to as atypia). Squamous dysplasia was documented over 50 years ago in association with carcinoma as well as in smokers without concurrent carcinoma. Central squamous lesions are flat, inconspicuous, and only rarely identified by white light fiberoptic bronchoscopy. They are somewhat better visualized with laser-induced fluorescence emission (LIFE) bronchoscopy. This method relies on a loss of autofluorescence and signal from dysplasias; it may be mimicked by fluorescence loss caused by mucosal hemorrhage and other reactive changes, so whereas sensitivity of LIFE bronchoscopy is high, specificity is low.
Prospective studies on the power of dysplasia to prospectively predict future central airway malignancy are few and face the forbidding challenges of inaccessibility of the bronchi and the prolonged follow-up that is required to achieve significant results in a sufficiently powered cohort. Sputum studies indicate that atypia of cells exfoliated from the central airways is associated with elevated risk for incidental carcinoma. Hazard ratio increases with proximity of sputum collection date to lung cancer diagnosis, but sputum atypia even 3 years before lung cancer diagnosis is associated with increased risk of lung carcinoma.
Bronchial biopsy is an alternative way to evaluate the status of the airway in high-risk smokers. A recent prospective study of 188 patients who underwent biopsy at mapped bronchial sites found that the persistence of high-grade dysplasia at the same bronchial site was associated with future squamous carcinoma but not adenocarcinoma, suggesting that persistent dysplasia may be a marker of progressive field change. This study also illustrates the multiyear timeline and multifocality of premalignant changes.
The corollary question of whether ablation of premalignant central airway lesions could reduce lung cancer mortality remains an open one. The findings of a chemoprevention study using iloprost, a prostacyclin agonist, have suggested that reversal of premalignant dysplasia morphological features may be possible, but the study was a small one and the end point was purely histological. The long-term effect on mortality of agents that may alter the premalignant morphologic characteristics of central airway epithelium remains unknown.
Atypical adenomatous hyperplasia (AAH), adenocarcinoma in situ (AIS), and pure lepidic carcinomas consist of alveolar epithelial cell proliferations that do not invade the alveolar wall or elicit a significant stromal response. AAH and AIS are small (<5 mm) solitary lesions that are usually recognized incidentally in screening trials or in clinical practice by their ground-glass appearance on CT imaging. AAH and AIS are characterized by indolent growth rate and 100% survival after resection. Larger solitary ground-glass lesions of pure lepidic carcinomas as well as some lesions with areas of consolidation and even multifocal ground-glass lesions also are characterized by slow growth and excellent prognosis and may persist for many months to years without changing size or evincing evidence of alveolar wall invasion. It appears that the time to progression of premalignant lesions in both central and peripheral airways is long and capricious, complicating assessment of prognosis and intervention strategies.
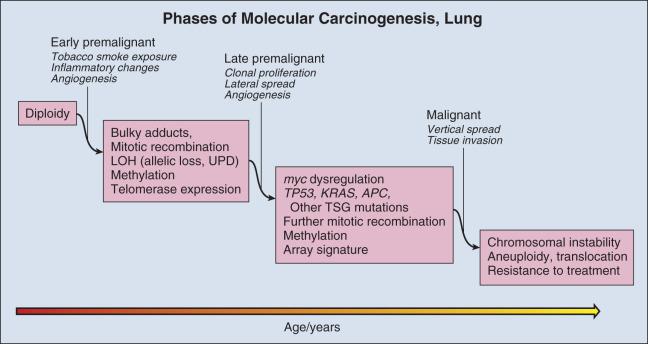
As in other organs, most notably the colon, molecular changes that accompany the morphologic changes of dysplasia have been investigated in considerable detail. These studies are informed by the well-established role of tobacco smoke as a carcinogen in lung cancer. The dominant carcinogenic compounds found in cigarette smoke are the polyaromatic hydrocarbons (PAHs) and nicotine-derived nitrosamine ketone (NNK). These compounds are metabolized to highly reactive intermediates that are inactivated largely by mitochondrial glutathione transferases (GSTs). However, repair is imperfect and a small proportion of the intermediates react directly with DNA, forming bulky adducts. Unless removed by the DNA repair system, adducts can create errors of replication. Over time, molecular alterations accumulate, providing a selective advantage to mutant cells for growth and survival.
Components of this pathway have been associated with increased risk for future lung cancer. Case control studies indicate that current smokers with tumors have higher levels of adducts in blood or tissue than non-tumor controls. However, bulky adducts appear to reflect ongoing smoking-related DNA damage rather than biologic changes specifically associated with malignant transformation. Clinically applicable adduct levels predictive of lung cancer have not been established.
GST and DNA repair enzyme mutations and polymorphisms that affect DNA repair activity could represent a risk factor for lung cancer because efficient deactivation of reactive carcinogens and repair of DNA damage caused by bulky adducts are important in protecting potential precursor cells from critical mutational changes. However, single-nucleotide polymorphism (SNP) association studies in many carcinogen-metabolizing and DNA repair enzymes have demonstrated only weak and inconsistent associations and have not been sufficiently informative to guide intervention in potentially affected individuals.
DNA damage in lung premalignancy engenders a number of genetic and epigenetic changes that provide a cellular survival or proliferative advantage to affected bronchial epithelial cells. Perhaps the earliest molecular events may be epigenetic and include DNA methylation, histone modification, and alterations in miRNA expression (reviewed by Jones and Baylin and Berger and colleagues ). The greatest experience regarding the use of epigenetic changes in the context of early detection of lung cancer has been obtained with regard to methylation. Methylation refers to the conversion of cytosine to 5′-methylcytosine by DNA methyltransferase (DNMT). In lung carcinogenesis, methylation preferentially occurs in CpG-rich regions (CpG islands) that are present in normally unmethylated promoter regions of critical genes (hypermethylation) and may silence gene expression. In lung tumors, interest has focused on genes such as p16, which are silenced in tumors but expressed in normal control tissue, and are considered to be TSGs. The development of a simple and sensitive assay based on the conversion of methyl cytosine to uracil in the presence of metabisulfite followed by PCR with primers specific for the methylated bases, methylation-specific polymerase chain reaction (MSP), has greatly facilitated the assessment of methylation status as a risk marker for lung carcinoma.
Methylation of key TSG promoter regions in cells exfoliated from bronchial surfaces into the sputum is nearly universally associated with prevalent lung carcinomas and frequently also occurs in sputum of smokers without tumor. TSG methylation is not present in sputum of never smokers. This finding initially led to optimism that methylation could be used to screen selected populations at risk for lung carcinoma. However, the predictive power of single-gene methylation or methylation of a small number of genes is limited by the ubiquity of methylation in smokers and the consequent high rate of false-positive results. This problem led to the strategy of testing promoter regions in large numbers of genes to estimate overall methylation burden. Identification of panels of genes to optimize the predictive power of methylation testing is a process that is still underway. In receiver operating characteristic (ROC) curve analyses, a seven-gene methylation profile selected from a 31-gene panel achieved a predictive accuracy of 71% to 77%. However, with a sensitivity set at 75%, the false-positive rate was 75%. Improved results were obtained in a case-control study of CT-detected nodules by using a three-gene combination having a sensitivity and specificity of 98% and 71%, respectively. Although TSG promoter methylation may prove problematic as a general screening tool, it may be useful in specific niche populations or to supplement other methods of screening such as LDCT, and ongoing studies aim to further evaluate the use of this approach.
A second molecular consequence of tobacco exposure is chromosomal instability, which is detected as aneuploidy with fluorescence in situ hybridization (FISH) in bronchial epithelial cells in situ and in sputum. Aneuploidy is nearly universal in lung carcinoma. It is also frequently observed in high-grade dysplastic lesions in the central airways. Aneuploidy in sputum has been associated with prevalent carcinoma and has recently been shown to have a sensitivity of 76% within a detection window of 18 months before the appearance of invasive carcinoma. However, FISH performed on sputum is labor-intensive and difficult to perform both in the specimen preparation and slide interpretation, requiring a high level of expertise at every step. This has led to the development of automated methods of FISH screening. However, to date these automated methods have not been broadly applied. It seems likely that FISH may most successfully be applied in restricted settings in conjunction with other technologies.
Mutations in several tumor-associated genes have been documented in adjacent nonmalignant airways or in subjects at high risk for carcinoma but without concurrent malignancy. Chief among these is TP53, which marks clonal populations that may spread laterally across the bronchial surfaces without invading stroma. Mutation in TP53 appears late in lung carcinogenesis; and although it is one of the most commonly mutated gene in lung carcinoma, it is mutated in only half of tumors and in fewer benign airways. It seems unlikely that single-gene mutation analysis will be sufficiently sensitive for use as a screening tool.
High-throughput methodologies have been suggested as possible early detection tools. Genome-wide association studies (GWASs) have identified an SNP variant at 15q15.2 that is significantly associated with cancer risk, with an overall odds ratio of 1.15. The low odds ratio suggests that the SNP variant itself will not represent a useful predictor. However, this region of the gene is still being explored for more powerful biomarkers.
Gene expression microarrays have identified consistent changes in the transcriptome of epithelium brushed from nonmalignant bronchial surfaces that distinguish smokers from nonsmokers. An additional set of changes are reported to identify subjects with cancer from smokers without cancer with an accuracy of 83%. Finally, large-scale genomic sequencing studies have identified differences in the “genomic landscape” between smokers and nonsmokers. It is expected that manageable numbers of biomarkers and clinically applicable testing platforms will emerge from these exploratory studies that will improve risk assessment and management of high-risk smokers as well as patients with already invasive carcinomas.
Premalignant cellular and molecular changes are described in several other tumors including breast, prostate, bladder, and esophagus. Detailed review of these changes may be found in appropriate chapters of this text. However, to date, identification of these changes has not led to mortality reductions.
The examples described above illustrate the uneven progress that has been made and the challenges that remain in reducing morbidity and mortality from cancer. What emerges from this review is that progress depends on improved understanding of site-specific carcinogenesis and intervention strategies that are informed by basic biology. Relatively simple technologies such as cytologic evaluation or HPV testing can profoundly reduce cancer burden. Understanding the biology of premalignancy provides a lead-time advantage that can be translated into greatly reduced cancer morbidity and mortality. Appropriate molecular strategies may supplement and eventually replace morphology but will require thorough validation.
Histology of tumor tissue has been the bedrock on which diagnosis of malignancy is established and is the basis for selecting appropriate testing to guide treatment. Histologic diagnosis, although simple in concept, is extremely complex in practice and crucially depends on the expertise of the examining pathologist. In solid tumors, a nearly universal feature of malignancy is the capacity of tumor cells to invade and replace stromal tissue, and nearly all of the following discussion refers to destructive, invasive malignant tumors that form solid masses. The primary tool of the pathologist remains the hematoxylin-and-eosin stained section, but this is now supplemented by an arsenal of immunohistochemical stains and other tissue testing procedures that permit much greater specificity of diagnosis than was previously possible. Diagnostic immunohistochemical tests are relatively simple and inexpensive and can be assessed in conjunction with in situ cellular architecture, which ensures a high level of specificity and permits an assessment of the relevance of results to the overall clinical and anatomic features. Interpretation of immunohistochemical data is now assisted by online databases that provide frequencies of immunohistochemical results in specific tumors and organ sites. Histologic diagnosis was discussed earlier in the context of early detection and is addressed elsewhere in this book in relation to individual tumor types.
Although histologic evaluation remains the primary method for diagnosis of solid tumors, molecular methods are increasingly influential not only in identification of treatment targets as discussed here, but also in tumor classification. In CNS tumors, for example, molecular findings are such powerful predictors of biologic behavior that molecular features have been formally incorporated into the diagnostic classification of this complicated group of neoplasms (see later). Both molecular and cellular diagnostics continue to depend not only on consistent and effective fixation and processing of tissue, but also on reliable accessioning and informatics.
Molecular and cellular tissue testing is a multistep process ( Fig. 15.7 ), and success requires tight control of collection, accessioning, fixation, dissection, extraction, and storage of tissue as discussed in the following sections.
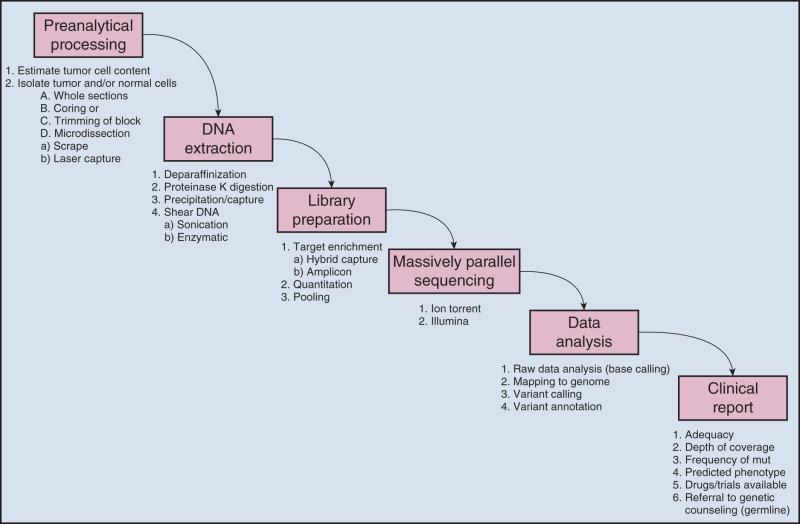
Registering patients and biopsy specimens is a demanding exercise that is often overlooked and underfunded. Although numerous and variable protocols are available for tracking specimens, virtually all tracking systems include a secure electronic database with password-limited access and reliable backup. Appropriate specimen identifiers, which today are increasingly bar-coded, access to signed consent forms when associated research studies are underway, and timely data entry are all essential for any well-run laboratory. A detailed discussion of laboratory information systems for molecular testing is beyond the scope of this chapter but must be a major consideration in the management and quality assurance programs of all clinical molecular testing laboratories.
Tissues obtained for solid tumor molecular testing may come from surgical resection or biopsies (fine-needle aspirates and cores). Most often, the tissue needed for molecular testing is derived from diagnostic material, unless special arrangements are made to specifically collect tissues for molecular studies. Issues regarding specific specimen types are discussed in the following sections.
Resection specimens are specimens obtained as a result of surgical procedures to remove tumors for therapeutic purposes. These specimens provide the largest amount of material for molecular testing. Several aspects of specimen collection must be borne in mind to optimize resection specimen quality and yield.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here