Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Advances in patient selection, surgical technique, and perioperative management have improved the outcomes of patients undergoing hepatic resection in recent years. As a result, the utilization of liver resection for patients with both benign and malignant disease has increased worldwide , (see Chapter 101A ). Marked improvements in operative and anesthetic techniques, improved patient selection, and the emergence of hepatobiliary surgery have reduced mortality rates following liver resection from over 20% to under 2% by experienced surgeons at high-volume centers. In addition, there is now a better understanding of liver anatomy, an enhanced knowledge of tumor biology, improvements in diagnostic imaging technology, and more effective systemic and liver-directed therapies for hepatic malignancies. These advances have facilitated personalized surgical approaches based on individual patient anatomy and tumor biology.
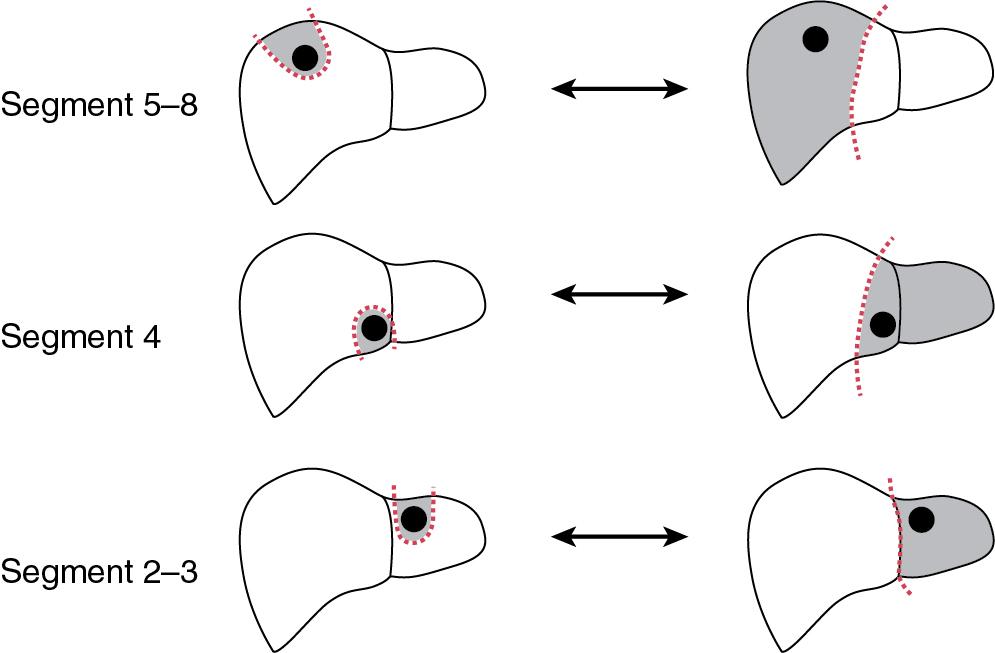
A parenchymal-sparing approach to hepatic resection has emerged as a novel method of optimizing the amount of normal hepatic tissue in order to minimize the risk of postoperative liver insufficiency and maximize the potential for liver-directed therapies in the future. The principle behind parenchymal-sparing hepatectomy (PSH) is to maximize the future liver remnant (FLR) by removing the minimal tissue necessary to achieve optimal oncologic outcomes. In contemporary practice, this principle is most frequently equated with techniques involving resection of one or more liver segments rather than an entire hepatic “lobe.” Anatomic resections , such as segmentectomy or sectionectomy, are often used in parenchymal-sparing approaches. Obtaining adequate tumor clearance can also be achieved by more limited nonanatomic resections, termed variously nonanatomic, atypical, or wedge resections. ,
This chapter reviews the rationale and indications for PSH for various primary and secondary hepatic malignancies. The short- and long-term outcomes of PSH are reviewed here; the technical aspects of surgical approach are reviewed in Chapter 118B .
Modern surgical liver anatomy is based on the Brisbane classification, which separates the liver into 8 Couinaud segments and 4 anatomic sections (right posterior, right anterior, left medial, left lateral) (see Chapter 2 ). Nevertheless, confusion persists regarding terminology to describe the extent of hepatic resection including major/minor, wedge, nonanatomic, and PSH. Traditionally, hepatic resections were categorized as either “major” (≥3 segments) or “minor” (<3 segments) based on the number of resected segments. This designation does not represent technical complexity; in fact, some traditionally “major” resections (e.g., left hepatectomy) can be technically less demanding than certain “minor” resections (e.g., right posterior sectionectomy). Resections are also commonly classified as “anatomic” or “nonanatomic” where anatomic resection refers to excision of an entire liver segment with its anatomic pedicle. The term parenchymal-sparing hepatectomy can be applied to either situation as it refers to a technique that maximally preserves healthy liver tissue. Whereas the term wedge resection connotes a nonanatomic minor resection, the term parenchymal-sparing hepatectomy does not imply whether the resection is anatomic or nonanatomic. Figure 102A.1 illustrates various parenchymal-sparing and anatomic resection options based on tumor location. Table 102A.1 lists the nomenclature proposed for various parenchymal-sparing operative procedures.
| TYPE OF RESECTION | SEGMENT(S) RESECTED |
|---|---|
| Grade 1 (Low Complexity) | |
| Peripheral wedge resection | Any isolated segment |
| Left lateral sectionectomy | S2 and S3 |
| Grade 2 (Medium Complexity) | |
| Right posterior sectionectomy | S6 and S7 |
| Grade 3 (High Complexity) | |
| Right anterior sectionectomy | S5 and S8 |
| Central hepatectomy | S4, S5, S8 |
| Right inferior bisegmentectomy | S5 and S6 |
| Right superior bisegmentectomy | S7 and S8 |
| Central anterior bisegmentectomy | S4b and S5 |
| Central posterior bisegmentectomy | S4a and S8 |
| Left medial sectionectomy | S4a and S4b |
Ahn et al. have demonstrated that tumors are not always located in the center of a segment; sometimes they are located within proximity to multiple portal branches. In these cases, an anatomic segmentectomy may not be oncologically sufficient, but at the same time, resection of multiple segments or hemihepatectomy may be unnecessarily extensive. For example, for a small tumor located on the ventral or dorsal aspect of segment VIII, a classic anatomic segment VIII resection may represent an overly extensive surgery and may not be feasible in patients with underlying liver disease because segment VIII can account for almost one quarter of total liver volume. Instead, systematic resection of either the ventral or the dorsal portion of segment VIII may be considered an oncologic surgical approach that maximizes the safety of hepatic resection. These considerations have led some authors to redefine “anatomic resection” as a surgical maneuver that removes the territory of one or more third-order portal branches.
Several innovative diagnostic techniques have been developed to assist in the reliable identification of hepatic segments and subsegmental anatomy. Intraoperative ultrasound (IOUS) is certainly critical for any hepatobiliary surgeon (see Chapter 24 ). In addition, novel near-infrared fluorescent imaging techniques using intrabiliary or intravascular injection of indocyanine green (ICG) paired with fluorescent imaging systems allow for intraoperative visualization of biliary and vascular anatomy and can aid surgeons in defining their intended resection plane with high accuracy. , A recently revisited technique, described as early as 1985, uses IOUS-guided dye injection of the portal venous branch feeding a tumor, in order to accurately delineate the extent of tumor-bearing parenchyma that should be resected. , In a series of 65 segmentectomies performed for hepatocellular carcinoma (HCC), Ahn et al. noted that 45% of resected tumors were fed by two or more segmental portal branches, with almost one-quarter having a complex pattern of portal venous supply. In all these cases, portal venous dye injection allowed the surgeon to establish a precise resection plane based on tumor–portal vein relationships. The authors were able to perform a limited nonanatomic resection while maintaining oncologic resection principles. Importantly, the long-term disease-free survival (DFS) and overall survival (OS) rates reported by this group were comparable with those reported by other series. ,
Despite the technically demanding nature of some of these techniques, the specialized equipment needed, and the additional time and training that may be required, these maneuvers enhance real-time identification of liver anatomy and facilitate anatomic-based parenchymal-preserving resections that are oncologically sound.
The primary rationale for parenchymal-sparing techniques is that maximizing the volume of liver remaining after a planned resection also maximizes functional parenchyma, which is highly predictive of postoperative morbidity, mortality, and liver dysfunction. , Resections removing up to 80% of functional parenchyma can be safely performed in the setting of normal liver function. However, an FLR of at least 40% is often necessary in patients with underlying liver disease, such as cirrhosis, steatohepatitis, chemotherapy-induced liver injury, or in the setting of biliary obstruction and jaundice. The relationship between liver volume and liver function is not static, particularly in the setting of underlying liver disease, where parenchymal-sparing techniques are often indicated. Accurate calculation of the volume and function of the FLR is imperative in order to determine resectability, discuss perioperative risk with the patient, and predict postoperative outcomes. The degree to which the underlying liver disease constitutes an absolute versus relative contraindication to hepatic resection depends on the anticipated volume and function of liver remaining after resection. Quantitative measures of liver function, including ICG clearance and lidocaine conversion tests, can assist in preoperative planning. Systematic measurement of liver remnant volume and careful assessment of liver function have improved the safety of major resections (see Chapter 4 ).
Several studies have found a decreased risk of posthepatectomy liver failure (PHLF) after hepatic resection through parenchymal-sparing techniques, particularly in patients with impaired liver function. Fisher et al. reported a large, retrospective, multi-institutional study of almost 600 patients undergoing formal right hepatectomy versus a parenchymal-sparing right posterior sectionectomy for benign and malignant disease; a right posterior sectionectomy was associated with a significantly lower rate of PHLF (1% vs. 8.5%), as defined by hyperbilirubinemia or significant ascites. These findings were confirmed by other studies associating extent of liver resection with increased rates of PHLF. , To that end, parenchymal-sparing techniques maximize the liver volume and liver function after resection and subsequently decrease the risk of PHLF compared with more extensive resections.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here