Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Myelodysplastic syndrome (MDS)/myeloproliferative neoplasm (MPN) overlap syndromes are a group of rare myeloid malignancies that paradoxically exhibit both dysplastic and proliferative hematopoietic cell characteristics. They share phenotypic features of both MDS, such as dysplasia and cytopenias, and of MPNs, including constitutional symptoms, splenomegaly, and terminally differentiated blood cell expansion. There are four adult MDS/MPN overlap syndromes: chronic myelomonocytic leukemia (CMML), atypical chronic myeloid leukemia (aCML), MDS/MPN with ring sideroblasts and thrombocytosis (MDS/MPN-RS-T), and MDS/MPN unclassifiable (MDS/MPN-U). There is also a pediatric syndrome, juvenile myelomonocytic leukemia (JMML). These disorders were historically classified as either an MDS or an MPN, but in 2002, the World Health Organization (WHO) first designated MDS/MPN overlap syndromes as an independent category of tumors of hematopoietic tissues, in recognition of the unique features of these myeloid malignancies. Recurrent mutations shared between the subtypes of MDS/MPNs, and the MPN and MDS highlight the interrelated nature of these diseases.
MDS/MPNs present challenges both from a diagnostic and therapeutic standpoint. As rare diseases, an accurate diagnosis can be challenging and often relies on referral to tertiary care centers with a team of experienced hematopathologists and clinicians. In addition, there is a paucity of clinical research and novel therapeutic approaches for treating these malignancies included within the MDS/MPN overlap syndromes. Treatment approaches are, therefore, largely borrowed from those used to treat MDS or the MPNs. Given advances in the molecular and pathobiologic understanding of MDS/MPNs overlap syndromes, novel therapeutic development is starting to advance, which has the potential to alter the generally poor outcomes seen with such patients.
The epidemiology of MDS/MPN overlap syndromes is poorly described. CMML is the most common MDS/MPN overlap syndrome, with an incidence of 0.35 cases per 100,000. As expected, the incidence of CMML increases significantly with age based on SEER (Surveillance, Epidemiology, and End Results) data. There is a male predominance, and the disease is more common in those of European descent than Blacks or Asian/Pacific Islanders.
Epidemiologic data are limited for aCML, although one study suggests a slight male predominance with a median age of onset of 69 years. There is no published epidemiologic information concerning MDS/MPN-RS-T or MDS/MPN-U. JMML has an estimated incidence of 1.2 cases per million annually and accounts for approximately 2% of leukemias occurring during childhood. Males are affected more frequently than females, and approximately 50% of cases occur in infants.
The diagnostic features for the MDS/MPN overlap syndromes, although diverse, essentially encompass a hybrid of both marrow cell dysplasia and myeloproliferation. The dysplastic features are similar to the classic features associated with MDS involving one or more of the hematopoietic lineages. The myeloproliferative component results frequently in monocytosis in CMML, thrombocytosis in MDS/MPN-RS-T, or granulocytosis in aCML. Establishing the diagnosis of a patient with an MPN/MDS overlap syndrome usually requires an integrated approach incorporating clinical presentation, laboratory data, peripheral blood and marrow morphologic evaluation, cytogenetics, and myeloid gene mutational profiling.
In general, special considerations should be taken when making the diagnosis of an MDS/MPN overlap syndrome. The diagnosis should be made prior to the patient receiving any therapy and other genetically-defined myeloid neoplasms should be excluded using cytogenetic analyses and molecular testing. In essence, MDS/MPN overlap syndromes are frequently diagnoses of exclusion.
MDS/MPN overlap syndrome patients present with abnormal blood counts with at least a single lineage cytopenia (the MDS component) or a single lineage cytosis (the MPN component). Of particular interest are the monocyte counts (associated with CMML), eosinophilia (may suggest myeloid neoplasm with PDGFRα , PDGFRβ , or FGFR1 abnormalities), and basophilia (may suggest chronic myeloid leukemia [CML] rather than MDS/MPN overlap syndromes). Clinically, patients experience symptoms of MDS including fatigue, frequent infections, and bleeding/bruising. In addition, features of the MPNs such as constitutional symptoms and extramedullary hematopoiesis (often manifested as splenomegaly) can be observed. However, there is a wide spectrum of clinical manifestations in MDS/MPN overlap syndrome patients ranging from the patients being asymptomatic to having debilitating symptoms.
CMML is characterized by sustained monocytosis in the setting of bone marrow (BM) cellular dysplasia. Monocytosis is defined by having an absolute monocyte count greater than 1 × 10 9 /L with greater than 10% monocytes being present in the white blood cell (WBC) differential count that persists for at least 3 months. An examination of the BM is critical to exclude acute myeloid leukemia (AML) from the diagnosis since CMML patients, unlike AML patients, have a blast cell percentage less than 20%. Many other entities can lead to a reactive form of monocytosis, which are discussed in detail in Chapter 49 , and must be excluded. Non-neoplastic causes of monocytosis include chronic infections, autoimmune diseases, and drug reactions, among others. Other myeloid neoplasms with associated monocytosis should be excluded as well as CML, primary myelofibrosis, and MDS. The WHO diagnostic criteria for CMML are listed in Table 73.1 . CMML can be further subtyped based on the degree of elevation of the peripheral blood WBC count into MPN-CMML (WBC ≥13 × 10 9 /L) and MDS-CMML (WBC <13 × 10 9 /L).
|
|
|
|
|
|
|
|
|
a Cases of MPN can be associated with monocytosis or they can develop it during the course of the disease. These cases may simulate CMML. In these rare instances, a previous documented history of MPN excludes CMML, whereas the presence of MPN features in the BM and/or of MPN-associated mutations ( JAK2 , CALR , or MPL ) tend to support MPN with monocytosis rather than CMML.
b Blasts and blast equivalents include myeloblasts, monoblasts, and promonocytes. Promonocytes are monocytic precursors with abundant light gray or slightly basophilic cytoplasm with a few scattered, fine lilac-colored granules, finely distributed, stippled nuclear chromatin, variably prominent nucleoli, and delicate nuclear folding or creasing. Abnormal monocytes, which can be present both in the PB and BM, are excluded from the blast count.
c The presence of mutations in genes often associated with CMML (e.g., TET2 , SRSF2 , ASXL1 , SETBP1 ) in the proper clinical context can be used to support a diagnosis. It should be noted, however, that many of these mutations can be age-related or be present in subclones. Therefore, caution would have to be used in the interpretation of these genetic results.
The marrow is typically hypercellular with myeloid hyperplasia and monocytosis. The dysplasia is common in the three lineages; however, dysplasia is more common and pronounced in the megakaryocyte lineage, with micromegakaryocytes and small megakaryocytes with hypolobated nuclei being a prominent feature ( Fig. 73.1 ). In rare cases where the dysplasia may not be overt, the diagnosis of CMML can be made if a cytogenetic or myeloid mutational abnormality is detected or if the monocytosis is persistent with no secondary causes identified. Mild BM fibrosis is not uncommon in CMML.
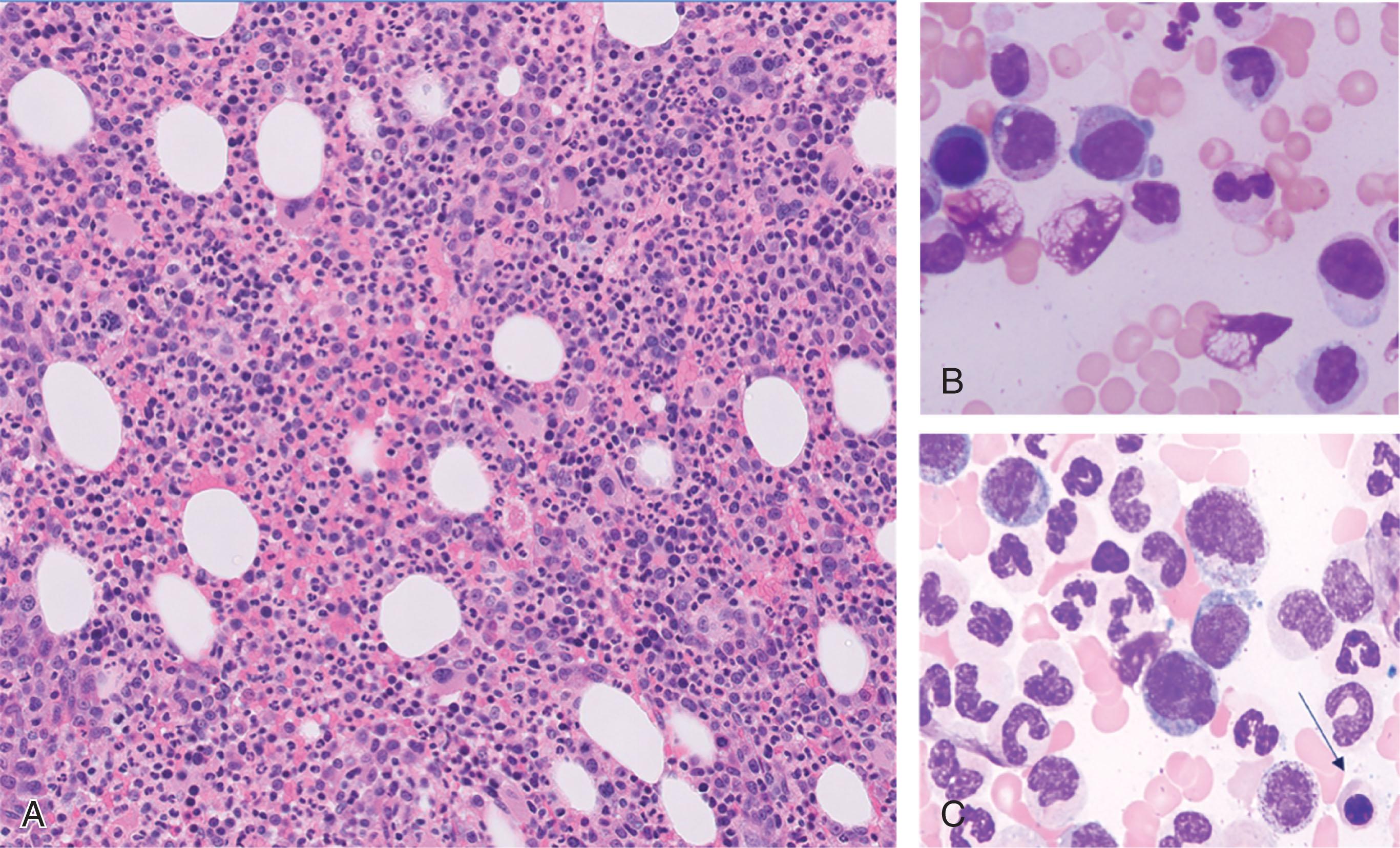
A plasmacytoid dendritic cell (PDC) proliferation (CD123-positive) is a feature of CMML that is observed in 20% of cases but is a non-specific finding and can be seen in other myeloid neoplasms. Of note, CMML is the most common myeloid neoplasm that is associated with systemic mastocytosis (see Chapter 75 ).
Recently, an entity termed “oligomonocytic chronic myelomonocytic leukemia” has been described. This condition is characterized by blood monocytes ≥10% of the WBC without an absolute monocyte count greater than 1 × 10 9 /L. These patients, when compared to CMML, are typically younger and have lower WBCs and absolute neutrophils counts. The molecular signature is similar to CMML with mutations in ASXL1, TET2 , and SRSF2 . Progression to overt CMML occurs in approximately 40% of cases over a median time of 12 months. Thus, these cases should not be classified as a type of MDS but rather to represent a forme fruste of CMML.
aCML is defined by left-shifted neutrophilic leukocytosis with severe dysgranulopoiesis and greater than 10% immature myeloid precursor cells present in the peripheral blood. Anemia and thrombocytopenia are common. These patients lack the BCR-ABL-1 fusion protein that is diagnostic of CML (see Chapter 69 ). Basophilia and eosinophilia are not typical features of aCML and should prompt careful consideration of a diagnosis of another myeloid neoplasm. aCML often presents with extramedullary disease, especially in the spleen and liver. The two other diagnoses that resemble aCML and should be ruled out are CMML (which exhibits monocytosis) and CML (which is BCR-ABL-1 -positive). The WHO diagnostic criteria for aCML are listed in Table 73.2 .
|
|
|
|
|
|
|
|
a Cases of myeloproliferative neoplasm (MPN) , particularly those in accelerated phase and/or in postpolycythemic or postessential thrombocythemic myelofibrosis, if neutrophilic, may simulate aCML. A previous history of MPN, the presence of MPN features in the bone marrow, and/or MPN-associated mutations (in JAK2 , CALR , or MPL ) tend to exclude a diagnosis of aCML. Conversely, a diagnosis of aCML is supported by the presence of SETBP1 and/or ETNK1 mutations. The presence of a CSF3R mutation is uncommon in aCML and, if detected, should prompt a careful morphologic review to exclude an alternative diagnosis of chronic neutrophilic leukemia or other myeloid neoplasm.
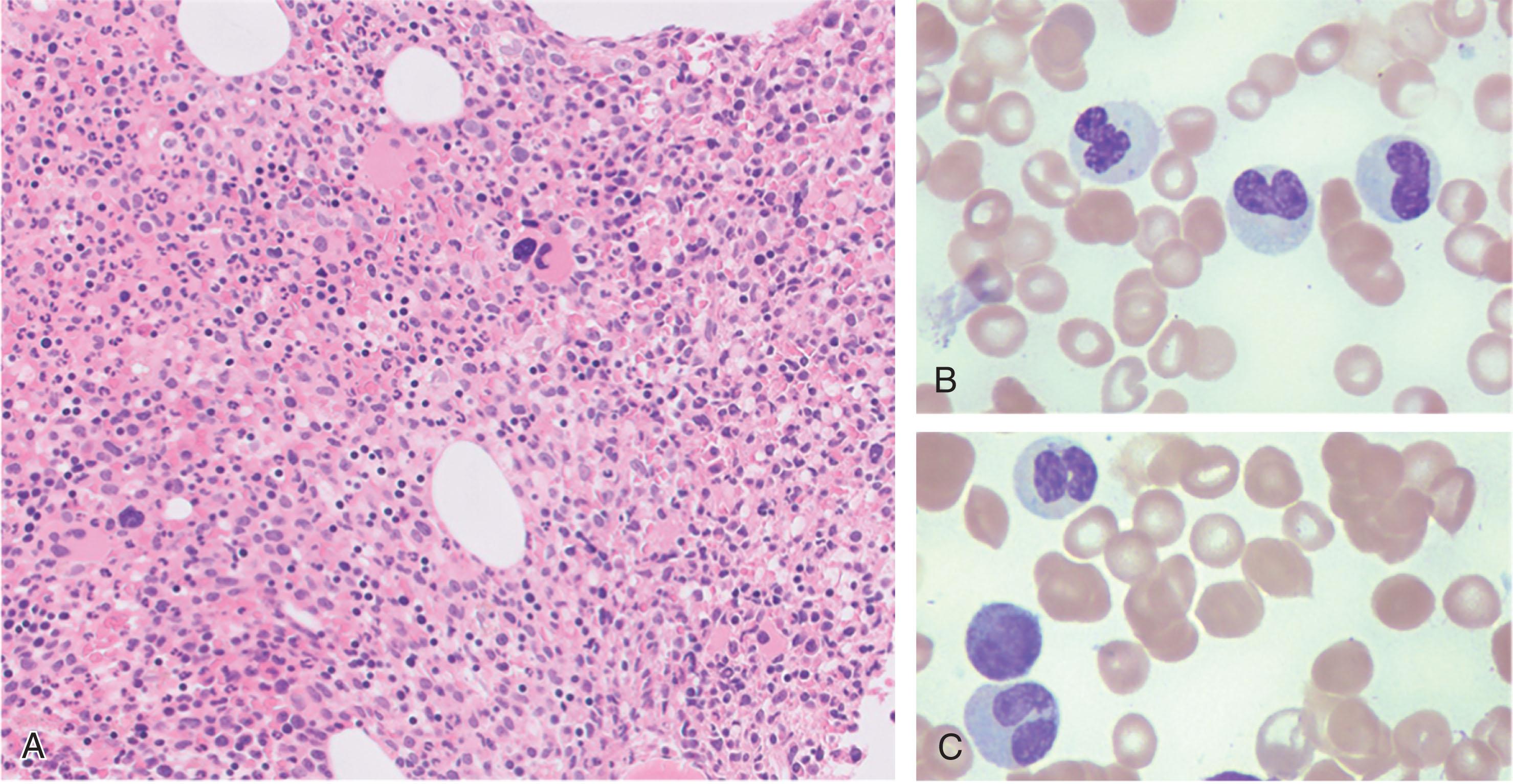
The BM in aCML is hypercellular with myeloid hyperplasia and a high myeloid-to-erythroid (M:E) ( Fig. 73.2 ). aCML is characterized by dysplastic hematopoiesis and the presence of immature myeloid precursor cells in the peripheral blood that distinguishes it from chronic neutrophilic leukemia (CNL) (see Chapter 72 ). In addition to the granulocytic dysplasia, erythroid and megakaryocytic dysplasia are also common. Importantly, aCML is characteristically associated with mutations in SETBP1 and/or ETKN1 . Since the CSF3R mutation is seen in less than 10% of patients with aCML, the presence of this mutation, in conjunction with the absence of BM dysplasia and immature myeloid precursors, suggests a diagnosis of CNL rather than aCML. The presence of MPN mutations involving JAK2 , MPL , or CALR tends to exclude the diagnosis of aCML
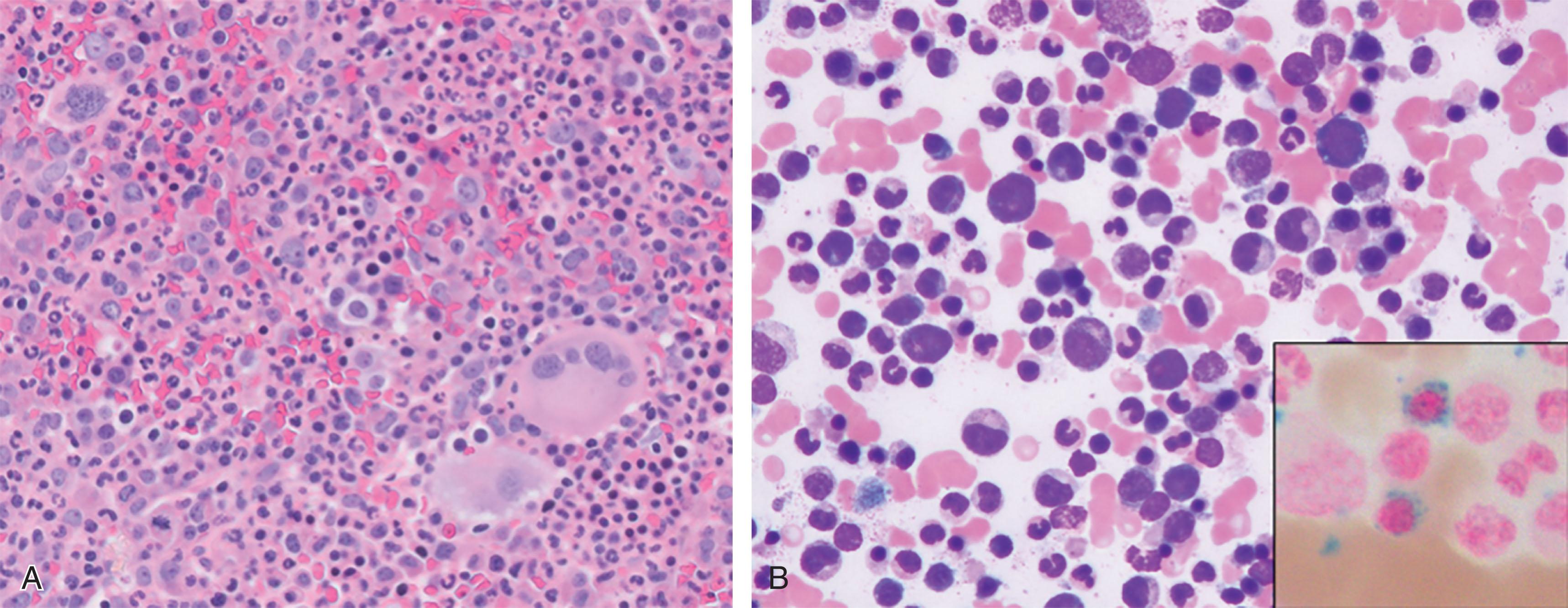
Patients with MDS/MPN-RS-T can present with vasomotor symptoms, such as migraine headaches, erythromelalgia, and paresthesias, or can have fatigue related to anemia. Additionally, some patients initially present incidentally after routine laboratory testing that reveals thrombocytosis and anemia. Table 73.3 lists the WHO diagnostic criteria for MDS/MPN-RS-T, which includes refractory anemia, ≥15% ring sideroblasts in the BM, and thrombocytosis. The presence of the spliceosome mutation SF3B1 co-mutated with an MPN driver mutation (in particular JAK2V617F ) is highly suggestive of MDS/MPN-RS-T in the appropriate clinical setting. Distinction between MDS with ring sideroblasts is essential, which in contrast to MDS/MPN-RS-T, lacks thrombocytosis and has hypolobulated, instead of hyperlobulated, atypical megakaryocytes ( Fig. 73.3 ). Other non-clonal disorders can result in the appearance of ring sideroblasts including lead or zinc toxicity, copper or pyridoxine deficiency, the use of particular medications (linezolid, isoniazid), excessive alcohol use, and congenital forms of sideroblastic anemias (see Chapter 39 ). Reactive causes of thrombocytosis, including infection, rheumatologic conditions, recent trauma/surgery, blood loss, iron deficiency, and hemolysis, should be ruled out (see Chapters 29 and 71 ).
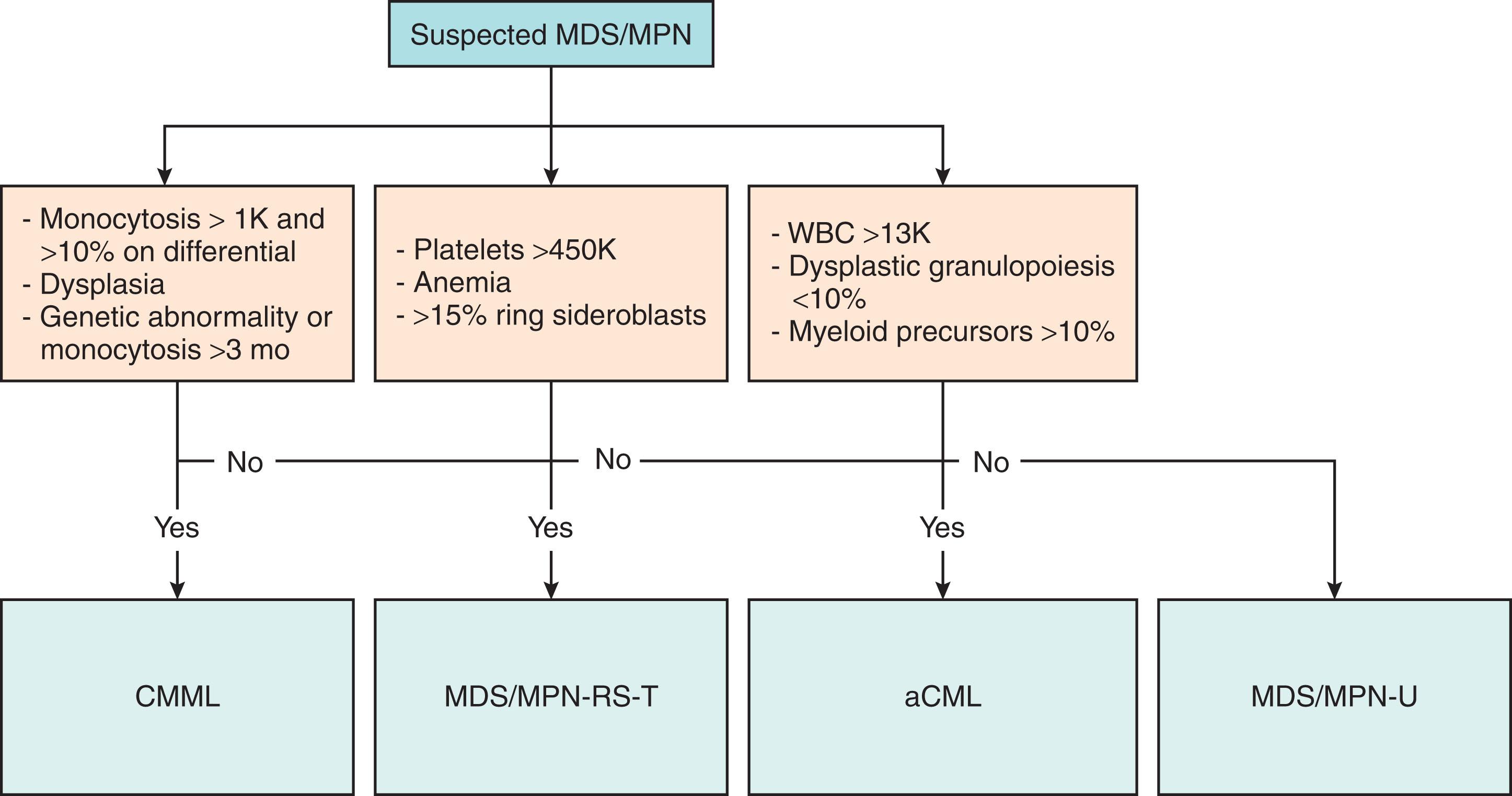
MDS/MPN overlap syndromes should be suspected in patients who display phenotypic properties of both bone marrow failure and myeloproliferation. Fig. 73.4 summarizes the diagnostic approach for patients with MDS/MPN overlap syndromes. As noted in the WHO diagnostic criteria ( Tables 73.1–73.4 ), a bone marrow biopsy is essential for the diagnosis of MDS/MPN overlap syndromes in order to distinguish among the diagnoses and from other myeloid malignancies and to determine the type of MDS/MPN overlap syndrome. Histopathology review by an expert hematopathologist is key for proper diagnosis. In all MDS/MPN overlap syndrome patients, exclusion of chronic myeloid leukemia (CML) with polymerase chain reaction (PCR) for BCR-ABL1 must be performed. CMML, the most common MDS/MPN, can be diagnostically challenging given that monocytosis is seen in other related hematologic disorders or can be reactive due to chronic infections, autoimmune disorders, or other systemic inflammatory processes. Therefore, persistent monocytosis lasting more than 3 months must be documented, unless there is a clonal cytogenetic or molecular marker specific for myeloid malignancies, such TET2 , SRSF2 , ASXL1 , or SETBP1 . The diagnosis of aCML should be considered in a patient with neutrophilia with greater than 10% immature myeloid cells being the differentiating feature among other MDS/MPN overlap syndromes. Careful consideration of CNL must also be given when considering a diagnosis of aCML given that both have a high frequency of the CSF3R mutation. Among the differentiating features, the presence of ≥10% immature granulocytes strongly favors aCML and the presence of less than 10% with greater than 80% neutrophils and bands favors CNL. MDS/MPN-RS-T is characterized by thrombocytosis, concurrent anemia, and ring sideroblasts which differentiates this diagnosis from ET. Cases that harbor features of both MDS and MPN but do not meet WHO diagnostic criteria for any MDS/MPN subtype can be categorized in the “grab-bag” category of MDS/MPN-U.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
a Germ line mutations (indicating Noonan syndrome) need to be excluded.
|
|
|
|
|
a At least 15% ring sideroblasts are required even if SF3B1 mutation is detected.
b A diagnosis of MDS/MPN-RS-T is strongly supported by the presence of SF3B1 mutation together with a mutation in JAK2 V617F, CALR , or MDS-RS, myelodysplastic syndrome with ring sideroblasts; MPL genes.
c In a case which otherwise fulfills the diagnostic criteria for MDS with isolated del(5q)-no or minimal absolute basophilia; basophils usually less than 2% of leukocytes.
MDS/MPN-U is, by definition, a diagnosis of exclusion, and the clinical presentation varies widely. By definition, MDS/MPN-U is characterized by at least one dysplastic lineage, less than 20% blasts in the marrow and/or peripheral blood, and absence of BCR-ABL-1 (which defines CML) and the fusions genes involving PDGFRA, PDGFRB , and FGFR1 (which define eosinophilia-associated myeloid/lymphoid neoplasms). In addition, there must be dysplastic and proliferative features that do not meet the diagnostic criteria for CMML, JMML, aCML, or MDS/MPN-RS-T U. This “wastebasket” category of MDS/MPN overlap syndromes can include patients who will eventually evolve to a definitive MPN/MDS overlap syndrome or a rare form of an MPN that is associated with dysplastic changes without documentation of a prior chronic phase.
The mutational landscape of MDS/MPN-U is heterogeneous, with ASXL1 being the most commonly mutated gene followed by TET2 and SRSF2. JAK2 mutations can be seen in up to 30% of patients with MDS/MPN-U, while this mutation is rare in MDS. The presence of a JAK2 mutation in the setting of dysplasia without features of the other MDS/MPN overlap syndromes strongly suggests a diagnosis of MDS/MPN-U. However, cases of MDS with isolated del(5q) may be associated with a JAK2 mutation and are better classified as MDS with isolated del(5q) and concurrent JAKV617F rather than MDS/MPN-U. In comparison to other MDS/MPN overlap syndrome diagnostic categories with strict definitions, the diagnostic criteria for MDS/MPN-U are poorly defined and therefore misclassification is common. Therefore, consultation with an expert hematopathologist is essential for the proper diagnosis of MDS/MPN-U ( Fig. 73.4 ).
JMML is a rare MPN/MDS overlap syndrome that occurs almost exclusively in infants and young children. It is characterized by an aggressive clinical course, with monocytic and granulocytic proliferation, organ infiltration with significant splenomegaly, and activating mutations in the Ras/Erk pathway ( Fig. 73.5 ). Ras signaling proteins are small guanosine triphosphatase (GTPase) molecules that are ubiquitously expressed. They play diverse cellular roles including regulation of proliferation, survival, and differentiation. Ras pathway activation stimulates a number of signaling pathways including Raf/MEK/ERK and PI3K/AKT/mTOR. Mutations in PTPN11, NRAS, KRAS, CBL, or NF1 account for approximately 90% of JMML cases. These canonical Ras mutations are associated with five clinically and biologically distinct subtypes of JMML. Germline mutations in Ras family genes, so-called RASopathies, are a clinically distinct group of genetic syndromes that are described below.
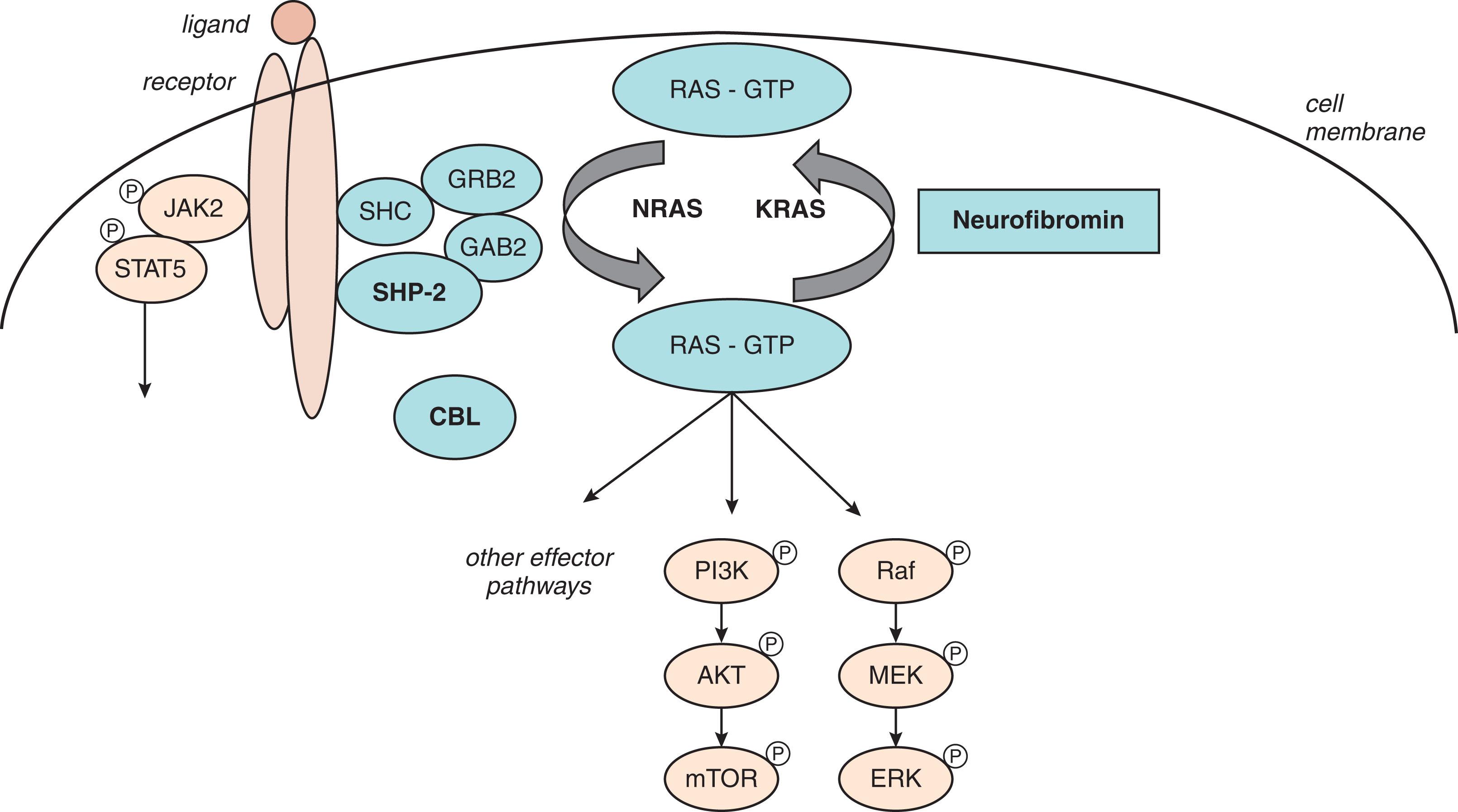
PTPN11 is the most common mutation found in JMML, encompassing approximately 40% of all cases. The PTPN11 gene encodes a cytoplasmic protein tyrosine phosphatase SHP-2, a dual-specificity phosphatase, which contains two tandem SH-2 domains at the N-terminus and a catalytic PTP domain at the C-terminus. All PTPN11 genes are gain-of-function, missense mutations resulting in constitutive Ras activation. PTPN11 -associated JMML is associated with an older age at presentation and a more aggressive course. Noonan syndrome is an autosomal-dominant condition associated with a germline PTPN11 mutation in approximately 50% of patients that causes abnormal development of multiple organs. Most individuals with Noonan syndrome have normal intelligence, but some may have special educational needs or intellectual disability. Noonan syndrome occurs in about 1 in 2500 births and is clinically associated with short stature, facial dysmorphism, neurodevelopmental problems, and lymphedema. Congenital heart disease is present in 80% of patients with Noonan syndrome, most frequently pulmonary stenosis, although atrial septal defects, and hypertrophic cardiomyopathy can also be seen. Cardiac manifestations usually present within the first year of life. Noonan syndrome can be associated with a transient myeloproliferative disorder that is more indolent than JMML but in some cases can progress to JMML (more commonly with certain PTPN11 or KRAS mutations). The PTPN11- associated MPN can be present at birth or develop during infancy or childhood.
NRAS and KRAS mutations are heterozygous somatic RAS mutations that lock RAS into the active guanosine triphosphate-bound state, thereby diminishing GTPase activity. NRAS or KRAS mutations are present in approximately 25% of JMML cases. A small proportion of patients with Noonan syndrome will harbor a mutation in NRAS or KRAS , with the latter associated with more severe intellectual disability. NRAS- mutated JMML has a highly varied clinical presentation and course, with some young infants experiencing an indolent course with spontaneous hematologic improvement being observed during a 2- to 4-year follow-up period. By contrast, KRAS -mutated disease is a particularly aggressive form of JMML.
CBL encodes a member of the Cbl family of proteins, which functions as an E3 ubiquitin ligase. CBL syndrome is an autosomal-dominant developmental disorder resulting from germline missense CBL mutations, which are characterized by impaired growth, developmental delay, cryptorchidism, vasculitis, and a predisposition to develop JMML. CBL mutations account for approximately 15% of cases of JMML. Many individuals with CBL syndrome experience spontaneous regression of their JMML. Importantly, JMML specimens from affected children are characterized by loss of the normal CBL allele through acquired isodisomy.
NF1 , located on chromosome 17q11.2, encodes neurofibromin, a GTP-ase activating protein that acts as a negative modulator of RAS function. Loss of a normal NF1 allele in leukemic cells therefore results in RAS hyperactivity. The syndrome characterized by NF1 mutations, neurofibromatosis 1 (NF1), is an autosomal-dominant disorder clinically characterized by café-au-lait macules, iris hamartomas (Lisch nodules), axillary and inguinal freckling, and neurofibromas. Germline loss-of-function mutations in NF1 are the molecular cause for neurofibromatosis type 1 is a so-called “cancer predisposition syndrome.” Additional tumors, including optic gliomas and soft tissue sarcomas, appear after the first year of life. Although these signs and symptoms are often present, children carrying germline NF1 mutations have a high likelihood of developing JMML, and JMML may, in some cases, actually be the presenting phenotype (without the presence of the aforementioned skin changes) that highlights the importance of NF1 and hyperactive RAS signaling in normal myeloid cell growth and leukemogenesis. NF-1 mutated JMML encompasses approximately 10% of JMML cases and is clinically characterized by higher platelet counts, higher marrow blast percentages, and older age at diagnosis as compared with other JMML subtypes.
It is important to distinguish germline versus somatic mutations given that germline mutations can be associated with MPNs that are transient and may resolve spontaneously. Searching for the characteristic mutations in non-hematopoietic tissues (such as fibroblasts) can determine the germline versus somatic nature of a RAS pathway mutation.
The diagnostic criteria for JMML ( Table 73.4 ) include a combination of clinical, hematologic, and genetic features. Patients must have an elevated peripheral monocyte count over 1 × 10 9 /L, blast numbers in the BM and peripheral blood below 20%, splenomegaly, and the absence of a BCR-ABL1 rearrangement. In addition to these features, a genetic mutation is required (a somatic mutation in PTPN11 , KRAS , or NRAS , a germline mutation in CBL or loss of heterozygosity of CBL , or a diagnosis of neurofibromatosis type 1 or a mutation in NF1 ). In patients without somatic mutations, a combination of other features is needed to establish a diagnosis, such as monosomy 7, or other chromosomal abnormalities in addition to an elevated hemoglobin F level for the patient’s age, myeloid or erythroid precursors observed on the peripheral smear, in vitro hypersensitivity of hematopoietic progenitor cells to granulocyte-macrophage colony-stimulating factor (GM-CSF), or hyperphosphorylation of STAT5.
Children presenting with JMML are often quite ill and present with disease features secondary to marrow and organ infiltration. Common presenting symptoms include fevers, failure to thrive, and splenomegaly. Hepatomegaly may also be present, and lung involvement due to leukemic infiltration manifesting as cough and dyspnea, with abnormal findings on chest X-ray, is possible. Blood tests frequently show leukocytosis with monocytosis, and thrombocytopenia. A small population of blasts may be seen in the peripheral blood.
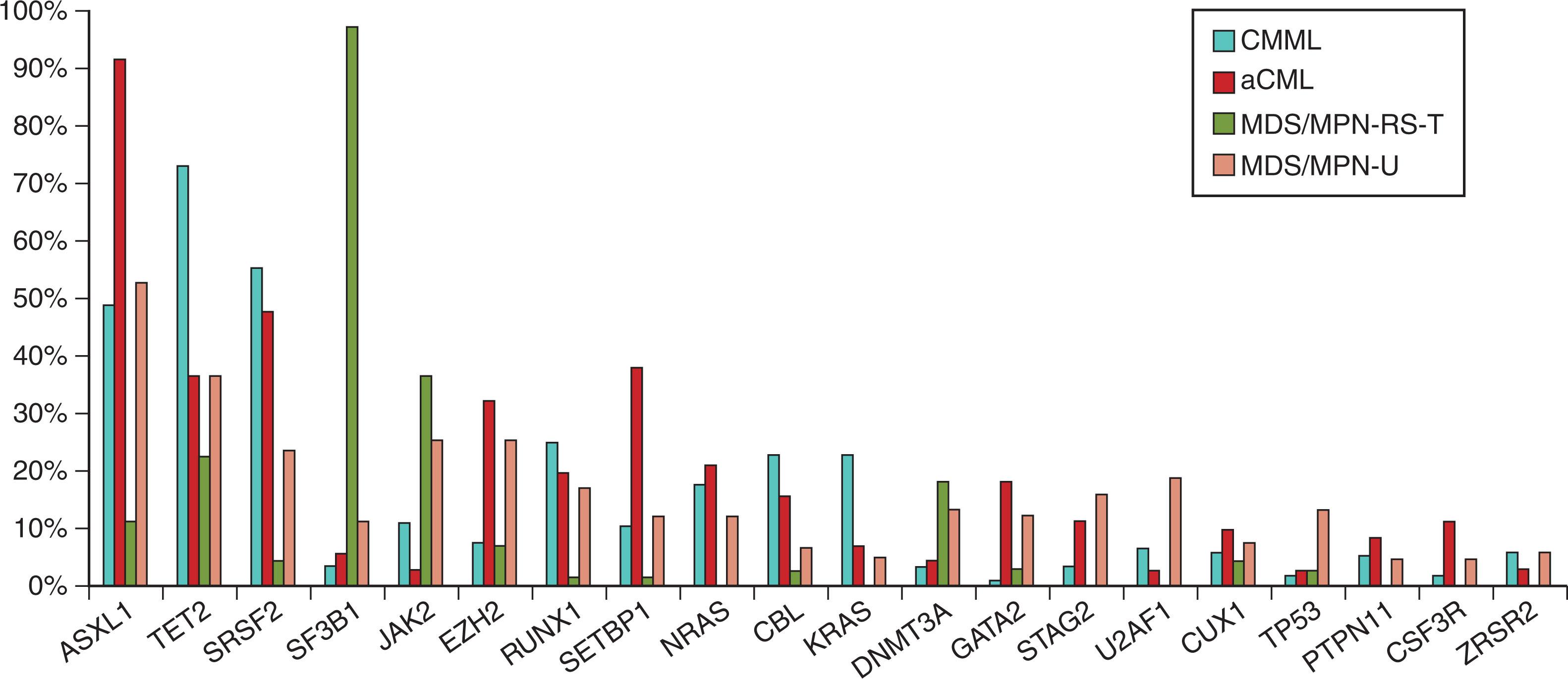
Despite clinical differences, the MDS/MPN overlap syndromes share a multistep pathogenesis, with alterations initially in genes typically associated with MDS, followed by additional mutations characteristic of other gene classes. Commonly mutated genes in MDS/MPNs include those involved in epigenetic regulation, transcriptional regulation, post-transcriptional modulation (in the form of splicesome mutations), and signaling pathways such as JAK-STAT. Notably, mutations in TP53 , while common in MDS and AML, are very rare in these patients, although they can be seen in up to 10% of MDS/MPN-U patients. Fig. 73.6 demonstrates the frequency of specific mutations in adult MDS/MPN overlap syndrome subtypes based on a study of 367 patients where 30 genes were recurrently mutated in ≥3% of patients. Among all MDS/MPN overlap syndrome patients, the most common mutated genes were ASXL1 (51%), TET2 (45%), SRSF2 (35%), SF3B1 (24%), JAK2 (19%), EZH2 (17%), RUNX1 (17%), SETBP1 (15%), NRAS (13%), CBL (13%), and KRAS (10%). Of note, patients with MDS/MPN-RS-T had a lower mean number of variants per patient compared with other MDS/MPN overlap syndrome subtypes. Certain mutations are highly enriched in different subtypes. For example, SF3B1 mutations are found in nearly all patients with MDS/MPN-RS-T and ASXL1 is identified in nearly all patients with aCML.
As described, monocytosis is the hallmark of both CMML and JMML. Interestingly, the two diseases usually reach a similar phenotype via different genetic mechanisms. A pre-disease CMML state is thought to occur when initiating mutations are acquired, most frequently in TET2 , SRSF2 , ASXL1 , or the Ras pathway associated genes. These mutations favor increased monocyte-precursors and monocyte production in the BM. However, the disease only presents itself after an additional second-hit mutation occurs. Mutations in the Ras pathway typify MPN-CMML, a more proliferative subtype of CMML, while additional MDS-associated mutations usually result in dysplasia and MDS-CMML phenotype.
Interestingly, deletion of TET2 in murine models leads to an increased Lin−Sca-1+c-Kit+ (LSK) cell pool. These cells have an increased hematopoietic repopulating capacity and abnormal cell differentiation pattern that skews heavily toward monocytic/granulocytic lineages. These mice develop a myeloid malignancy resembling CMML, suggesting that TET2 functions as a tumor suppressor to regulate hematopoietic stem cell (HSC) differentiation.
JMML is characterized by either germline or somatic mutations in the Ras pathway, as previously described. Germline mutations (i.e., RASopathies) in NF1 ( NF1 germline mutation), Noonan syndrome ( PTPN11, NRAS , or KRAS germline mutations), or CBL syndrome ( CBL1 germline mutation) often have different functional effects. The PTPN11 mutations that are germline have a weaker gain-of-function effect as compared with somatically acquired mutations. Interestingly, some somatically acquired Ras mutations can occur very early in life, even in utero. In a study of patients who developed JMML at ages 2 to 19, sequencing of dried blood from Guthrie cards taken at birth detected acquired mutations in PTPN11 , NRAS , and KRAS , suggesting that the JMML clone originated in utero in these children.
Importantly, mutations in the RASopathy genes ( PTPN11, NRAS, KRAS, CBL, and NF1 ) are mutually exclusive within a given patient, implying that a mutation in any of these genes is sufficient to drive the JMML disease phenotype. In patients who lack one of these five Ras pathway mutations, there may be abnormal Ras signaling in the BM microenvironment that can result in JMML. For instance, a murine model where PTPN11 mutation is present in the BM stroma cells, and not hematopoietic cells, results in a phenotype that is indistinguishable from JMML where PTPN11 is present in hematopoietic cells.
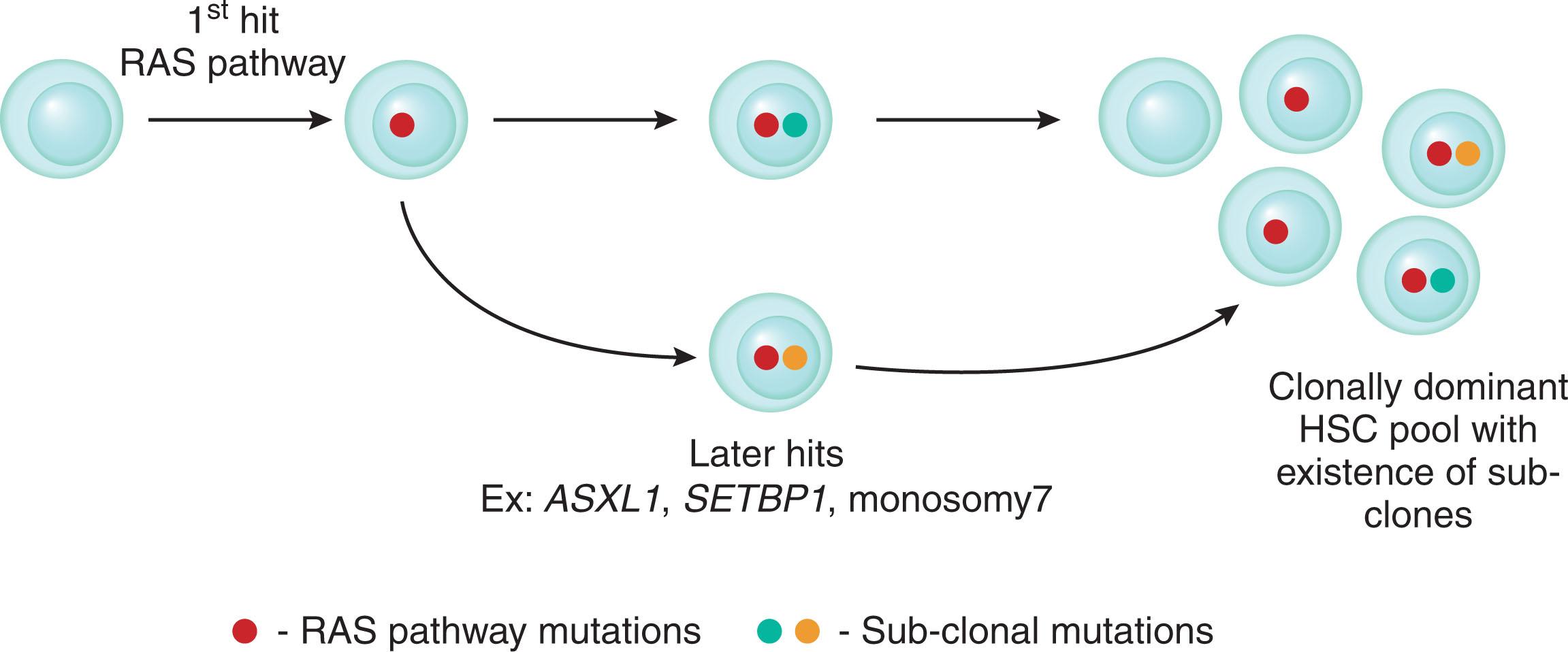
Recently, the origin of the leukemia stem cell (LSC) in JMML has been studied by Louka et al. using primary patient specimens, FACS, and single-cell RNA sequencing revealed that the LSC is markedly heterogeneous in origin. Specifically, the granulocyte monocyte progenitors (GMP, Lin-CD34+CD38-CD90+CD45RA-) and a novel double-positive cell population (Lin-CD34+CD38-CD90+CD45RA+) were aberrantly increased in JMML samples as compared with healthy pediatric controls. Using an in vivo xenotransplantation model, the authors demonstrated that the LSC resided within the JMML HSC compartment, in addition to the GMP and the novel double-positive cell population. Single-cell index sorting of Lin-CD34+ stem cell followed by colony genotyping demonstrated that the JMML HSCs and progenitor cells contain clonally dominant RAS pathway mutations, as well as subclones with additional canonical myeloid mutations such as ASXL1 , SETBP1 , and monosomy 7. They proposed model for LSC clonal evolution in JMML that proposes that “first hit ”occurs within the RAS pathway, followed later by additional hits leading to a phenotypically heterogeneous HSC pool ( Fig. 73.7 ).
MDS/MPN-RS-T is thought to develop in a similar fashion as MDS-RS, with mutations in SF3B1 occurring initially, as 85% to 95% of patients with this disease entity harbor this mutation. A second mutation, most commonly JAK2V617F , is then required to convert the MDS-RS into the MDS/MPN-RS-T phenotype. aCML and MDS/MPN-U do not appear to closely follow the above pathogenic paradigm, with the later disease especially difficult to define due to its heterogeneity. A study to evaluate the genetic signatures of the different MDS/MPN subsets from a large and diverse cohort of 177 adult MDS/MPN overlap syndrome patients revealed similarities between aCML and CMML, with MDS/MPN-RS-T having a distinct transcriptome, and MDS/MPN-U showing features of both disorders. In another large study of 367 adult patients with MDS/MPN overlap syndrome that utilized genome-wide sequencing, the authors were able to reclassify MDS/MPN-U cases into molecular subtypes that resembled other MDS/MPN overlap syndromes. For instance, a co-mutation in SRSF2 with either TET2 or RUNX1 were termed CMML-like and had similar hematologic parameters as pathologically defined CMML (i.e., monocytosis). Similarly, co-mutation of ASXL1 and SETBP1 in MDS/MPN-U patients displayed hematologic features similar to aCML, and the presence of SF3B1 and JAK2 -mutations resembled MDS/MPN-RS-T, with a higher percentage of ringed sideroblasts and an excellent prognosis. Overall, 61% of all MDS/MPN-U cases in this study could be genomically reclassified as another MDS/MPN overlap syndrome subtype even though they did not meet the standard pathologic diagnostic criteria. This study highlights that genomic features can be useful in reclassifying pathologically ambiguous cases of MDS/MPN-U.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here