Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Necrotizing soft-tissue infections (NSTIs) are highly fatal infections that share features, including the presence of necrotic tissue and the need for surgical debridement (among other therapies). NSTIs are under the category of complicated skin and soft-tissue infections (SSTIs). The nomenclature of these infections has typically been complicated and confusing because different terms are used to describe specific types of NSTI based on location, causative organisms, and other features, which may ultimately delay diagnosis and/or surgical intervention. The use of the term necrotizing soft-tissue infection is advocated because it groups all of the categories together and helps establish a common pathway to diagnosis and management. Advanced NSTIs are relatively easy to recognize based on characteristic local findings, severe systemic derangement, and are associated with a high mortality rate. Early diagnosis is key to improved clinical outcomes, but it is not always straightforward. A high index of suspicion coupled with biochemical and radiological studies can help confirm or rule out the diagnosis. Surgical exploration is the ultimate diagnostic (and therapeutic) strategy. Management of NSTIs should include early debridement, broad-spectrum antimicrobial therapy, and supportive care. Prognostic factors have been identified and prognostic scores developed; these tools may help in selecting patients who may benefit from a more aggressive surgical strategy and/or novel treatments.
A 40-year-old male with a history of type 2 diabetes mellitus presents with a 2-day history of severe left arm pain to the ED. He states the left upper extremity pain is associated with redness. The patient explained that the area has been getting progressively “more red” and painful throughout the day. Social history is significant for heavy alcohol use and IV drug use on the weekends.
Vitals BP 160/82, HR 112, O 2 stat 97% on room air, RR 18, Temperature 38°C.
Physical exam within normal limits, except for mild distress on initial examination. 5×3 cm area of erythema on left antecubital fossa—without sharp margins, tender to palpation, edema involving the whole extremity. Weakened strength in left upper extremity, but normal ROM.
Labs reveal an elevated WBC 16 k/uL, hemoglobin 12.1 g/dL, hematocrit 23%, sodium 134 mmol/L, potassium 4.0 mmol/L, glucose 305 mg/dL, creatinine 1.0 mg/dL, C reactive protein (CRP) 15 mg/dL, lactate 0.8 mmol/L.
Differential diagnosis: NSTI versus cellulitis. Other: abscess versus phlebitis versus hidradenitis suppurativa
The Laboratory Risk Indicator for Necrotizing Fasciitis (LRINEC) score = 7. The patient was started on meropenem, vancomycin, and clindamycin, and general surgery completed an emergent surgical debridement for complete diagnosis and treatment. Intraoperative findings revealed necrotic pale appearing tissues, thrombosed vessels, easy dissection, thin exudate without clear purulence, and dull gray appearance of fascia. Blood culture was positive for CA-MRSA. Procalcitonin (PCT) POD 1= 12.9 ng/mL, PCT POD 2= 11.5 ng/mL; PCT ratio= 1.12. Based on these results and persistent erythema and pain, the patient was taken back for serial debridements.
A 63-year-old female with a history of a strangulated ventral hernia is status post laparoscopic converted to open exploratory laparotomy, lysis of adhesions, and bowel resection. The patient started to develop worsening abdominal pain at the surgical site on POD 2. The patient was evaluated and found to have purulent drainage when staples were removed at the surgical site. The following day, the patient developed induration and worsening erythema at the surgical site associated with fever and tachycardia.
Vitals: BP 155/62, HR 104, O 2 stat 95% on room air, RR 19, Temperature 39°C
The physical exam was within normal limits, except for mild distress on initial examination. Erythema and edema along laparotomy and laparoscopic incisions on abdomen, mild purulent drainage, soft, tender to palpation, bowel sounds × 4, distension difficult to assess due to body habitus.
Labs revealed WBC 17.29 k/uL, hemoglobin 9.3 g/dL, hematocrit 35%, sodium 135 mmol/L, potassium 3.8 mmol/L, glucose 201 mg/dL, creatinine 1.3 mg/dL, CRP 19.6 mg/dL, lactate 0.7 mmol/L.
Differential diagnosis: postoperative wound infection versus NSTI
A computed tomography (CT) of the abdomen and pelvis was performed and revealed subfascial fluid collection and areas of inflammation and subcutaneous emphysema at the areas of port sites. The LRINEC score= 6. GI surgery removed the remaining staples and examined the wound. Residual purulent fluid from the surgical sites was evacuated and the fascia was found to be necrotic with dehiscence. The patient was started on meropenem and clindamycin, and general surgery completed an emergent surgical debridement for complete diagnosis and treatment. Intraoperative findings revealed residual purulent drainage associated with necrotic fascia and infection extending through the tissue planes. The tissues had foul-smelling drainage that appeared thin and serosanguinous, and there was woody induration of the tissue. PCT POD 1 = 10.8 ng/mL, PCT POD 2 = 9.7 ng/mL; PCT ratio: 1.11. The patient had to be taken for serial debridement × 3 and ultimately needed a negative pressure wound therapy dressing placed. Blood culture × 2 negative. Fluid culture revealed Clostridium perfringens .
NSTIs include a wide range of skin and soft-tissue infections ( Fig. 48.1 ) characterized by the presence of necrotic tissue and the need for debridement. Common terms include necrotizing cellulitis/fasciitis, “flesh-eating bacteria,” gas gangrene, and Fournier gangrene. There is no known geographic distribution for NSTIs, except those related to specific risk factors.
Given the rarity of NSTIs, it is hard to estimate accurate disease burden. Population-level studies using administrative and insurance-based databases have estimated an incidence of 4 per 100,000 person-years and an estimated 500 to 1500 new cases diagnosed yearly in the United States. As a consequence of the increasing number of Staphylococcus -related SSTIs, the incidence of NSTIs appears to have increased over time.
Despite a lack of evidence-derived data on the impact on health and cost of NSTIs, it is well known that NSTIs are infections with high risk for fatal outcome or limb loss, and when successfully treated, they are associated with significant effects on quality of life, including prolonged hospitalizations, disfiguring procedures, and the need for long-term rehabilitation. Furthermore, these effects carry substantial healthcare costs.
Few studies have compared patient and other baseline characteristics of NSTI with those of nonnecrotizing SSTI populations. Two such studies were able to identify intravenous drug use, as well as diabetes mellitus, as conditions more commonly associated with NSTI. Intravenous drug users with muscle or subcutaneous injections are particularly susceptible to developing NSTI. NSTI outbreaks have been reported in patients with drug use associated with contaminated heroin lots (e.g., “black tar heroin”). Other series evaluating NSTI populations, although not methodologically able to identify risk factors, have found associations with older age, chronic comorbidities, obesity, alcohol and drug use, malnutrition, immune suppression, and specific medications (steroids and nonsteroidal antiinflammatory drugs [NSAIDs], among others). These conditions do appear to be commonly associated with patients presenting with NSTI; however, to date, there is no evidence suggesting their predictive, risk-related, or causative association. Although adequate epidemiologic studies have identified an association between NSTIs and NSAID use, it is more likely that patients with NSTIs take NSAIDs during the initial phase of their infection than NSAID use predisposes individuals to developing NSTI. It is important to remember that NSTIs can occur in all populations.
Clinical manifestations of NSTI vary significantly based on the timing of presentation and the extent of the infectious process. The necrotizing component can involve any layer of the skin and soft tissues, including the skin, subcutaneous tissue, superficial/deep fascia, and/or muscle (see Fig. 48.1 ). Different anatomic areas can be involved, including, in order of frequency, the extremities (lower more than upper), perineum, trunk, and head and neck.
The vast majority of patients with early NSTI start with innocuous signs and symptoms. A preceding event localized to the involved area can be recalled, although in up to 20% of cases, no precipitating event is identified. Initial symptoms are difficult to differentiate from those of nonnecrotizing SSTIs (e.g., cellulitis, erysipelas) and include warmth, erythema, and pain with or without fever and tachycardia. Progression of the infection is variable and can be prolonged but usually occurs over 2 to 5 days. Once the necrotizing component starts to spread, the more ominous findings ensue and progress rapidly, leading to an overwhelming systemic infection. It is essential to identify these NSTIs early in their course, in order to prevent uncontrolled spread and systemic involvement. Progression from the time of the inciting event can be extremely rapid. Severe and advanced NSTIs are characterized by tense edema extending beyond erythema, ecchymosis, bullae or blisters, erythema without sharp margins, pain out of proportion, and crepitus. At these advanced stages, systemic manifestations, including fever, tachycardia, hypotension, multisystem organ dysfunction, and shock, are the sine qua non of NSTI ( Fig. 48.2 ).
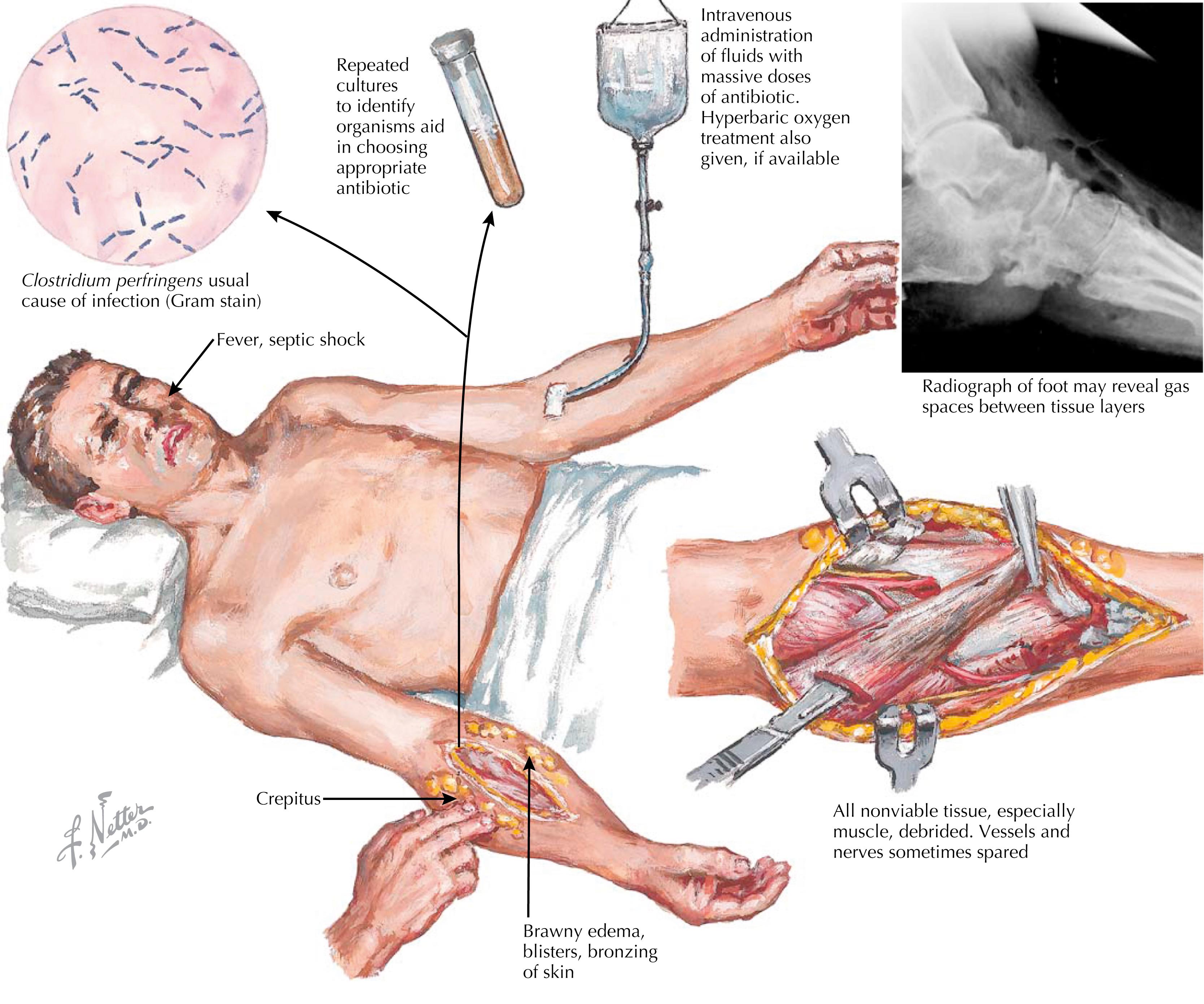
Clostridial and group A Streptococcal infections (GAS), as well as those caused by mucormycosis and Vibrio species, are characteristically aggressive, with rapid local progression and severe systemic derangement such as signs of systemic inflammation response or sepsis features, and are associated with the highest mortality rates. Clostridial infections are usually characterized by thin serosanguinous fluid and woody induration of the involved tissue. Upon exploration, there is typically thin, “dishwater” appearing fluid.
One of the most challenging aspects of managing patients with NSTI is the ability to diagnose it early. Early diagnosis with adequate surgical debridement is the most important therapy. The diagnosis of NSTI is purely clinical, and when in doubt, it should be confirmed or ruled out with surgical exploration. This is a crucial concept that allows early identification with timely debridement, maximizing the opportunity to control the infection and its systemic manifestations and leading to better overall outcomes.
Multiple studies have focused on different tools that may help identify patients with NSTIs early. The most important step in achieving this is to have a high index of suspicion based on risk factors and clinical presentation. Once NSTI is considered as a potential diagnosis, biochemical and radiological studies can be used. In a study by Wall et al. (2000), the authors found that a white blood cell count (WBC) greater than 15,400 and serum sodium level less than 135 mmol/L were associated with NSTI. Although the positive predictive value (PPV) was 26%, the negative predictive value (NPV) was 99%, making these values useful for ruling out NSTI when neither of these criteria is present. Wong and colleagues (2004) identified a series of factors associated with NSTI and designed a diagnostic score based on their presence: WBC, hemoglobin, serum sodium, serum glucose, serum creatinine, and C-reactive protein. The score is referred to as the LRINEC. The PPV and NPV of this score were 92% and 96%, respectively. Although useful for confirming the index of suspicion, the LRINEC score and other laboratory tests have limited sensitivity. One should be careful not to use them as standalone tests to rule out NSTIs. Lab results such as left shift, acidosis, and coagulopathy may be more useful for specific circumstances. It may be beneficial to obtain creatinine kinase and aspartate transaminase levels when there is concern for muscle or fascial involvement.
Radiological tests can also help establish the diagnosis of NSTI and are useful in patients with equivocal findings and no evidence of sepsis or shock. Radiographs, ultrasound, CT, and magnetic resonance imaging (MRI) have all been studied. In general, evidence of subcutaneous gas is pathognomonic of NSTI and should prompt emergency surgical debridement; however, this is a late finding and is less frequent than previously thought, present in only 24% of patients. Additional radiological findings include thickening of the underlying soft tissues and decreased enhancement of the deep fascial layers. These findings are not specific for NSTI but can increase the suspicion and direct further diagnostic efforts or surgical exploration for diagnosis.
Other studies have focused on evaluating the transcutaneous oxygen saturation with good PPV and NPV. However, this test is limited to patients with adequate underlying vasculature and hence is less useful for those with peripheral vascular disease. Finally, frozen section biopsy has been used to confirm the diagnosis of NSTI before formal surgical debridement. Typical microscopic findings include leukocyte infiltration, thrombosis of small arteries and veins, and necrosis. This test is limited by the subjective interpretation and availability of the pathologist, and when there is sufficient clinical suspicion to perform biopsy, the diagnosis is usually evident to the naked eye.
The most important strategy in diagnosing NSTI is a high index of suspicion followed by immediate surgical exploration. This cannot be emphasized enough and constitutes the difference between early infection control versus extensive delayed debridement in the setting of septic shock, multisystem organ failure, and a significantly higher risk of death. Intraoperative findings supporting the diagnosis of NSTI include the presence of necrotic tissue, easy finger dissection through normally fixed planes, foul-smelling “dishwater” purulence, thrombosed vessels, and lack of bleeding during transection of involved tissues.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here