Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
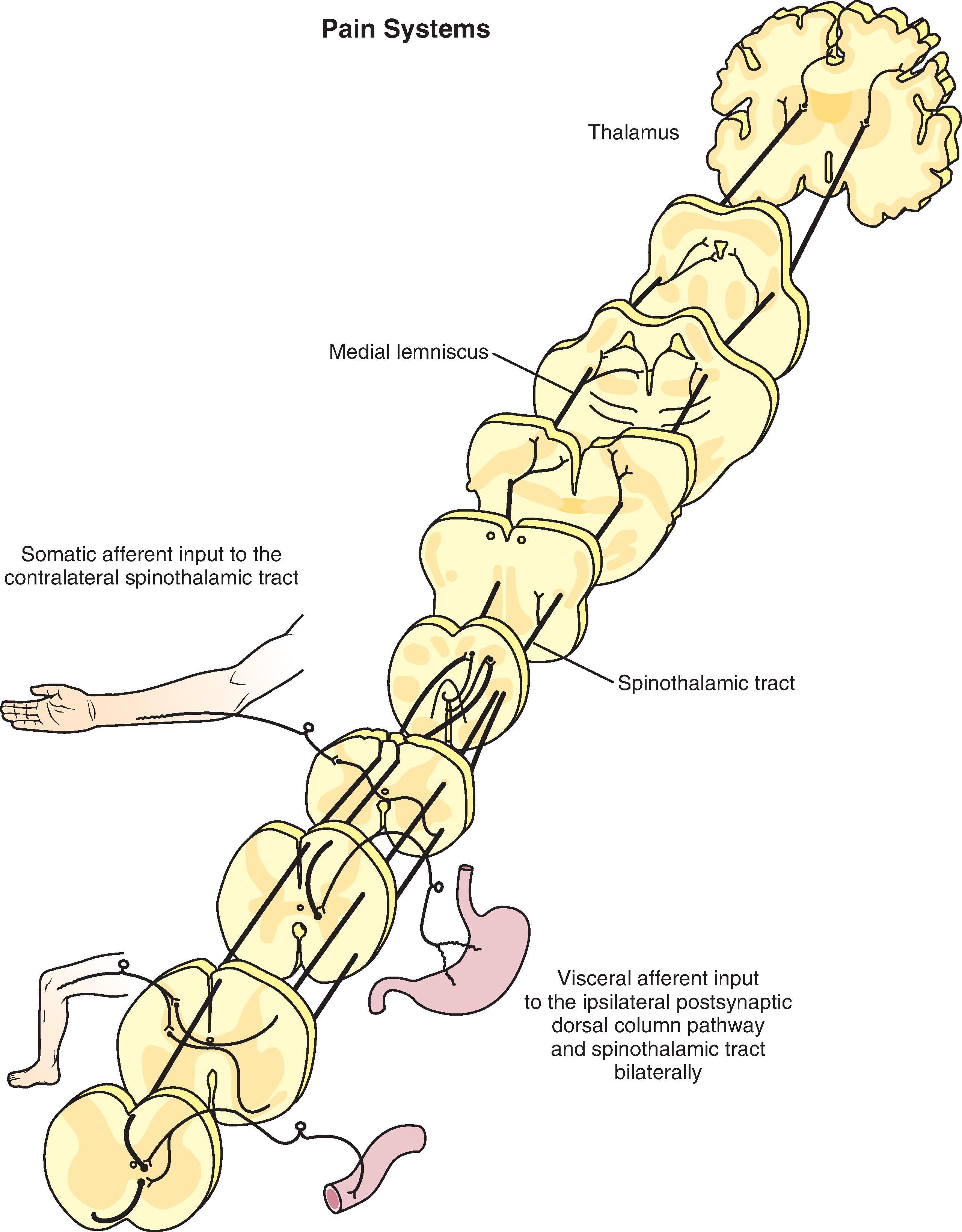
The neural circuits that are responsible for pain and the reactions to pain can be termed the pain system, or perhaps more appropriately, the pain systems . The pain systems include (1) peripheral neurons with a set of peripheral receptive elements, the nociceptors, (2) numerous central neuronal relay pathways, and (3) sets of integrative neurons that impose excitatory or inhibitory influences on nociceptive information at numerous levels of the neuraxis.
While functioning under normal conditions provides warning information vital to tissue protection, and if pain persists, maladaptive neuroplastic reconfiguration occurs at the synapse and in specific brain regions creating a chronic pain state. When expansion to other systems such as affective, aversive, learning, and memory occurs, chronic pain, anxiety, and depression can result.
The initial reception of inputs perceived by the brain to be painful occurs at the peripheral terminations of sensory axons, the nociceptors that transduce noxious mechanical, thermal, and chemical stimuli into action potentials. Sensory nerve axons transmit information about internal or external stimuli that are noxious, distressing, or damaging to the second order neurons located at the corresponding spinal cord or brainstem level (i.e. lumbar spinal cord for leg input, thoracic spinal cord for stomach lining input, and trigeminal spinal nucleus for face input). The spinal excitatory (glutamatergic) and inhibitory ([g]-aminobutyric acid-ergic [GABAergic] and/or glycinergic) interneurons provide a complex integrative network circuitry influencing the incoming nociceptive input received by nociceptive spinal dorsal horn projection neurons. As examples, the projection neurons and the inhibitory neurons have excitatory drive from spinal interneurons and/or from primary afferent nerve fibers. Spinal inhibitory interneurons may mediate pre-synaptic inhibition at the terminals of primary afferent nerve fibers as well as pre- and post-synaptic inhibition of spinal excitatory interneurons. Nociceptive spinal dorsal horn projection neurons are modulated further by long, descending facilitatory and inhibitory pathways.
Nociceptive signals are then transmitted by the pain system projection neurons to other integration sites in the brainstem and higher brain via collateral input. The primary integration of site-specific sensory information in the brain occurs in the thalamus that then relays the information for cortical site perception. A variety of coordinated responses are generated in response to relay of nociceptive information to numerous other brain neuronal sites and circuitry activating integrative excitatory and inhibitory mechanisms. Beyond cortical awareness of pain, processing occurs to provide protective somatic and autonomic reflexes, endocrine actions, emotional responses, as well as learning and memory about the event.
In addition to pain and pain reactions, the brain centers that receive nociceptive information provide either negative or positive feedback that can reduce or accentuate pain and pain reactions. Negative feedback to the spinal cord circuitry is mediated by descending pathways that are often called the “endogenous analgesia system.” The mechanism and pathways responsible for the accentuation of pain and pain reactions, referred to as central sensitization, can involve increased responsiveness at all levels of the pain systems, including the peripheral nociceptors, sensory neurons, the spinal cord, brainstem, and higher centers. The net effect of the positive and negative circuitry alterations leads to the perceptual experience of “pain,” but over time the discomfort of continuing pathologic chronic pain can result in increased anxiety, depression, and disability.
The initial reception of noxious inputs perceived to be painful occurs at the specialized endings of primary afferent sensory neurons known as nociceptors . Reception of noxious input occurs at the functionally specialized free nerve endings of the skin, muscle, joints, , viscera, , and dura ( Fig. 8.1 ). Nociceptive endings are also located in the fascia and adventitia of blood vessels. Nociceptor subtypes respond best to mechanical (mechanical nociceptors); mechanical and thermal (mechano-thermal nociceptors); or mechanical, thermal, and chemical stimuli (polymodal nociceptors). , Common types of cutaneous nociceptors are Aδ mechanoreceptors and C polymodal nociceptors, which relay the transduced information about potentially harmful input via Aδ and C-fibers, respectively. , Glutamate receptors, as well as μ and δ-opiate, substance P (SP), somatostatin, and vanilloid receptors, have been identified immunohistochemically on the peripheral endings of cutaneous nerve fibers. Neuroactivators released by tissue injury (ATP, bradykinin, nitric oxide, H+) and cytokines (tumor necrosis factor [TNF]α, interleukin [IL]-1β, IL-6) released by macrophages, neutrophils, and mast cells drawn to an injury site produce amplified peripheral nerve responses or peripheral sensitization (i.e. enhanced nociception).
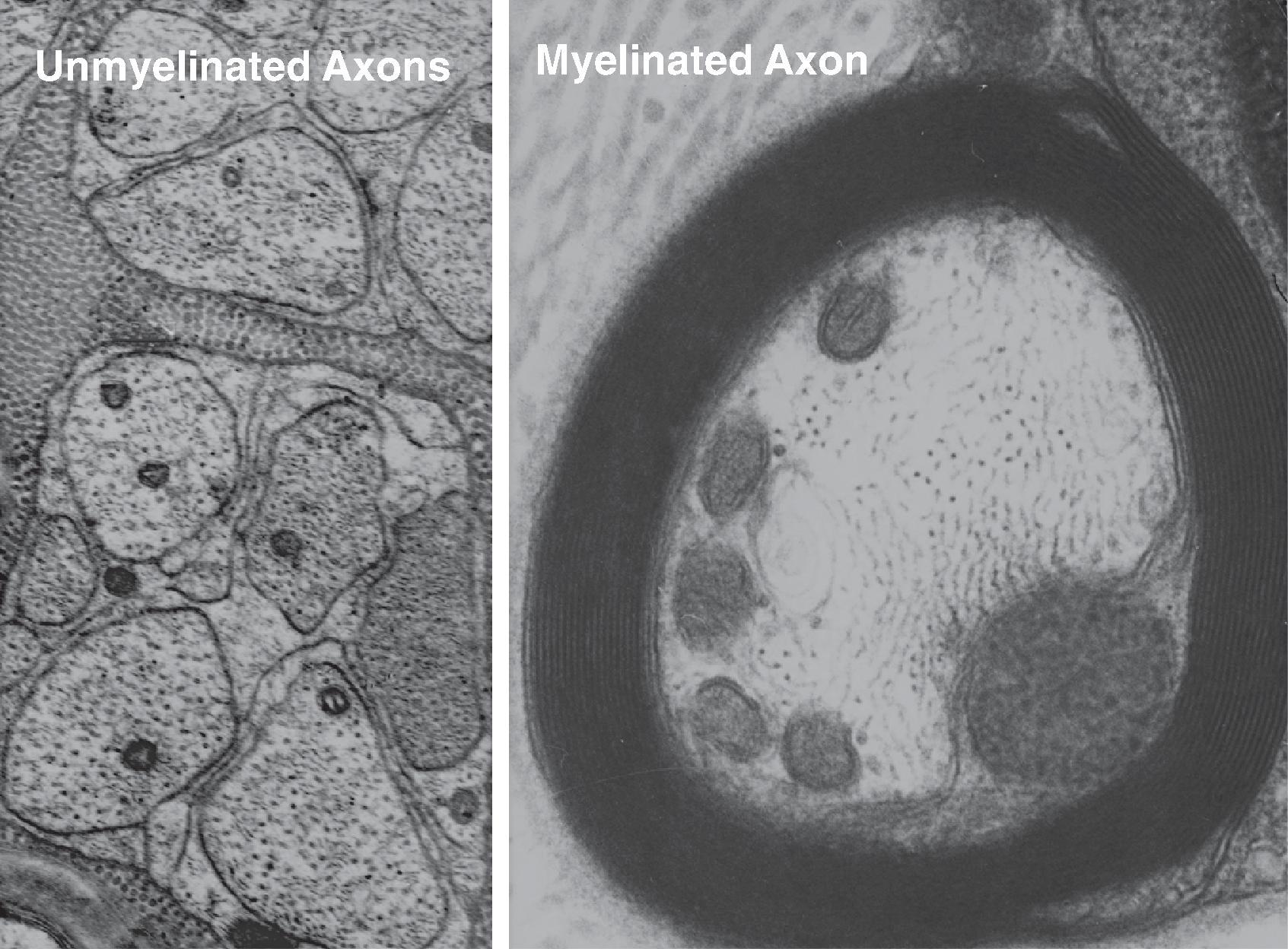
The small axons that relay information about noxious input from the skin and other tissues to the central nervous system (CNS) fall characteristically into the range of unmyelinated fibers with conduction velocities less than 2.5 m/s in the case of C-fiber (or group IV) nociceptors ( Fig. 8.2 ), and small fibers wrapped in a thin layer of myelin produced by Schwann cells with a conduction velocity of 4–30 m/s in the case of the Aδ fibers (or group III). Primary afferent C-fibers are more numerous than myelinated primary afferents in peripheral nerves. For example, in dorsal roots, the ratio of C-fibers to A fibers is about 2.5:1, and in joint nerves (after sympathetic postganglionic axons are removed by sympathectomy), the ratio of C-fibers to A fibers is 2.3:1.
While peripheral nerves have both sensory and motor functions, all sensory afferent fibers, whether they innervate cutaneous, deep, vascular, or visceral tissue, separate from motor fibers near the spinal cord and join to form the dorsal root before entering the spinal cord on the dorsal surface ( Fig. 8.3 ). Primary afferent fibers providing input from visceral structures and the vasculature travel along with the sympathetic efferent nerves. However, they pass directly through the sympathetic trunk to join other afferent fibers entering the dorsal horn of the spinal cord ( Fig. 8.4 ). They remain silent unless provoked by inflammation or abnormal distension.
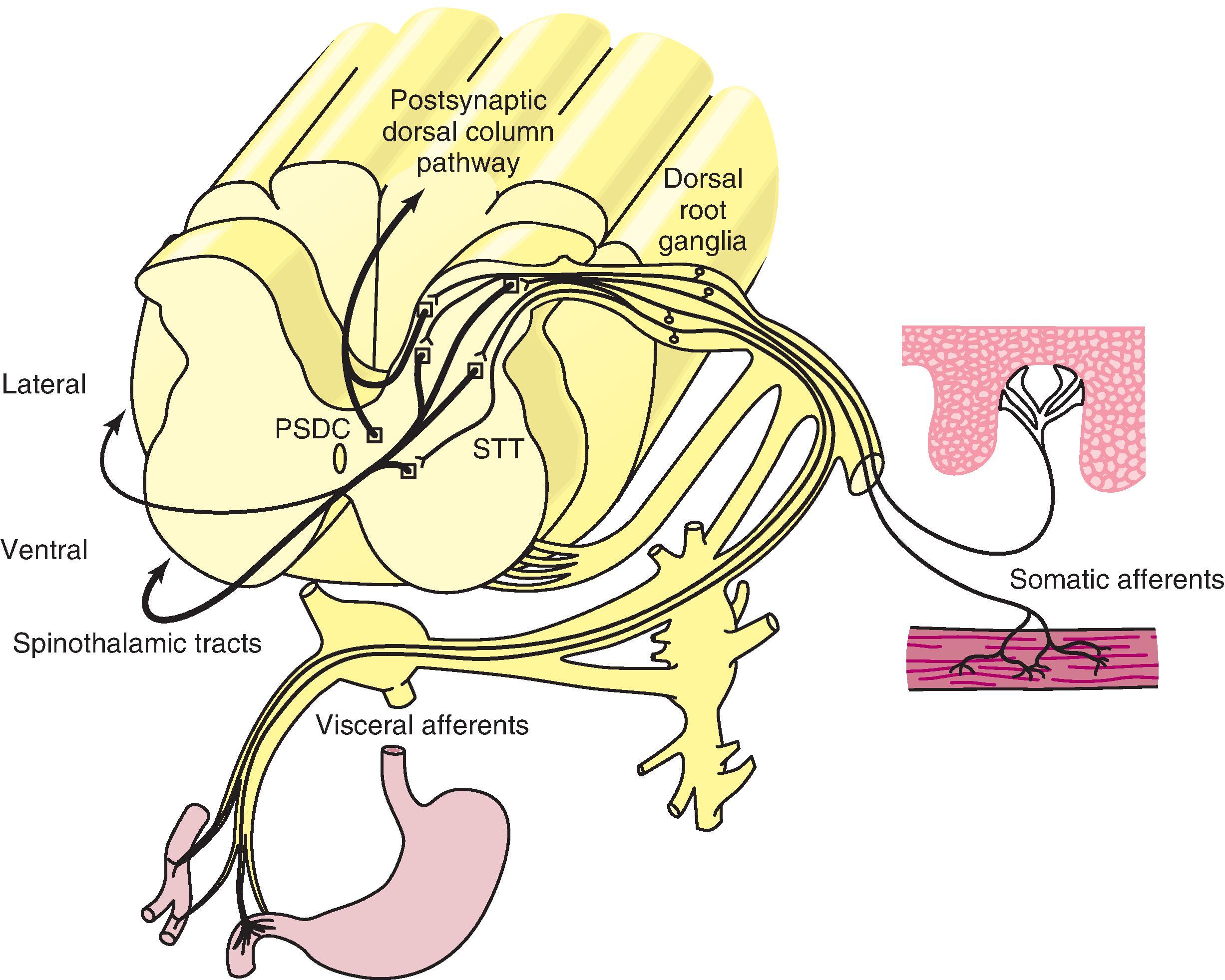
The cell bodies of the nociceptive primary afferent nerve fibers lie just outside the spinal column clustered in the dorsal root ganglia (DRG). Nociceptive DRG neurons are small and medium sized but lie among the large dorsal root ganglion cells activated by touch, pressure, and vibration. , , They extend their axons centrally to the spinal cord as dorsal roots. As in most central neuronal circuits, glutamate is the primary neurotransmitter substance in primary afferent nociceptors. Its action is modulated by neuropeptides co-released at their terminal endings and activation molecules. Calcitonin gene-related peptide (CGRP), SP, neurokinin A, galanin, and somatostatin are just a few examples of the many neuropeptides that provide neurogenic modulation and inflammation peripherally. , Modulatory neuropeptides produced in the DRG are transported quickly, both to the terminal endings in the periphery and to the spinal cord, and thus may not necessarily be evident in the cell body without experimental manipulations. The small satellite glia, the other primary component of the DRG, provide support but are capable of providing modulatory influences on the neurons that promote peripheral and central sensitization. The DRG, as well as afferent nerve endings both centrally and peripherally, are activated by pro-inflammatory mediators such as TNFα, IL-1, and IL-6. , Other activators include the small molecules adenosine, TRPV1, ATP, nitric oxide, and other reactive oxygen species. All of these pro-inflammatory substances have been identified as being present at nerve terminal sites, in the DRG, and in the dorsal horn.
Transcriptomic profiling of single sensory ganglia neurons provides further functional classification diversity and molecular drug targets. As an example, ribonucleic acid (RNA) sequence data for visceral sensory neurons has been investigated. Seven distinct subtypes of visceral sensory neurons of the splanchnic and pelvic sensory nerves retrogradely traced from their innervation of the colon have been distinguished using combined single-cell RNA sequencing, calcium imaging, qRT-PCR, and immunohistochemical validation as reviewed previously. Five subtypes enter the thoracic level from the splanchnic nerve, and two almost exclusively from the pelvic nerve are located in the lumber five (L5) DRG. Like the distinct differences in somatic and visceral sensation, the molecular phenotype of visceral transient receptor potential channel (TRP)-sensitive mechanosensors differs from analogous somatosensory mechanosensors. While both isolectin B4 positive (IB4+) and IB4- somatosensory neuron mechanoreceptor subtypes from naïve mice have similar broad tetrodotoxin (TTX)-resistant action potentials, proprioceptive neurons relaying position sense have TTX-sensitive, narrow action potentials. Transcriptional profiling has revealed that these differences in firing characteristics can be attributed to the very different ion channel expression patterns of these neurons. Nociceptive neurons are highly enriched for Cacna2d1 (α2δ1) and Cacna2d2 (α2δ2), the pharmacologic targets of gabapentin and pregabalin. Both IB4+ and IB4- somatosensory neurons express sodium ion channel Nav1.7, which, when congenitally absent in individuals, renders them without pain sensation and unprotected from injury.
The afferent fibers enter the gray matter of the spinal cord through the dorsal root entry zone and primarily innervate regions of the spinal cord within the same or adjacent spinal segments matching that spinal nerve. The two major populations of small diameter primary afferent nerves carrying pain related information to the dorsal horn are classified as 1) peptidergic containing CGRP ( Fig. 8.5 ) and SP terminating in Lamina I-II outer , and 2) non-peptidergic neurons expressing P2X3 and binding IB4 terminating in Lamina II inner . Functional distinction between these nociceptors has been noted for their involvement in inflammatory and acute pain (IB4-), while the IB4+ neurons have more involvement in mechanical and chronic pain. The IB4- neurons also express GDNF, TrkA, TRPV1, and other acid-sensing ion channels (ASIC) and have larger heat-evoked currents. Neurons that are strongly IB4+ are C-nociceptor type with higher density of Nav1.9 voltage-gated TTX-resistant Na+ channel expression defining their longer action potential durations and rise times, slower conduction velocities, and negative membrane potentials. , , A-d low threshold mechanoreceptive neurons are weakly IB4+ or IB978-0-323-71101-2.
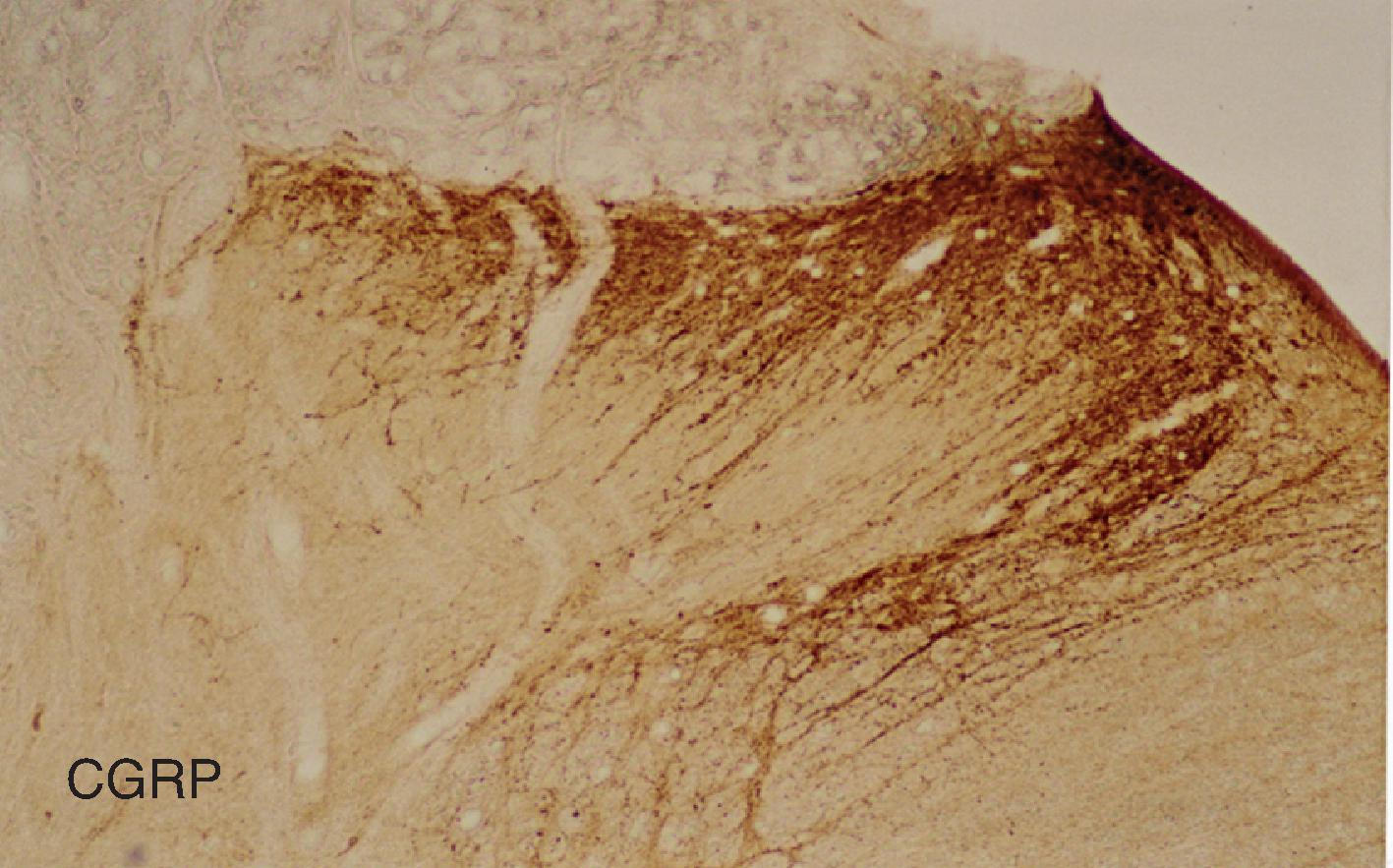
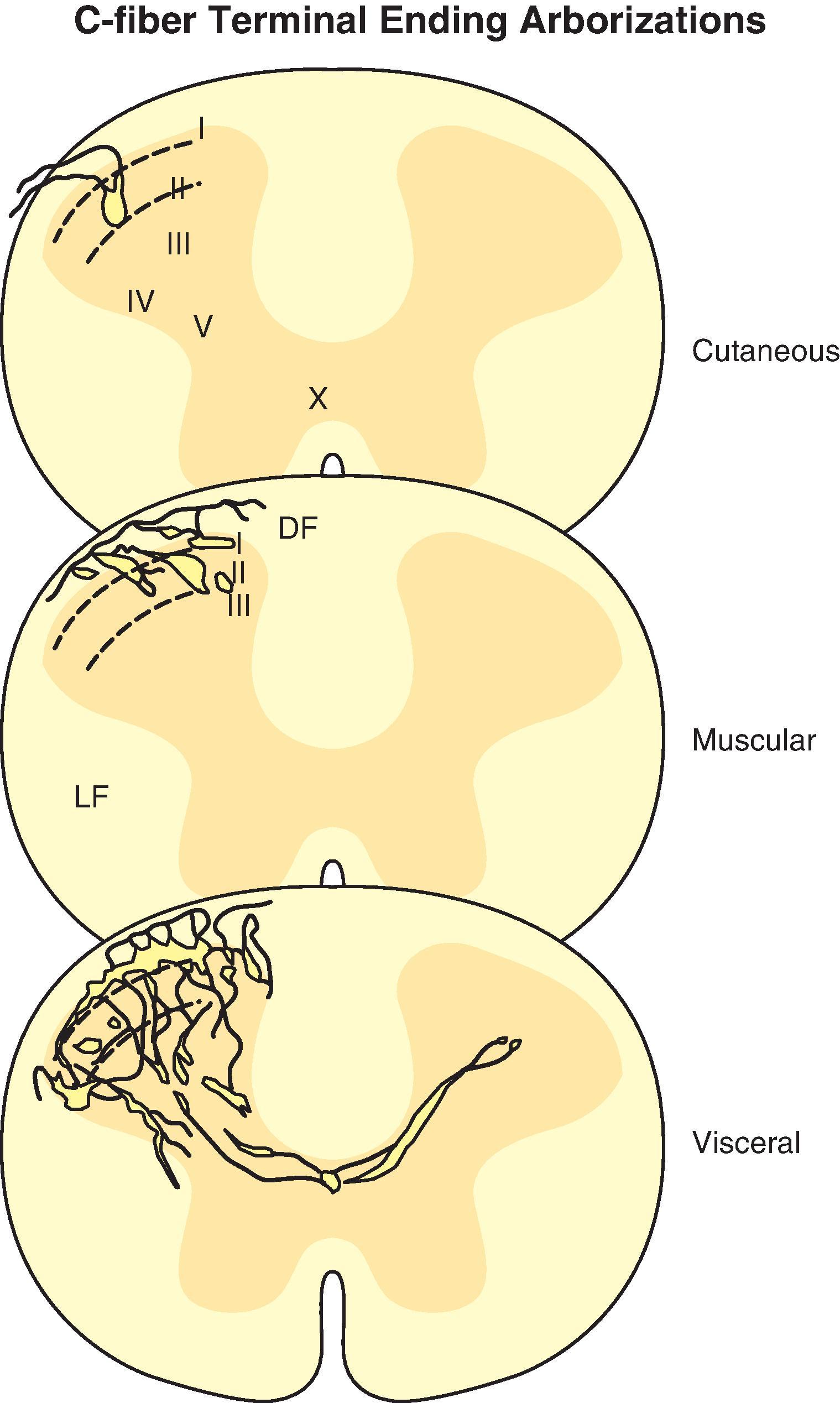
The smaller myelinated and thinly unmyelinated axonal fibers carry information about temperature, and nociceptive input perceived as pain in humans enters the spinal cord segmentally through the dorsal roots. The fibers enter Lissauer’s tract, then primarily innervate the superficial gray matter laminae I–II of the spinal cord where neuronal cell bodies and dendrites receive their arborized synaptic endings ( Figs. 8.3 and 8.4 ). The afferent fibers may also ascend rostrally or descend caudally through Lissauer’s tract for a few spinal segments. For example, CGRP fibers extend extensive fiber terminations up and down the superficial spinal cord dorsal horn through as many as six spinal levels and can even cross the midline in small numbers. , Numerous other neuropeptides have been localized in the dorsal horn terminals and neurons. Visceral afferents are rich in vasoactive intestinal polypeptide (VIP), bombesin, CGRP, and SP , and would account for some of the population of these peptides in the deep dorsal horn laminae. Other neurotransmitters and neuromodulators are discussed more thoroughly in Chapter 9.
The outer marginal layer or lamina I of the spinal cord contains nets of spiny dendrites of cells that are longitudinally arranged along the length of the cord. Somatic nociceptor C-fiber afferent endings are distributed mainly to laminae I and II in the same and adjacent segments. In contrast, visceral C-fiber afferents can extend for more than five segments before they terminate and are widely distributed in laminae I, II, V, and X ipsilaterally and in laminae V and X contralaterally (Fig. 8.4). , In addition to the afferent endings, fiber terminations of the descending pathways and the local interneurons also heavily innervate the superficial dorsal horn laminae I–II, adding to the dense crescent of terminal endings observed when stained immunocytochemically for many neurotransmitters and receptors. Cutaneous Aδ mechanical nociceptors terminate in the ipsilateral laminae I and V, and they may also have endings in lamina X and the contralateral dorsal horn. ,
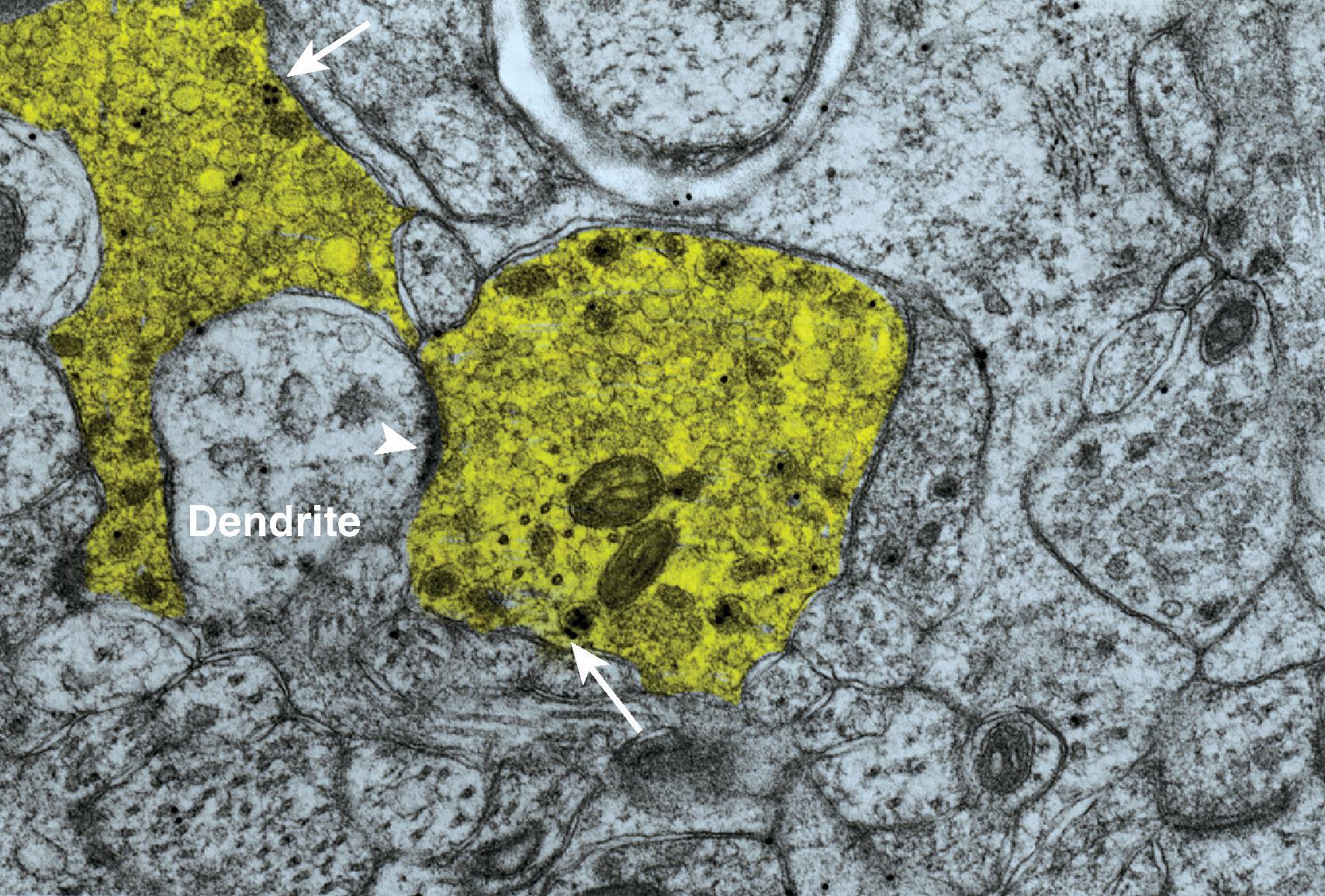
Primary afferent endings in the spinal cord dorsal horn contain small, round, clear glutamate containing vesicles released upon nerve activation for synaptic transmission and relay of nociceptive information to spinal cord neurons. The large, glomerular type afferent endings typically contain large, dense cored vesicles containing peptides ( Fig. 8.6 ). CGRP receptors on post-synaptic membranes of spinothalamic tract neurons are found opposite glomerular, elongated, and dome-shaped synaptic endings in the superficial dorsal horn. In high activation states such as after tissue damage, increased release of glutamate further increases the activation of dorsal horn neurons as it does in other sites of neural integration. The increased release is accompanied by the release of small molecule modulatory peptides expressed by primary afferent neurons, including SP, CGRP, VIP, and cholecystokinin (CCK). , These modulators, along with cytokines (TNFα, IL-1β, IL-6) released from adjacent microglia and astrocytes in the spinal cord, generate central sensitization in the case of intense and/or prolonged nociceptive stimulation. ,
When afferent nerve endings are activated persistently after an injury, increased release of glutamate and other excitatory amino acids in the dorsal horn activates spinal cord interneurons. The post-synaptic activation results in the increased firing of GABA-ergic inhibitory interneurons as well as the firing of excitatory interneurons. The gamma aminobutyric acid (GABA) hyperpolarizes the neuronal membrane that dampens incoming afferent firing. Pre-synaptic inhibition also occurs as the excessive glutamate activates autoreceptors on the afferent endings. The depolarized membrane also causes the inactivation of voltage-gated calcium channels, preventing calcium influx at the synapse. The net result of pre-synaptic inhibition is reduced amplitude of the action potential as it invades pre-synaptic terminals. This results in less calcium influx and therefore less transmitter release. However, as the pre-synaptic inhibition continues, other changes in the inhibitory control process result in prolonged membrane hyperpolarization and chloride conductance that can actually depolarize the afferent nerve endings themselves. This occurs because of the activation of GABA A receptors that open Cl − channels allowing chloride to enter the cell passively as it follows the chloride equilibrium gradient. Eventually, this will result in Cl − efflux as the equilibrium potential is exceeded, resulting in depolarization of the afferent terminal. This process is referred to as primary afferent depolarization. The depolarization can be blocked by antagonists of GABA A receptors. These findings and the initial work by John Eccles on pre-synaptic inhibition have since been extended in various ways. For example, it was later explained that with persisting afferent nerve activation, the potassium-chloride co-transporter (N + -K + -2Cl − , NKCC1) becomes insufficient to remove the influx of chloride ions from the afferent terminals. When the intercellular chloride ion concentration exceeds the equilibrium potential, a reversal occurs, and there is an efflux of chloride ions. After that, GABA has a depolarization effect. Another contributor proposed is the increase in the extracellular concentration of K + following neural activity.
Activation of nociceptive neurons resulting in primary afferent depolarization of the dorsal horn nerve ending initiates action potentials in the central afferent terminals. The action potentials generated travel back out the afferent nerve toward the periphery, where they have been recorded in A-b, A-d, and C-fibers. These “dorsal root reflexes” release neurotransmitters peripherally into the damaged tissue, such as glutamate and peptides, contributing to the increase in inflammation and peripheral blood flow, but it can be blocked centrally by GABA A and non-NMDA antagonists. Likewise, the action potentials contribute further to the increased excitatory transmitter release in the dorsal horn. These events set up a reverberating positive feedback loop, amplifying nociceptive input and increasing both peripheral and central sensitization that can promote the establishment of chronic pain if it continues.
Most of the spinal cord neurons are interneurons (~90%). The gray matter at the core of the spinal cord dorsal horn that receives sensory nerve terminations is comprised of excitatory and inhibitory integrative interneurons forming the first tier of sensory information processing, integration, and modulation of sensory inputs arising from the peripheral or internal environment. Spinal interneurons also receive input from other interneurons and descending pathways that together impact and integrate their responses.
Noxious input relayed by the primary afferent fibers is received by the dendritic arbors of neurons in the dorsal horn gray matter of the spinal cord. The gray matter is a matrix of synaptic terminations, neurons, and glial cells subdivided topographically as 10 laminae by Rexed based on histologic appearance ( Figs. 8.4 and 8.7 ). , The site of afferent fiber type terminations centrally in spinal gray matter laminae is relevant to function. The importance of the laminar organization has been upheld by other phenotypic findings, including chemogenetic, optigenetic, , and gene expression patterns. Prominent among overrepresented genes in spinal neurons are transcription factors (30%–38%) and neuropeptides (24%–26%) relevant to a specific function.
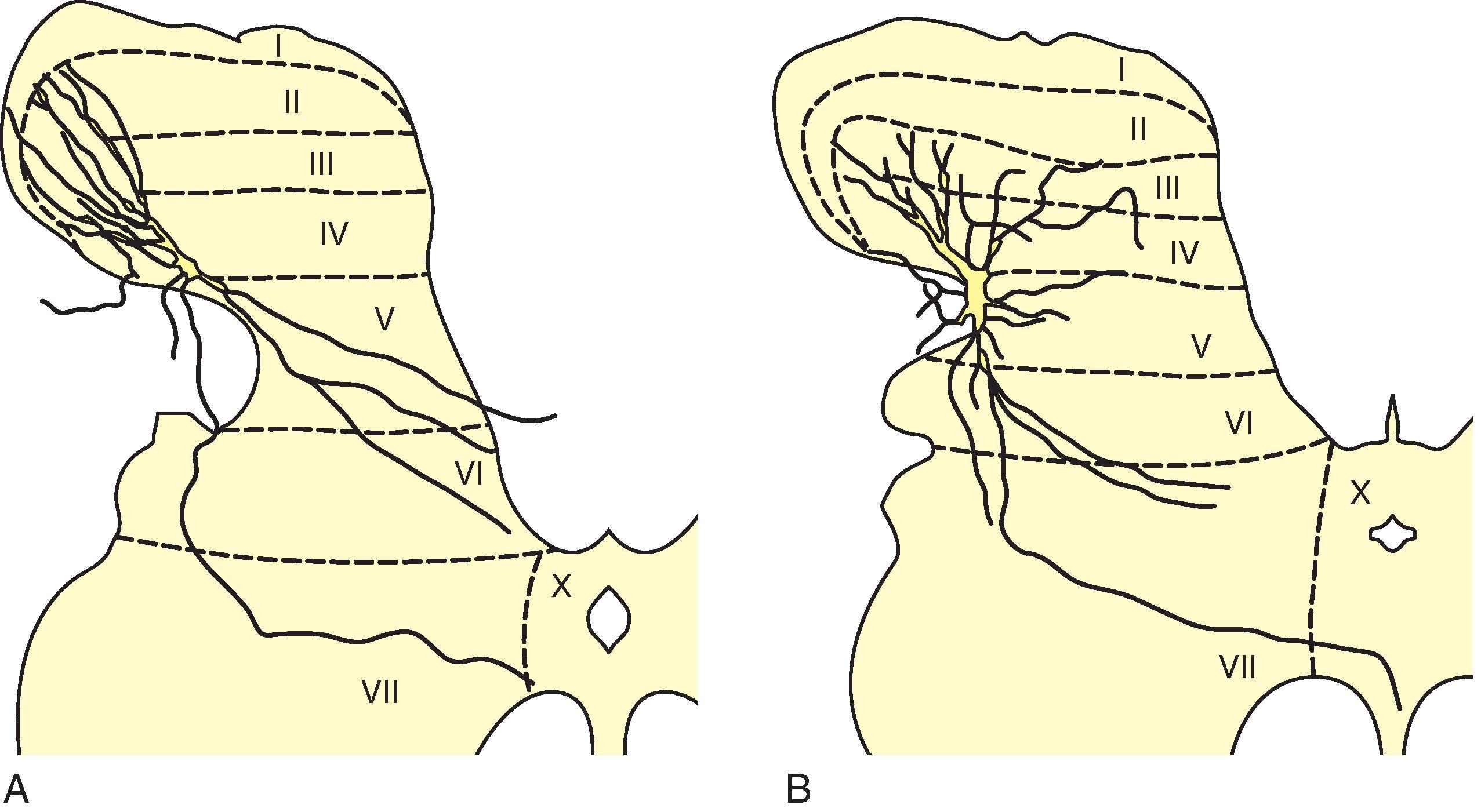
Excitatory neurons utilize glutamate neurotransmission (60%–70%) and inhibitory interneurons (30%–40%), GABA, and glycine. Fifteen types of excitatory and 15 types of inhibitory interneurons have been identified using single-cell RNA sequencing in the mouse spinal cord. Validated by spatial transcriptomics, this laminar distribution report classified heat and cold sensitive excitatory and inhibitory neuronal sets. In addition to glutamate, GABA, and glycine, interneurons contain many neuroactive substances that are presumably neurotransmitters or modulators involved in nociceptive processing by the local interneuronal circuits of the dorsal horn. These substances include adenosine, choline acetyltransferase, cholecystokinin, corticotropin releasing factor, dynorphin, enkephalin, galanin, neurokinin B, neuropeptide Y, neurotensin, protein kinase C g, somatostatin, SP, and thyrotropin-releasing hormone. , Additional information on the molecular content is described in Chapter 9.
Excitatory interneurons predominate in the dorsal horn, although lamina II or the substantia gelatinosa contains both excitatory and inhibitory interneurons that modulate projection neurons. Two non-overlapping populations have been identified characterized by either their content of gastrin-releasing peptide (GRP) or SP (radial neurons with many dendrites). The SP neurons generally show delayed firing and respond to noradrenaline and serotonin with an outward current under voltage clamp conditions, suggesting that they respond to descending monoamine modulatory input, but they have no response to A-d opioid receptor agonist. The GRP interneurons do not respond to 5-HT, or NA, but respond with an inhibitory outward current to μ-opioid receptor agonist DAMGO under voltage clamp conditions. They do not respond to d or k agonists. The GRP interneurons have local intersegmental connectivity. Typical of lamina II are the large glomerular synaptic complexes sometimes referred to as scalloped primary afferent endings that contact multiple dendrites of dorsal horn cells simultaneously ( Fig. 8.6 ). , The terminals contain large dense core vesicles, which are identified as neuropeptide-containing, with CGRP in particular. Dorsal horn protein kinase C gamma (PKCγ) containing excitatory interneurons (30%) with islet-like morphology and abundant spines have been characterized by delayed firing patterns and receive synaptic inputs from both Aδ and C low threshold mechanoreceptors. Six other excitatory and four inhibitory interneurons receiving low threshold mechanoreceptor information were identified using genetic tools in that study.
The GABA inhibitory interneurons take the form of stalked (limiting) cells and the inhibitory containing islet (central cells) interneurons. , GABA interneurons contain parvalbumin (70%), and those found in laminae I and III can also contain another inhibitory amino acid, glycine, , with dendrites extending both dorsally into lamina I and ventrally into deeper laminae of the dorsal horn. The orientation of intralaminar connectivity among the excitatory dorsal horn interneurons in lamina II is extensive in the rostrocaudal plane compared to the inhibitory interneuronal connectivity, and both have limited dendritic spread in the mediolateral plane. The central, radial, and islet cell populations receive extensive input almost entirely from within lamina II, including substantial input from both IIo and IIi. The vertical cell population receives additional input from the deep dorsal horn lamina III–IV and has been speculated to be highly modifiable in states of continued nociceptive activation.
The many interneurons in the spinal cord circuitry are also highly involved in spinal modulation that decreases the impact of noxious input. Inhibition of spinal transmission by weak mechanical stimulation can occur either through activation of segmental or supraspinal circuitries. The events that can inhibit nociceptive transmission can occur by reducing neurotransmitter release from nociceptor terminals. While physiologists have conveniently explained “surround” type inhibition of dorsal horn synaptic transmission in terms of dorsal horn axo-axonic synaptic transmission, only a few anatomic figures of this type have been described for the dorsal horn. , , Rather, the anatomic arrangement of synapses for the Gate Theory of Pain proposed by Melzack and Wall is likely mediated through axodendritic arrangements of CGRP primary afferent terminals innervating the dendrites of GABA interneurons in lamina II and reciprocal interactions that are difficult to image reliably. Fine myelinated and unmyelinated CGRP labeled afferent fibers have been observed synapsing with GABAergic dendritic profiles (islet cells), which have also been shown to contain glycine. GABA interneurons are uniquely qualified to provide the “pre-synaptic” inhibition of nociceptive input either through contacts provided by their dendrites back onto CGRP primary afferent endings or through axonal contacts back onto other dorsal horn neurons. Other inhibitory neurons in the dorsal horn contain dynorphin and glycine. Similarly, an interposed excitatory interneuron, such as one containing glutamate, would provide an excitatory boost to spinal nociceptive processing. Interestingly, neurons in lamina II, the substantia gelatinosa, do not respond to the release of SP since they lack NK 1 (SP) receptors. The SP terminal endings are located on nociceptive projection cells in both lamina I and the deep dorsal horn, including lamina I cells with NK1 receptors that rapidly internalize the receptors upon nociceptive stimulation. ,
The interneurons in the dorsal horn have been found to be highly involved in the activation of central sensitization when the nociceptive pathway is exposed to repeated noxious stimuli. Dysregulation of the balance of excitatory and inhibitory interneurons results in sustained nociceptive transmission to higher brain levels. Further discussion is provided below since glia are intimately involved in this process.
While signals from nociceptors are relayed across at least one synapse to activate spinal neurons prior to arriving at higher brain regions, input for non-noxious tactile discriminative sensory information (tactile, pressure, vibratory sense) is relayed directly to the dorsal column nuclei in the brainstem ( Figs. 8.1 and 8.3 ). However, in states of intense or prolonged overactivation, the large myelinated primary afferent fiber collateral input to the dorsal horn becomes involved in nociceptive processing. Their collateral input activates deep dorsal horn laminae III–IV neurons that then become sensitized in conditions of central disinhibition. The percentage of superficial dorsal horn interneurons that receive polysynaptic A-β input is normally relatively low but can be increased by inhibitory transmitter blockade. Increase in A-β input to the superficial dorsal horn in animal models of inflammatory or neuropathic pain hypersensitivity has also been reported. This contributes to hypersensitivity, including thermal and mechanical allodynia.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here