Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Surgery for burn injury is a key component to the multidisciplinary care of the burn patient. It usually involves early procedures such as escharotomy and fasciotomy when indicated, followed by burn excision. Early intervention in the form of early burn wound excision and grafting has dramatically changed the outcome and survival of the burn patient.
Following burn injuries, tissues affected, including skin, promote an inflammatory response at the junction between the eschar and the underlying viable tissue. The presence of microorganisms within the eschar attracts polymorphonuclear leukocytes that release large amounts of proteolytic enzymes and inflammatory mediators. Subsequent enzymatic action results in separation of the eschar, leaving granulation tissue. In large surface area burns, the inflammatory response at the burn injury site develops into a systemic response.
Mediators such as prostanoids, thromboxane, histamine, cytokines, and tumor necrosis factor are produced and released from the burn site. The serum levels of these mediators increase proportionally to the skin surface area of the burn. The hypermetabolic response with increased protein catabolism, increased energy expenditure, weight loss, poor wound healing, and immune depression continues until the outpouring of mediators abate.
Early excision and skin grafting to achieve wound closure have been shown to reduce infection rates, reduce length of hospital stay, and improve survival in burn patients. Pediatric burn patients in particular have benefited from timely surgical intervention. There has been a remarkable increase in the total burn surface area (TBSA) associated with a 50% mortality risk over recent decades. It is now fairly unusual for a child to succumb to burn injuries of any size, even in association with an inhalation injury ( Tables 12.1 and 12.2 ). Early prompt fluid resuscitation, appropriate critical care, nutritional support, and treatment of infections have also played a major role in this achievement.
| Age [years] | LA50 (% TBSA) | ||
|---|---|---|---|
| 1942–1952 | 1980–1991 | 1992–2004 | |
| 0–14 | 49 | 98 | 99 |
| 15–44 | 46 | 70 | 88 |
| 45–64 | 27 | 46 | 75 |
| >65 | 10 | 19 | 33 |
| Years | MORTALITY SORTED BY BURN SIZE (% TBSA) | |||||||
|---|---|---|---|---|---|---|---|---|
| <20% | n | 21%–40% | n | 41%–60% | n | 61%–100% | n | |
| 1980–1985 | <0.1% | 889 | 1% | 230 | 8% | 105 | 33% | 95 |
| 1986–1990 | <0.1% | 571 | 1% | 224 | 4% | 117 | 19% | 88 |
| 1991–1995 | <0.1% | 522 | 2% | 192 | 8% | 94 | 20% | 78 |
| 1996–2000 | <0.1% | 635 | 1% | 222 | 3% | 133 | 19% | 114 |
| 2001–2004 | 2% | 83 | 2% | 121 | 26% | 91 | ||
Early operative intervention, however, remains the major contributor to this major advance in burn care. In 1974, Burke and colleagues reported on the results of total excision of full-thickness burns where allografts were applied to cover burn wounds following excision; allograft rejection was controlled using immunosuppressants.
Improved mortality, shorter hospital length of stay, and fewer metabolic complications were noted when early excision was retrospectively compared with late excision. In another study, 32 children with an average age of 7 years and mean burn size of 65% (TBSA) who underwent either total excision or serial debridement were studied. Mortality, overall blood loss, and cumulative operating time were equivalent. The early excision group, however, had their length of hospital stay almost halved (97 ± 8 days vs. 57 ± 5 days). Since then, hundreds of children with burns of more than 30% TBSA treated with early excision have demonstrated a hospital length of stay of less than 1 day/% TBSA burned.
Tompkins et al. demonstrated that mortality of adult burn patients at Massachusetts General Hospital improved from 24% in 1974 to 7% in the time period from 1979 to 1984 after implementation of prompt eschar excision and immediate wound closure. This study was later expanded to include 85 patients aged 17–55 years. Those patients aged 17–30 years without inhalation injury showed significantly reduced mortality of 9% if treated by early excision, compared to 45% when treated conservatively. Patients with a concomitant inhalation injury or aged over 30 years, however, had no survival benefit from early excision.
In children with burn injuries, Xiao-Wu et al. showed that delays in the excision of extensive burn were associated with longer hospitalization, delayed wound closure, increased rates of invasive wound infection, and increased incidence of sepsis.
Munster et al. demonstrated a statistically significant decrease in hospital length of stay correlating with a decrease in the interval between surgical interventions over a 14-year period. Other variables such as burn size, inhalation injury, and age remained unchanged during the same time period; mortality rate decreased significantly.
Elderly burn patients have also been shown to benefit from early excision. Deitch et al. demonstrated that early surgical excision in patients with an average age of 68 led to 40% reduction in mean hospital length of stay compared to the national average and reduced mortality. Many studies show that early excision can be safely performed in the elderly with clear benefits of reducing hospital length of stay and the number of septic episodes.
Hypertrophic scar formation is common following burn injuries, with dark-skinned patients more prone to hypertrophic scar development. However the most important factor in the development of hypertrophic burn scars is delayed wound healing. Deitch et al. demonstrated that in wounds requiring more than 10 days to heal, the risk of hypertrophic scarring is significant and rises to 80% if healing is delayed beyond 21 days. Operative treatment also limits the duration of pain caused by the burn wound.
In conclusion, early burn wound excision is lifesaving, offers improved cosmetic and functional outcomes, and is cost-effective.
Operative intervention is indicated without delay once the burn injury is determined to be a “deep” injury. Deep burn injuries are clearly either full-thickness or deep partial-thickness burns that are unlikely to heal within 3 weeks and are typically flame or contact burns. Heimbach et al. noted that deep partial-thickness burns did not convert to full-thickness burns when topical antimicrobials were used to control infection. Although these wounds eventually healed after several weeks, they showed persistent blistering, pruritus, hypertrophic scar formation, and poor functional outcomes. These observations prompted a prospective trial of early excision and grafting versus nonoperative treatment of indeterminate depth in burns of less than 20% TBSA. Shorter hospitalization, lower cost, and reduced time away from work, but greater use of blood products were seen in patients treated with early excision. Those patients treated nonoperatively required more late skin grafting to achieve wound closure and developed more hypertrophic scars.
Tangential excision, as a technique, is the meticulous removal of burned skin while preserving underlying viable tissue. Body contours are better preserved with tangential excision than with fascial-level excision, which removes underlying subcutaneous fat; hence, it is the standard excision technique for burns.
Tangential excision was originally described by Janzekovic, who observed that deep skin graft donor sites could be grafted with thinner split-thickness skin grafts taken from another area. Later she extended this concept to partial-thickness burns by repeatedly excising thin layers of the burned skin until viable tissue was reached ( Fig. 12.1 ). Then split-thickness skin grafts were immediately applied.
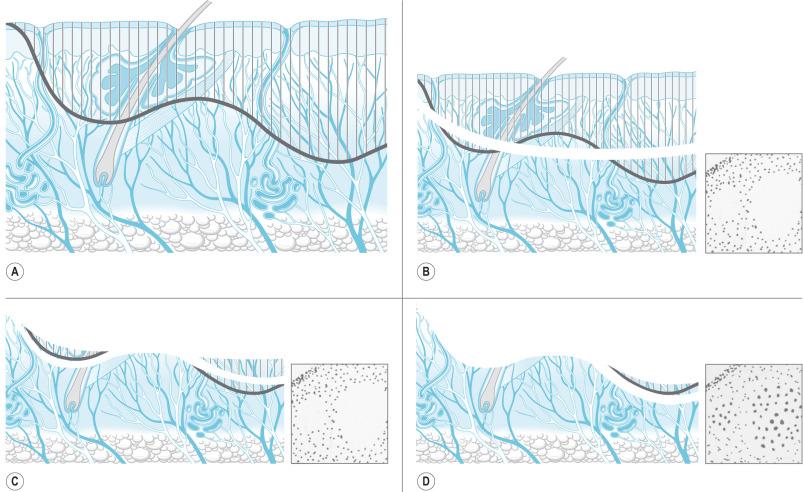
The technique of tangential excision and autografting of partial-thickness burns was a major advance in burn care at the time. Prior to its introduction, only full-thickness burns were excised, including subcutaneous fat and accompanying lymphatics down to the underlying layer of investing fascia ( Fig. 12.2 ). Janzekovic analyzed the results of the use of tangential excision in more than 2000 patients. She found that, compared to fascial excision, hospital length of stay, pain, and reconstructive procedures were decreased.
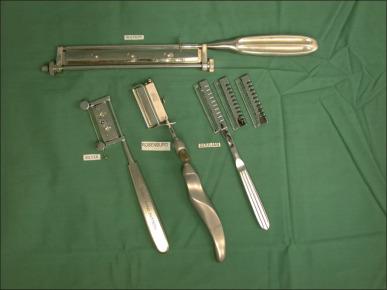
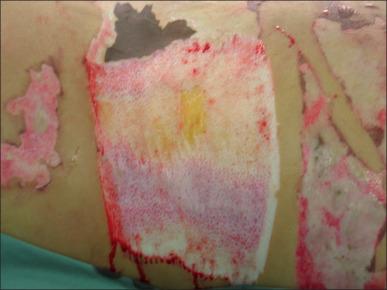
A number of different instruments can be used to perform tangential excision of the burn wound. The Goulian knife, Watson knife, and the Versajet Hydrosurgery System are used for tangential excision ( Fig. 12.3 ). Deep partial-thickness burns are debrided down to white, viable dermis with punctate bleeding. In full-thickness burns, excision continues layer by layer until viable subcutaneous tissue with a yellow glistening appearance is encountered. Dull yellow color, purple discoloration, or thrombosed vessels indicate nonviable tissues that are not suitable for grafting and require deeper excision ( Fig. 12.4 ). When excision is performed on limbs with a pneumatic tourniquet, these features are particularly important.
There are noninvasive imaging tools available for predicting burn depth and healing time in adult burns. These include laser Doppler imaging (LDI), which is the current gold standard; infrared thermography (IRT); and spectrophotometric intracutaneous analysis (SIA). LDI uses a low-intensity laser beam to scan across the tissue surface. Laser is both absorbed and scattered by tissue matrix and the blood flow in the vessels. Flux is then determined by processing the photocurrent resulting from both absorbed and scattered light. The device generates a colored map of the wound correlating with the extent of the injury. LDI can be a useful tool in surgical decision-making in indeterminate-depth burns.
IRT uses the heat signature resulting from tissue perfusion to predict the depth of burn injury. Superficial burns retain a bright color compared to the surrounding normal tissue due to increased perfusion, whereas deeper burns appear darker in color due to decreased or lack of perfusion. SIA is used in the diagnosis of pigmented skin lesions such as melanoma. It uses spectrally filtered images using polarized light ranging from 400 to 1000 nm. A complex set of mathematical algorithms determines the melanin and hemoglobin content of the epidermis and papillary dermis, then presents them in high-resolution images. The additional analysis of pigment available using SIA may more accurately indicate the depth of burn compared with perfusion alone.
Ultrasounds with frequencies higher than 20 MHz have shown promising results in quantitative assessment of burn depth in animal models. Currently only LDI has been validated for use in burn depth assessment. It has been shown to reduce wound healing time in patients requiring surgery with a potential cost saving of about $1200 per patient. Unfortunately LDI costs in excess of $70,000 in addition to annual servicing costs.
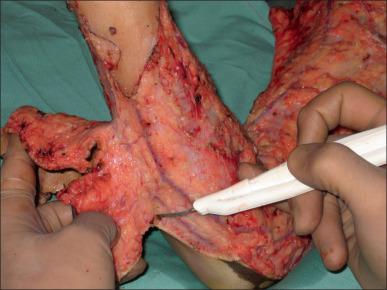
Skin and subcutaneous tissue are excised en-bloc using electrocautery in fascial excision. This involves excision of the full thickness of the integument, including all subcutaneous tissues down to the investing deep fascia. It is usually performed to reduce blood loss in extensive burns or in cases of severe infection to control the source of infection.
Fascial excision can limit the amount of blood loss because control of the deeper perforating vessels is achieved during the excision, bypassing the extensive capillary network present in the skin and subcutaneous tissue. Fascial excision is also required for areas where subcutaneous tissue has been involved in the burn injury. The advantages of this technique include improved graft take on fascia and reduced blood loss.
Fascial excision is also indicated in cases of life-threatening invasive wound infection or sepsis, particularly fungal infections such as Aspergillus and Mucor , and in cases of failed graft take in extensive burns in critically ill patients. Episodes of sepsis lead to ischemic necrosis of the subcutaneous fat due to poor peripheral perfusion and microvascular stasis. This represents a considerable problem in patients with extensive burns leading to late graft loss, and the ischemic areas are transformed into portals for invasive wound infection. The disadvantages of fascial excision are lymphedema due to the interruption of the subcutaneous lymphatics and contour deformities due to the loss of subcutaneous fat.
Excision of large burns can result in considerable, if not massive, blood loss. Tangential excision can lead to large blood loss unless measures are taken to limit hemorrhage. The simplest measure is to operate within 24 h of the burn injury. Vasoactive metabolites, in particular the potent vasoconstrictor thromboxane, are in abundance during this time to limit blood loss. Desai et al. demonstrated in a prospective trial of 318 pediatric patients with burns of more than 30% TBSA that early excision of burns is associated with an overall reduction in blood loss. The average blood loss per surface area excised (mL/cm 2 ) was compared at various post-burn excision time points. The total blood loss during excision performed within the first 24 hours after burn was 0.4 mL/cm 2 excised, compared to 0.75 mL/cm 2 excised if performed between days 2 and 16 post burn. The blood loss dropped to 0.49 mL/cm 2 excised if the procedure was performed after the 16th day. Overall mortality was 5% for an average burn size of 60% TBSA. Early excision had no adverse effect on mortality. Very early excision led to a halving of blood loss for both large and small burns ( Fig. 12.5 ).
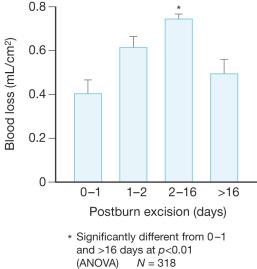
Other factors associated with increased blood loss during excision of burns are old age, male gender, larger body size, large full-thickness burns, high wound bacterial count, total area excised, and operative time.
Several techniques to reduce blood loss during burn wound excision can be employed in the operating room. Adjunctive measures to limit blood loss include tourniquets for the extremities, pre-debridement tumescent infiltration with epinephrine solutions, topical application of epinephrine 1: 10 000–1: 20 000, fibrin sealant, autologous platelet gel, calcium-enriched alginate sheets, and immediate bandaging with delayed grafting.
The application of tourniquets to the extremities is a very effective method of minimizing blood loss, especially for excision of burns involving hands and digits. As with tumescent infiltration, the absence of bleeding can make the estimation of the right depth of excision difficult. To counteract this, the tourniquet can be released briefly to check the adequacy of excision and then reinflated. The larger vessels are controlled by electrocautery or ligation and the wound is covered with sheets of calcium alginate or sponges, which are soaked in epinephrine solution. Following the release of the tourniquet, the limb should be elevated for about 10 min prior to graft application.
Tumescent solution made of epinephrine containing saline can be injected into the burn wound prior to excision. This technique is particularly useful for burn excision of areas such as the trunk, scalp, and face; 1.6 mL of 1 : 1000 epinephrine (0.8 mL in pediatric patients) is added to 500 mL of 0.45% normal saline solution. The resulting local vasoconstriction will minimize blood loss, and one-way syringes, such as the Multi-Ad Fluid Dispensing System or a pneumatic infuser, can make the injection process easier ( Fig. 12.6 ).
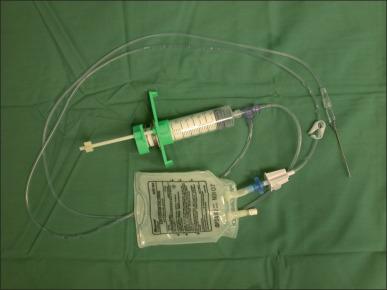
Monitoring the patient's hemodynamics is necessary because epinephrine may cause tachycardia and hypertension, which can worsen bleeding. Another drawback of this technique is that the lack of bleeding from the wound bed may make the adequacy of excision difficult to assess.
When dealing with large areas of disrupted capillaries, one of the most effective ways to safely limit blood loss is rapid excision followed by coverage with epinephrine-soaked sponges or gauze and compressive bandages.
Delayed grafting after excision is used in some centers to limit blood loss. In case of delayed skin grafting, the wound bed needs to be kept moist and clean prior to skin grafting. The wound can be covered with bulky cotton dressings with tubes through the dressing to the wound surface for continuous or intermittent irrigation with antibiotic solution. The patient returns to the operating room within 24 h and undergoes a second procedure of procurement and application of skin grafts. This may be a practical approach in cases of large surface area excision, where coagulopathy can develop due to large blood loss or hypothermia. The authors of this chapter adhere to a hemoglobin (Hgb) threshold of 7 for transfusion. One study suggested a possible decrease in infection rate when compared to a more liberal transfusion strategy.
Once procured, the autograft skin can be used in different ways depending on the size of body surface area burned that requires grafting.
Autografts are classified as full- or split-thickness depending on the depth of dermal layer included when harvesting the skin. Full-thickness autografts have better cosmetic results and reduced scarring compared to split-thickness autografts because the dermis provides flexibility and elasticity. The main caveat is that the increased amount of dermis in the full-thickness autograft can compromise its own viability during the imbibition, inosculation, and neovascularization phases of graft take. The donor site completely devoid of dermis must be closed primarily or grafted. Therefore, split-thickness skin grafts are mainly used in the treatment of extensive burn injuries.
If the burn is small, skin grafts can be applied without meshing as a “sheet.” The advantage of nonmeshed skin is better cosmetic outcomes. The disadvantage is the possible accumulation of seroma or hematoma underneath the skin graft that can compromise the apposition of the skin graft to the wound bed. Seroma or hematoma should be identified and removed by aspiration with a 25- or 27-gauge needle. The skin graft can be secured to the wound bed with quilting sutures or the application of a bolster dressing, which can reduce shearing of the skin graft. These grafts are best laid perpendicular to the long axis of limbs, particularly across joint flexural creases, which is in line with the general rule of placing potential scars perpendicular to the dominant muscle contraction in the area, thus reducing the degree of contracture if it occurs. Possible exceptions are the dorsum of the hand and forearm, where one may contend that longitudinally placed grafts are cosmetically superior.
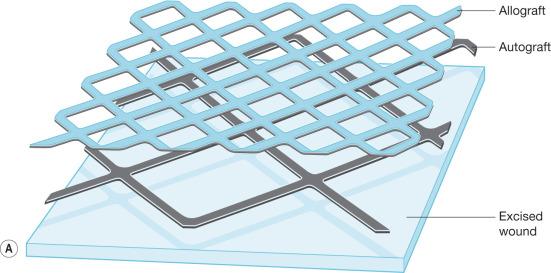
In large burns where donor sites are limited, autografts are usually meshed. This allows the autograft skin to be expanded to cover a wider area compared to its original size. The most commonly utilized mesh ratios are 2 : 1 and 4 : 1, although other ratios of expansion can be employed. The main advantages of 2 : 1 meshed autograft skin is the ease of handling and application and the extensive network of slits that allows drainage of seroma or hematoma. The main disadvantage is that the interstices leave a visible pattern once healed. Much larger areas can be covered when autografts are meshed 4 : 1 or greater and widely expanded. This widely meshed skin graft requires an overlay coverage of nonexpanded allograft skin to decrease the risk of graft loss, and this is applied over the autograft skin in a sandwich pattern ( Fig. 12.7 ). The large interstices, which require a considerable amount of time to heal by epithelialization; the need for an allograft skin overlay; and the less desirable cosmetic result limit the use of 4 : 1 meshed autograft skin to massively burned patients. Even in massive burns, the face, neck, and hands are still grafted with nonmeshed split-thickness skin graft due to the unacceptable outcome of using widely meshed skin grafts in these areas.
The Meek technique, which was later modified by Kreis et al., is another tool to cover extremely large burn surface areas. It uses autografts cut into small squares with the aid of a dermatome and a cork board. The squares are then pressed onto prefolded pleated gauze that is expanded in all four directions, leaving uniformly distributed islands of autograft skin that are then applied to the wound bed. Ratios of expansion can vary from 3 : 1 to 9 : 1. This technique has similar disadvantages to 4 : 1 meshed autografts.
The flexibility, elasticity, and strength of normal skin are provided mainly by the dermis. Excision of full-thickness burn removes the entire dermis, and the lack of the dermal layer prevents the healed skin from having the properties of normal skin. The use of dermal replacements in the treatment of burns can provide the highly desired characteristics of normal dermis. Integra is a dermal substitute composed of a porous matrix of cross-linked bovine collagen and glucosaminoglycans providing a scaffold for cellular invasion and capillary growth. It is applied to the wound bed following burn excision, and the matrix is fully incorporated into the wound bed in 2–3 weeks; then, thin split-thickness autograft skin is applied. Except for a possible increased risk of infections, the use of Integra is safe and effective. Skin graft take is usually aided by the use of negative-pressure wound therapy such as VAC Therapy ( Fig. 12.8 ).
Another dermal analog available for the treatment of full-thickness burns is Alloderm, consisting of cadaveric dermis devoid of cells and epithelial elements. Its applications are similar to other dermal analogs, and it has shown favorable results.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here