Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
A 38-year-old woman presents to her general gynecologist with a complaint of a vaginal “cyst” at the introitus. She notes some fullness at the opening to the vagina and feels that this interferes with penetrative intercourse. She denies pain or drainage. She is G1P1 with a history of one prior uncomplicated vaginal delivery. She has an otherwise unremarkable medical and surgical history. On exam, the gynecologist notes a 2 cm firm nodule in the midline at the level of the introitus. The mass is minimally fluctuant and mobile and is located just deep to the dermis. The gynecologist counsels the patient, and they agree to perform an excision of the mass, presuming benign etiology. The mass is excised, and the pathology reveals a deep, aggressive angiomyxoma. What are the next steps in management of this patient? How would her treatment differ from treatment of other, more common vulvar cancers? What factors should a clinician keep in mind in order to identify uncommon vulvar cancers at the time of presentation?
Vulvar cancer is a rare gynecologic malignancy, accounting for approximately 6000 new cases and 1350 deaths annually. This represents approximately 0.3% lifetime risk of vulvar cancer among American women. The rate of vulvar cancer diagnosis has increased steadily over the past 30 years, with incidence of 2.1/100,000 in 1992 to 2.5/100,000 in 2018; however, the death rate has remained stable at approximately 0.5/100,000 women. The median age of vulvar cancer diagnosis is 68 years.
The majority of vulvar cancers, approximately 75%, are squamous cell carcinomas (SCCs). However, numerous other histologic tumor types have been described. Many of these have clinical courses which differ from that of SCC. In this chapter, we will discuss several rare vulvar malignancies—HPV-independent vulvar cancers, verrucous carcinoma, aggressive angiomyxoma, and yolk sac tumors (YSTs)—and describe how their treatment may differ from the treatment of other, more common vulvar cancers ( Table 20.1 ).
| Characteristic | HPV associated vulvar squamous cell carcinoma | Non-HPV–associated vulvar squamous cell carcinoma |
|---|---|---|
| Age of diagnosis | 30–50 years | 40–60 years |
| Precursor Lesion | High grade squamous intraepithelial lesion | Differentiated vulvar intraepithelial neoplasia (dVIN) |
| Immunohistochemical characteristics | P16 abnormalities | P53 abnormalities |
| Prognosis | Favorable compared to non-HPV associated vulvar carcinomas | Poor compared to HPV-associated carcinomas |
Vulvar squamous cell carcinoma (SCC) is the most common vulvar cancer. Recent investigation into the pathogenesis of this disease has identified two distinct entities; carcinomas which arise in the presence of HPV; and carcinomas which arise in the absence of HPV. Sixteen to 40% of vulvar SCC is caused by HPV infection, whereas approximately 60% are HPV-independent. HPV-associated vulvar carcinomas are typically diagnosed in women who are younger (in their 30s–50s), have a precursor high-grade squamous intraepithelial lesion, and express p16 histopathologically. By contrast, HPV-independent tumors are diagnosed in older women, (age 40–60 years), are associated with chronic inflammatory conditions of the vulva such as lichen sclerosis, have a precursor lesion of differentiated vulvar intraepithelial neoplasia (VIN) (discussed in the pathology section of this chapter), and are associated with p53 alterations pathologically. Importantly, HPV-independent SCCs have a worse prognosis compared with HPV-associated tumors. McAlpine and colleagues published their experience evaluating the survival outcomes of nearly 200 women with vulvar SCC and found that HPV-independent tumors were associated with worse overall, progression-free and disease-specific survival compared with similarly matched HPV-associated disease.
Verrucous carcinomas are another rare type of SCC. While HPV infection has been demonstrated in patients with vulvar verrucous carcinoma, the pathogenesis of this entity is not clearly established. Verrucous carcinomas tend to grow into large tumors, but rarely metastasize to the lymph nodes. For this reason, they may be associated with improved survival compared with other SCCs. Nodal or distant recurrence rates are very rare, and local recurrence rates of 12%–17% have been reported.
Aggressive angiomyxomas are very rare vulvar tumors that tend to occur in young women (in their mid-30s–40s). No known precursor lesions or risk factors have been identified. Patients have a high rate of local recurrence, ranging from 36% to 72%, though the rate of lymph node and distant metastasis is very low.
YSTs represent a very rare subset of extraovarian germ cell tumors. The incidence of all extragonadal germ cell tumors among males and females is 1.8–3.4 per 1 million. The incidence of vulvar YSTs is rarer still, with fewer than 20 cases reported in the published literature. The median age of diagnosis is 19 years, though cases have been reported in multiple age groups, from infancy to the 50s. Interestingly, though perhaps merely coincidentally, a preponderance of reports describe tumors arising in the right labia majora.
The most recent classification of preinvasive and invasive squamous neoplastic lesions of the vulva is now broadly classified as HPV-dependent and HPV-independent. Only the less common HPV-independent category will be discussed below.
This category of vulvar intraepithelial neoplasia (VIN) has been evolving rapidly over the last few years. It was initially thought that differentiated vulvar intraepithelial neoplasia (dVIN) was the sole precursor to HPV-independent SCC but recently two other lesions, now thought to be precursors, have been described. These lesions have been termed differentiated exophytic vulvar intraepithelial lesion (DE-VIL) and vulvar acanthosis with altered differentiation (VAAD). The discussion below will focus predominately on dVIN.
HPV-independent VIN, often arises in the setting of lichen sclerosus or lichen simplex chronicus and can be uni- or multifocal. The lesions can be ulcerated and erythematous or can present as a slightly raised, white lesion.
The lesion is characterized by pleomorphic keratinocytes with ample eosinophilic cytoplasm and large vesicular nuclei with prominent nucleoli confined to the basal and parabasal layers with normally differentiated cells in the superficial layers of the epithelium. For this reason, in the past it was referred to as dVIN which will be used interchangeably with HPV-independent VIN. The epithelium can have range of findings from atrophic or acanthotic; however, it often demonstrates elongation and fusion of the rete pegs ( Fig. 20.1 A). The squamous epithelium has spongiosis, and the atypical cells often show pronounced intercellular bridges and abrupt keratinization. Dyskeratosis and keratin pearls at the base of the rete ridges i.e. paradoxical maturation is characteristic. The atypical basal and parabasal cells ( Fig. 20.1 B) frequently demonstrate atypical mitotic figures and may be hyperchromatic.
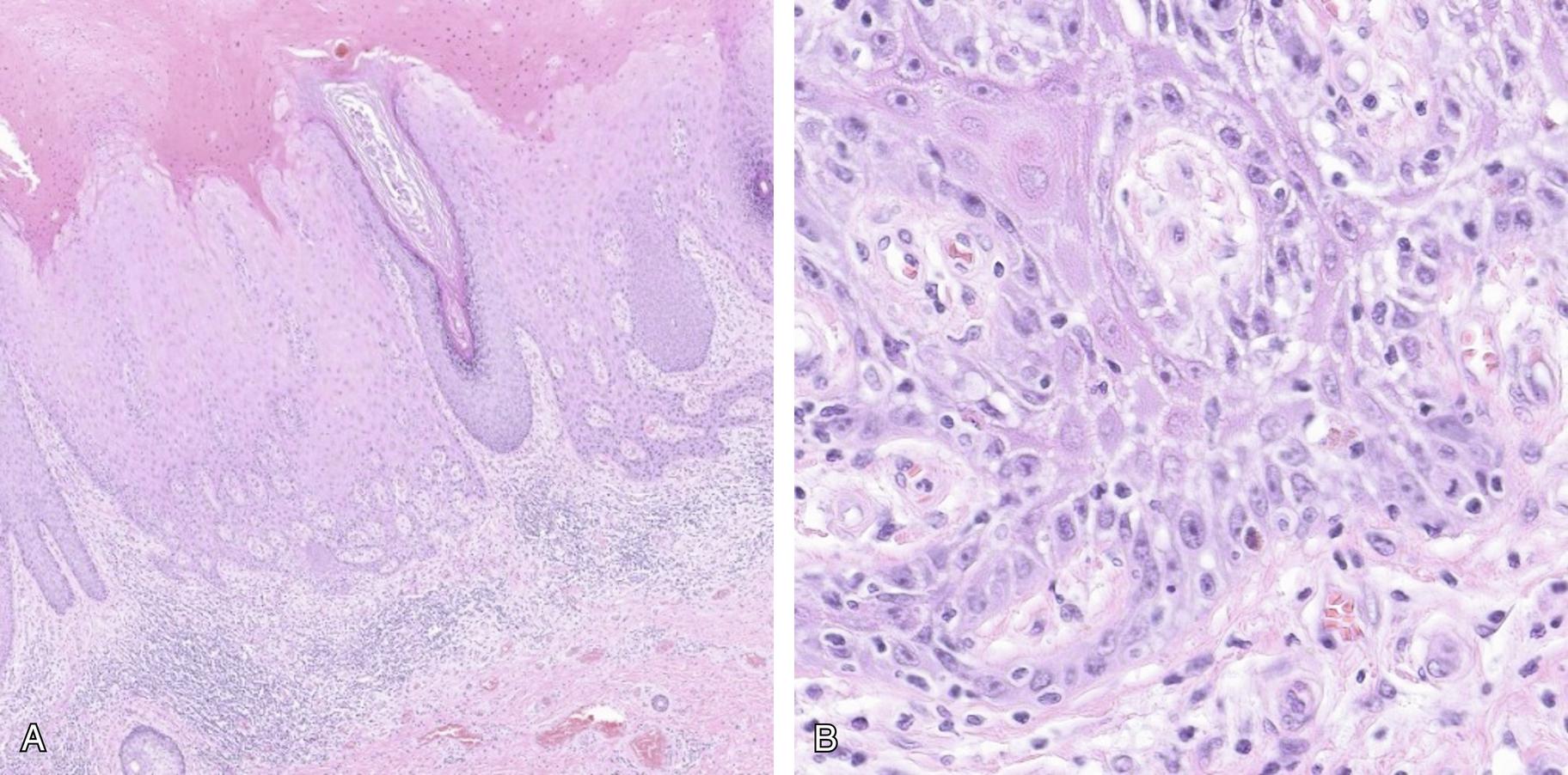
The identification of this form of VIN is difficult as the findings can be subtle and overlap with other more common entities with which it is often associated, including lichen simplex chronicus and hypertrophic lichen sclerosus. In addition, it can be confused with reactive changes that have associated pseudo-epitheliomatous hyperplasia. In small biopsies distinguishing reactive squamous atypia from dVIN is particularly challenging and in some cases multiple sequential biopsies are necessary prior to making the correct diagnosis. dVIN can also be mistaken for HPV-dependent high grade VIN, when they have a more basaloid appearance, and ancillary studies (see below) can aid in this distinction. Finally, as mentioned above, two other types of HPV-independent VIN have been recently identified, namely DE-VIL and VAAD, which share some features with dVIN; however, they both lack significant basal atypia and TP53 mutations and are thought to be precursors to the more uncommon form of HPV-independent squamous carcinoma called verrucous carcinoma.
This category consists of two histologically distinct forms of carcinoma. The most common is referred to as HPV-independent vulvar squamous cell carcinoma (VSCC), while the other less common form is called verrucous carcinoma (VC). The discussion will focus primarily on VSCC. However, the section on molecular findings will cover VC as well.
Invasive squamous carcinoma can present as focal ulcers or hyperkeratotic, exophytic masses. They can be unifocal or multifocal. Unlike HPV dependent SCC, patients can present with rapidly enlarging tumors.
These tumors are typically well-differentiated and demonstrate prominent keratinization with keratin pearl formation. The invasive front of the tumor is composed of wide-spread infiltrating tongues of neoplastic squamous epithelium ( Fig. 20.2 A). VSCC can also show a variety of other patterns including a warty or basaloid appearance and can lack keratinization. A note must be made about the challenges with measuring depth of invasion. In exophytic tumors and those that are not well oriented accurately measuring depth of invasion is challenging. As the required depth for distinguishing stage IA from IB is 1 mm, in microscopic lesions, there can be interobserver variability in measuring depth of invasion, i.e., < 1 mm versus > 1 mm. In a subset of cases, adjacent lichen sclerosus and dVIN may be present. In contrast, verrucous carcinoma is characterized by broad, pushing invasive borders ( Fig. 20.2 B) and basal atypia is minimal. The diagnosis of verrucous carcinoma and distinction from precursor lesions such as DE-VIL and VAAD is often not possible and conservative excision of the mass is recommended for definite diagnosis.
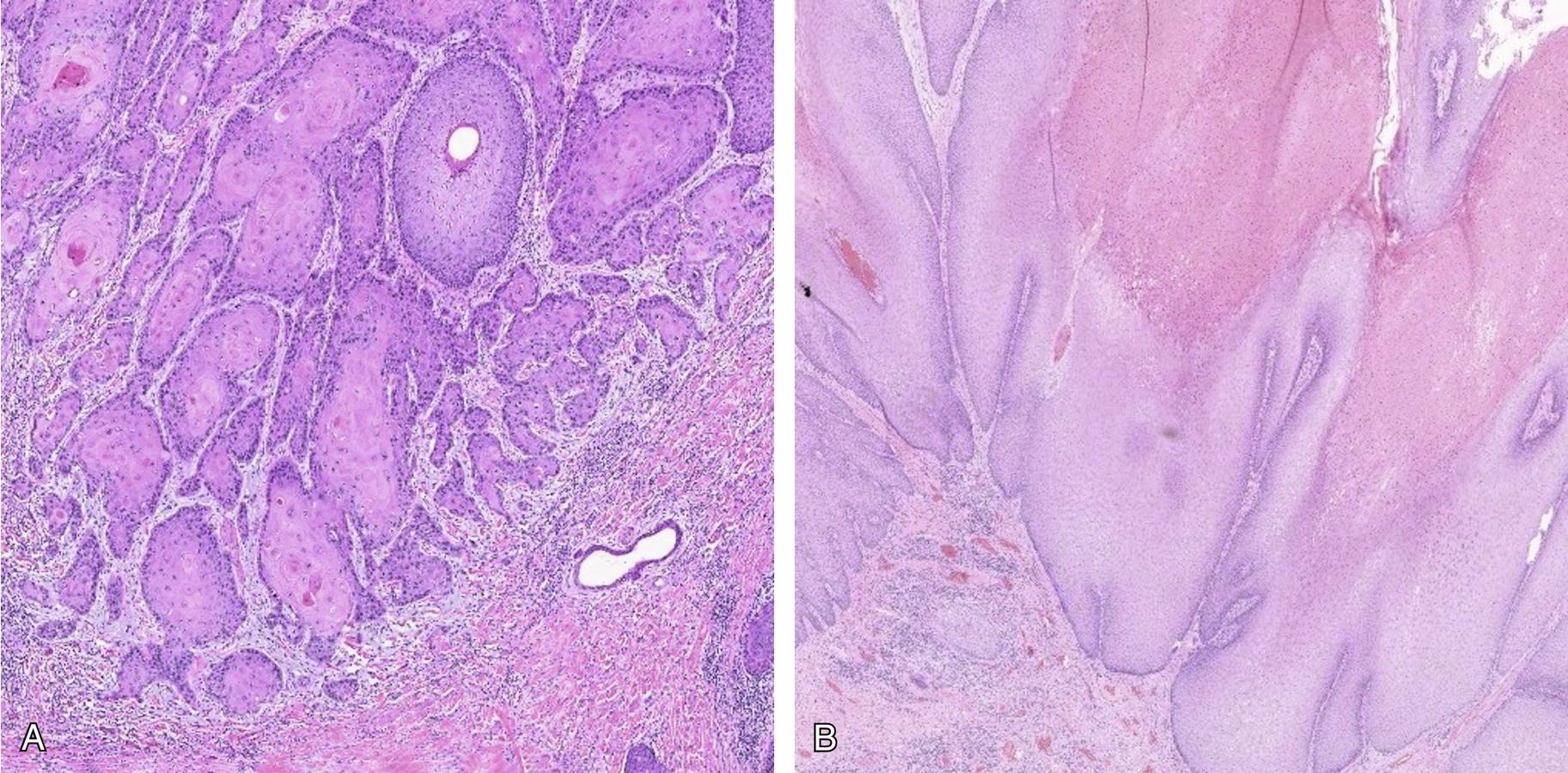
Because of the wide variety of histologic patterns and their overlap with HPV-dependent SCC, a reliable distinction between the two types of tumors is often not possible based solely on morphology. However, the presence of a precursor lesion or the background changes in the vulva can aid in the distinction, as HPV-independent tumors may have associated dVIN and arise in the background of lichen sclerosus or lichen planus. Immunohistochemical stains can be helpful in the identification of HPV-independent VIN and squamous carcinoma. Since TP53 mutations are frequently present in dVIN and VSCC, immunohistochemistry for p53 can be used to help in the identification and classification of the lesions. The p53 staining can take on two appearances in dVIN. It can show diffuse, strong staining in the basal and parabasal zone with nearly all of the cells in the regions showing intense staining. A null p53 staining pattern can also be seen where there is a complete lack of staining in the cells of the basal and parabasal zone. In normal or reactive squamous epithelium p53 shows heterogeneous staining with negative and positive cells with varying intensity of staining. The same holds true for SCC which can show either strong, diffuse nuclear staining or a null staining pattern of p53, both indicative for the presence of a mutation. This is helpful in distinguishing the lesions from HPV-dependent SCC. However, if the lesion has definitive features of dVIN or SCC without abnormal p53 then the diagnosis can still be made, as not all dVIN or HPV-independent SCCs harbor TP53 mutations. For the acanthotic, hyperkeratotic lesions that have overlap with HPV-dependent lesions, p16 immunohistochemistry can be helpful as the latter show diffuse, block-like staining which is not a typical feature of dVIN or HPV-independent SCCs. If HPV RNA in situ hybridization is available, it can also be useful in the distinction of the two types of carcinomas and precursor lesions.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here