Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Infections with bacteria of the species Neisseria meningitidis are the most common cause of community-acquired bacterial meningitis in young adults.
The disease may occur in seasonal epidemics and it affects both children and adults worldwide.
Severe flu-like symptoms predominate initially, with nausea, vomiting, headache, severe myalgias, rash, and possibly sepsis, with or without meningeal signs.
50% develop a petechial or purpuric cutaneous eruption with predilection for the trunk and limbs.
Early suspicion and treatment with empiric cephalosporin therapy will decrease mortality.
Meningococcemia, meningococcal sepsis, meningococcal meningitis, cerebrospinal fever, epidemic cerebrospinal meningitis
Meningococcemia presents as a flu-like illness in an otherwise-healthy child or young adult. Initial symptoms may include a petechial or purpuric rash, headache, nausea, vomiting, myalgia, and fever with or without meningeal signs. The gold standard for diagnosis is isolation of N. meningitidis from sterile body fluids, to include cerebrospinal fluid (CSF) or blood. Treatment with appropriate systemic antibiotic agents must be initiated without delay if the diagnosis of systemic meningococcal infection is suspected. Aggressive management of shock and disseminated intravascular coagulation (DIC) in an intensive care unit setting may also be required. This condition carries an approximate mortality rate of 10–15% if treated promptly, but a 90% fatality rate if left untreated. Recognition of cutaneous findings by the health-care provider may help expedite the diagnosis and prevent delay in treatment.
Meningococcal disease was first described in 1805 after a meningitis outbreak occurred in Geneva. However, it was not until 1882 that Anton Weichselbaum isolated the responsible pathogen from the CSF of an infected patient In 1909, immunologically distinct serotypes of the bacteria were identified. The advent of sulfonamides and penicillin and their use for chemoprophylaxis and treatment drastically reduced the incidence of disease and mortality rates during and after World War II.
N. meningitidis exclusively infects humans and causes both endemic and epidemic infection. These attacks occur with seasonal variation, exhibiting the highest frequency in February and March, and the lowest in September. There is no sex predisposition. Large-scale epidemics still occur with devastating consequences in Africa, parts of Asia, countries of the former Soviet Union, and South America, but remain endemic in most western developed countries.
There are at least 13 serogroups of N. meningitidis , determined by polysaccharide capsule composition, with the overwhelming majority of worldwide disease caused by serogroups A, B, C, Y, and W-135. The largest outbreaks, which originated in northern China and spread globally, were caused by strains of serogroup A. This serogroup still accounts for the majority of disease in Asia. In 1987 serogroup A strains spread from China to the Middle East, causing a massive epidemic among pilgrims during the Haj in Mecca. Today, major African epidemics are associated with serogroups A and C; however, the W-135 serogroup caused an epidemic in 2000 and 2001 among pilgrims returning from the Haj in Mecca, Saudi Arabia. In 2002, serogroup W-135 also emerged in Burkina Faso, killing approximately 1500. In most industrialized countries, serogroup B strains have been the predominant infectious strains. In northwestern Europe, periodic hyperendemic infections have occurred in the last three decades. Similar group-B isolates have also emerged in Brazil, the USA, Israel, Australia, New Zealand, China, Thailand, Spain, North Africa, and northern Europe. Most recently, serogroups B, C, and Y are the major causes of meningococcal disease in the United States, divided almost equally among the three strains.
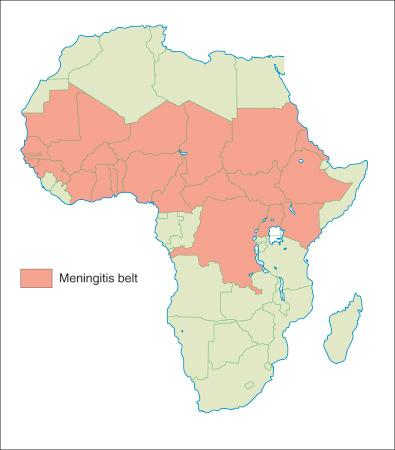
An equatorial region of sub-Saharan Africa, referred to as the meningitis belt, displays a high burden of invasive meningococcal disease. The countries of Burkina Faso, Ghana, Togo, Benin, Niger, Nigeria, Chad, Cameroon, Central African Republic, The Sudan, Ethiopia, Mali, Guinea, Senegal, and Gambia comprise this region ( Fig. 20-1 ). Outbreaks due to several strains, but especially serogroups A and C, occur annually in this hyperendemic region, causing predictable, devastating disease. In 2015, 8500 suspected cases of N. meningitis serogroup C swept across Niger, resulting in over 570 reported deaths.
Epidemic spread of meningococcal disease is not entirely understood, but it appears that the occurrence of invasive meningococcal disease is determined by the virulence of certain bacterial strains, the susceptibility of the population, and certain risk factors ( Box 20-1 ) that enhance transmission. The most important of these risk factors is nasooropharyngeal carriage. 8 Personal risk factors that further increase the likelihood of disease include: preceding viral or bacterial infections, chronic underlying disease, terminal complement deficiency, asplenia, active or passive smoking, and household crowding. The annual incidence of meningococcal disease in the United States has declined drastically over the last several decades from 1.1 to 0.4 cases per 100,000 population between 1996 and 2011.
Nasooropharyngeal carriage
Host risk factors
Smoking
Preceding respiratory tract infection
Neisseria meningitidis , a fastidious bacterium, is a Gram-negative kidney-bean-shaped diplococcal obligate aerobe. Of particular importance are virulence factors ( Box 20-2 and Fig. 20-2 ), which allow the organism to invade tissue and evade immune responses. Phase switching and antigenic variation of these virulence factors further magnify the pathogenicity of these organisms.
Opacity (adhesion) proteins
Antigenic variation
Immunoglobulin A protease
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here