Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
In the United States, nasopharyngeal carcinoma is uncommon, occurring at a rate of less than one case per 100,000 people. In contrast, in southern China and Hong Kong, the incidence is 25 to 50 per 100,000 people and among the Inuit in Alaska and Greenland, 15 to 20 per 100,000 people.
A potential link between Epstein-Barr virus (EBV) and nasopharyngeal cancer was first described more than 30 years ago; advances in molecular biology have furthered our understanding of this association. The rate of positive EBV serology is higher with nonkeratinizing or poorly differentiated versus well-differentiated lesions, and EBV DNA is detected less frequently in patients with keratinizing squamous cell carcinomas.
Evaluation for nasopharyngeal cancer includes detailed palpation of neck nodes, flexible nasopharyngoscopy with biopsy, computed tomography (CT), magnetic resonance imaging (MRI) of the head and neck, a complete blood count, a chemistry panel, and a chest radiograph. Positron emission tomography (PET) should be considered as well.
When treated with radiotherapy as a single modality, patients with nasopharyngeal carcinoma have a similar 10-year survival rate in the United States, Denmark, and Hong Kong (34%, 37%, and 43%, respectively). Advancing technology promises to allow better optimization of radiotherapy, improving target coverage and minimizing morbidity. The Intergroup study 0099 showed an advantage to the addition of concurrent plus adjuvant chemotherapy to radiotherapy for stages III and IV disease, with a survival advantage at 3 years (78% vs. 48%; p = 0.005). The advantage of concurrent chemotherapy for advanced disease is now well established, although the benefit of adjuvant chemotherapy remains unclear.
For persistent or recurrent nodal disease, surgical resection is the preferred treatment if technically possible, but recurrent disease at the site of the primary lesion is difficult to approach surgically. Radiation remains the mainstay of salvage therapy and in the era of modern techniques can achieve local control without excessive risk of toxicity. Reirradiation for recurrent disease appears to be most effective in patients with a prolonged disease-free interval and small tumor volume treated to a high reirradiation dose (≥ 60 Gy).
Advances in clinical and basic science research have led to a better understanding of the carcinogenesis of nasopharyngeal carcinoma (NPC) and to better treatment of this condition; however, such advances have also raised many new questions about this disease. This chapter provides an overview of the current understanding of etiological factors, such as Epstein-Barr virus (EBV), and the present diagnostic and therapeutic interventions for NPC. Therapeutic controversies are highlighted, and the data relevant to controversial issues are reviewed to guide treating physicians in applying clinical data to individual patients.
NPC more commonly occurs in men than women, with up to a two- to threefold difference in incidence. It is an uncommon tumor in the United States, where the incidence is less than 1 case per 100,000 people. In comparison, the incidence is considerably greater in southern China and Hong Kong (25-50 cases per 100,000 people) and among the Inuit residing in Alaska and Greenland (15-20 cases per 100,000 people). The incidence of nasopharyngeal cancer is also higher in other parts of Southeast Asia (Taiwan, Vietnam, and Thailand), the Philippines, and Malaysia and in some Mediterranean and North African populations (8-12 per 100,000 people). These areas of increased incidence of nasopharyngeal cancer are considered endemic areas; the incidence outside endemic areas is much lower and more often associated with tobacco use. Although incidence of nasopharyngeal cancer has declined in some endemic areas over the past 20 years (Hong Kong, Taiwan), it has remained stable in other endemic regions.
Several geographic-specific etiological factors have been implicated in the incidence variations throughout the world. The high consumption of salted fish in southern China and Hong Kong has been implicated as a possible etiologic factor for NPC in these areas. It has been suggested that various macromolecular lignins associated with these foods in southern China (and perhaps other areas, such as Alaska) may activate EBV, which has also been identified as a probable etiological factor in NPC. Studies by Bouvier et al. involved the fractionation of harissa, a homemade spice mixture used in Tunisia on various foods, including salted fish. Harissa was separated into various macromolecular fractions by column chromatography. The lignin-containing complexes extracted from harissa induced the EBV promoter in Raji cells. The possibility that these environmental factors are important in the etiology of NPCs is further supported by the finding that incidence rates decrease for successive generations of people who originally emigrated from southern China to California.
Other potential environmental etiological factors that have been associated with NPC include alcohol consumption and exposure to dust, fumes, formaldehyde, and cigarette smoke. Cigarette smoke and alcohol have long been associated with many other head and neck carcinomas, but their association with NPC has been controversial. Some studies have suggested that alcohol consumption and cigarette use were not associated with NPC. Nam et al. conducted a case-control study using a National Mortality Follow-Back Survey based on death certificates and found that cigarette smoking and alcohol consumption are independent statistically significant risk factors for NPC. The increased risk of NPC with heavy smoking (adjusted for alcohol consumption) was threefold, and an excess risk of 80% was demonstrated for heavy alcohol consumption (adjusted for cigarette use).
In addition to environmental factors, genetic factors have also recently been discovered that may explain some of the risk of developing nasopharyngeal cancer. Multiple investigators have found genetic variation in human leukocyte antigen (HLA) to be associated with the increased incidence of NPC in southern China. Simons et al. originally described an association in Chinese patients with NPC and the HLA-A2 antigen with a deficit of the second antigen at the second locus (B locus). Two large genome-wide association studies have subsequently confirmed the role of HLA locus in mediating risk of development of NPC. It remains unclear whether specific HLA alleles may influence development of NPC directly, possibly by affecting the immune reaction to EBV, or whether, and perhaps more likely, the genetic susceptibility to NPC is encoded by a locus in close linkage disequilibrium. Several other single-nucleotide polymorphisms have also been implicated to risk for NPC, including genes involved in DNA repair and metabolism of various toxins. However, these latter studies have generally been smaller and have not yet been replicated.
The nasopharynx cannot be visualized externally. Tumors of this area often present after they have eroded into a vital structure and produced a presenting symptom. The association of EBV and nasopharyngeal cancer has led some investigators to hypothesize that serological screening (EBV titer) may be useful in certain high-risk populations to identify groups of patients who might benefit from frequent nasopharyngeal examinations. In a study from China of 338,868 patients who underwent serological screening for EBV titer, 9367 persons had immunoglobulin (Ig) A antibodies to EBV. Of these patients, 306 were positive for IgA to EBV early antigen. Nasopharyngeal cancer was detected in 113 of the 9367 patients (1.2%) and in 63 of the 306 (20.5%) who were positive for IgA to EBV early antigen. Most of the tumors (> 85%) were early-stage lesions. Serological EBV IgA screening is currently being used in endemic areas.
In addition to EBV serological screening as a means of early detection and possible prevention of advanced-stage disease, several environmental and genetic predisposing factors are being explored as possible markers that could identify groups of patients at high risk for nasopharyngeal cancer. Real-time polymerase chain reaction (PCR) techniques show promise as screening tools for nasopharyngeal cancer. EBV DNA detection—in particular, detection of EBV nuclear antigen—eliminates many false-positive results and improves the sensitivity and specificity of EBV IgA serological screening. In a prospective study involving 1318 asymptomatic volunteers ages 40 to 60 years, three individuals with NPC were detected who were all positive for plasma EBV DNA analysis, whereas only one of the patients was positive for EBV IgA serology. A retrospective analysis of blood samples from patients with disease relapse from previous clinical trials indicates that EBV nuclear antigen screening by PCR would have led to earlier detection of distant failure but, because of the quality of current imaging, would not have significantly affected detection of local recurrences. Additionally, another screening study including patients from Hong Kong and Toronto yielded a sensitivity of 98.9% and specificity of 99.3% using transoral brush biopsies and quantitative PCR for EBV DNA in the detection of NPC. Another potentially beneficial biomarker is the EBV latent membrane protein 1 (LMP1), an oncoprotein encoded by EBV that is expressed in nearly all primary NPC tumors and mediates many oncogenic signaling pathways, which makes it a promising target in both the preventive and therapeutic settings. Real-time PCR screening for EBV LMP1 in nasopharyngeal swabs has also been shown to be a promising screening tool for high-risk populations, with a sensitivity of 87% and specificity of 98%. These techniques are likely to replace serum IgA screening in endemic areas and have a promising role in screening for relapse in EBV-positive patients. Current efforts are being made to establish harmonized EBV DNA assays that may demonstrate clinical utility in various settings (e.g., pretreatment, posttreatment, diagnosis, and monitoring). The further study of other environmental and genetic markers may lead to the identification of patient populations that could benefit from screening or chemoprevention.
Chemoprevention has been tested extensively in patients with a prior diagnosis of head or neck malignant tumors because second primary tumors in the respiratory and digestive tracts are common in that population. Hong et al. and Benner et al. conducted a prevention trial for patients with head and neck carcinoma after curative surgery or radiotherapy, or both. Patients were randomly assigned to receive 1 year of 13-cis-retinoic acid (isotretinoin) as a chemoprevention agent (see Khuri et al. for a discussion of the biological factors) or placebo. The treated group had significantly fewer second primary tumors (4%) than the placebo group (24%; p = 0.005); however, there was no difference in rates of survival or incidence of relapse between the 2 groups. In addition, the chemopreventive effect appeared to abate after 3 years; the incidence of second primary tumors was similar in the 2 groups after 3 years. Acute toxicity was significant.
The results of the trial by Hong et al. were not confirmed by a controlled trial of 316 patients by Bolla et al., in which patients were randomly assigned to 2 years of etretinate (a retinoic acid similar to isotretinoin) at a maintenance dosage of 25 mg/day. Subsequently, a large intergroup effort was mounted—1302 patients with previously treated head and neck malignant tumors were randomly assigned to receive 3 years of isotretinoin or placebo. Because of unacceptable toxicity seen with higher doses of isotretinoin (100-200 mg/m 2 [≈150-400 mg total]) in prior studies, the dose was decreased to 30 mg/day in this trial. No difference was seen in the occurrence of second primary tumors (4.6% in both arms), which was the primary endpoint of the study. A decrease in local recurrence was seen, however, and 13-cis-retinoic acid remains a topic of investigation.
The issue of whether retinoids or other chemopreventive agents should be used in the setting of premalignant lesions of the oral cavity has been considered. Many trials have suggested that retinoids can induce considerable responses in oral leukoplakia. Toxicity has been a limiting issue in these trials, however. It will be important to determine whether these chemoprevention strategies, developed for leukoplakia, can be extrapolated to prevention of nasopharyngeal cancer in subgroups of patients at high risk for such malignant tumors.
Blockade of cyclooxygenase-2 (COX-2) has also shown some promise as a chemopreventive strategy; COX-2 inhibitors were recently the topic of investigation for various malignant tumors, including head and neck cancers. A Phase II study at Fox Chase Cancer Center evaluated a 3-month course of celecoxib in patients with biopsy-proven dysplastic or hyperplastic leukoplakia, with results pending (NCT00101335). In another pilot, a randomized study of celecoxib in oral premalignant lesions conducted by the M. D. Anderson Cancer Center (MDACC), doses of 100 and 200 mg twice daily were shown to be ineffective in controlling these lesions. Most recent COX-2 inhibitor cancer prevention trials have been closed, except in very-high-risk settings such as familial polyposis, because of the increased rate of cardiovascular events seen in a few of the celecoxib and rofecoxib prevention trials. Non–COX derivatives of nonsteroidal antiinflammatory drugs have recently shown some promise as cancer prevention agents and may prove to have a superior toxicity profile.
Malignant tumors of the nasopharyngeal area are generally carcinomas (90%), with lymphomas making up approximately 5% of lesions. Histological classification of carcinomas continues to evolve as we find out more about the biological characteristics of these tumors. The most current pathological classification of NPCs by the World Health Organization (WHO) includes three major categories: keratinizing squamous cell carcinoma (formerly WHO type 1), nonkeratinizing squamous cell carcinoma (including differentiated, or former WHO type 2 lesions, and undifferentiated, or former WHO type 3 lesions), as well as recently described basaloid squamous cell carcinoma.
On histological testing, keratinizing squamous cell carcinomas appear to be well differentiated with intercellular bridges. These lesions make up approximately 20% of carcinomas. Differentiated nonkeratinizing carcinomas, representing 30% to 40% of NPCs, lack well-defined squamous cell characteristics but continue to show a “pavement stone pattern” characteristic of squamous cell histology. Undifferentiated nonkeratinizing carcinomas, comprising 40% to 50% of carcinomas, also formerly known as lymphoepitheliomas, are characterized by a lymphoplasmacytic infiltrate. On cytological testing, the cells appear uniform, with round to oval nuclei and prominent nucleoli. The frequency of the different histologic types varies with geographic area. In the United States, the majority of NPCs have keratinizing squamous cell histological findings, whereas in Asia, the undifferentiated histological type is by far the most common ( Table 40.1 ).
| STUDY | |||
|---|---|---|---|
| Sanguineti et al. | Johansen et al. | Lee et al. | |
| Location | United States (MDACC) | Denmark | Hong Kong |
| Study size | 378 patients | 167 patients | 5037 patients |
| Histologic type | |||
| Well differentiated | 5 patients | 8% | 0.3% a |
| Moderately differentiated | 36 patients | 5% | — |
| Poorly differentiated | 109 patients | 87% | — |
| Squamous cell carcinoma | 193 patients | — | — |
| Not specified | 43 patients | — | — |
| Lymphoepithelioma | 154 patients | — | — |
| Undifferentiated | — | — | 99.7% |
| Unknown | 31 patients | — | — |
| N0 category | 21% | 25% | 39% |
| Primary treatment | 60.2-72 Gy | 57-68 Gy at 1.8-2 Gy/day | 65 Gy b |
| Neck treatment | Yes | Yes | 906 of 1290 N0 patients not treated |
| Primary control | 66% (10-y) | 67% (10-y) | 87% (initial CR) |
| Nodal control | 83% (10-y) | 86% overall, 60% if no irradiation | 87% (initial CR) |
| Survival | 48% (5-y) | 37% (10-y) | 52% (initial CR) |
| Complications | See Table 40.7 | Xerostomia Edema Dysphagia Skin fibrosis/necrosis Osteonecrosis Radiation myelitis |
9% neurological damage 7% significant functional disturbance (soft-tissue necrosis) 1% death |
a Well differentiated or nonkeratinizing type.
b Median equivalent dose using time and dose fractionation (TDF) tables. Large (> 2 Gy) fraction sizes were often used.
The nasopharynx is a musculofascial tube that connects the nasal cavity and oropharynx. The anatomic borders of the nasopharynx are (1) the anterior border, consisting of the posterior nasal apertures and nasal septum; (2) the posterior border, the pharyngeal mucosa; (3) the superior border, the pharyngeal mucosa and body of the sphenoid; and (4) the inferior border, the oropharynx. The lateral wall of the nasopharynx contains the pharyngeal opening of the auditory tube. The medial cartilaginous extension of the auditory tube forms a protrusion from the lateral wall of the nasopharynx at the superior and posterior aspects of the opening of the auditory tube. This protrusion, with its overlying mucosa, creates the torus tubarius. Just posterior to the torus tubarius lies the pharyngeal fossa, or the fossa of Rosenmüller, which is formed by the junction of the lateral and posterior walls of the nasopharynx ( Fig. 40.1 ).
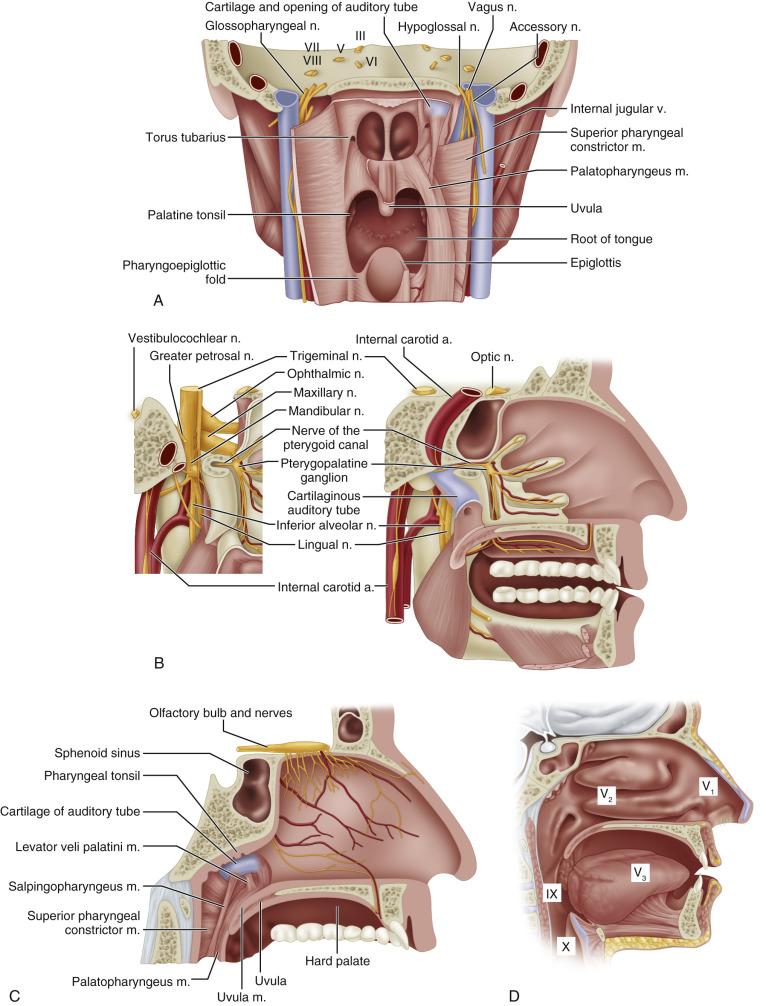
The superior wall of the nasopharynx warrants review. Extension of tumors superiorly often leads to invasion of structures, the symptoms of which are typical of the initial clinical presentation of patients. The sphenoid bone lies superior to the mucosa and pharyngobasilar fascia of the nasopharynx. Several cranial nerves exit the base of skull in this area. Critical foramina housed in the sphenoid include the superior orbital fissure (cranial nerves III, IV, V [ophthalmic division], and VI), the foramen rotundum (cranial nerve V [maxillary division]), the foramen ovale (cranial nerve V [mandibular division]), and the foramen spinosum (recurrent branch of cranial nerve V [mandibular division]). The foramen lacerum is formed by the junction of the sphenoid and temporal bones. The internal carotid artery passes over the superior opening of the foramen lacerum but does not traverse through the foramen. The nerve of the pterygoid canal and the meningeal branch of the ascending pharyngeal artery course through the foramen lacerum. Deep to the mucosa of the superior aspect of the lateral wall of the nasopharynx lies the superior pharyngeal constrictor muscle. Beyond this muscle lie cranial nerves IX, X, and XI, which exit the base of the skull from the jugular foramen in the temporal bone, and cranial nerve XII, which exits the base of the skull through the hypoglossal canal of the temporal bone.
The sensory innervation is shown in Fig. 40.1D . The maxillary division of the trigeminal nerve supplies the upper nasopharynx, the posterior part of the nasal cavity, and most of the palate and upper gums of the oral cavity. The general sensory branches of the lingual and pharyngeal branches of the glossopharyngeal nerve supply the sensory innervation of the lower part of the nasopharynx, the posterior third of the tongue, and part of the soft palate and oropharynx.
Understanding the anatomic relationships of the nasopharynx and adjacent structures is important to correctly diagnose patients with NPC. The importance of superior extension into the sphenoid bone of the base of the skull is exemplified by the fact that cranial nerves can be involved at presentation. The frequency of involvement of cranial nerves has been described in two large series. Cranial nerves V and VI are the most commonly involved. These nerves traverse the sphenoid bone and can be involved when tumors erode superiorly through bone. Tumors of the nasopharynx can also gain access to the cranial nerves of the base of the skull by eroding superiorly through the foramen lacerum, which is bordered superiorly by the internal carotid artery ( Fig. 40.1B ).
Lateral extension of NPCs can lead to erosion of the medial opening of the auditory tube and the medial pterygoid plate, which can involve cranial nerves IX, X, XI, and XII ( Figs. 40.1B and 40.1C ). Lateral extension can lead to involvement of the carotid artery and internal jugular vein.
Lymph node involvement is common; 65% to 80% of patients present with clinically involved cervical neck nodes. The level 5A and level 2 lymph nodes are commonly involved. The frequency of involvement of these areas in two large series is presented later in the “Clinical Manifestations” section. Recent studies have shown that, for clinically N0 or N1 disease, microscopic involvement of the inferior neck and posterior neck (levels 4 and 5B) is less frequent.
The potential etiological link between EBV and NPC was first described more than 30 years ago. Recent advances in molecular biology have shed further light on this association. Earlier work focused on the serological response to EBV, showing elevated levels of IgA and IgG antibodies to the viral capsid antigen and a replication protein called early antigen in patients with NPC. Other studies have demonstrated the presence of EBV DNA and encoded proteins directly in the NPC tissue samples. Latent infection of cells with EBV, initially studied in B lymphocytes, is associated with regulated expression of several viral genes, including LMP1, LMP2A, and LMP2B; 6 EBV nuclear antigens (EBNAs), EBNA1, EBNA2, and EBNA3A to EBNA3C; LP; and two small noncoding nuclear RNAs (EBERs). Some of the molecular mechanisms by which expression of these genes can lead to transformation have been elucidated. For example, LMP1 induces expression of epidermal growth factor receptor (EGFR); therefore, it may influence cellular growth. A restricted pattern of these EBV antigens is present in nasopharyngeal cancer cells, which may explain in part why immunotherapy directed against these antigens has been more successful in other EBV-associated diseases. Allogeneic cytotoxic T cells to various EBV antigens have been developed and show some promise for treating locally recurrent NPC.
Several studies have suggested that serological responses to EBV may be more specific for the nonkeratinizing carcinoma (former WHO types 2 and 3). However, this was not confirmed by another group. Using the Southern blot technique to detect the viral DNA, Raab-Traub et al. showed the presence of EBV in all former histological variants of NPC, but the EBV copy number was lowest in keratinizing squamous cell carcinomas. The presence of EBV in keratinizing squamous cell carcinoma has been consistently detected by other groups. The differences in the rate of EBV detection by different investigators may stem from variability in the sensitivity of different assays. Alternatively, it may reflect differences in the sampled populations. The detection rate of EBV infection in keratinizing squamous cell NPC is highest in endemic areas.
Several groups have examined measuring circulating EBV DNA as a biomarker of tumor burden and response to treatment. The rationale for measuring circulating EBV DNA is that tumor cells are hypothesized to spill EBV DNA into the circulation and, as such, pretreatment EBV DNA levels would serve as a measure of burden of disease, and postradiotherapy EBV DNA levels might serve as an indicator of residual local or systemic disease. Lo et al. demonstrated that, using real-time quantitative PCR, cell-free EBV DNA was detectable in 96% of patients with nasopharyngeal cancer. Investigators from Hong Kong then showed that EBV DNA levels 1 week postradiotherapy correlated with poor outcome, with a 2-year overall survival (OS) of 56.3% for those with detectable EBV DNA compared with 96.7% for those with undetectable EBV DNA. In a recent meta-analysis of 23 studies, including 10,732 patients with NPC, hazard ratios (95% confidence intervals [Cis]) of pretreatment EBV DNA levels and posttreatment plasma EBV levels for OS were 2.78 (95% CI, 2.19-3.55) and 5.43 (95% CI, 2.72-10.82), respectively. Multiple other groups have also confirmed the prognostic value of posttreatment circulating EBV DNA. At present, several Phase III studies are underway investigating whether the presence of circulating EBV DNA after definitive chemoradiotherapy can predict for the need of adjuvant chemotherapy.
Beyond examining the role of EBV in nasopharyngeal cancer, others are examining genomic alterations in nasopharyngeal cancer via integrated genomic profiling with sequencing, expression, and copy number analysis as part of the International Cancer Genome Consortium. To date, most investigations have focused either on copy number changes in nasopharyngeal tumors or alterations in gene expression. Deletions of the short arm of chromosome 3 (3p25, 3p14) and of chromosome 9 (9p21-22) are the most common cytogenetic changes. An extended genome-wide association study with an analysis of 7046 NPC cases and 8570 controls resulted in 2 SNP associations with genome-wide significance, TERT-CLPTM1L at chromosome 5p15 and CIITA at chromosome 16p13. Candidate genes in these and other locations of interest are being identified. These findings may lead to a greater understanding of the genetic basis of NPCs and the interplay of environmental factors with these genetic factors. A full catalog of commonly mutated genes in nasopharyngeal cancer has yet to be determined.
As mentioned, NPC is rare in the United States; therefore, it is not often suspected as a possible cause of a patient's early symptoms. In addition, the list of possible early symptoms of NPCs includes many symptoms that could have more common causes. In a series of 378 patients from the MDACC, the presenting symptoms included a neck mass in 41%; hearing loss, ear drainage, or otalgia in 27%; nasal bleeding or obstruction in 21%; cranial nerve deficits in 8%; and other nonspecific symptoms in 8%. Investigators from Washington University noted that the typical presentation involved multiple symptoms. In this series of 143 patients, presenting symptoms included otitis in 43%, throat pain in 39%, nasal obstruction in 37%, a neck mass in 35%, nasal bleeding in 29%, cranial nerve involvement in 24%, and trismus or other symptoms in 5%.
Similar to these data, most series have shown that neck masses, obstructive ear symptoms, and cranial nerve deficits are common presenting symptoms for NPC. In the Washington University series, 66% of patients were found to have ipsilateral neck masses, and 28% had contralateral neck masses on examination, although only 35% of patients had reported a neck mass as the reason for seeking medical advice. Of the patients with clinically involved neck disease, 60% had enlarged ipsilateral level 2 lymph nodes and 32% had enlarged ipsilateral level 5 lymph nodes. The most frequently involved cranial nerves in the series were cranial nerve VI in 15% of patients, V in 7.7%, and VIII, X, and XII in 5.6% of patients each.
In the series from the MDACC, the level 5A lymph nodes were most commonly enlarged (54% of patients), followed by the level 2 nodes (49% of patients). The level 3 and lower level 5A/upper level 5B groups were involved in 24% and 22% of patients, respectively. The level 4 lymph nodes, level 5B lymph nodes, and supraclavicular lymph nodes were involved in 10%, 13%, and 10% of patients, respectively. Similar to the series from Washington University, cranial nerve VI was the most frequently involved nerve (6% of cases).
The initial evaluation of patients with NPC should include a history and physical examination, with special attention to the level of nodal involvement (if any; Fig. 40.2 ). It is helpful to diagram or digitally photograph the clinically involved lymph nodes in the neck because it can be helpful when the radiotherapy boost is considered.
Patients should undergo flexible fiberoptic nasopharyngoscopy to determine the involvement of mucosal surfaces. A diagram or digital photograph of these findings should also be made. Biopsy of the primary lesion should be attempted first. If this procedure gives indeterminate results, a needle biopsy of a potentially involved lymph node should be performed.
Computed tomography (CT) and magnetic resonance imaging (MRI) of the head and neck are useful in the evaluation of both erosion of tumor into the bony structures of the base of the skull and retropharyngeal and cervical lymphadenopathy. Although the same information is often provided by CT and MRI, some investigations have suggested that MRI may be more useful in delineating soft-tissue invasion outside the nasopharynx and the extent of retropharyngeal lymph node involvement, whereas CT may be most useful in delineating skull base erosion. Given the level of anatomic information required for the current T staging system, MRI is a critical imaging study in the evaluation of nasopharyngeal tumors.
Positron emission tomography (PET) can be useful for staging as well as for posttherapy management in which other imaging is unclear. PET/CT, which provides additional anatomic information, is more commonly used now and may be helpful for radiotherapy planning. Further PET/CT helps evaluate for distant metastatic disease before beginning definitive locoregional treatment.
The completion of the diagnostic evaluation involves acquiring routine complete blood counts, a chemistry panel, and a chest radiograph (see Fig. 40.2 ). Further evaluation of possible metastases should be done on the basis of the clinical presentation of the patient.
After a pathological assessment has been made, the workup or evaluation leads to a clinical stage ( Table 40.2 ). Clinical staging has been a matter of controversy; thus, the prognostic significance of the various TNM (tumor, node, metastasis) staging criteria remains under investigation. Several staging systems have been in use throughout the world. The most commonly used systems in North America and Europe are those of the American Joint Committee on Cancer (AJCC) and the International Union Against Cancer (Union Internationale Contre le Cancer [UICC]), which have become essentially analogous. In Asia, where NPC is endemic, the Ho classification system was initially developed in Hong Kong. In addition, in 1992, Chinese physicians adopted an independent system similar to the Ho system. All four systems, shown in Table 40.2 , have limitations and continue to evolve, with researchers often drawing on each other's experience. Updated versions of the Chinese system and the AJCC system were released in 2008 and 2010, respectively. The AJCC/UICC system remains the most commonly used in the English literature.
| Classification | AJCC, 7th ed. | 2002/1997 AJCC | Ho | 2008 Chinese |
|---|---|---|---|---|
| T1 | Confined to nasopharynx or extends to oropharynx and/or nasal cavity | Confined to nasopharynx | Confined to nasopharynx | Confined to nasopharynx |
| T2 | With parapharyngeal extension | Soft-tissue invasion A: Without parapharyngeal extension B: With parapharyngeal extension |
Nasal fossa, oropharynx, muscle, or nerves below base of skull | Nasal cavity, oropharynx, parapharyngeal extension |
| T3 | Bony or paranasal sinus extension | Bony or paranasal sinus extension | A: Bone involvement below base of skull B: Involves base of skull C: Cranial nerves D: Orbits, laryngopharynx, or infratemporal fossa a |
Skull base or medial pterygoid extension |
| T4 | Intracranial extension, or cranial nerve or infratemporal fossa, a hypopharynx, or orbital involvement | Intracranial extension, or cranial nerve or infratemporal fossa, a hypopharynx, or orbital involvement | — | Cranial nerves, paranasal sinuses, masticator space a (excluding medial pterygoid muscles), intracranial (cavernous sinus, dural meninges) extension |
| N1 | Unilateral, ≤ 6 cm and/or unilateral or bilateral retropharyngeal ≤ 6 cm | Unilateral, ≤ 6 cm | Upper neck above thyroid notch | A: Retropharyngeal B: Unilateral level 1b, 2, 3, and 5a or ≤3 cm |
| N2 | Bilateral, ≤ 6 cm | Bilateral, ≤ 6 cm | Below thyroid notch above line joining end of clavicle and superior margin of trapezius muscle | Bilateral level 1b, 2, 3, and 5a or > 3 cm or with extranodal spread |
| N3 | A: > 6-cm node B: Supraclavicular involvement | A: > 6-cm node B: Supraclavicular involvement | Supraclavicular fossa b or skin involvement | Level 4 or 5b involvement |
| M1 | — | Metastases | Metastases | Metastases |
| Stage I | T1N0M0 | T1N0M0 | T1N0 | T1N0M0 |
| Stage II | T1N1 T2N0 T2N1 |
A: T2aN0M0 B: T1-2aN1M0 or T2bN0M0 |
T2 and/or N1 | T1N1a-1bM0 or T2N0-1bM0 |
| Stage III | T1N2M0 T2N2M0 T3N0-2M0 |
T1-2bN2M0 T3N0-2M0 |
T3 and/or N2 | T1-2N2M0 or T3N0-2M0 |
| Stage IV | A: T4N0-2M0 B: Any TN3M0 C: Any T any NM1 | A: T4N0-2M0 B: Any TN3M0 C: Any T any NM1 | N3 (any T) | A: T1-3N3M0 or T4N0-3M0 B: Any T any NM1 |
| Stage V | — | — | M1 | — |
a Masticator space is defined as infratemporal fossa.
b Supraclavicular fossa as described by Ho is a triangular space defined by three points: (1) the superior margin of the sternal end of the clavicle, (2) the superior margin of the lateral end of the clavicle, and (3) the point where the neck meets the shoulder. This includes the caudal portion of levels 4 and 5b lymph nodes.
A well-recognized advantage of the Ho classification system lies in its approach to lymph node metastasis, namely, use of lymph node location in N-stage assignment and a more even distribution of nodal disease in stage grouping (see Table 40.2 ). Nodal involvement appears in stage II, and supraclavicular nodal metastasis, which is known to carry a worse prognosis, is represented in a separate stage IV category (see Table 40.2 ). The 1992 AJCC system used lymph node size and laterality in N-stage assignment, both of which carry some prognostic significance. However, only some patients with N1 disease were included in stage III, and most patients with nodal involvement were grouped into stage IV together with patients with metastatic disease. Modifications to the AJCC classification implemented in 1997 and maintained in 2002 and 2010 have incorporated lymph node location into N-category assignment, designating involvement of the supraclavicular fossa, as originally defined by the Ho system, as N3 disease, and have placed all N3M0 lesions in a separate stage IVA grouping. In addition, stage groupings were changed to provide a more even-stage distribution. These changes improved risk stratification of the 1997 AJCC classification in comparison with both the prior version of the AJCC system and the 1978 Ho system.
Important changes to the most recent AJCC system targeted T-category assignment. Invasion into the soft tissues of the nasopharynx was used in the 1997 and 2002 AJCC classification to segregate T1 and T2 lesions, but studies have shown that this carries no prognostic significance. The parapharyngeal space involvement used to segregate T2a and T2b lesions does, however; therefore, the 2010 AJCC system segregates lesions with parapharyngeal involvement into the T2 subgroup, whereas all tumors either confined to the nasopharynx or extending into surrounding subsites but without parapharyngeal involvement are now in the T1 subgroup. Furthermore, cranial nerve involvement carries a significantly worse prognosis than base-of-skull involvement, but both were aspects of the T4 subgroup in the 1997 and 2002 AJCC systems and of the T3 subgroup in the Ho system. In the 2003 and 2010 versions, akin to the 2008 Chinese classification system, base-of-skull involvement has been downstaged to T3. Furthermore, data are emerging that extensive cranial nerve involvement, orbit involvement, and intracranial extension are associated with worse outcomes within the T4 subgroup. Therefore, some of the proposed modifications to the AJCC system include a separation of the T4 subgroup into two categories. T4a would include tumors with involvement of the masticator space (infratemporal fossae), asymptomatic radiographic cranial nerve involvement, and involvement of the hypopharynx. T4b would include tumors with intracranial extension, orbital involvement, and symptomatic cranial nerve palsies. However, more data need to be collected to validate this proposed modification.
With respect to N-category classification, the 2010 seventh edition of the AJCC staging manual for the first time includes disease that has spread to the retropharyngeal lymph nodes in staging, placing it in the N1 category. On MRI evaluation, these nodes are involved in 83% of patients with NPC compared with 74% involvement of the levels 2 to 4 nodes and are considered to be the first echelon of nodal spread. Therefore, their inclusion in the staging system is expected to improve prognostic accuracy.
The evolution of technology—specifically, CT and MRI—has allowed for more precise evaluation of tumor invasion into surrounding structures and lymph node involvement, which will undoubtedly lead to improvement in staging accuracy and risk stratification.
Traditionally, the nasopharyngeal area has not been easy to examine without fiberoptic technology and has been difficult to approach surgically. Surgical exposure of the area and resection of tumors with adequate tumor margins have long been challenging. For these reasons, primary surgical intervention fell out of favor in the 1950s; primary treatment has generally consisted of radiotherapy for early-stage disease and chemoradiotherapy for locally advanced disease.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here