Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Neisseria gonorrhoeae , which is predominantly a sexually transmitted infection, is a common cause of urethral discharge in men and cervical discharge in women. Because N. gonorrhoeae may infect mucosal epithelial cells, it also may cause pharyngeal and rectal infection in adults and conjunctival infection in the newborn. Complications of N. gonorrhoeae infections include epididymitis in males and pelvic inflammatory disease in females. Chronic or recurrent pelvic inflammatory disease may result in uterine tubal scarring and subsequent infertility. Additional complications may include disseminated infection with resultant skin lesions, arthritis, tenosynovitis, and rarely, endocarditis or meningitis. Gonococcal infections also may increase the risk of acquiring and transmitting the human immunodeficiency virus (HIV) infection.
N. gonorrhoeae are gram-negative bacteria typically observed microscopically as diplococci. The organism is composed of an inner cytoplasmic membrane, a middle cell wall composed of peptidoglycan, and an outer membrane. The outer membrane contains various components that interact with the host, thereby enabling invasion and infection, as well as mutant surface proteins and lipo-oligosaccharides that facilitate the evasion of host immunity. N. gonorrho eae have a genome of circular DNA of 2.1 million base pairs. The genome contains 2069 genes, many of which are involved in DNA repair pathways.
N. gonorrhoeae is a sexually transmissible organism. Very rarely it can be acquired through the inadvertent inoculation of infected secretions onto a susceptible anatomic site (e.g., causing gonococcal orbital cellulitis or be associated with household transmission). Mother-to-child transmission from passage of the fetus through an infected birth canal can result in neonatal gonococcal conjunctivitis, which can lead to blindness if untreated.
The infectivity of gonorrhea is about 50%, which on its own would not allow the maintenance of endemic infection without core groups of individuals who have a higher than average number of sexual contacts. The likelihood that an infected man will transmit infection to a woman during a single episode of unprotected vaginal intercourse is about 50 to 70%, and the risk that a woman will infect a man is about 20%. The risks of transmission associated with fellatio and cunnilingus are not well defined. Anal intercourse is also an efficient mode of transmission, but the absolute risk of transmission per contact is uncertain. Evidence also suggests gonococcal transmission from the use of infected saliva as a sexual lubricant.
After changes in sexual behavior due to the wide availability of oral contraceptive agents and the rise of the gay liberation movement in the 1970s, cases of gonorrhea soared in the United States. The discovery of HIV and the resultant fear of AIDS in the 1980s led to changes in sexual behaviors that reduced the incidence of gonorrhea to an all-time low by the mid-1990s. Over the past two decades, cases of gonorrhea have increased again, primarily in men who have sex with men, but also in younger minority populations and in groups with poor access to health care. Infections now are most common among females ages 15 to 24 years and males ages 20 to 29 years. Currently, gonorrhea is the second most frequently reported disease in the United States, with an estimated 1.6 million new infections per year.
Globally, N. gonorrhoeae is the most commonly notifiable infection after Chlamydia trachomatis , with an estimated annual incidence of 87 million new cases and a population-level prevalence of about 0.7 to 0.9%. In pregnant women in sub-Saharan Africa, N. gonorrhoeae has a prevalence of 2.3% overall, with higher frequencies in Southern versus Central Africa. Antimicrobial-resistant N. gonorrhoeae infections are widespread and increasing.
N. gonorrhoeae contain a variety of virulence and growth factors. Their characteristic filamentous structures, called pili, serve to attach to membranes of human mucosal and uroepithelial cells. Variations in pili contribute to immune evasion and tissue tropism. N. gonorrhoeae also have porins, which are proteins on the outer membrane, that enable the transport of ions and nutrients essential for bacterial survival. Certain porins, such as porin B isoform 1A, have been associated with increased resistance to host immunity and an increased capacity for dissemination. Opacity-associated proteins are integral outer membrane proteins that determine the invasive capacity of N. gonorrhoeae . Gonococci may also evade host immunity by expressing enzymes such as IgA proteases, which cleave mucosal antibody and use several molecular mechanisms to vary surface antigens such as lipo-oligosaccharides.
Select complement deficiencies ( Chapter 37 ) predispose a patient to dissemination of N. gonorrhoeae because of decreased complement-mediated killing of N. gonorrhoeae . Up to 50% of patients with C6, C7, or C8 complement deficiencies may develop bacteremia with pathogenic Neisseria species. Complement deficiencies also predispose patients to recurrent and perhaps subacute or even chronic infections. Eculizumab, a monoclonal antibody to C5 used in conditions such as complement-mediated hemolytic uremic syndrome ( Chapter 158 ), also appears to increase the risk of disseminated gonococcal infection.
The clinical manifestations of gonorrhea are highly dependent on the anatomic site of infection and host factors related to inflammation. N. gonorrhoeae may infect the mucosal tissue of the urethra, cervix, throat, or rectum. Infections at these sites may produce an inflammatory response with subsequent purulent discharge or pain (often burning in character) with urination or defecation, or, as is often the case in pharyngeal infection, either mild symptoms (e.g., sore throat) or no symptoms whatsoever. After a brief incubation period, typically just 1 or 2 days, acute genital infections are almost always (>90%) symptomatic in men but much less so (about 50%) in women, a difference that partly explains why asymptomatically infected women contribute to ongoing transmission.
Clinical manifestations in women may include pain with intercourse, vaginal bleeding, and lower abdominal pain and tenderness with or without fever, consistent with pelvic inflammatory disease ( Chapter 264 ). Repeated pelvic inflammatory disease increases the risk for tubal infertility. Infection in pregnancy can lead to adverse birth outcomes including prematurity, low birthweight, and neonatal mortality. In HIV-infected pregnant women, gonococcal coinfection can increase the mother-to-child transmission of HIV infection. In men, prolonged infection may lead to testicular infection and epididymitis.
Rarely, when infection does not cause acute symptoms that lead to diagnosis and effective therapy, N. gonorrhoeae may disseminate in the blood stream and seed normally sterile sites, including the joints, cerebrospinal fluid, and heart valves. Disseminated gonococcal infection may also be associated with a pustular skin rash and inflammation of the affected joints and tendons.
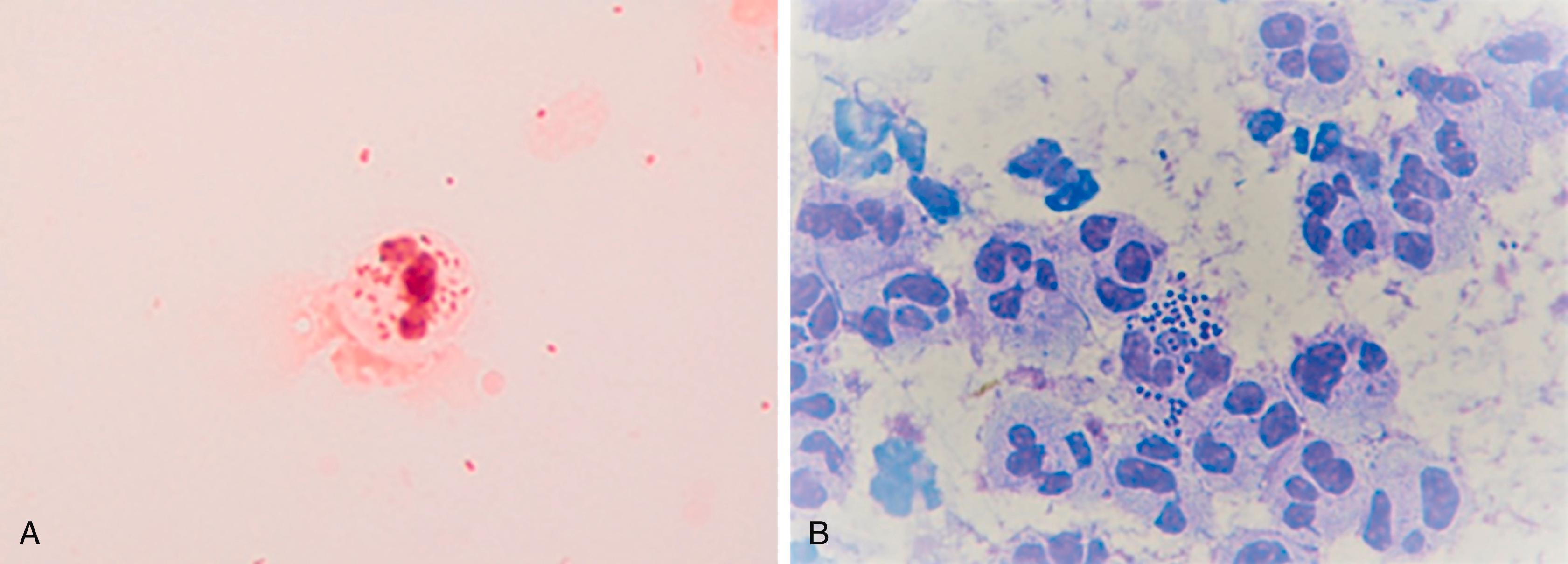
Diagnosis is based on recent sexual exposure, clinical signs, and symptoms. Laboratory confirmation of infection is made by the use of nucleic acid–based molecular testing (of a swab from the urethra, cervix, vagina, throat, or rectum; or of urine), antigen-based immunoassays, or the observation of characteristic organisms with the distinct morphology and coloration on point-of-care Gram staining. After heat-fixing the urethral smear slide, the multistep Gram stain procedure allows visualization of the nearly pathognomonic gram-negative intracellular diplococci upon oil-immersion microscopy ( Fig. 275-1A ). A methylene blue/gentian violet stain ( Fig. 275-1B ), which does not require heat fixation and takes only 1 minute, is an equally good alternative. Because N. gonorrhoeae are typically restricted to specific anatomic sites of infection, testing for infection must occur with specimens from each anatomic site of sexual exposure. Urine tests detect urethral infections but not infections at other anatomic sites. Culture is rarely required except in cases of possible treatment failure or to diagnose disseminated gonococcal infection, abscesses, endocarditis, or meningitis. Rarely, other Neisseria species (e.g., Neisseria meningitides ; Chapter 276 ) may also be present in urethral secretions and may be associated with a similar syndrome.
Urethritis is the most common clinical manifestation of gonococcal infection in men. After an incubation period of 2 to 6 days, men develop a profuse, purulent urethral discharge that is usually white, yellow, or green in color ( Fig. 275-2A ). In contrast, the discharge in non-gonococcal urethritis is usually thin and clear or gray in color ( Fig. 275-2B ). Dysuria is common; meatal irritation, meatal erythema, and penile lymphedema can also occur. Depending on the infecting strain, 10% of men have an asymptomatic infection that is only detected by screening. Because the diplococci of N. gonorrhoeae adhere to mucosal columnar and cuboidal epithelial cells of the urethra and cervix, women can also have symptomatic urethritis with dysuria.
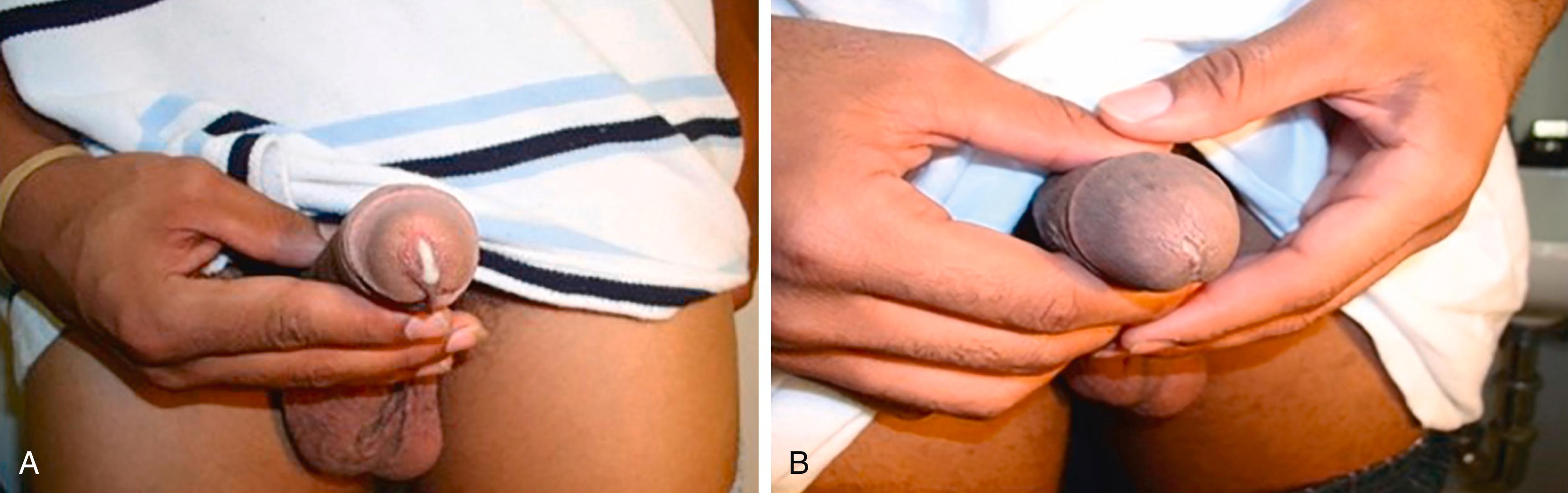
The diagnosis of gonococcal urethritis should be suspected based on dysuria with a urethral discharge (i.e., burning with urination). Gonococcal urethritis may be confirmed with point-of-care Gram stain, methylene blue stain, or nucleic acid detection testing of the urethral discharge by a penile meatal or urethral swab. In women, nucleic acid testing of urine or self-collected vaginal swab specimens has replaced the need to collect cervical swab specimens.
Cervicitis is the most common clinical manifestation of gonococcal infection in women, but up to 50% of infections are asymptomatic. When symptomatic, women report vaginal discharge, irritation, or itching. Postcoital bleeding, pain with vaginal sexual intercourse, and abnormal vaginal bleeding are also associated with cervicitis. The physical examination may show a purulent or mucopurulent cervical discharge ( Fig. 275-3 ), an erythematous or edematous cervix, a friable cervix with easily induced bleeding, or, occasionally, cervical motion tenderness.

Nucleic acid amplification testing is the mainstay for detecting urogenital N. gonorrhoeae from the cervix, vagina, and urine in women because other methods are not sufficiently sensitive for detecting the organism. Other organisms that may be detected by nucleic acid testing ( Chapter 264 ) include Chlamydia trachomatis ( Chapter 294 ), Mycoplasma genitalium ( Chapter 293 ), Trichomonas vaginalis ( Chapter 324 ), herpes simplex viruses ( Chapter 345 ), and Candida species ( Chapter 310 ), but no etiology is found in 50% of women with cervicitis.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here