Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Prostatic intraepithelial neoplasia (PIN) is a noninvasive neoplastic proliferation of the acinar epithelium that arises within preexisting prostatic acini and ducts. PIN glands have a benign architecture but are lined with cytologically atypical cells characterized by cellular stratification, papillary formation, nuclear enlargement, and mitotic activity. Originally, PIN was divided into three grades (PIN I, II, and III); PIN now has been condensed into low-grade (LGPIN) and high-grade (HGPIN) types, based on the degree of cytologic atypia. There is no proven association between LGPIN and prostate carcinoma. HGPIN, on the other hand, is strongly associated with prostate carcinoma and is accepted as a precursor lesion for prostate carcinoma.
The prevalence rate of HGPIN increases with age. In addition, higher prevalence rates and more extensive HGPIN cases are observed in black Americans compared with white Americans. An autopsy study identified HGPIN in 7%, 26%, 46%, 72%, 75%, and 91% of black Americans between the third and eighth decade, compared with 8%, 23%, 29%, 49%, 53%, and 67% in white Americans.
The prevalence of HGPIN ranges from 4% to 16% in prostate needle biopsy specimens and from 2% to 3% in specimens obtained with transurethral resection of the prostate (TURP). HGPIN is found in 85% to 100% of radical prostatectomies performed for prostate carcinoma, reflecting a strong association between the two.
A significant subset of HGPIN and prostatic carcinoma harbors similar molecular alterations. The incidence rate of aneuploidy in HGPIN is 50% to 70%, which is slightly lower than or similar to the rate in invasive prostate carcinoma. Genetic changes tend to be very similar to those in prostate carcinoma. The most common change involves chromosome 8, including loss of 8p and gain of 8q. Other common changes include gains of chromosomes 10, 7, 12, and Y and loss of 10q, 16q, and 18q. HGPIN has decreased expression of NKX3.1, a prostate-specific homeodomain transcription factor, and p27, a cell cycle regulatory protein. TP53 gene mutation and protein accumulation, and c-myc (MYC) gene amplification are identified in some HGPIN. GSTP1, a gene involved in detoxification of genotoxins, is inactivated by promoter hypermethylation in 70% of HGPIN. Overexpression of α-methylacyl-coenzyme A racemase (AMACR) is seen in most HGPIN specimens. TMPRSS2:ERG gene fusions, the most common molecular subtype of ETS family gene fusions, occur in approximately 50% of prostate carcinomas and 20% of HGPIN intermingled with adjacent cancer, demonstrating identical gene fusions.
Prostatic intraepithelial neoplasia does not result in abnormality on digital rectal examination (DRE), nor does it lead to elevation of serum prostate-specific antigen (PSA). It can only be diagnosed with histological examination of prostatic tissue.
High-grade PIN may appear as a hypoechoic lesion, indistinguishable from prostate carcinoma, on ultrasound scan.
Prostatic intraepithelial neoplasia is not grossly visible.
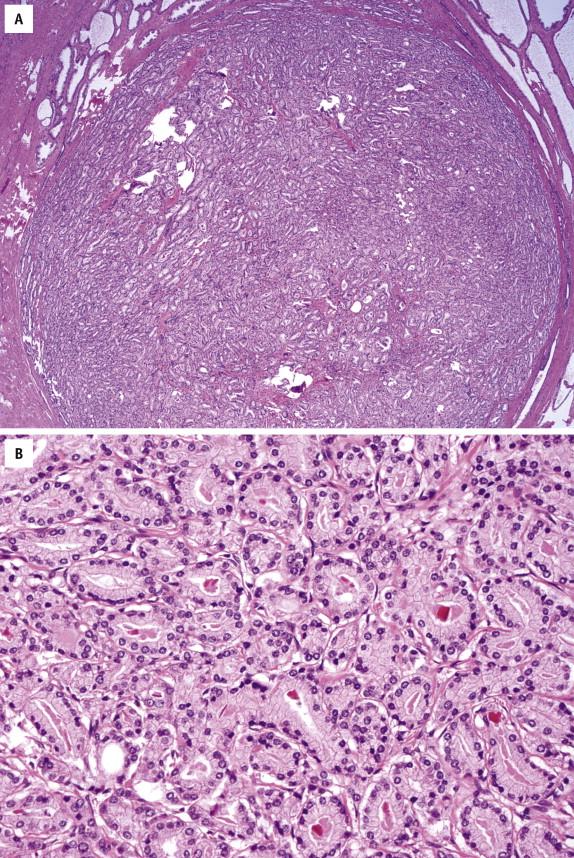
The histologic diagnosis is based on architectural and cytologic features. At low scanning power, glands that harbor PIN appear darker than normal prostatic glands as a result of higher nuclear density and increased cytoplasmic eosinophilia or amphophilia ( Fig. 2-1 , A ). LGPIN shows crowding of luminal secretory cells, irregular nuclear spacing, and stratification. Nuclei are enlarged and vary in size, although the chromatin appears normal and nucleolar prominence is rare or absent ( Fig. 2-1 , B ). The basal cell layer is intact. Because LGPIN is difficult to reproducibly distinguish from normal or hyperplastic epithelium, a diagnosis of LGPIN is not recommended.
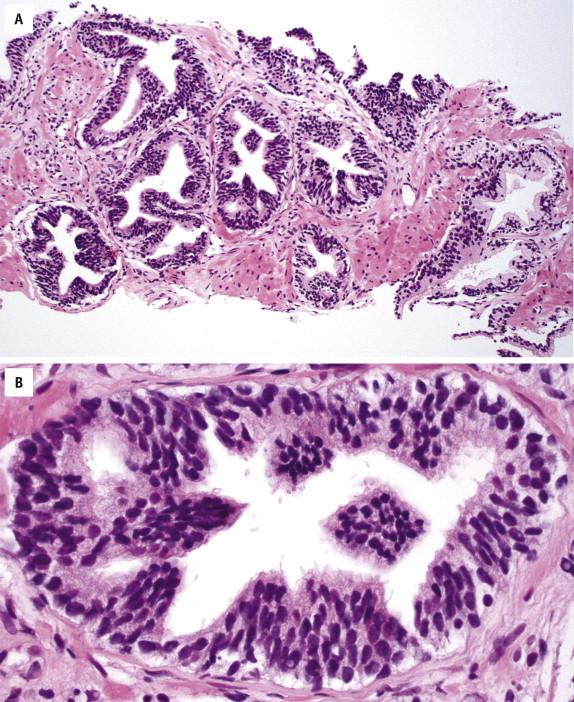
In HGPIN, the individual cells are uniformly enlarged with only a minimal degree of nuclear size and shape variation. Many cells show large and prominent nucleoli, as well as hyperchromatic and clumpy chromatin ( Fig. 2-2 ), similar to that seen in prostate carcinoma. The basal cell layer is often discontinuous and occasionally is absent in small HGPIN glands. Mitoses may be found occasionally.
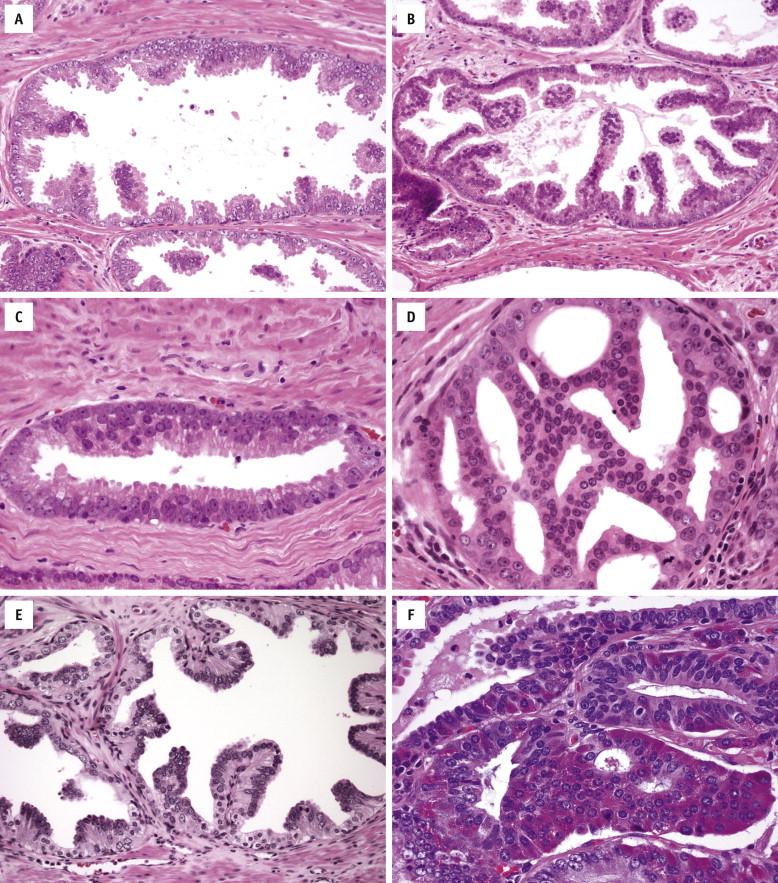
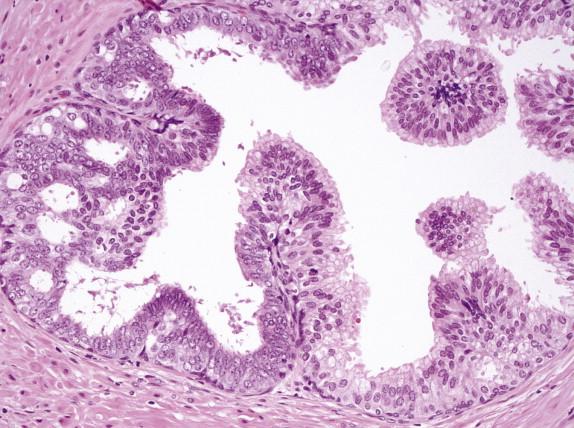
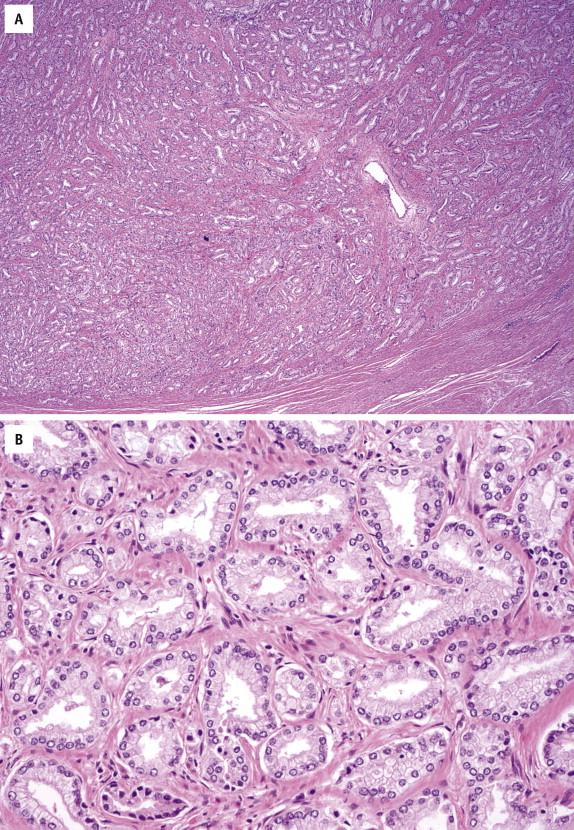
Prominent nucleoli are the best discriminator between LGPIN and HGPIN. No consensus exists as to what constitutes prominent nucleoli. However, if distinct nucleoli can be visualized at ×20 magnification, even when only a few cells are involved, the nucleoli qualify as prominent; therefore a diagnosis of HGPIN can be established. In contrast, LGPIN may have nuclear enlargement and stratification; however, it only has inconspicuous or tiny nucleoli at higher magnification. In the absence of prominent nucleoli, the presence of mitosis and significant nuclear pleomorphism can also be used to diagnose HGPIN. Four major structural patterns of HGPIN—tufting, micropapillary, flat, and cribriform—and several minor variations have been described, although these patterns are important only for diagnostic consideration and in general have no clinical significance. In a tufting pattern, which is most common, cells pile up to form undulating mounds of nuclei ( Fig. 2-2 , A ). Micropapillary pattern, the second most common pattern, has columns of atypical cells without fibrovascular cores ( Fig. 2-2 , B ). Flat pattern has no significant architectural changes ( Fig. 2-2 , C ). Cribriform pattern, the least common type, has more complex architecture, with epithelial arches and cribriform formations ( Fig. 2-2 , D ). Other rare patterns include solid and inverted variants. The inverted type is characterized by polarization of enlarged nuclei toward the luminal surface rather than the basal aspect ( Fig. 2-2 , E ), usually of tufting or micropapillary type. HGPIN may partially involve a prostatic gland ( Fig. 2-3 ), and different patterns may exist in the same gland.
High-grade PIN glands are composed of dysplastic cells that are morphologically similar or identical to prostate carcinoma. Histologic variants that correspond to virtually all the morphologic variants of prostate carcinoma have been described, including signet-ring, mucinous, foamy gland, and small cell neuroendocrine variants ( Fig. 2-2 , F ).
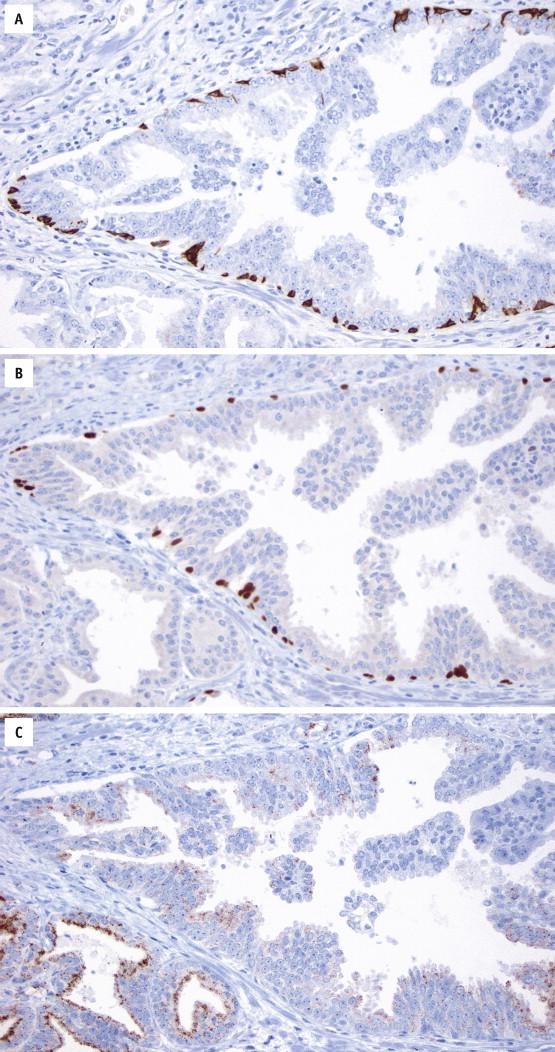
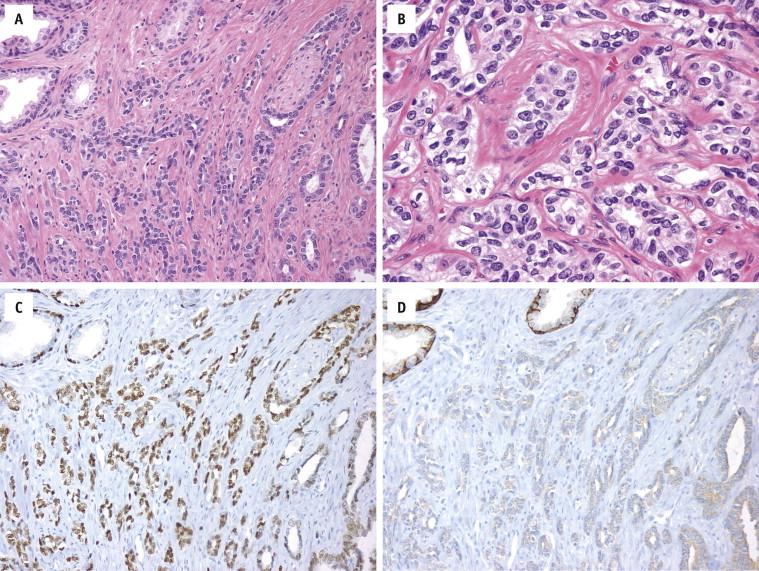
High-grade PIN is positive for pancytokeratins (AE1/3 and Cam5.2) and prostate-specific markers (prostate specific antigen [PSA] and prostate-specific acid phosphatase [PSAP]). Stains for basal cells (high–molecular-weight cytokeratin [HMWCK or 34βE12] and p63) show complete or discontinuous, occasionally absent, basal cell layers ( Fig. 2-4 , A and B ). Expression of AMACR is detected in most HGPIN samples ( Fig. 2-4 , C ). ERG protein is expressed in 15% to 20% of HGPIN adjacent to fusion positive cancer.
Normal central zone glands have architectural complexity, with eosinophilic cytoplasm and nuclear stratification, but lack the nuclear atypia that characterizes HGPIN. Seminal vesicle or ejaculatory duct epithelium does not show the characteristic nuclear changes of HGPIN, in particular, the prominent nucleoli. It often has pleomorphic hyperchromatic nuclei of degenerative nature scattered among normal-appearing nuclei. Nuclear pseudoinclusions are frequent. Golden-brown lipofuscin pigment is almost invariably present, although similar pigment can occasionally be found in HGPIN.
Prostate glands and ducts adjacent to inflammation or infarction may show nuclear atypia, including prominent nucleoli, but they do not have the prominent architectural changes of HGPIN. Benign glands after irradiation often show basal cell prominence and multilayering and streaming of cells. Squamous metaplasia is also common. Although usually pronounced, the nuclear atypia appears degenerative and varies from gland to gland. Transitional cell metaplasia is usually multilayered. However, the nuclei are frequently oval to slightly elongated with nuclear grooves. Nuclear atypia, including prominent nucleoli, is absent.
Basal cell hyperplasia often appears as small and solid nests, although some may retain lumina, in contrast to the medium-caliber to large-caliber glands affected by HGPIN. The term atypical basal cell hyperplasia is applied to basal cell hyperplasia with prominent nucleoli. However, the nucleoli are in basal cells rather than in secretory cells. Secretory cells are often found on top of the hyperplastic basal cells. Immunostains for basal cell markers (p63 or HMWCK) can sometimes aid in establishing a definitive diagnosis: basal cell hyperplasia is positive for these markers, whereas in HGPIN, the apical secretory cells are negative and only residual basal cells are highlighted by such stains.
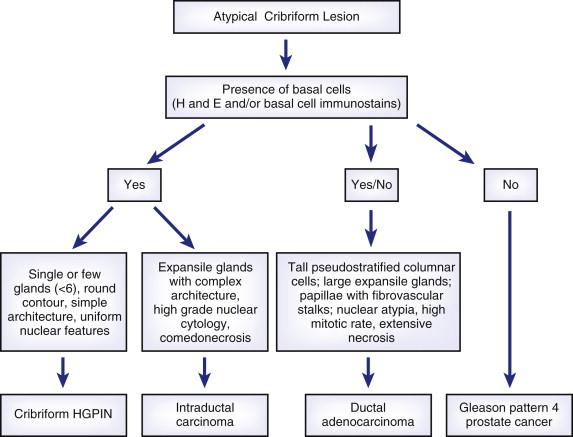
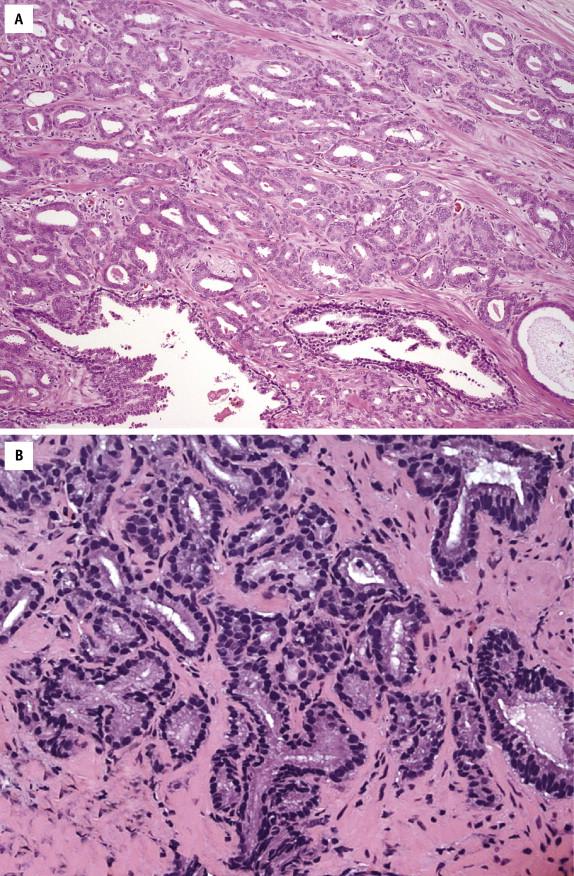
Clear cell cribriform hyperplasia is a benign condition that most commonly is found in the transition zone. It has cribriform architecture at low magnification. Cytologic atypia is absent. In addition, the basal cells are often prominent and form a collarette around the gland. Differentiation of HGPIN from invasive cribriform prostate carcinoma, ductal adenocarcinoma, and intraductal carcinoma is more problematic. All three entities consist of atypical cells that span the diameter of the involved prostatic glands. Figure 2-5 provides an algorithm for working up atypical cribriform lesions in prostate needle biopsy. Intraductal carcinoma exhibits solid or dense cribriform architecture. Comedonecrosis may be present. Markedly pleomorphic nuclei that are at least six times the size of adjacent benign nuclei may also be found in intraductal carcinoma. Unlike HGPIN, cribriform adenocarcinoma exhibits higher architectural complexity, including presence of large expansile glands, back-to-back glands, and consistent cribriforming and associated solid nests with or without intraluminal necrosis. Invasive cribriform prostate carcinoma lacks basal cell lining. The most salient histologic feature that distinguishes intraductal carcinoma from HGPIN is the presence of multiple cribriform glands with prominent nuclear atypia and comedonecrosis. When a diagnosis of intraductal carcinoma is made, a note should be included in the diagnosis to indicate that it is commonly associated with an invasive prostate carcinoma component. Ductal adenocarcinoma usually involves the prostatic urethra and verumontanum and extends into periurethral ducts, sites that are uncommonly affected by HGPIN. However, ductal adenocarcinoma may occasionally arise in the peripheral zone, or ordinary acinar prostate carcinoma may focally have ductal adenocarcinoma features. In addition, ductal adenocarcinoma may retain basal cells. Ductal adenocarcinoma is composed of tall columnar cells that are frequently pseudostratified. Unlike HGPIN, ductal adenocarcinoma may have large expansile glands and papillae with true fibrovascular stalks; the cells may show significant nuclear atypia with a high mitotic rate and extensive necrosis, features uncommon in HGPIN.
Prostatic intraepithelial neoplasia–like adenocarcinoma is characterized by stratified epithelium and may form medium-sized glands that resemble HGPIN, particularly flat or tufted patterns. Unlike HGPIN, PIN-like adenocarcinoma has no basal cells and may be associated or mingled with typical acinar carcinoma pattern.
Urothelial carcinoma that involves prostatic ducts and acini replaces the ductal-acinar epithelium with predominantly solid, highly atypical, and mitotically active neoplastic cells. The cytoplasm of these cells is dense or hard and may show squamous differentiation compared with the granular cytoplasm of HGPIN. Immunohistochemically, urothelial carcinoma, unlike HGPIN, expresses GATA3, HMWCK, and p63 but is negative for PSA and PSAP.
High-grade PIN in prostate needle biopsy is a risk factor for detection of prostate carcinoma in subsequent biopsies, whereas LGPIN is not. The incidence rate of detection of invasive cancer on rebiopsy ranges from 36% in patients who were diagnosed with HGPIN in the early 1990s to 22% in patients who were diagnosed after 2000. The cancer risk is 18% after an LGPIN diagnosis and 15% to 19% after a diagnosis of benign prostatic tissue. Between 80% and 90% of prostate carcinomas are detected on the first rebiopsy after an HGPIN diagnosis, and rebiopsy may detect persistent HGPIN in 5% to 43% of cases.
The number of cores involved with HGPIN is the one pathologic parameter that predicts a higher risk of subsequent carcinoma on rebiopsy. HGPIN on two or more cores is associated with such a sufficiently high risk of subsequent cancer that rebiopsy within a year of the initial HGPIN diagnosis is warranted. Rebiopsy should sample the entire gland because HGPIN confers an increased risk for cancer throughout the gland. For cases with a single core of HGPIN on needle biopsy, recommendations are that men do not need a routine repeat needle biopsy within the first year after the diagnosis of HGPIN in the absence of other clinical indicators of cancer.
Atypical glands adjacent to HGPIN (PINATYP) may represent a microinvasive prostate cancer budding off the HGPIN gland or tangential sectioning of the HGPIN gland ( Fig. 2-6 ). PINATYP seems to confer a risk of cancer equivalent to that after a diagnosis of “atypical glands suspicious for carcinoma” and higher than HGPIN alone, with a risk of approximately 50%. All patients with PINATYP should undergo rebiopsy within 3 to 6 months.
Noninvasive neoplastic proliferation of the acinar epithelium that displays moderate cytologic atypia within the preexisting prostatic acini and ducts
Prevalence increases with age
Higher prevalence and more extensive HGPIN in black Americans compared with white Americans (7%, 26%, 46%, 72%, 75%, and 91% in black Americans versus 8%, 23%, 29%, 49%, 53%, and 67% in white Americans between the third and eighth decades)
Does not result in abnormal DRE findings or elevated PSA level
Diagnosed only with histological examination
May appear as a hypoechoic lesion, indistinguishable from prostate carcinoma
Presumptive premalignant lesion
HGPIN in needle biopsy denotes a 20% to 25% risk of finding prostate carcinoma in subsequent rebiopsy; consider rebiopsy within a year of the initial HGPIN diagnosis if two or more biopsy cores are involved
No treatment within a year for HGPIN as an isolated finding in needle biopsy (in the absence of other clinical indicators of cancer)
DRE, Digital rectal examination; HGPIN, high-grade prostatic intraepithelial neoplasia; PSA, prostate-specific antigen.
Not visible grossly
Luminal cell crowding, irregular spacing, piling-up with chromatin hyperchromasia, clumping and prominent nucleoli
Architectural patterns: flat, tufting, micropapillary, and cribriform
Histologic variants: signet-ring, mucinous, inverted, and small cell neuroendocrine
Secretory cells + for pancytokeratins and low–molecular-weight cytokeratin; + but often reduced expression of PSA and PSAP; 60% to 80% + for AMACR (P504S)
HMWCK (34βE12) and p63 demonstrate complete or discontinuous basal cell layers
ERG protein is expressed in 15% to 20% of HGPIN adjacent to fusion-positive cancer
Prostatic central zone glands
Seminal vesicle/ejaculatory duct epithelium
Reactive atypia from inflammation, infarction, or radiation
Metaplasia (transitional cell, squamous cell)
Hyperplasia (clear cell cribriform hyperplasia, basal cell hyperplasia)
Invasive prostate carcinoma with cribriform pattern
Ductal adenocarcinoma
Intraductal carcinoma
PIN-like adenocarcinoma
Urothelial carcinoma
AMACR, α-Methylacyl-coenzyme A racemase; HGPIN, high-grade prostatic intraepithelial neoplasia; HMWCK, high–molecular weight cytokeratin; PSA, prostate-specific antigen; PSAP, prostate-specific acid phosphatase.
Prostate carcinoma is the second most common malignant disease in men and the sixth leading cause of cancer death in men worldwide, with 238,590 new cases and 29,720 deaths from prostate cancer estimated in the United States in 2013. The incidence and mortality rates have increased in the last several decades, in part because of the introduction of newer diagnostic tests, such as PSA screening. However, prostate carcinoma is a less prominent cause of cancer death, accounting for 10% of all cancer deaths in men in the United States. The incidence varies greatly with geographic location, ethnic background, and age. The highest incidence rate is seen in the United States, Canada, Australia, western Europe, and Scandinavian countries, whereas prostate carcinoma is relatively uncommon in south central and eastern Asia and northern Africa. In the United States, blacks have much higher incidence and mortality rates than do whites. The incidence rate rises dramatically with age. The clinical disease is uncommon before the age of 50. The hazard of detection is 40 times greater for men older than 65 years, compared with younger men. One autopsy study showed an 83% prevalence rate among patients 90 years of age and older.
Multiple genetic and environmental factors are involved in prostate carcinogenesis. Age, family history, and race are definitive risk factors. A family history of prostate carcinoma confers a higher risk. The degree of risk is related to the age and number of affected relatives, with the greatest risk conferred by a father or brother with onset before 40 years of age. These observations strongly support a genetic basis for prostate carcinoma. Many susceptibility loci and several candidate genes have been identified for hereditary prostate cancer. Racial background, with American blacks having higher incidence, higher grade, and more extensive cancer, may be related to various genetic and environmental factors.
Dietary fat and sex hormone levels are probable risk factors for prostate carcinoma. High fat intake, especially from red meat, is strongly associated with development of prostate carcinoma. On the other hand, fruit and vegetable consumption may protect against prostate carcinoma.
Sex hormones play an important role in the development and growth of prostate carcinoma. Testosterone is converted to more potent dihydrotestosterone (DHT) by the enzyme 5-α-reductase. Testosterone and DHT then bind to the androgen receptor (AR), which is a transcription factor that regulates androgen-dependent cell growth. Genetic polymorphisms in 5-α-reductase and AR genes affect the enzyme and receptor activities.
Other potential risk factors include increased plasma insulin-like growth factor I expression, exposure to herbicides, exposure to cadmium, obesity, and low levels of vitamin D.
Recent studies suggest that hereditary prostate cancer is a complex disease that involves multiple susceptibility genes with variable phenotypic expression. Family-based studies have identified four strong candidate susceptibility genes involved in the hereditary form of prostate cancer: RNAse L/HPC1 on 1q24-25, ELAC2/HPC2 on 17p11.2, MSR1 on 8p22-23, and HOXB13 on 17q21-22. These genes do not account for all the hereditary prostate cancer, and more than 10 other genes also have been implicated. Of particular importance to the inherited susceptibility for prostate carcinoma are the common polymorphisms in a number of low-penetrance genes, or genetic modifiers. Specific combinations of these variants, in the proper environmental setting, are hypothesized to profoundly affect the risk of development of prostate carcinoma. Mutations in any of the classic oncogenes and tumor suppressor genes are detected only infrequently in sporadic prostate carcinomas. They harbor nonrandom somatic genomic alterations. The most common chromosomal alterations include losses at 1p, 6q, 8p, 10q, 13q, 16q, and 18q and gains at 1q, 2p, 7, 8q, 18q, and Xq. Many genes have been implicated in prostate carcinogenesis and progression, including GSTP1, NKX3-1, PTEN, CDKN1B, KLF6, Rb, p53, c-myc, bcl-2, c-kit, and STAT5 . Monoallelic and biallelic PTEN losses have been reported in approximately 42% and 10% of prostate cancers, respectively. Rearrangement in chromosome 22 between TMPRSS2 and ERG is the most common event in prostate cancer, seen in about half of cases. A significant co-occurrence of TMPRSS2:ERG rearrangement and PTEN loss has been reported in prostate cancer. SPOP mutations represent the most common point mutations in primary prostate cancer, detected in 6% to 13% of multiple independent cohorts. SPOP mutations are mutually exclusive with TMPRSS:ERG fusion and other ETS rearrangements. SPINK1 protein is overexpressed in a subset of ETS-negative prostate cancers. Alterations in the AR gene itself and in interacting proteins that can modulate the activity of the AR and its downstream target genes are recurrent lesions implicated in the development and progression of prostate cancer.
Most prostate carcinomas are asymptomatic. About three fourths of prostate carcinomas arise in the peripheral zone, and some can result in abnormal findings on DRE. Rarely, prostate carcinoma can lead to urinary obstruction when a large tumor mass arises in the transition zone or extends into the transition zone from the peripheral zone or invades the bladder neck. Locally aggressive prostate carcinoma that involves the bladder and rectum can cause hematuria, rectal bleeding, or obstruction.
Rarely, patients present with symptoms and signs that are related to prostate carcinoma metastatic to different anatomic sites, most commonly bone, regional lymph nodes, lung, and brain.
Currently, most prostate carcinomas are diagnosed on prostate biopsies performed for elevated serum PSA levels or abnormal DRE results. In approximately one fourth of patients, the serum PSA value is not elevated and abnormal DRE results are the reason for prostate biopsy.
Prostate-specific antigen is a serine protease secreted by the epithelial cells of prostatic ducts and acini to liquefy the seminal coagulum. Benign and malignant prostatic tissue produces PSA. Only a small percentage of PSA in serum is free; most is complexed with α-1-antichymotrypsin and α-2-macroglobulin. Patients with prostate carcinoma in general have higher serum PSA values than those without the disease. In the recent past, a serum PSA level greater than 4 ng/mL was considered abnormal and a reason for patients to undergo prostate biopsy. However, serum PSA lacks sensitivity and specificity for prostate carcinoma. Approximately 70% to 75% of prostate carcinomas have serum PSA values greater than 4 ng/mL. The positive predictive value of serum PSA is higher when the concentration is greater than 10 ng/mL or accompanied by abnormal DRE findings. In addition, some nonmalignant conditions, including benign prostatic hyperplasia (BPH), inflammation, infarction, instrumentation, and ejaculation, also increase the serum PSA level. The current American Urologic Association (AUA) guidelines for use of PSA emphasize that PSA is one among many factors to be considered when estimating an individual’s risk for prostate cancer. The other factors to be considered are percentage of free PSA, PSA velocity, DRE findings, family history, ethnicity, and biopsy history. The AUA no longer recommends a PSA threshold cutoff value for biopsy as recommended in the past. The National Comprehensive Cancer Network (NCCN) recommends a baseline PSA and DRE at age 40 years. Provided the DRE results are normal, if the baseline PSA is less than 0.6 ng/mL, then annual screening may begin at age 45 years. If the baseline PSA is more than 0.6 ng/mL, then screening should begin annually at age 40 years. The NCCN recommends prostate biopsy if the PSA is more than 2.5 ng/mL.
Other modifications of PSA testing have been introduced to improve the test’s specificity, including the ratio of free to total PSA, which is decreased in prostate carcinoma; the PSA density, which is the ratio of total PSA to prostate glandular volume as measured with transrectal ultrasound scan (TRUS) and is higher in prostate carcinoma; and the PSA velocity, or the rate of PSA change over time, which is increased in prostate carcinoma.
The PCA3 assay is a recently introduced genetic-based diagnostic test that quantitatively detects PCA3 messenger RNA (mRNA) expression in urine and prostatic fluid with transcription mediated amplification. PCA3 is expressed only in prostatic tissue and is significantly overexpressed in malignant prostate tissue. PCA3 mRNA is highly overexpressed in more than 95% of prostate cancer tissue. A PCA3 score is calculated as the ratio: PCA3 mRNA/PSA mRNA × 1000. A PCA3 score of 35 has been adopted as a cutoff for biopsy. The lower the PCA3 score, the lower the risk of a positive repeat biopsy. The sensitivity of PCA3 in urine is 66% (∼62% probability of positive biopsy); the specificity is 76% (versus 47% specificity for serum PSA). PCA3 is independent of prostate volume, number of prior biopsies, and patient age. The test is unaffected by benign prostatic hyperplasia and prostatitis. PCA3 test is not used as the only sufficient screening tool for prostate cancer detection because cancer has been detected in prostate biopsies despite a negative PCA3 result and, on the other hand, excessively high PCA3 score has been reported despite absent detectable tumor on biopsy.
Prostate-specific antigen testing has significantly affected the clinical and pathologic attributes of detected prostate carcinoma. Most prostate carcinomas detected with PSA screening are of intermediate grade and confined to the prostate. In addition, 5% to 27% of prostate carcinomas are considered potentially clinically insignificant.
Preoperative PSA is often used in combination with needle biopsy Gleason score and clinical stage to predict pathologic stage, including the probability for organ-confined disease, extraprostatic extension, seminal vesicle invasion, and lymph node metastasis, and the likelihood of disease recurrence after therapy. These probability estimates are helpful in discussions with patients about risks versus benefits in choice of treatment options.
Prostate-specific antigen levels may also be used to monitor the response to various treatments. After radical prostatectomy, the serum PSA should drop to an undetectable level; elevated PSA levels signal recurrence or residual disease. After radiation therapy, three consecutive increases in PSA level after it initially reaches a nadir indicate a biochemical failure.
The standard method for detection of prostate carcinoma is with TRUS-guided sextant core biopsies that systematically sample the parasagittal midlobe regions of apex, middle, and base. Several modifications of the sextant protocol have been made. More recently, the standard has been to sample the prostate gland with approximately 10 to 12 cores, in particular from the more lateral aspect of the peripheral zone, where most prostate carcinomas arise. In cases with high suspicion for cancer despite negative biopsy results, “saturation biopsy” may be performed, with collection of 10 to 15 biopsies from each side of the prostate. The addition of transition zone biopsies is also used typically only when the initial biopsy results are negative and the clinical suspicion for cancer is high. More needle cores may also be taken for larger prostates.
Information concerning the anatomic locations of needle biopsies should be preserved by either embedding the samples in separate blocks or inking them in different colors. This information is critical for prognostication and therapy planning. It is also important when a diagnosis of “atypical glands suspicious for prostate carcinoma” (ATYP) is made. Rebiopsy strategy entails concentration in the initial atypical region in addition to sampling of the rest of the prostate. At least three levels of each biopsy core should be cut for hematoxylin and eosin (H&E) staining. An option is for one or two intervening sections to be cut on positive charged slides for potential immunohistochemical studies.
Transurethral resection of the prostate mainly detects transition zone prostate carcinoma and bulky peripheral zone prostate carcinoma that extends into the transition zone. Initially, eight cassettes should be submitted in random fashion. In younger patients (<65 years of age), submission of all the tissue is justified to identify all T1a lesions (i.e., those in which tumor occupies <5% of the total specimen). If T1b disease (in which tumor occupies >5% of the total specimen) is found on the initial eight slides, it is not necessary to submit additional tissue. However, if T1a prostate carcinoma is found on the initial eight slides, the remaining tissue should be submitted for review.
The sonographic appearance of prostate carcinoma is not specific, with 70% to 75% of prostate carcinomas being hypoechoic and the remaining being isoechoic and indistinguishable from surrounding tissue. Many significant prostate carcinomas are missed with TRUS. TRUS remains as a means of guidance for prostate needle biopsies. Innovative ultrasound techniques (including ultrasound contrast agents, three dimensional [3-D] and four-dimensional [4-D] sonography, elastography, and harmonic sonography) promise benefits in comparison with standard TRUS to accurately diagnose prostate cancer.
Computed tomographic scan does not play a significant role in the diagnosis of primary prostate cancer; however, new scanners improve the accuracy of prostate cancer staging. The combination of conventional and functional magnetic resonance imaging (MRI) techniques (including diffusion-weighted imaging, dynamic contrast-enhanced MRI, and magnetic resonance spectroscopy) can provide information for differentiating prostate cancer from noncancerous tissue and can be used for MRI-guided biopsies, especially in patients with persistent elevation of serum PSA and previous negative TRUS-guided biopsies.
A bone scan provides the most sensitive method for detection of bony metastasis. ProstaScint (Cytogen Corporation, Princeton, NJ), a radioisotope-labeled monoclonal antibody against prostate-specific membrane antigen (PSMA), may detect microscopic metastasis. The sensitivity can be improved with use in combination with PSA, biopsy Gleason score, and clinical stage data.
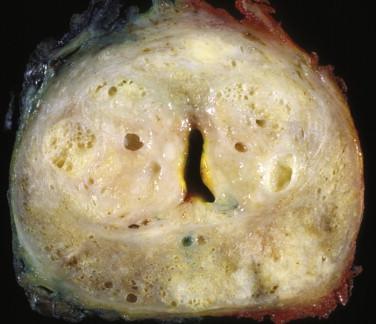
Gross examination of TURP specimens is of little significance because benign processes can mimic prostate carcinoma. On radical prostatectomies, grossly identifiable prostate carcinoma is typically of higher grade and stage and larger diameter. In contrast to the adjacent normal benign prostate tissue, which appears tan and spongy, grossly evident prostate carcinoma is firm and solid and ranges in color from white-gray to yellow-orange ( Fig. 2-7 ). Prostate carcinomas discovered with PSA screening are less visible grossly; these cancers are often small (<5 mm) and of lower grade and stage.
Local extraprostatic extension typically occurs anteriorly for transition zone cancer and posteriorly and posterolaterally for peripheral zone cancer. Prostate carcinoma can also spread superiorly into the bladder neck. Rarely, it can penetrate Denonvilliers’ fascia posteriorly to involve the rectum. Metastatic prostate carcinoma most commonly involves regional lymph nodes and bones of the pelvis and axial skeleton, where a characteristic osteoblastic response is often elicited.
Prostate carcinoma has a constellation of architectural, cytoplasmic, nuclear, and intraluminal features.
Gland-forming prostate carcinomas are more crowded than benign glands and typically exhibit a haphazard growth pattern, with malignant glands oriented perpendicular to each other and irregularly separated by smooth muscle bundles. They also display infiltrative growth pattern, with malignant glands situated between or flanking benign glands ( Fig. 2-8 , A ). When prostate carcinoma becomes less differentiated, it loses glandular differentiation and forms cribriform structures, fused glands, poorly delineated glands, solid sheets or cords, or even single tumor cells.
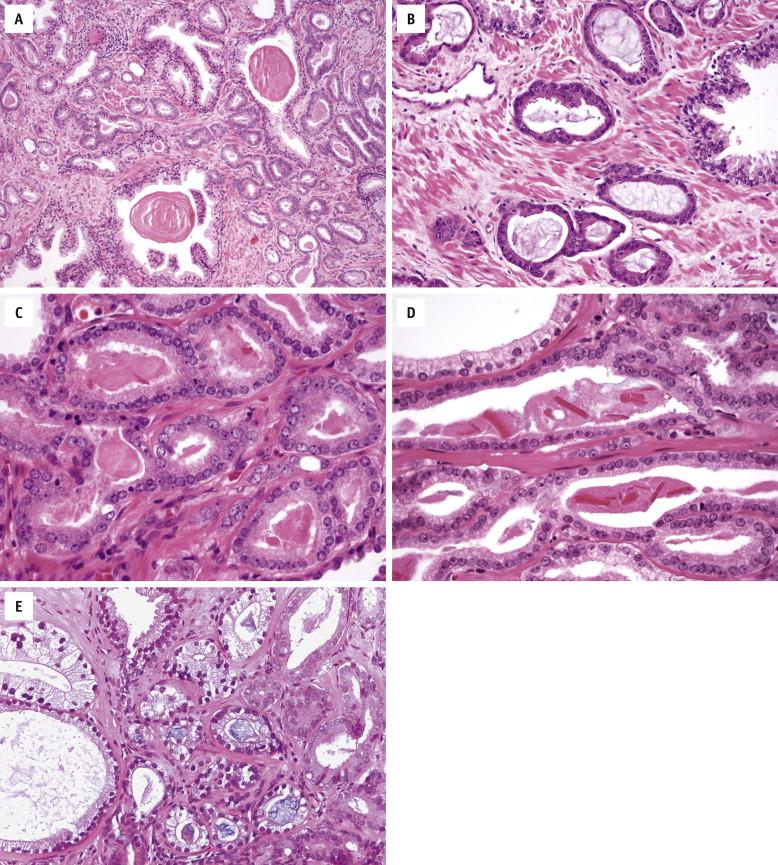
In contrast to benign glands with irregular and undulating luminal borders, prostate carcinoma glands are smaller and have straight luminal borders. They may have amphophilic, or darker, cytoplasm that is evident even at low magnification ( Fig. 2-8 , B ). However, low-grade prostate carcinoma often has pale-clear cytoplasm, indistinct from benign glands. Prostate carcinoma typically lacks lipofuscin pigment.
Typically, prostate carcinoma displays nuclear characteristics distinct from surrounding benign glands, including enlarged nuclei and prominent nucleoli ( Fig. 2-8 , C and D ). Some prostate carcinomas lack prominent nucleoli yet have enlarged and hyperchromatic nuclei. Mitoses and apoptotic bodies are more common in prostate carcinoma, although they are rarely found in benign glands and are still not frequently seen in malignant glands. Cancer nuclei, even in poorly differentiated glands, show little variation in size and shape.
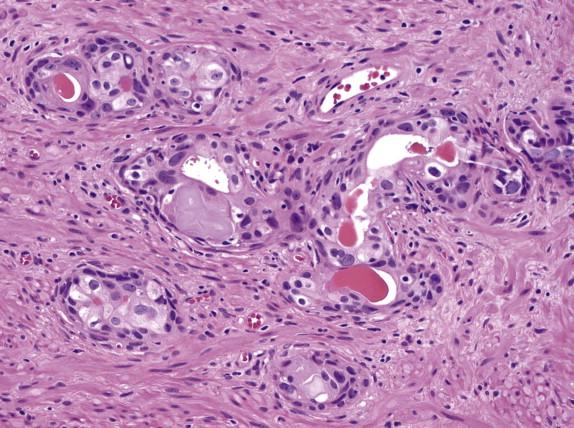
Crystalloids (dense eosinophilic, crystal-like structures within the glandular lumina; Fig. 2-8 , D ) are found more commonly in cancer than in benign glands. However, they are also frequently found in adenosis, a benign condition that mimics low-grade prostate carcinoma. Intraluminal pink, acellular, dense secretions ( Fig. 2-8 , C ) and blue-tinged mucin ( Fig. 2-8 , E ) are additional findings seen preferentially in prostate carcinoma. In contrast, corpora amylacea are common in benign glands and are only rarely seen in prostate carcinoma.
Ordinary prostate carcinoma does not elicit a stromal inflammatory or desmoplastic response. Ductal adenocarcinoma of the prostate, however, may induce such stromal reactions with fibrosis-containing hemosiderin-laden macrophages.
Three histologic features are diagnostic of prostate carcinoma because they have not been described in benign glands. Mucinous fibroplasia, or collagenous micronodules, occurs as a delicate fibrous tissue with ingrowth of fibroblasts within or adjacent to cancer glands ( Fig. 2-8 , F ). Glomeruloid formation is created by intraluminal proliferation of malignant cells and is often surrounded by a crescentic space that resembles a renal glomerulus ( Fig. 2-8 , G ). Perineural invasion with cancer glands completely or nearly completely encircling the nerve is pathognomonic of prostate carcinoma ( Fig. 2-8 , H ). Benign glands can occasionally be found to abut a nerve; however, circumferential extension of benign glands entirely around a nerve has not been described.
Histologic variants of prostate carcinoma account for 5% to 10% of all the carcinomas in the prostate and are typically seen in association with ordinary acinar prostate carcinoma. These variants often differ from the latter in clinical, immunophenotypic, ultrastructural, and genetic features. Many also differ in prognosis and may prompt a different therapeutic approach. The following is a brief description of the histologic features of several variants. Other variants, including ductal adenocarcinoma, urothelial carcinoma, small cell carcinoma, squamous cell carcinoma, basal carcinoma, and intraductal carcinoma are discussed separately.
The cancer glands have scant cytoplasm ( Fig. 2-9 , A ) and may be confused with benign atrophy. The diagnosis is based on several features. First, atrophic prostate carcinoma displays an infiltrative growth pattern, with atrophic cancer glands intermingling with larger benign glands. In contrast, benign atrophy usually has a lobulated configuration. Atrophic prostate carcinoma has significant cytologic atypia, namely, nuclear enlargement and prominent nuclei ( Fig. 2-9 , B ). Finally, atrophic prostate carcinoma is often intermixed with nonatrophic ordinary prostate carcinoma.
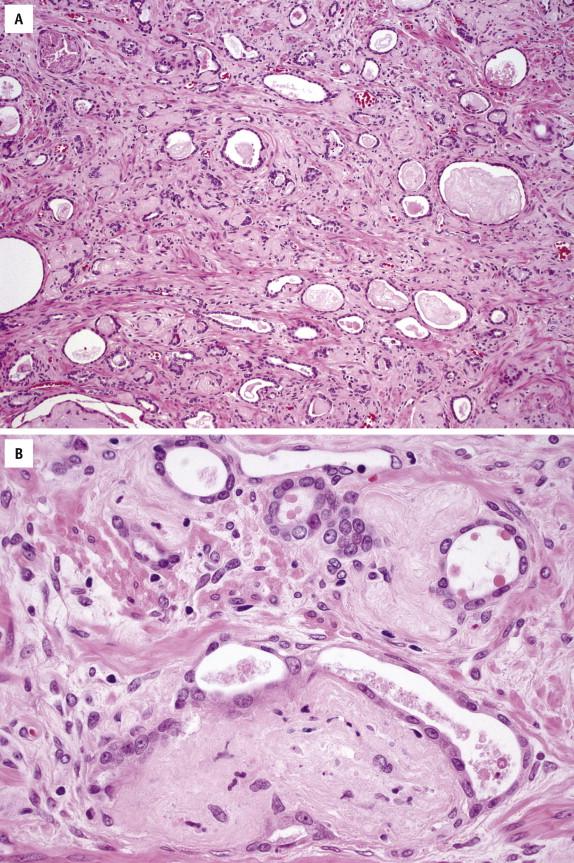
Resembling benign prostatic glands, pseudohyperplastic prostate carcinoma glands are large with branching and papillary infoldings ( Fig. 2-10 , A ). However, the malignant glands are much more closely packed than benign glands, and they display malignant nuclear features typical of prostate carcinoma ( Fig. 2-10 , B ). The diagnosis of pseudohyperplastic prostate carcinoma in needle biopsy is often difficult and necessitates immunohistochemistry to verify the absence of basal cells ( Fig. 2-10 , C ). Despite its benign appearance, pseudohyperplastic prostate carcinoma may be associated with typical intermediate-grade cancer and can exhibit aggressive behavior.
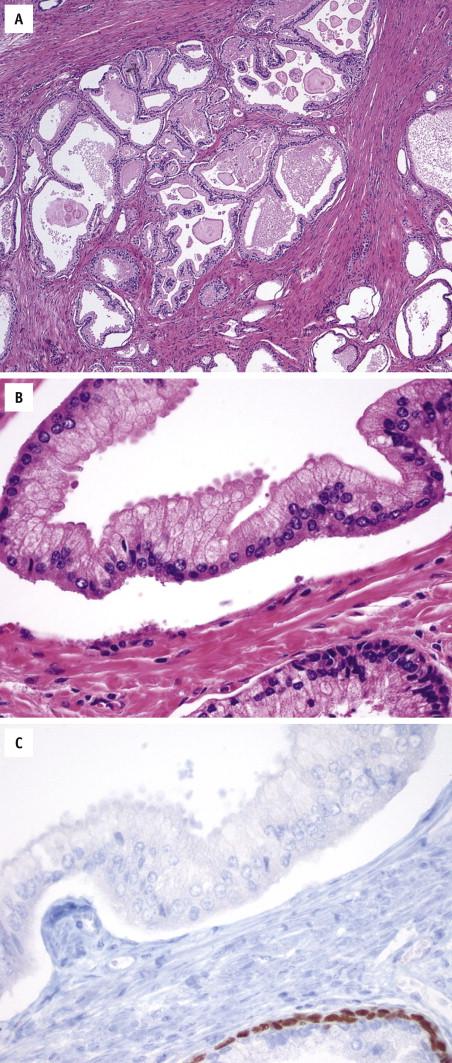
Microcystic adenocarcinoma represents a form of atrophic or pseudohyperplastic variants. It is characterized by cystic dilation and rounded expansion of glands, with a flat luminal lining. Microscopically, the mean glandular size is 10-fold greater than that of usual acinar adenocarcinoma. Intraluminal crystalloids and intraluminal blue mucin are commonly seen ( Fig. 2-11 ). It should be assigned a Gleason score 6.
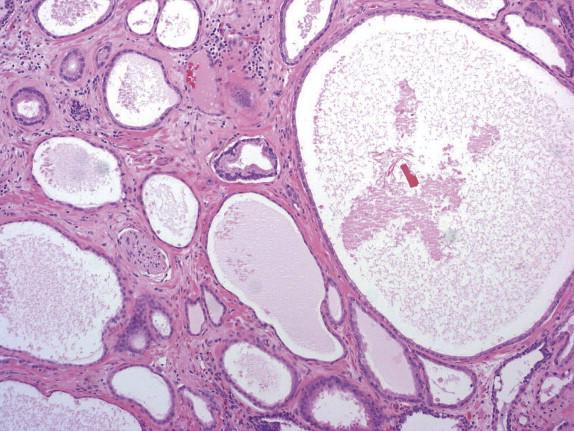
Cancer cells have abundant foamy or xanthoma-like cytoplasm with a very low nuclear/cytoplasmic ratio ( Fig. 2-12 , A ). Although the cytoplasm is xanthomatous in appearance, it contains empty vacuoles rather than lipid. The nuclei of foamy gland prostate carcinoma cells are typically small and hyperchromatic and lack the cytologic features of ordinary prostate carcinoma ( Fig. 2-12 , B ). The diagnosis of foamy gland prostate carcinoma is based on its architectural pattern of crowded or infiltrative glands, abundant foamy cytoplasm, and frequent intraluminal, dense, pink, acellular secretions. Basal cells are absent with immunohistochemistry. Despite the bland cytology, most of the cases that include an associated nonfoamy cancer are Gleason score 6 or greater.
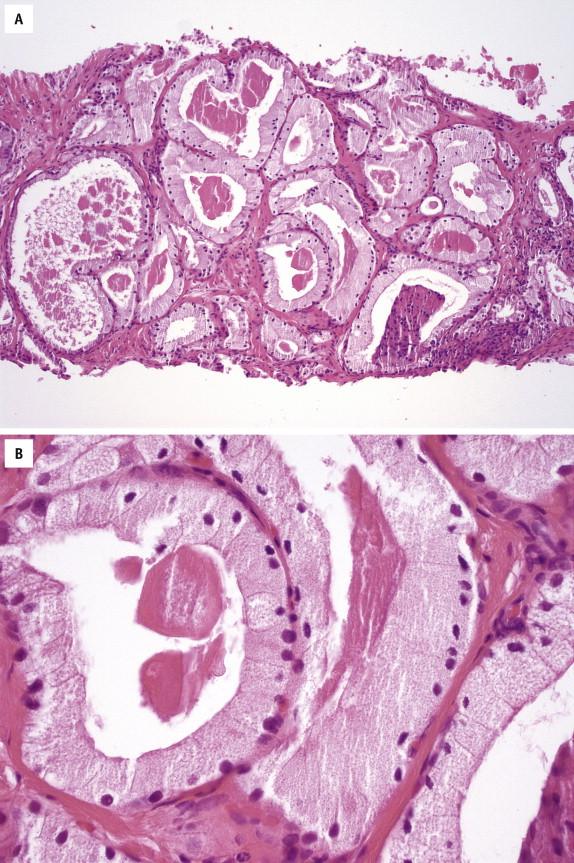
Mucinous prostate carcinoma is defined as a cancer in which 25% or more of the tumor consists of lakes of extracellular mucin. Prostate carcinoma with less than 25% mucinous component should be classified as having mucinous features, whereas prostate carcinoma with intraluminal mucin without lakes of extracellular mucin is not considered as mucinous prostate carcinoma. Clinically, the average age for mucinous prostate carcinoma is similar to that for the ordinary acinar prostate carcinoma, although the clinical stage at presentation is often advanced, with locally advanced or metastatic disease. Microscopically, tumor cells float in lakes of extracellular mucin that are sharply demarcated from the stroma ( Fig. 2-13 , A ). Tumor cells are arranged in cribriform configurations, cords, strands, acini, or tubules. Cytologically, the cells appear bland with occasional prominent nucleoli ( Fig. 2-13 , B ). They are positive for PSA and PSAP.
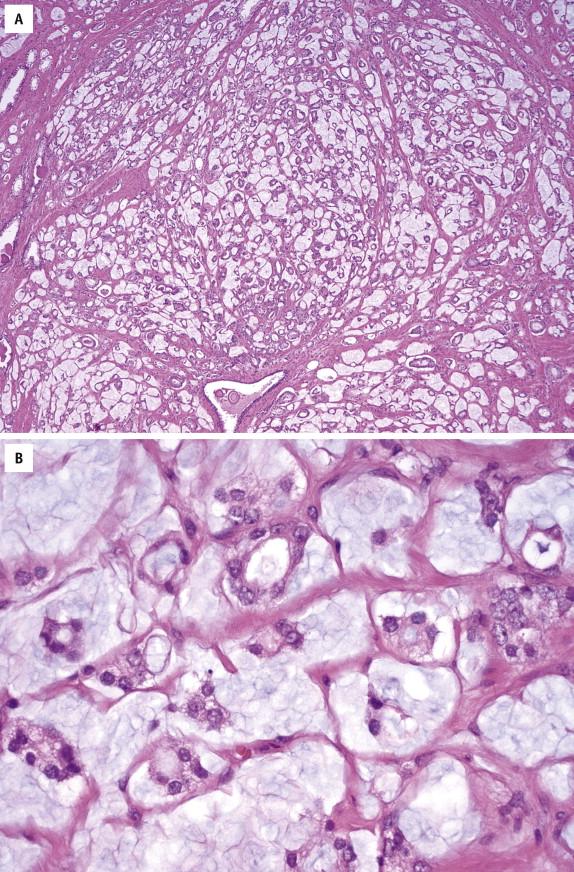
Prostatic intraepithelial neoplasia–like adenocarcinoma is a recently recognized unusual variant of prostatic adenocarcinoma that cytologically and architecturally resembles high-grade PIN. Microscopically, this variant of adenocarcinoma is characterized by noncribriform stratified epithelium similar to flat, tufted, or micropapillary patterns of PIN ( Fig. 2-14 , A ). Immunohistochemical stains for basal cells are completely negative ( Fig. 2-14 , B ). In addition, a greater degree of glandular crowding or disorder is found than is usually seen in PIN. The term PIN-like ductal adenocarcinoma has been used to describe some cases that display high columnar cells ( Fig. 2-14 , C ), like ductal adenocarcinoma. The assigned Gleason score is 6.
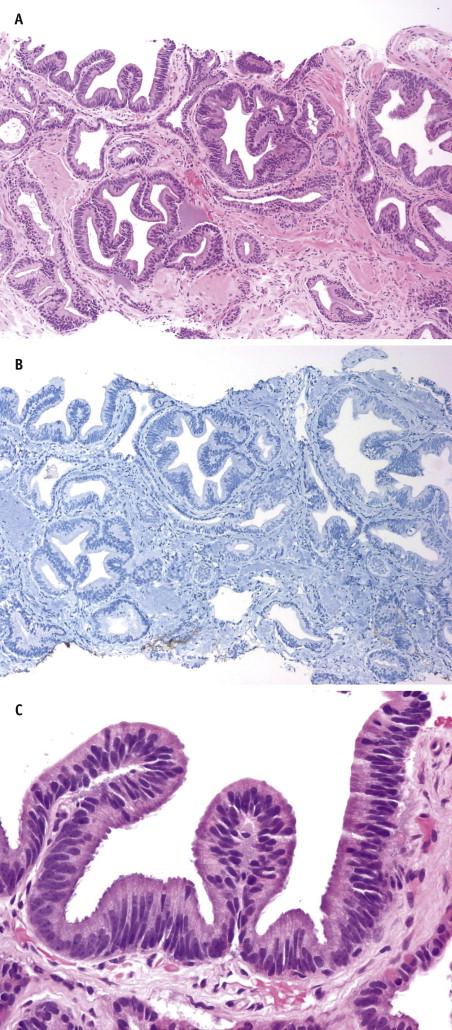
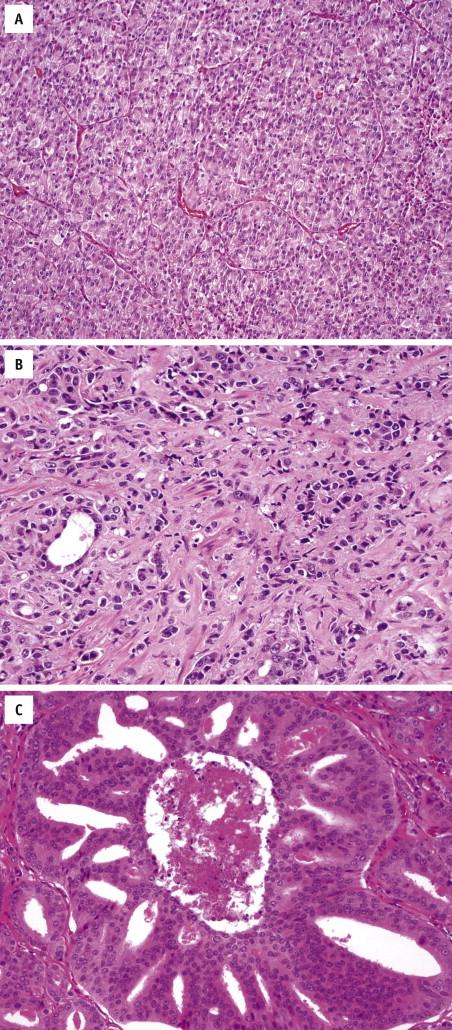
Defined as 25% or more of tumor mass consisting of signet-ring–appearing cells, this histologic variant is a rare entity with an aggressive clinical course. Microscopically, the signet-ring–like tumor cells display nuclear displacement and indentation with clear cytoplasmic vacuoles ( Fig. 2-15 ). In most cases, these vacuoles contain lipid rather than mucin, as with true signet cells. The cancer cells grow in sheets, in small clusters, and as single cells. They are invariably mixed with ordinary acinar prostate carcinoma components. Immunostains for PSA and PSAP are positive in most cases, and stains for CK7, CK20, and HMWCK are negative in all cases. Before a diagnosis of prostatic signet-ring carcinoma is established, a metastasis from other anatomic sites, including stomach, lung, colon, and pancreaticobiliary system, must be excluded. On the other hand, a prostatic signet-ring carcinoma should be considered when one encounters a signet-ring–cell carcinoma of unknown primary, especially if mucin stains are negative.
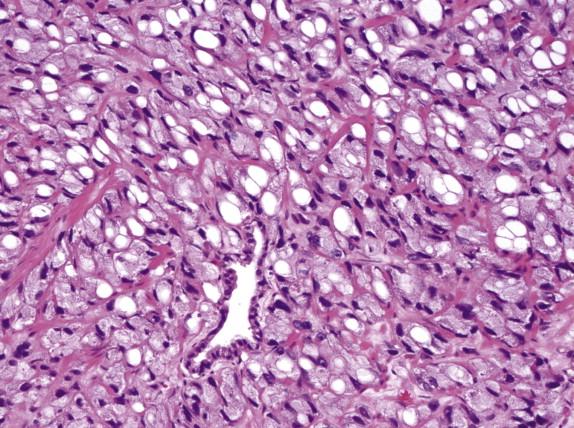
Sarcomatoid prostate carcinoma is composed of both malignant epithelial and spindle cell elements. It may be a de novo diagnosis, or patients may have a history of prostate carcinoma treated with radiation or hormonal ablation therapy or both. Serum PSA values are within normal limits in most cases, despite the frequent presence of nodal and distant metastases. The 5-year survival rate is less than 40%. Microscopically, sarcomatoid prostate carcinoma is biphasic, with a glandular component that shows variable Gleason patterns and a sarcomatoid component that often exhibits nondescript malignant spindle cell proliferation ( Fig. 2-16 ). Specific mesenchymal differentiation can also be present, including osteosarcoma, chondrosarcoma, and rhabdomyosarcoma. Immunohistochemically, the epithelial elements are positive for PSA or pancytokeratins, whereas the sarcomatoid elements react with markers of corresponding mesenchymal differentiation and variably express cytokeratins.
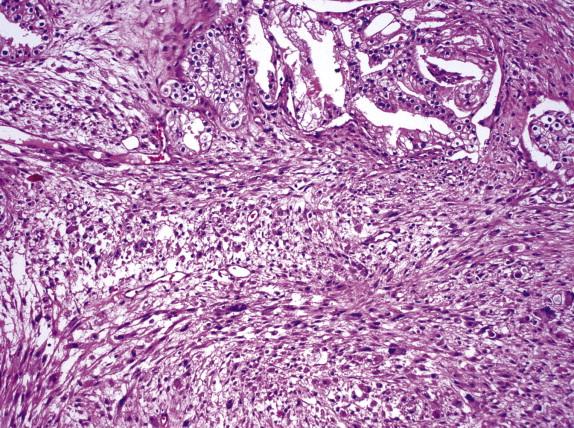
This extremely rare variant of prostate cancer has histological features that are indistinguishable from those of large cell carcinoma of the lung. Most of the patients reported had a history of prostatic adenocarcinoma treated with hormonal therapy. Microscopically, the tumor is characterized by sheets and ribbons of cells with large nuclei, coarse chromatin, and prominent nucleoli admixed to acinar adenocarcinoma. The prognosis is dismal. Tumor cells show focal immunoreactivity for PSA and PSAP and diffuse staining for chromogranin, synaptophysin, CD56, and CD57.
Pleomorphic giant cell carcinoma is a rare (less than 10 cases reported), highly aggressive histologic variant composed of high-grade prostatic adenocarcinoma with prominent bizarre, anaplastic giant cells. In addition to the pleomorphic giant cell component, multiple coexistent histologic components have been reported, including high Gleason score conventional prostate cancer, small cell carcinoma, squamous carcinoma, and ductal adenocarcinoma. Some of the patients present with distant metastases. Both acinar adenocarcinoma and anaplastic giant cell components display immunoreactivity for cytokeratins AE1/AE3 or CAM 5.2; staining for PSA is variable in the giant cell component. Most cases are reported in patients with a history of prostate cancer treated with androgen ablation and radiation. The disease course is aggressive.
This uncommon, poorly differentiated carcinoma histologically resembles lymphoepithelioma of the nasopharynx but does not seem to be related to Epstein-Barr virus (EBV) infection. Few cases have been reported in the prostate, with a mean age of 76 years (range, 69 to 82 years). Patients present with obstructive symptoms, elevated PSA values, and locally advanced disease. Tumor has a syncytial growth pattern with indistinct cell border. A dense lymphocytic infiltrate is present admixed with plasma cells, neutrophils, and occasional eosinophils. In all cases, the lymphoepithelioma-like component is admixed with acinar adenocarcinoma. The carcinoma cells are positive for PSA, PSAP, and AMACR. The clinical outcome is poor.
This recently described unusual variant of prostate cancer is characterized by p63 expression of the tumor cells in a nonbasal distribution ( Fig. 2-17 , C ) and lack of basal cell stain for HMWCK and CK5/6 ( Fig. 2-17 , D ). Microscopically, the tumor is composed predominantly of glands, nests, and cords of cells with atrophic cytoplasm, hyperchromatic nuclei ( Fig. 2-17 , A ), and visible nucleoli ( Fig. 2-17 , B ). Cases range from Gleason patterns 3 to 5. Most of the tumors reported so far were organ confined at radical prostatectomy.
Treatment for prostate carcinoma, especially radiation and androgen ablation, can have profound effects on both benign and malignant prostate tissue.
Both cancer and benign glands respond variably to radiation, with some glands showing marked radiation effect and others showing no such effect. For benign prostatic tissue, the classic alterations are extensive glandular atrophy with stromal predominance. Glands retain their lobular architecture, although individual glands often assume irregular, angulated contours. Secretory cells are atrophic, and basal cells become hyperplastic and multilayered, with scattered, markedly atypical nuclei that appear degenerated ( Fig. 2-18 ). These cells are positive for basal cell immunohistochemical markers. Radiation-treated prostate carcinoma, on the other hand, has a variable appearance, from no or minimal radiation effect to significant radiation-induced changes. The classic radiation-induced changes in prostate carcinoma are a decrease in number of cancer glands, poorly formed glands, and single cancer cells; abundant vacuolated cytoplasm; nuclear pyknosis; and stromal fibrosis ( Fig. 2-19 ). Architecturally, the key feature for recognition of the radiated cancer is closely packed glands with a haphazard infiltrative growth pattern and the presence of infiltrating individual cancer cells—architectural findings that are inconsistent with a benign process. Up to 30 months may be needed for cancer to clear after radiation therapy. Therefore rebiopsy should be performed no sooner than this time. Findings on rebiopsy predict the prognosis, with positive biopsy results that show no treatment effect having a worse outcome than negative biopsy results and cancer with treatment effect having an intermediate prognosis.
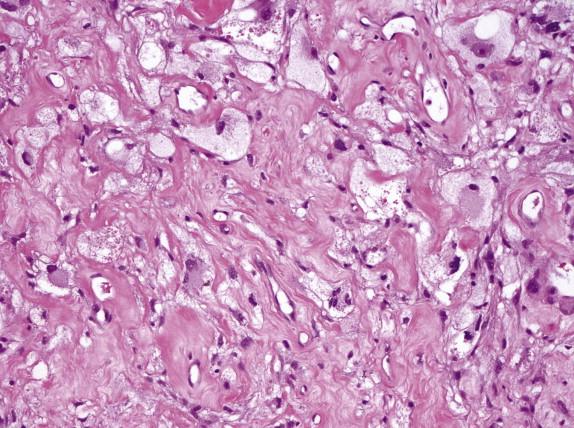
Androgen deprivation via surgical castration or pharmacologic blockade has become a common treatment modality for patients with locally advanced or metastatic prostate carcinoma. More recently, preoperative (neoadjuvant) hormonal ablation is sometimes used for clinically localized prostate carcinoma, to retard tumor growth before surgery if there is a delay between biopsy and definitive resection. In benign prostate tissue, androgen ablation results in pronounced glandular atrophy and stromal predominance. Luminal secretory cells become atrophic and exhibit nuclear pyknosis and cytoplasmic clearing. Basal cells undergo hyperplasia and squamous metaplasia. The histology of prostate carcinoma may be significantly altered after hormonal therapy. The neoplastic glands tend to be smaller and atrophic, with compressed or obliterated lumina ( Fig. 2-20 , A ). Tumor cells develop pyknotic nuclei and abundant foamy or clear vacuolated cytoplasm ( Fig. 2-20 , B ). Sometimes, single tumor cells are widely scattered within the stroma, resembling histiocytes. Complete dissolution of tumor cells may also be seen, leaving empty clefts or mucin aggregates ( Fig. 2-20 , C and D ). Immunohistochemically, cancer cells after hormonal therapy are positive for pancytokeratins, PSA, and AMACR but negative for HMWCK, although the staining intensity for PSA and AMACR may be reduced.
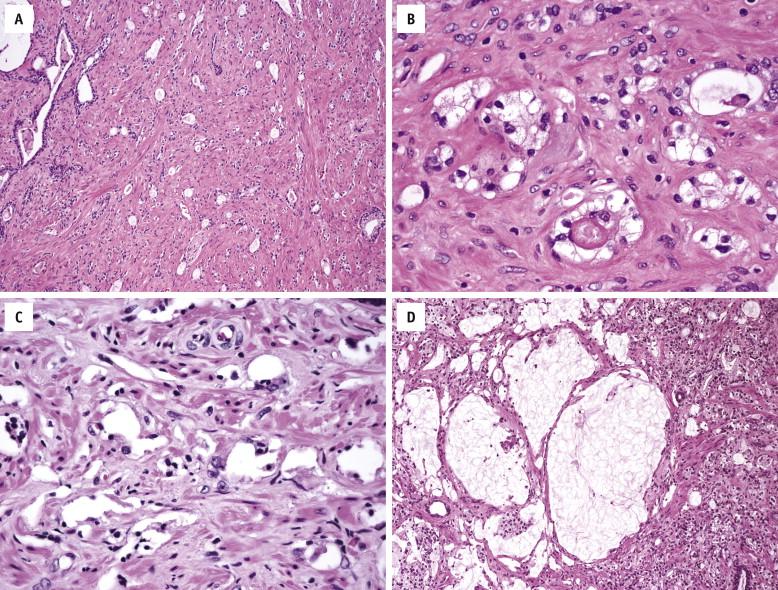
Cryoablation of the prostate is a promising technique in the treatment armamentarium for clinically localized prostate cancer and for localized recurrent prostate cancer after radiation therapy. Cryosurgical ablation refers to rapid deep freezing of the prostate. Multiple cryoprobe needles filled with circulating liquid nitrogen transform the prostate gland into an ice ball, which results in tissue destruction and death of benign and malignant cells. Postcryosurgery prostate tissue shows typical features of repair, including marked stromal fibrosis and hyalinization ( Fig. 2-21 , A ), hemosiderin deposition ( Fig. 2-21 , B ), and stromal hemorrhage, inflammation, and squamous metaplasia. Coagulative necrosis ( Fig. 2-21 , C ) may be present between 6 and 30 weeks from therapy, followed by patchy chronic inflammation. Dystrophic calcification is infrequent and usually appears in areas with the greatest reparative response. Biopsies after treatment may reveal no evidence or residual tumor, even in some patients with elevated PSA levels. Ghosts of malignant cells may be appreciated in areas that show coagulative necrosis ( Fig. 2-21 , C ). In some cases, the cancer shows no evidence of treatment effect or immune response, which suggests lack of inclusion of that area in the ablation killing zone.
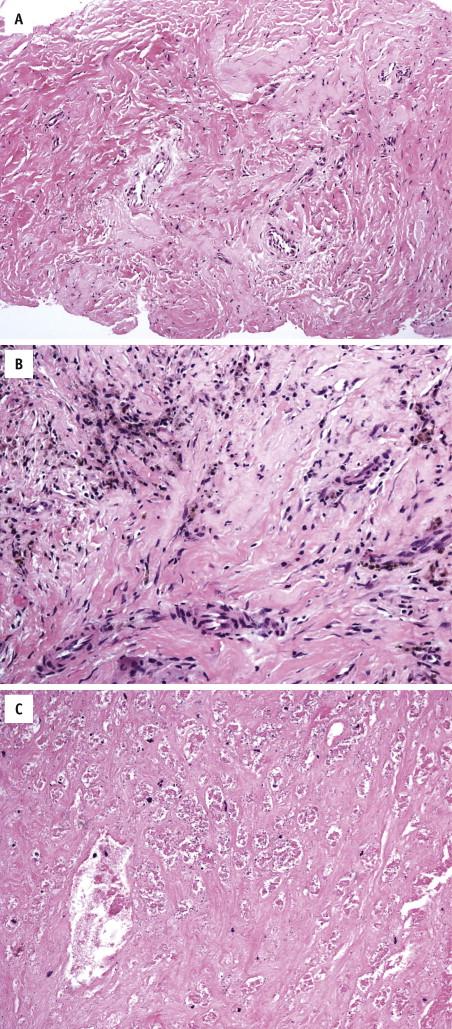
High-intensity focused ultrasound scan (HIFU) destroys target tissue through the thermal and mechanical effects of nonionizing, acoustic radiation (i.e., sound waves). As the tissue compresses and expands with exposure to the acoustic waves, gas is extracted, creating bubbles that oscillate violently, collapse, and disrupt cell membranes. The high temperature also induces the creation and release of chemically reactive free radicals, responsible for the induction of apoptosis and activity on nuclear DNA. HIFU-induced acute tissue damage manifests as areas of sharply delineated intraprostatic coagulative necrosis. Vacuolated cytoplasm, ruptured cell membranes, and destroyed cellular organelles as signs of definitive cell death have been routinely observed in treated tissue regardless of their structure. Careful histological examination within the area of coagulative necrosis has revealed no remnant viable cells that could potentially escape the therapeutic effect of HIFU. Recent studies have reported acute and chronic inflammation, glandular atrophy, hemosiderin deposition, focal coagulative necrosis, dense stromal fibrosis, and edema in benign tissue after HIFU treatment. In areas of viable tumor, no post-HIFU histological changes appear to preclude the use of Gleason scoring.
Prostate-specific antigen is detected in secretory cells of benign prostate glands in all anatomic zones, but not in basal cells, seminal vesicle/ejaculatory duct epithelium, or prostatic urothelial cells. Most prostate carcinomas also express PSA ( Fig. 2-22 , A ), although considerable intratumoral and intertumoral heterogeneity is seen and the expression is decreased in a minority of high-grade prostate carcinoma. After androgen deprivation and radiotherapy, some cancers can lose PSA expression. PSA immunoreactivity can be detected to variable degrees in some nonprostatic tissues and tumors, including urethral and periurethral glands, cystitis cystica and glandularis, urachal remnants, bladder adenocarcinoma, and extramammary Paget disease of the penis.
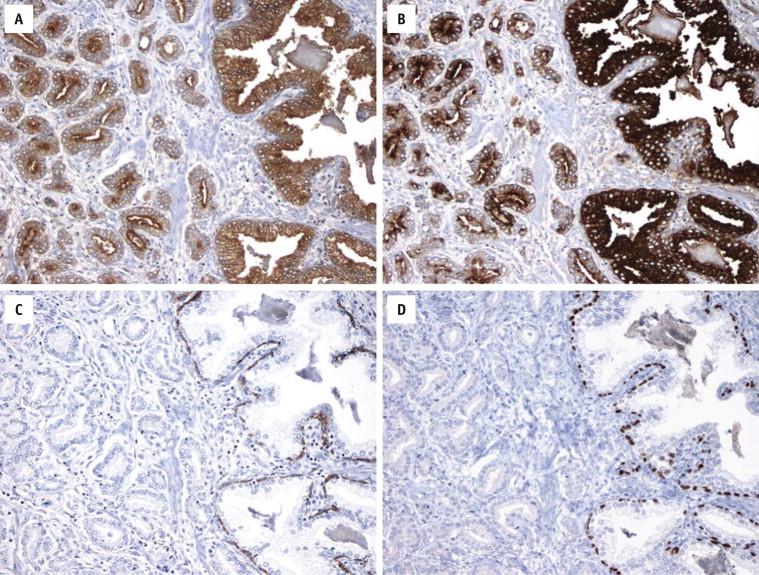
Prostate-specific acid phosphatase has diagnostic utility similar to that of PSA, although it is in general more sensitive and less specific than the latter ( Fig. 2-22 , B ). Nonprostatic tumors that are reported to be positive for PSAP include some neuroendocrine tumors (pancreatic islet cell tumors and gastrointestinal carcinoids), urothelial carcinoma, and anal cloacogenic carcinoma.
Benign prostatic secretory and basal cells are immunoreactive for antibodies to broad-spectrum and low–molecular-weight cytokeratins (CKs). Negative staining for both CK7 and CK20, which is typical of prostate cancer, is useful to differentiate prostate carcinoma from urothelial carcinoma, which is typically positive for both markers.
High–molecular-weight cytokeratin, detected with the antibody clone 34βE12 that recognizes CK1, CK5, CK10, and CK14 or with the antibody cocktail that recognizes CK5 and CK6, is expressed only by prostate basal cells, and not by secretory cells ( Fig. 2-22 , C ). Prostate carcinoma uniformly lacks a basal cell layer and consequently is negative for HMWCK ( Fig. 2-22 , C ). Absence of a basal lining is demonstrated by lack of immunostain for HMWCK and supports a diagnosis of prostate carcinoma in the context of suspicious architectural or cytologic features on routine H&E stains. However, prostate carcinoma can occasionally contain sparse tumor cells that are positive for HMWCK yet not in a basal cell distribution, especially after radiation or hormonal therapy. Intraductal spread of prostate carcinoma or entrapped benign glands may also be mistaken as residual basal cells in prostate carcinoma. Conversely, some benign conditions, including adenosis (atypical adenomatous hyperplasia) and partial atrophy, can sometimes have a discontinuous or even absent basal cell lining. p63 is a nuclear protein expressed in basal cells of pseudostratified epithelia, including prostate. It has similar diagnostic utility ( Fig. 2-22 , D ) and pitfalls to HMWCK but with the following advantages: (1) it stains a subset of basal cells that are negative for HMWCK; (2) it is less susceptible to staining variability, particularly in TURP specimens with cautery artifact; and (3) it is easier to interpret because of its strong and sharp nuclear staining. An additional pitfall is that in a rare variant of prostate cancer (adenocarcinoma with diffuse aberrant p63 expression), tumor cells expression p63 in a nonbasal distribution (see Fig. 2-17 , C ).
The AMACR (or P504S) enzyme is involved in the metabolism of branched-chain fatty acids and bile acid intermediates. It is overexpressed in most prostate carcinomas ( Fig. 2-23 , A and B ). Because of its intratumoral heterogenous expression patterns, AMACR is positive in only 80% of prostate carcinomas in prostate needle biopsies. Several histologic variants of prostate carcinoma, such as foamy gland, atrophic, and pseudohyperplastic prostate carcinoma, show lower AMACR expression. AMACR is not entirely specific for prostate carcinoma because it is present in HGPIN (>90%), adenosis (∼20%), partially atrophic glands, and occasionally morphologically benign glands. AMACR can be used as a confirmatory staining for prostate carcinoma, in conjunction with H&E histology and basal cell markers.
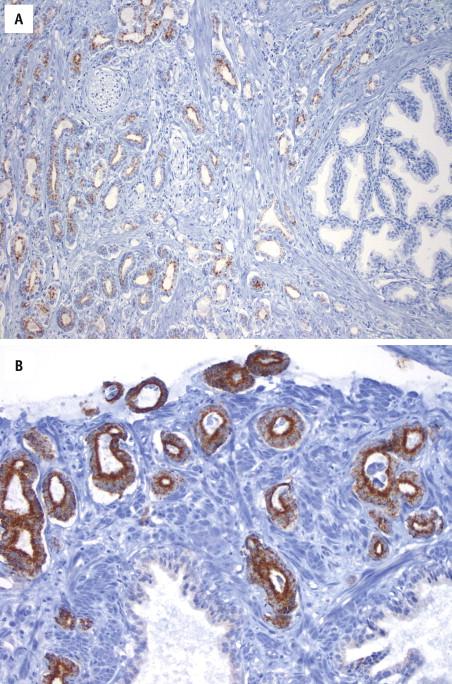
Basal cell markers and AMACR can be combined in a single immunostaining reaction ( Fig. 2-24 ). Such “cocktail” staining may be advantageous for working up a small focus of cancer that might be present only in one tissue section.
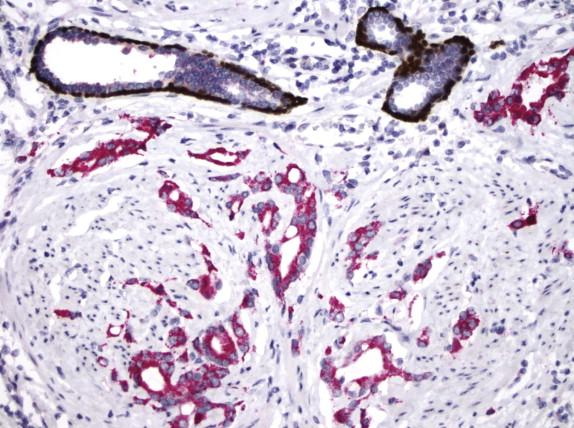
NKX3.1 is an androgen-regulated tumor suppressor gene located on chromosome 8p largely specific to prostate. Staining for NKX3.1 protein is positive in most primary prostatic adenocarcinomas, but it may be downregulated in high-grade prostate cancers and lost in some metastatic prostate cancers. The use of NKX3.1 is recommended in the differential diagnosis of poorly differentiated prostate cancer versus urothelial carcinoma ( Fig. 2-25 , A and B ), particularly when tumor exhibits equivocal/weak or negative staining for PSA and negative/focal staining for p63 and HMWCK, and also in the evaluation of metastases from unknown primary site.
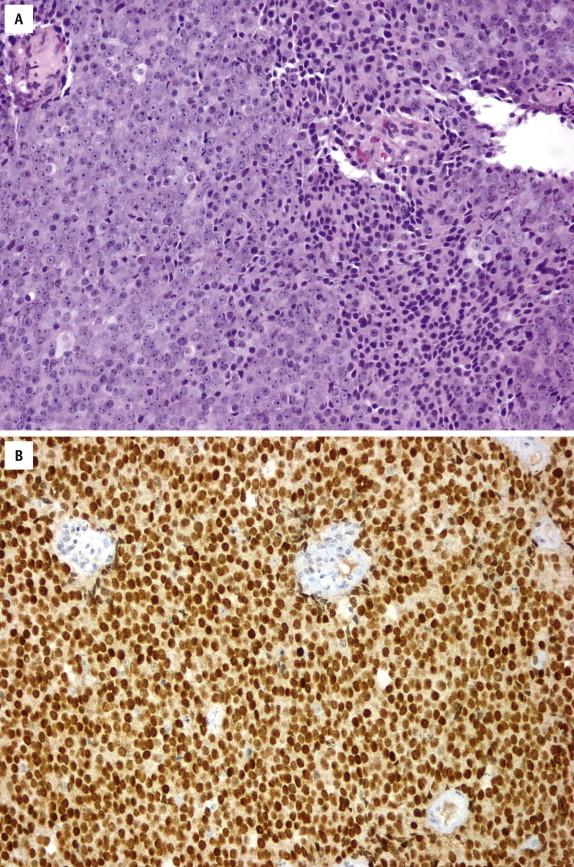
Prostein (or P501S) is a prostate-specific protein localized to the Golgi complex, uniquely expressed in prostatic tissue (normal and cancerous) and unrelated to Gleason grade. Similar to NKX3.1, the use of prostein is recommended in the differential diagnosis of poorly differentiated prostate cancer versus urothelial carcinoma ( Fig. 2-26 , A and B ) and in the metastatic setting from unknown primary site.
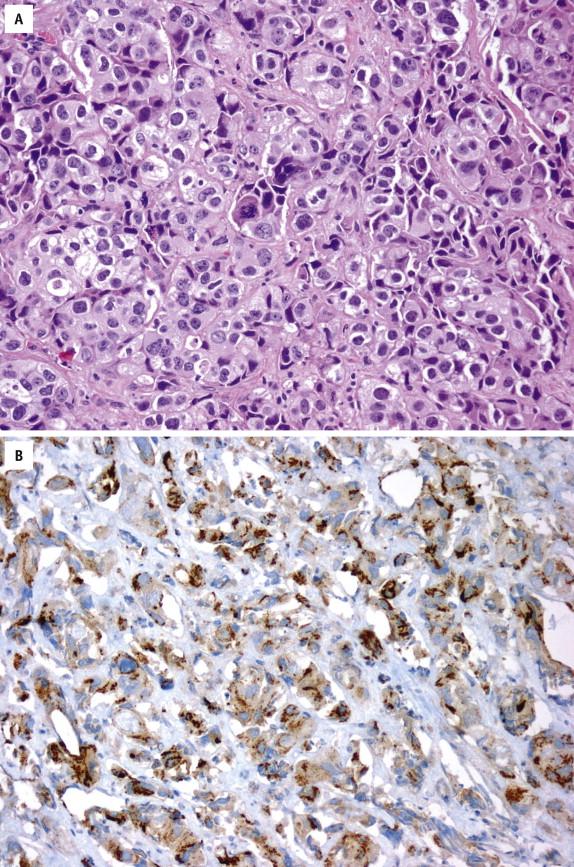
Immunostaining for ERG is used as a surrogate for TMPRSS2-ERG gene fusion, a specific molecular event seen in 40% to 50% of prostate cancers and in approximately 20% of HGPIN that intermingles with cancer. Given the high specificity of ERG protein expression for prostate cancer, positive ERG staining may help establish a diagnosis of limited prostate cancer on challenging needle biopsies ( Fig. 2-27 , A and B ) in conjunction with other markers (basal cell markers, AMACR) and may be useful in the diagnosis of metastasis from unknown primary site.
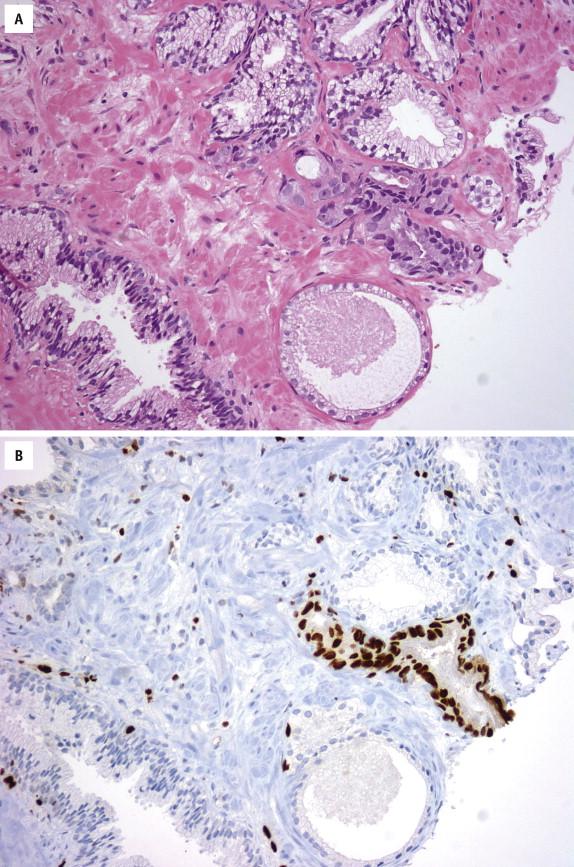
The differential diagnosis of prostate carcinoma is complex ( Table 2-1 ). In many instances, the differential is with normal prostatic and nonprostatic structures, including seminal vesicles/ejaculatory duct epithelium, Cowper gland, paraganglia, and mesonephric duct remnants. A wide variety of benign pathologic processes, such as inflammation, atrophy (simple atrophy, partial atrophy, and postatrophic hyperplasia), metaplasia (urothelial, squamous, and mucinous), basal cell hyperplasia, BPH, and radiation and hormonal treatment effects, can simulate prostate carcinoma to varying degrees. The prostate gland can rarely be involved by primary urothelial, small cell, mucinous, and signet-ring–cell carcinoma. However, such a diagnosis should be made only after a metastasis from other sites is diligently excluded. On the other hand, prostate carcinoma can also mimic benign conditions. For example, a well-differentiated Gleason score 2 to 4 prostate carcinoma should always be differentiated from adenosis. Cribriform prostate carcinoma should be distinguished from benign cribriform clear cell hyperplasia, cribriform HGPIN, and intraductal carcinoma. Atrophic and foamy gland prostate carcinoma may be confused with benign atrophy and xanthoma, respectively. Pseudohyperplastic prostate carcinoma shares some architectural features with BPH, although the former invariably has significant nuclear atypia. Careful evaluation of the architectural and cytologic features and prudent use of basal cell markers and AMACR lead to a correct diagnosis.
| Differential Diagnosis | Architectural Features | Cytologic Features | Immunohistochemistry |
|---|---|---|---|
| Normal prostatic/nonprostatic structures | |||
| Seminal vesicle or ejaculatory duct | Central irregular lumen with surrounding clusters of smaller glands | 1. Scattered cells that show nuclear atypia | 1. Basal cell + by p63 and prominent degenerative HMWCK |
| 2. Golden-brown lipofuscin pigment | 2. Secretory cells – PSA (monoclonal antibody) | ||
| 3. + MUC6 | |||
| Verumontanum mucosal | 1. Closely packed small acini beneath urethral mucosa | 1. Prominent nucleoli – | Basal cells + by HMWCK gland hyperplasia |
| 2. Orange-brown dense luminal secretion | 2. Lipofuscin pigment + | ||
| 3. Basal cells + | |||
| Cowper glands | 1. Noninfiltrative, lobular pattern | Acini with voluminous, pale cytoplasm | PSAP – |
| 2. Dimorphic population of ducts and mucinous acini | PSA – or + in heterogeneous, clumpy fashion in minority of cases | ||
| 3. Intermixed with skeletal muscle fibers | |||
| Paraganglia | 1. Small cluster or nest of clear cells with prominent vascular pattern | 1. Clear or amphophilic, granular cytoplasm | Neuroendocrine markers + PSA, PSAP – |
| 2. Intimately associated with nerve | |||
| 3. Most common in periprostatic soft tissue | 2. Inconspicuous nucleoli | ||
| Benign prostatic lesions | |||
| Mesonephric remnants | 1. Lobular arrangement of small regular tubules | Tubules lined with single layer of epithelium | PSA, PSAP – HMWCK + |
| 2. Dense, eosinophilic intraluminal secretion | |||
| Partial atrophy | 1. Lobular configuration often maintained | May have mild nuclear atypia | Basal cell markers + but often patchy, or even absent in some glands |
| 2. Pale glands with irregular or angulated contour | |||
| 3. Atrophy involving some glands, or part of a gland | |||
| Postatrophic hyperplasia | Central dilated atrophic duct surrounded by fibrosis and clustered smaller atrophic glands | 1. Atrophic cytoplasm | Basal cell markers + |
| 2. Inconspicuous nucleoli | |||
| Urothelial metaplasia | 1. Stratification of elongated cells underneath the secretory cells | 1. Elongated nuclei with nuclear grooves | Basal cell markers + |
| 2. Cells perpendicular to basement membrane | 2. Perinuclear clearing | ||
| Squamous metaplasia | 1. Associated with inflammation, infarction, or hormonal ablation therapy | 1. Intercellular bridges | Basal cell markers + |
| 2. Squamoid cytoplasm | |||
| 2. Small, solid nests with admixed inflammation | 3. Keratin pearls may be present | ||
| 4. Immature form may have prominent nucleoli | |||
| Basal cell hyperplasia | 1. More common in transition zone | 1. Bland oval or elongated cells | PSA, PSAP – HMWCK, p63 + |
| 2. Acinar, cribriform and solid growth patterns | |||
| 3. Lined with ≥ 2 layers of basal cells | |||
| 4. May have squamous metaplasia or intraluminal calcifications, or intracytoplasmic eosinophilic globules | 2. Occasionally have prominent nucleoli | ||
| Adenosis | Small crowded glands admixed with larger glands in lobular configuration | 1. Small glands share cytologic features with larger benign glands | Basal cells, detected with p63 or HMWCK antibody, may be patchy or absent in some small glands |
| 2. Prominent nucleoli absent | |||
| Sclerosing adenosis | Mixture of well-formed glands, cords, or single cells and cellular spindle cells | Hyaline collarette around glands | Basal cells + for p63, HMWCK, S100 and actin |
| Nonspecific granulomatous prostatitis | 1. Acini or duct-centered process | 1. Epithelial cells may have conspicuous nucleoli | PSA, PSAP, and pancytokeratin – CD68 + |
| 2. Mixed inflammatory infiltrates containing lymphocytes, histiocytes, neutrophils, eosinophils, plasma cells, and giant cells | 2. Histiocytes may have nucleoli | ||
| BPH | Nodular arrangement of small and large acini with papillary infoldings and projections | 1. No cytologic atypia | Basal cell markers + |
| 2. Basal cells + | |||
| Radiation effect in benign glands | 1. Lobular configuration | Scattered cells with marked degenerative nuclear atypia | Basal cell markers + |
| 2. Individual glands with irregular, angulated contour | |||
| 3. Multilayered cells | |||
| HGPIN | 1. Glands similar to adjacent benign glands in size and shape and separated by stroma | 1. Nuclear enlargement | Basal cell markers highlight basal cell layers |
| 2. Smooth glandular contour | 2. Coarse chromatin | ||
| 3. Basal cell layer may be intact or discontinuous | 3. Nucleolar prominence | ||
Sometimes a gland or a focus of glands is suspicious for prostate carcinoma, yet a definitive cancer diagnosis cannot be established because of the lack of sufficient architectural and cytologic atypia. The terms atypical small acinar proliferation (ASAP) and focal atypical glands have been used. Unlike HGPIN or prostate carcinoma, ASAP is a diagnostic term rather than a defined disease entity. It encompasses such lesions as HGPIN, benign mimickers of prostate cancer, reactive atypia, and many cases that in retrospect demonstrate focal cancer. ASAP found in needle biopsy denotes a high risk (∼50%) of detection of prostate carcinoma in subsequent biopsies.
The choice of therapeutic modalities for prostate carcinoma depends on the stage of the cancer, the patient’s life expectancy, and comorbidity. Expectant management, or active surveillance, in which patients are under close follow-up and definitive therapies are deferred until there is sign of disease progression, is most often selected for older men with a life expectancy of less than 10 years who have low-grade or intermediate-grade (Gleason score, <7), low-volume prostate carcinoma that is confirmed after extensive prostate sampling. For a man with clinically localized prostate carcinoma in whom curative intent is desired, radical prostatectomy and radiation therapy are options. Radical prostatectomy is the mainstay of primary treatment for clinically localized prostate carcinoma. It can also be performed after hormonal therapy and as salvage treatment after failure of radiation therapy. Radiation therapy, with external beam radiotherapy or implantation of radioactive seeds (brachytherapy), can also be used as a primary modality to treat prostate carcinoma. Hormonal ablation therapy, either with surgical castration (orchiectomy) or with pharmacologic blockade of androgen effect with luteinizing hormone–releasing hormone (LHRH) analogs or antiandrogen compounds, is the main treatment modality for men with disseminated prostate carcinoma. However, such treatment is only palliative and not curative. Locally advanced prostate carcinoma is frequently managed with a combination of irradiation and hormonal ablation.
The prognosis for patients with prostate carcinoma is highly variable and depends on a variety of host, tumor, and treatment parameters. The College of American Pathologists classifies the prognostic factors into three categories. Category I includes factors that are proven to be of prognostic importance and useful in clinical management, including preoperative serum PSA, Gleason grade, tumor node metastasis (TNM) stage, and surgical margin status. Category II includes factors that have been extensively studied biologically and clinically but whose importance remains to be validated in large multicenter trials. Factors included in this category are DNA ploidy, tumor volume, and histologic subtypes. Category III factors have not been sufficiently studied to demonstrate their prognostic value; they include perineural invasion, neuroendocrine differentiation, microvessel density, nuclear features other than ploidy, proliferation markers, and a variety of molecular markers.
The Gleason grading system, designed by Dr Donald Gleason, is the predominant grading system for prostate carcinoma. Grading should be undertaken at low magnification (×40 to ×100). It is based on the glandular architecture; nuclear atypia is not evaluated. The grading system defines five histologic patterns with decreasing glandular differentiation ( Fig. 2-28 , A ). The primary and secondary patterns (in order of prevalence) are added to obtain a Gleason score or sum. If a prostate carcinoma only has one pattern, then that pattern is doubled to obtain the Gleason score. The primary and secondary patterns should be reported along with the Gleason score. In 2005, several modifications were made to the original Gleason grading scheme in an effort to adapt this grading system to present-day practice in a uniform manner. The modified Gleason grading system is shown in Figure 2-28 , B. The significant changes include a stricter definition of Gleason pattern 3 cribriform glands and grading of ill-defined glands with poorly formed glandular lumina as pattern 4 (see subsequently). Recently, the validity of including cribriform glands as a component of Gleason pattern 3 has been questioned, and all tumors that show cribriform architecture are recommended to be classified as Gleason pattern 4 ( Fig. 2-28 , C ).
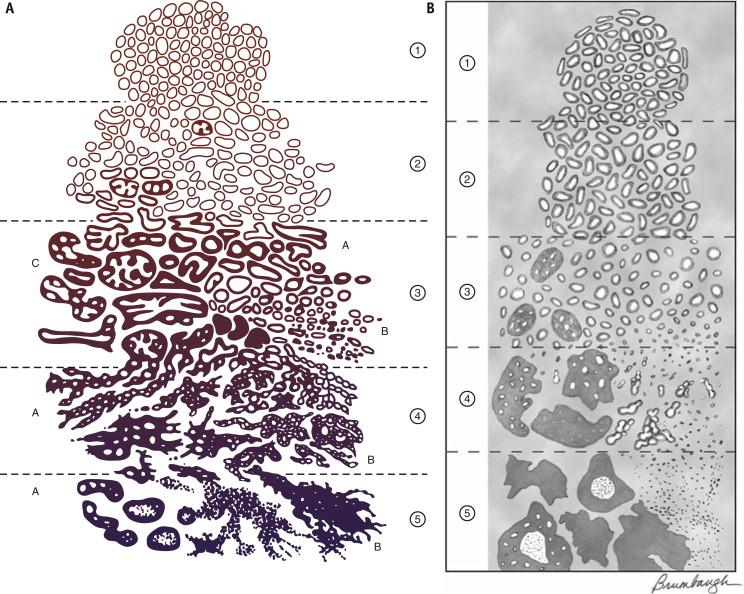
This pattern is composed of a very well-circumscribed nodule of closely packed but separate glands that do not infiltrate into adjacent tissue ( Fig. 2-29 ). The glands are of intermediate size and are similar in size and shape. This pattern is exceedingly rare and is usually only a minor component of prostate carcinoma when present. It may be seen in transition zone prostate carcinoma.
This pattern has a less well-circumscribed nodule of medium-sized glands, with some degree of variation in size and shape and looser arrangement. Minimal invasion of cancer glands into surrounding tissue may be found ( Fig. 2-30 ). Although the cytoplasm is not evaluated in the Gleason grading system, in Gleason patterns 1 and 2 cancer glands, it tends to be abundant and pale-clear. Gleason pattern 2 cancer may be seen in the transition zone. The new modified Gleason grading system states that for needle biopsies, patterns 1 and 2 should be diagnosed rarely, if ever. Gleason score 1 + 1 = 2 tumors also were acknowledged to almost always represent adenosis, with basal cell demonstrable after immunohistochemical staining.
This pattern is the most common. The cancer glands often infiltrate between the adjacent benign glands. They vary in size and shape and are often angular. Small glands are typical ( Fig. 2-31 , A ), but they may be large with angulations and branching ( Fig. 2-31 , B ). All cribriform glands should be graded as Gleason pattern 4. Poorly formed glands, originally considered Gleason pattern 3, are now considered Gleason pattern 4.
Glands appear fused, cribriform, or poorly formed. Fused glands are composed of a group of glands that are no longer separated by stroma. Cribriform glands may be small and have smooth, round, well-circumscribed contours ( Fig. 2-32 , A ) or may be large with irregular contours and jagged edges ( Fig. 2-32 , B ). The intraluminal cellular proliferation spans the entire diameter of the lumen. Poorly formed glands still have glandular configuration, but they have ill-formed glandular lumina ( Fig. 2-32 , C ). The hypernephromatoid pattern is a rare variant composed of fused glands with clear or very pale cytoplasm ( Fig. 2-32 , D ).
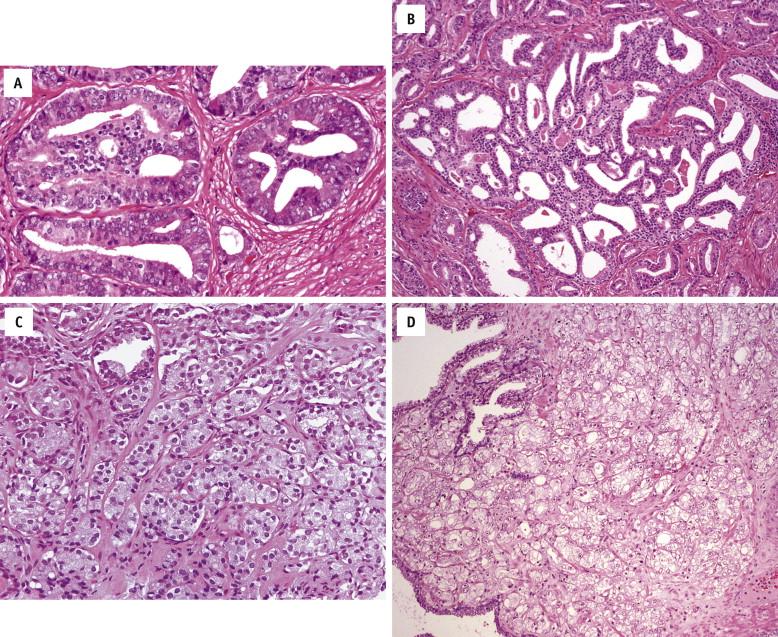
Cancer cells form solid sheets ( Fig. 2-33 , A ), strands, or single cells ( Fig. 2-33 , B ) that invade the stroma. Comedonecrosis may be present ( Fig. 2-33 , C ).
Tertiary Gleason pattern refers to a minor pattern that occupies less than 5% of the tumor volume. In radical prostatectomy, the presence of a high-grade tertiary pattern adversely affects the prognosis. For example, the presence of a tertiary pattern 5 in a Gleason score 4 + 3 = 7 prostate carcinoma worsens the prognosis, compared with the same tumor without a tertiary high-grade component. However, the prognosis is not as poor as that of a 4 + 5 = 9 cancer. In prostate needle biopsies that harbor three patterns when the worst pattern is the least common, the highest pattern should be incorporated as the secondary pattern.
Morphologic variants of prostate carcinoma are uncommon and often are mixed with ordinary prostate carcinoma. Grading such variants should be based on the underlying cancer glandular architecture. Cancers with pseudohyperplastic, atrophic, and microcystic features are graded as Gleason pattern 3. Ductal adenocarcinoma is graded as Gleason pattern 4 or 5 if comedonecrosis is present. Mucinous (colloid) adenocarcinoma can be graded as Gleason pattern 3, if composed of round discrete glands within mucin pools, or Gleason pattern 4, if composed of irregular, cribriform, or fused glands. Signet-ring–cell and sarcomatoid variants are aggressive tumors, comparable with Gleason score 9 or 10. On the other hand, squamous cell carcinoma, urothelial carcinoma, small cell carcinoma, and basaloid/adenoid cystic carcinoma are not assigned a Gleason grade. Prostate carcinoma treated with hormonal ablation or radiation can appear artifactually to be of higher Gleason grade; therefore Gleason grade should not be assigned to such cases. If no effect of the therapy is evident, a Gleason grade can be assigned. Prostate carcinoma displays remarkable intratumoral grade heterogeneity; therefore the biopsy Gleason grade may in some cases represent undergrading or overgrading compared with the radical prostatectomy. Nevertheless, the concordance between biopsy and prostatectomy Gleason scores is within one Gleason score in most cases. Multiple studies have shown that the Gleason grade is currently the most powerful prognostic indicator for prostate carcinoma. It correlates with all the important pathologic parameters in radical prostatectomies and with prognosis after radical prostatectomy and radiation therapy. The distinction between Gleason scores 6 and 7 is critical. Gleason 7 prostate carcinoma behaves significantly worse than Gleason 6 cancer but better than Gleason score 8 to 10 cancer. The importance of Gleason grade is evidenced by the use of various nomograms to predict pathologic stage and disease progression after surgery and radiotherapy. These nomograms, including Partin tables and Kattan nomograms, use preoperative biopsy Gleason score, tumor extent, clinical stage, and serum PSA to predict the risk of extraprostatic extension, seminal vesicle invasion, and lymph node metastasis and the probability of disease recurrence after treatment.
In addition to preoperative serum PSA, pathologic staging parameters in radical prostatectomy specimens, such as Gleason score, extraprostatic extension (EPE), seminal vesicle invasion, positive surgical margin, lymphovascular invasion, and lymph node status, are significant predictors of clinical and biochemical recurrence. Pathologic staging ( Table 2-2 ) is important in the determination of the most appropriate treatment options for each individual patient with prostate cancer and also in prediction of the likelihood of disease recurrence. There is no pathologic T1 classification. Organ-confined disease is classified as pathologic stage T2 (pT2). In the past, pT2 prostate cancers were subclassified in three categories to parallel the clinical staging system: (1) pT2a for tumors that occupy less than one half of one lobe; (2) pT2b for tumors that occupy greater than one half of one lobe; and (3) pT2c for tumors that involve both lobes. Although some pathologists still report pT2 substages, a consensus at the recent International Society of Urological Pathology (ISUP) conference was that this practice should be optional because subdivision of pT2 disease is problematic and has not proven to be of prognostic significance. Pathologic stage T3 disease (non–organ-confined disease) is subdivided into two categories: pT3a is determined by the presence of EPE in any location or microscopic invasion of bladder neck and pT3b by the presence of seminal vesicle invasion with or without EPE. Microscopic bladder neck invasion, defined as malignant cells or glands that invade thick smooth-muscle bundles in the bladder neck section ( Fig. 2-34 ), is currently classified as pT3a, rather than together with gross (macroscopic) bladder invasion (pT4). Extraprostatic extension is defined as the presence of prostate cancer glands beyond the confines of the gland. Cancer glands admixed with periprostatic adipose tissue is the most easily recognized manifestation of EPE ( Fig. 2-35 , A ). Posterolaterally, the finding of cancer glands within loose connective tissue or perineural spaces of the neurovascular bundle, even in absence of direct contact between tumor cells and adipocytes, represents EPE ( Fig. 2-35 , B ). A distinct prostate cancer nodule within desmoplastic stroma that bulges beyond the normal gland contour represents EPE ( Fig. 2-35 , C ). At the apex, EPE can be diagnosed when cancer glands touch an inked surgical margin where benign glands have not been similarly cut across or when malignant glands extend beyond the contour of the normal prostate ( Fig. 2-35 , D ). Prostate cancer within skeletal muscle does not constitute EPE. When EPE is identified, location and extent (focal versus established) should be documented. Focal EPE ( Fig. 2-35 , E ) refers to the finding of a few neoplastic glands outside the prostate and established to anything more than a few glands. Seminal vesicle invasion is defined as tumor that infiltrates the muscular wall of the seminal vesicle ( Fig. 2-36 ). Pathologic stage T4 prostate cancer is defined by direct rectal involvement, gross invasion of urinary bladder, external sphincter, levator muscles, or pelvic wall, with or without fixation. A positive surgical margin is defined as tumor (extraprostatic or intraprostatic) that extends to the inked surface (surgical tissue plane) of the prostate gland ( Fig. 2-37 ). Documentation of the location and the extent of positive margins is important. Although in practice many pathologists do report some measure of tumor volume and size, there is no one recommended method for doing so. Lymphovascular invasion requires the presence of tumor cells within endothelial-lined spaces conforming to the contour of the space and, when possible, with attachment to the endothelium ( Fig. 2-38 ). The number of regional lymph nodes identified in the specimen submitted for pathologic evaluation and the number involved by tumor should be routinely included in the pathology report. Reporting of the size/diameter of the largest metastatic focus is recommended because it appears to be one of most significant predictors of cancer-specific survival. Prostate cancer tends to spread to regional lymph nodes (N1; Fig. 2-39 , A ) and bone ( Fig. 2-39 , B ) and, to a lesser degree, to lung, liver, and brain. Metastases in other locations are exceptional. Involvement of lymph nodes lying outside the boundaries of the true pelvis is classified as M1a disease. Osteoblastic bone metastases (M1b) are the most common nonnodal site of PCA metastasis. Lung and liver metastases are usually identified late in the course of the disease and classified as M1c category.
Invasive carcinoma with prostatic secretory cell differentiation
Most common noncutaneous malignancy disease in United States
238,590 new cases in 2013
70% to 75% in peripheral zone, 15% to 20% in transition zone, 10% in central zone
Incidence rises dramatically with age; uncommon before 50 years of age
Highest incidence in United States, Canada, Australia, western Europe, and Scandinavian countries; relatively uncommon in south central and eastern Asia and northern Africa
Blacks have much higher incidence and mortality rates than whites in United States
Most asymptomatic
Some with abnormal DRE findings
Occasionally obstructive urinary symptoms
Rarely symptoms and signs related to metastasis to bone, brain, and other sites
70% to 75% has PSA >4 ng/mL
Expectant management (active surveillance)—localized prostate carcinoma with life expectancy <10 years
Radical prostatectomy/radiation therapy—localized prostate carcinoma
Hormonal ablation + radiation—locally advanced prostate carcinoma
Hormonal ablation—metastatic prostate carcinoma
Prognosis highly variable and depends on host, tumor, and treatment parameters
DRE, Digital rectal examination; PSA, prostate-specific antigen.
Firm, solid, white-gray to yellow-orange, in contrast to tan, spongy benign prostatic tissue
PSA-detected cancer often not grossly visible
Architectural features: haphazard glandular arrangement; infiltrative growth; less differentiated glands with cribriform, fused glands, cords, sheets, or single tumor cells
Typically small glands with straight luminal border
Cytologic features: pale to amphophilic cytoplasm; no lipofuscin pigment
Nuclear features: enlargement, hyperchromasia, variably prominent nucleoli; mitosis and apoptotic bodies can be present
Intraluminal features: crystalloids; blue mucin; pink amorphous secretion
Cancer-specific features: mucinous fibroplasia (collagenous micronodules); glomeruloid formation; perineural invasion
Atrophic carcinoma
Pseudohyperplastic carcinoma
Microcystic adenocarcinoma
Foamy gland carcinoma
Mucinous (colloid) carcinoma
PIN-like adenocarcinoma
Signet-ring carcinoma
Sarcomatoid carcinoma (carcinosarcoma)
Large-cell neuroendocrine carcinoma
Pleomorphic giant cell carcinoma
Lymphoepithelioma-like carcinoma
Adenocarcinoma with diffuse aberrant p63 expression
Ductal adenocarcinoma
Intraductal carcinoma
Urothelial carcinoma
Squamous cell carcinoma
Basal carcinoma
Small cell carcinoma
Normal prostatic/nonprostatic tissue (seminal vesicle/ejaculatory duct, verumontanum glands, Cowper glands, paraganglia, mesonephric remnants)
Benign conditions (atrophy, partial atrophy, postatrophic hyperplasia, urothelial/squamous metaplasia, basal cell hyperplasia, adenosis, sclerosing adenosis, inflammation, nonspecific granulomatous prostatitis, BPH, cribriform clear cell hyperplasia)
Cribriform HGPIN
Intraductal carcinoma
Treatment effect (radiation atypia)
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here