Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Nematodal helminths represent infectious agents that are not very infectious, but have developed a delicate strategy to infect human hosts. As such, they manage to overcome the epidermal barrier, enter the human body, and travel by several methods to different places in the body, where they cause disease. These diseases are endemic in Asia, Africa, and Latin America.
Despite the existence of effective and rather cheap therapies, many endemic countries cannot provide these therapies for the vast majority of infected patients. Until this changes, strong emphasis needs to be placed on prevention and patient education.
Guinea worm disease, dracunculiasis, dracontiasis, medina worm
Dracunculosis is a very common skin disease in the tropics worldwide due to Dracunculus medinensis .
It causes physical and psychological incapacitation.
It is preventable, curable, and can be eradicated.
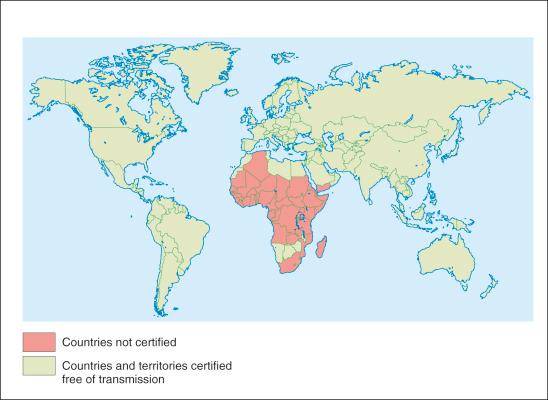
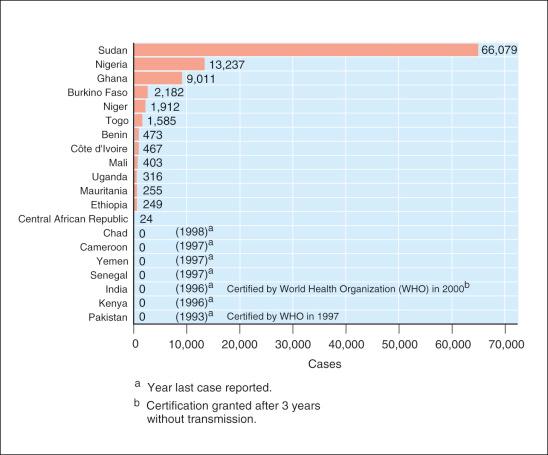
Dracunculosis is a parasitic dermatosis that has plagued humans for centuries. Earliest texts date from Ebers papyrus about 1500 bc and particular descriptions of the disease have been found in the work of Greek, Roman, and Arab physicians in the tenth and eleventh centuries ad . In the New World it persists in the tropics and is endemic in parts of sub-Saharan Africa ( Figs. 8-1 and 8-2 ).
The disease does not have any gender predisposition. However, it is less common in children.
Dracunculosis is a chronic nematode infection of humans by the female worm Dracunculus medinensis .
Thousands of larvae are released into water from females extruding from an ulcerated nodule of an infected human (definitive host). These larvae are then ingested by cyclopoid copepods (tiny aquatic crustaceans, approximately 2–3 mm long) that serve as intermediate hosts, where they mature into infective larvae ( Fig. 8-3 ). Humans unintentionally swallow the crustacea in unsafe (unfiltered, unboiled) drinking water, especially from surface waters – ponds, lagoons, valley dams, reservoirs, streams, or rivers.
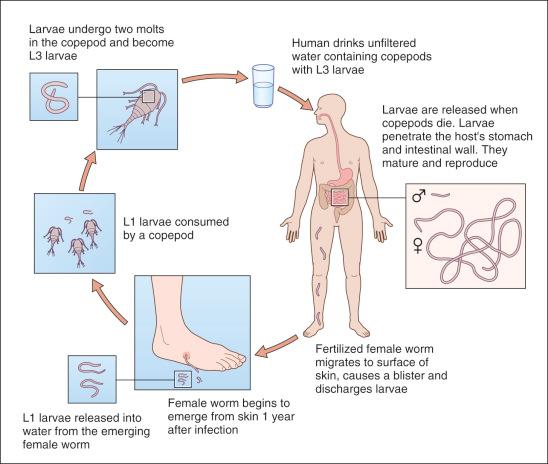
The crustaceans are dissolved in the acidic gastric environment, releasing infective larvae that penetrate transluminally into loose connective tissue, often the peritoneal cavity. Larvae then migrate through the interstitium and lymphatics, homing to subcutaneous tissues where they grow, mature, and fertilize in about 1 year (10–18 months). The adult female is 70–120 cm long.
Fertilized females migrate epicutaneously (over approximately 4 weeks) where on contact with water they release larvae to complete the life cycle, before they die (see Fig. 8-3 ).
This condition is asymptomatic during incubation until maturity and fertilization. Symptoms and signs occur due to epicutaneous migration of fertilized females. Systemic features may precede cutaneous manifestations and include nausea, vomiting, diarrhea, syncope, and dyspnea.
The earliest dermatological manifestations are urticaria and erythematous papulonodular lesions, which are painful but may be preceded by pruritus.
Papules and nodules may develop into vesiculobullous lesions with surrounding induration ( Fig. 8-4A ), which cause an intense burning sensation that is relieved by submerging the affected body parts in water.
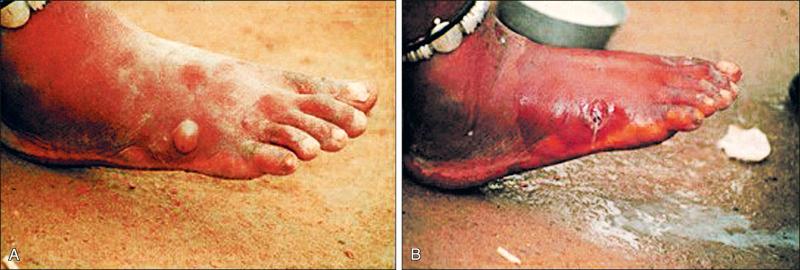
Subsequent skin ulcerations may be colonized by bacterial infections, often leading to pyodermas ( Fig. 8-4B ).
If the worm ruptures in the tissue, it causes cellulitis or abscess formation or heals with calcification.
The lower legs and feet are the most commonly affected sites but the parasites may extrude from thighs, trunk, upper limbs, or head. They have also been found in soft tissues like pericardium, testicles, and ligaments.
A patient may have more than 20 worms extruding at a time, but usually there are fewer than five. No protective immunity develops and reinfection is therefore common.
Typically, a worm extrudes from a skin lesion ( Fig. 8-5 ).
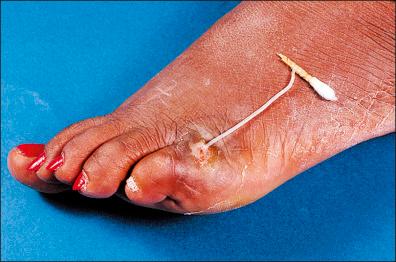
Suspect ulcer: wet smears showing motile larvae on microscopy.
Typical clinical findings.
Increased erythrocyte sedimentation rate, immunoglobulin E (IgE), and eosinophil count.
Histology reveals mixed inflammatory infiltrate and granulation tissue at the anterior end of the worm with or without fibrosis.
The differential diagnosis includes other filarial diseases before the typical worm emerges.
Slowly coil out the parasite around a small rod approximately 2–5 mm / day until the worm has come out completely.
The lesion should be widely excised to avoid complications, though care must be taken to check that all of the worm has been excised, as remnants may induce a severe inflammatory response.
Thiabendazole (tiabenzadole; mebendazole) 50 mg / kg per day divided into two doses, for 3 days, and metronidazole 250 mg tid for 10 days are not parasiticidal, but are antiinflammatory and aid healing.
Dracunculosis often causes physical and psychological incapacitation, which, though rarely permanent, causes significant loss of work output. Studies in Nigeria and Thailand revealed 21–84% and 28–34% of physical task incapacitation in affected patients, respectively.
Prevention includes avoiding the ingestion of contaminated water through prior sieving and boiling.
Eradication is possible and has been proven in Asia and previously endemic African countries: Senegal, Cameroon, Chad, and Kenya (see Fig. 8-2 ). Only 13 are still affected and statistics are encouraging, as in Uganda, which reduced its incidence to 4 in 2010 from 130 000 in 1990. On a larger global perspective, progress has been significant in other countries such as South Sudan, Chad, Mali, and Ethopia). To reach eradication, increased prioritization of public and medical knowledge of the management and preventability of dracunculosis and intermediary host / vector control is required, especially as this is a disease where a small number of water contacts or low proportion of risk behavior is sufficient to maintain transmission.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here