Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Major strides have been made in targeted cancer therapy over the past decades due to better understanding of underlying cancer biology, identification of cancer vulnerabilities, and integration of multiple therapy approaches . These advances have led to the development of small-molecule inhibitors and monoclonal antibodies that target dysregulated pathways and malfunctioning or overexpressed molecules. For example, imatinib mesylate, the ABL tyrosine kinase inhibitor, has been successfully used to treat chronic myeloid leukemia patients with the BCR – ABL translocation and is associated with very high cytogenetic and hematologic response rates . The monoclonal antibody dinutuximab against the disialoganglioside GD2 has been approved by the Food and Drug Administration (FDA) for patients with high-risk neuroblastoma. Dinutuximab facilitates the recognition and elimination of GD2 expressing neuroblastoma cells by the immune system, which results in improved event-free survival (EFS) of patients receiving this immunotherapy . Therapies have also been directed toward downstream signaling molecules, epigenetic mechanisms, and metabolic pathways within tumor cells or inhibition of angiogenesis and modulation of interactions with immune cells in the tumor microenvironment ( Fig. 3.1 ).
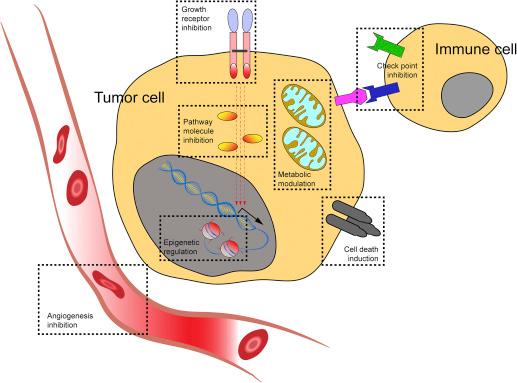
Although these new therapies have very specific mechanisms of action, therapy escape, and immune evasion, driven by the gain of new mutations in the tumor or change in epitope expression, respectively, pose a major challenge for molecularly targeted treatments. For example, approximately one-third of patients with neuroblastoma, while receiving monoclonal antibody therapy, experience treatment failure , and little is known about immune escape mechanisms such as a change in human leukocyte antigen or GD2 expression during the therapy. Real-time genomic and biologic testing during treatment can help unravel the mechanisms of clonal selection and aid the development of more effective therapies in the future.
Despite the highly effective infrastructure of clinical trials, conducting clinical studies in pediatric oncology has been inherently challenging because of low disease incidence, insufficient preclinical models and testing programs, and different perspectives on therapy success (i.e., striving for cure vs. prolonged survival). The heterogeneous clinical behavior of neuroblastoma adds yet another layer of complexity. Neuroblastoma is a rare childhood malignancy with an incidence of 11 per million children under the age of 15 years . It is the most common cancer in infants and toddlers , affecting patients at a median age of 17 months . Neuroblastoma is a developmental tumor that originates from primitive cells of the autonomic system . The heterogeneous biology of this tumor is reflected by disproportionate survival rates among patients. For example, in children with disseminated disease (stage 4), infants with stage 4S (special) disease may have spontaneous tumor regression and cure rates greater than 90% with very limited medical intervention . However, infants with MYCN amplification and those aged 18 months or older with stage 4 disease, irrespective of MYCN status, have dismal survival rates and account for 15% of childhood cancer deaths . Given this disparity in survival, several clinical features and molecular markers that predict biological behavior and outcome in neuroblastoma have been considered for developing a more accurate risk stratification system. Risk group assignment dictates the intensity of treatment and depends on clinical features such as age and initial tumor stage as well as biological factors, most importantly MYCN status . As such, it is important to note that despite sharing the same diagnosis, patients may respond differently to molecularly targeted therapy. Thus, further refinement in grouping might be necessary to identify the subset of patients in whom the effect of these agents can be maximized.
In past decades, some promising treatments for neuroblastoma have been studied and advanced in clinical trials. Treatments include tyrosine kinase inhibitors (e.g., anaplastic lymphoma kinase [ALK] inhibitors) and norepinephrine-targeted agents (e.g., 131 I-metaiodobenzylguanidine [MIBG]). Other strategies seek to improve immunotherapy by adding immune modulators (e.g., lenalidomide) or by specifically enhancing T cell-mediated cytotoxicity (e.g., cancer vaccination or chimeric antigen receptor [CAR] T cells). This chapter reviews the biological rationale of promising treatments that are currently under different stages of development and discusses their potential clinical implications.
The classic tyrosine kinase ALK belongs to the superfamily of insulin receptors and is important for neurogenesis and learning processes across multiple species . Its role in oncogenesis first emerged in the 1980s, when several research groups independently reported the t(2; 5) (p23; q35) translocation in anaplastic large cell lymphoma (ALCL) , which was later characterized as the pathognomonic nucleophosmin-ALK (NPM-ALK) fusion . Subsequently, other oncogenic ALK rearrangements were identified in B-cell non-Hodgkin lymphomas and nonlymphoid cancers, including non-small-cell lung cancer (NSCLC) and inflammatory myofibroblastic tumor (IMT) . In neuroblastoma, activating mutations in ALK have been identified in 8% of all patients with familial and sporadic disease combined , which by risk group affects 14% of children with high-risk, 6% with intermediate-risk, and 8% with low-risk disease . These patients have significantly poorer survival (5-year overall survival [OS] 59% ± 3.6%) than those without ALK aberrations (5-year OS 78% ± 1.8%), reinforcing the need for a better understanding of the role of ALK in neuroblastoma for developing new therapeutic strategies for this subgroup .
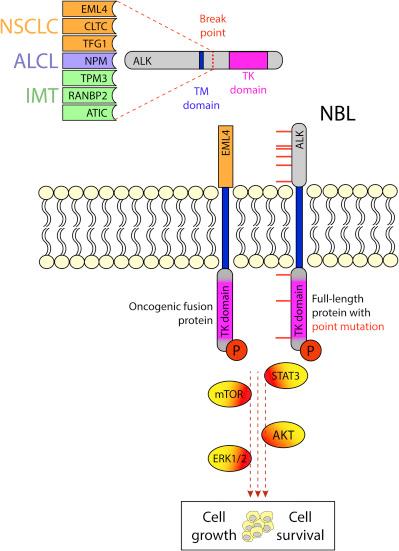
In most cancers, alterations in ALK involve chromosomal rearrangements that engage the intracellular tyrosine kinase domain of ALK and lead to constitutive activation of downstream signaling ( Fig. 3.2 ). However, patients with neuroblastoma have point mutations in ALK at three hotspot residues R1275 (43%), F1174 (30%), and F1245 (12%), which account for the vast majority of ALK mutations in this group . These alterations result in autophosphorylation of ALK and activation of the full-length ALK receptor. Mutated ALK has been shown to be the primary driver of neuroblastoma growth in preclinical studies, wherein targeted expression of mutated ALK induced tumor development in transgenic mice and knockdown of ALK resulted in proliferation arrest of neuroblastoma cells in vitro . Given these findings and the poor prognosis associated with ALK-mutant neuroblastoma, research efforts over the past years have concentrated on testing ALK kinase inhibitors that have made their way into the clinic for many adult cancers with ALK aberrations.
Oral crizotinib (PF-02341066) is a first-generation adenosine triphosphate (ATP)–competitive ALK inhibitor that is the ultimate in clinical testing and use. The compound was originally developed as an inhibitor of the mesenchymal-epithelial transition growth factor but was also found to inhibit ALK and ROS1 receptor tyrosine kinases . It acts by competitively blocking the ATP-binding pocket of ALK and prevents autophosphorylation and ALK activation. Crizotinib has been tested against various ALK rearranged and mutated tumors in vitro and in vivo and successfully translated into the clinic as first-line and subsequent-line therapy for NSCLC .
Crizotinib was first tested in the Children's Oncology Group (COG) Phase I dose-escalation trial ADVL0912 . In addition to accruing patients with relapsed solid tumors by using a rolling-six design, children with ALK -mutated tumors were eligible to participate and receive crizotinib at any point during the enrollment period. Crizotinib was tolerated up to a recommended Phase II dose of 280 mg/m 2 , which is nearly twice that of the dose recommended for adults. Hematologic and hepatic toxicities were the most common grade four adverse effects. This trial also included a Phase II expansion cohort for ALK -driven ALCL, neuroblastoma, and other cancers such as IMT. Dose-dependent response rates in this subcohort were 90% for ALCL at the recommended Phase II dose and 86% for IMT across all dose levels tested . In contrast, objective responses of the 11 patients with neuroblastoma and known ALK mutation were much lower; one patient achieved a complete response, and two patients had stable disease. Interestingly, two of the patients with responses carried an R1275Q germline mutation, which has been reported to confer sensitivity to crizotinib in preclinical studies . Of seven patients with progressive disease, three carried a sporadic F1174L mutation that has been reported in tumors with intrinsic resistance to crizotinib . Although initial biochemical studies concluded that changes in ATP-binding affinity of F1174-mutant ALK were responsible for crizotinib resistance , structural analysis later revealed that the F1174 residue was located outside of the ATP-binding pocket and therefore unlikely to induce resistance by this mechanism . It was not surprising that second-generation ALK inhibitors with a 20-fold higher ATP-binding affinity and potency than crizotinib could still not fully overcome the problem of drug resistance in F1174-mutant tumors . As discovered later, the substitution of phenylalanine by a smaller leucine residue causes a very subtle change in the ALK protein structure, primarily affecting the geometry of the αC helix and resulting in a catalytically active conformation change of ALK that will not respond to inhibition by crizotinib . This example highlights the need for more differential therapeutic targeting of ALK and raises the question of whether evaluating the conformation change of ALK rather than drug–protein interactions is more effective to assess preclinical drug sensitivity to ALK inhibitors in this subset and those with other genetically related tumors.
Current early phase clinical trials for neuroblastoma are summarized in Table 3.1 and include testing of second-generation ALK inhibitors such as entrectinib (RXDX-101; ClinicalTrials.gov identifier NCT02650401 ) and lorlatinib ( NCT03107988 ). Results from these studies are pending. In the next Phase III COG trial for newly diagnosed high-risk neuroblastoma, one treatment arm will be dedicated to ALK mutated tumors and evaluate 3-year EFS in patients receiving crizotinib in addition to standardized multimodal therapy. The trial plans to accrue patients with confirmed activating mutations or ALK copy number changes in the tumor as detected by ALK tyrosine kinase domain sequencing and fluorescence in situ hybridization, respectively. Correlating the genetic aberrations in these patients with therapy response may identify mutations associated with therapy failure and provide opportunities to understand and ultimately overcome therapeutic challenges related to resistance.
| Agent | NCT Number | Phase | Status | Sponsor |
|---|---|---|---|---|
| Ceritinib | NCT02780128 | I/Ib | Recruiting | CHOP |
| Crizotinib | NCT00939770 | I/II | Closed | COG |
| Ensartinib | NCT03155620 | II | Recruiting | NCI |
| Ensartinib | NCT03213652 | II | Recruiting | NCI |
| Entrectinib | NCT02650401 | I/Ib | Recruiting | Ignyta, Inc. |
| LDK378 | NCT01742286 | I | Recruiting | Novartis Pharmaceuticals |
| Lorlatinib | NCT03107988 | I | Recruiting | NANT |
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here