Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The term measles is applied most appropriately to “rubeola” (also known as “morbilli”), an extremely contagious, prolonged respiratory and systemic viral illness characterized by high fever, an erythematous maculopapular rash, cough, coryza, and conjunctivitis. A member of the family Paramyxoviridae, within the genus Morbillivirus , the measles virus exhibits at least 24 distinct genotypes but behaves as a single serotype because natural infection or adequate immunization confers broad protection against all types. ,
Measles may be the most contagious pathogen known to humans, and it has no other reservoir. Its basic reproduction number, the R 0 , which represents the number of naïve contacts potentially infected by exposure to one contagious individual, has been estimated to be 12 to 18, an R 0 far higher than the R 0 of two to three for Ebola ( Chapter 351 ) and influenza ( Chapter 332 ). Key challenges to control of the spread of measles include the immunization status of a population, their density, and the influence of migration and travel.
In the 1950s and early 1960s, measles was ubiquitous in the United States, with more than 500,000 cases reported annually, and nearly everyone was infected by adulthood. By 2000, a two-dose, live-attenuated vaccine led to endemic measles being declared “eliminated” from the United States. From 2001 to 2016, a median of 28 imported measles case were reported annually in the United States. More recently, measles cases in the United States increased dramatically, peaking in 2019 with 1282 confirmed cases, mostly in regional outbreaks in close-knit and underimmunized communities exposed to measles brought back by international travelers. , However, the number of confirmed cases dropped dramatically to just 13 in 2020, an unexpected benefit of the restriction of social interactions and use of masks in response to the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) coronavirus disease 2019 (COVID-19) pandemic ( Chapters 335 , 336 , and 337 ). This benefit may be short-lived because the pandemic’s disruptions also resulted in reductions in routine immunizations by 22% in children ages 12 to 23 months and 63% in children aged 2 to 8 years. Though rates rose quickly after the lifting of the most severe SARS-CoV-2 restrictions, data so far have not shown an effective “catch up” of the deficits encountered. Nevertheless, outbreaks can still occur, such as 85 locally acquired cases in Central Ohio in 2022, perhaps triggered by unvaccinated local residents who had recently returned from East Africa where measles outbreaks were ongoing.
The global incidence of measles decreased from an estimated 145 cases per million population in 2000 to just 18 per million in 2016, subsequently rose to 120 per million in 2019, then declined again to 17 per million in 2021 during the height of the COVID-19 pandemic. Annual global measles deaths decreased from over 500,000 in 2000 to 109,000 in 2017, rose to over 200,000 in 2019, and were 128,000 in 2021. The Americas are no longer free from endemic measles transmission, and the ambitious goal of eliminating measles in 5 of 6 world regions by 2020 has not been met.
Global protection by at least one measles immunization flattened after 2010, at a rate of about 86% until it fell to 81% in 2021, though the numbers of individuals receiving the required two immunizations rose from 48% in 2012 to 71% in 2019. In 2021, 47% of all WHO member countries achieved the goal of greater than 90% first-dose coverage, but an estimated 24.7 million children did not receive even an initial measles vaccine. About 60% of the missed initial doses occurred in just 10 countries, including Nigeria, India, Democratic Republic of the Congo, Ethiopia, Indonesia, Pakistan, Philippines, Angola, Brazil, and Tanzania.
Measles also can be associated with long-term complications. For example, the country of Georgia has suffered from endemic measles for years, with widespread outbreaks in 2004-2005 and 2013-2015. Between 2008 and 2017, 16 confirmed survivors developed devastating subacute sclerosing panencephalitis, 92% of whom were children infected prior to 2 years of age, at a mean interval of 6 years after the infection. Other than COVID-19, measles is the fourth leading worldwide cause of death due to infectious disease, following only tuberculosis ( Chapter 299 ), HIV/AIDS ( Chapter 353 ), and malaria ( Chapter 316 ).
Measles virus is spread very effectively by contact with large droplets or inhalation of aerosolized small droplets, which can remain suspended for hours after an infected individual has exited. Unlike other paramyxoviruses, which primarily infect and inflame respiratory epithelia, measles virus targets the immune system. Measles virus initially infects cells in the respiratory tract or conjunctiva, then migrates to regional lymph nodes where it infects CD150/SLAM1-expressing T and B cells. Infected mononuclear cells then travel to other lymphoid tissues, including the spleen, tonsils, and other lymph nodes to propagate infection. Virus may enter the central nervous system (CNS) through infected lymphocytes, although the exact mechanism remains unclear. Systemic infection of the gastrointestinal (GI) system, kidney, liver, and skin occurs by circulation of infected immune cells. Measles virus later infects the respiratory epithelium through the Nectin-4 receptor, spreads from cell to cell to cause damage and sloughing, and then is expelled as infectious droplets by coughing to repeat the cycle.
Measles virus initially evades immune recognition by inhibiting type 1 interferons, so extensive viral replication and systemic spread precedes clinical disease. The buccal mucosal lesions (Koplik spots) and the classic rash result from infection of capillary endothelium by measles virus, with syncytial cell formation as well as local production of nitrous oxide and tumor necrosis factor-α. Measles virus–specific T-cell host-immune responses lead to eventual necrosis of infected cells with viral clearance. Poor measles virus–specific T-cell responses are observed in individuals who are severely malnourished and immunocompromised, with reduced cellular necrosis and delayed viral clearance, thereby explaining their less obvious but prolonged skin changes.
The most dramatic aspect of measles virus infection may be its transient yet profound suppression of the immune system, possibly by targeting CD150(SLAM)-expressing cells. While inducing a cytotoxic T-cell response that clears infected cells and induces measles virus–specific memory B cells, measles virus concurrently depletes subsets of both regional and circulating T and B cells, with a loss of immune memory. The depletion of lymphocytes in gut and respiratory epithelium leads to increased morbidity and mortality from other infections and explains the increase in all-cause mortality for an after-measles infection.
CNS involvement includes several clinicopathologic syndromes. The mechanism of viral entry into the CNS is poorly understood, because human neurons do not usually express the most common receptors for measles virus (SLAM or Nectin-4), though entry may be facilitated by viral binding to Nectin-4–positive nasal or meningeal epithelial cells adjacent to neuronal tissue. Infection can cause an acute measles inclusion body encephalitis that is believed to be caused by persistent viral replication in the brain tissue of patients who are unable to mount an adequate immune response because of deficient cellular immunity. The name derives from histologic evidence of viral persistence, which is seen by intracytoplasmic and intranuclear inclusions caused by nucleocapsids.
CNS infections can also trigger an immune response. In acute demyelinating encephalitis, an aberrant host response to myelin basic protein is thought to lead to brain inflammation and perivenous demyelination. In subacute sclerosing panencephalitis, patients are infected with mutated forms of the measles virus. Mutations in the M (matrix) gene may allow for long-term escape from host neutralizing antibody, and an altered F (fusion) gene leads to expression of an F protein with “hyperfusogenicity,” which in turn allows for highly efficient cell-to-cell spread in neuronal tissues. Intranuclear inclusion bodies are seen in the neurons and glial cells of the white matter and neurons of the brainstem. An exaggerated host mononuclear inflammatory response occurs, with a vigorous leptomeningeal and parenchymal perivascular inflammation, which may be compounded by a degree of autoimmunity due to cross-reactivity between viral and myelin antigens. Early demyelination is followed by late stage cortical atrophy.
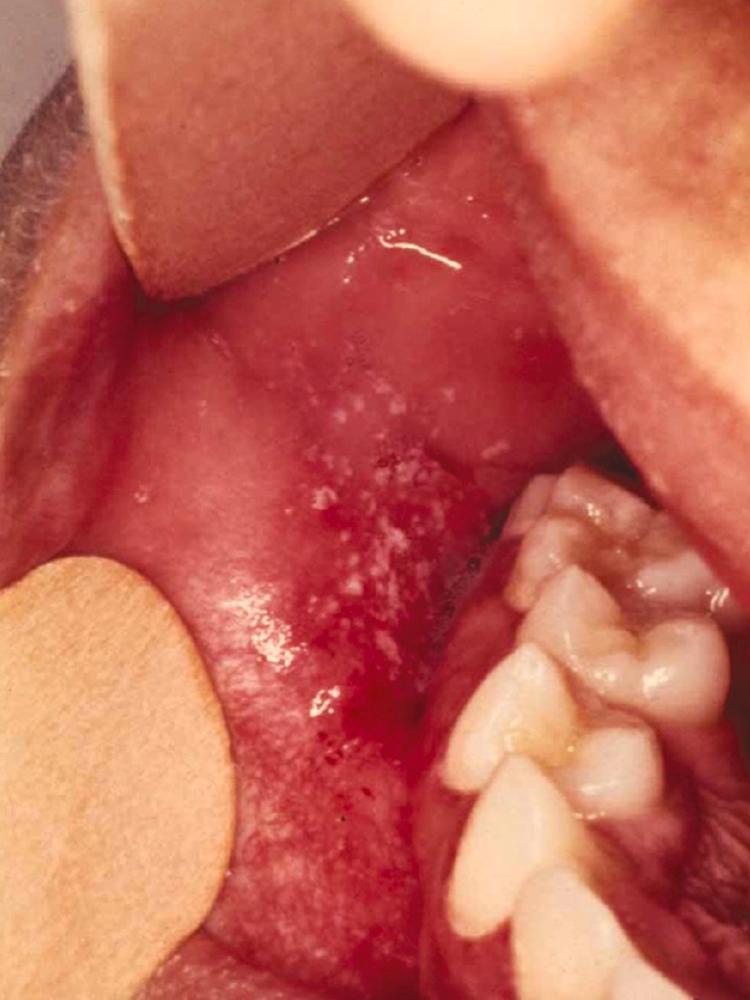
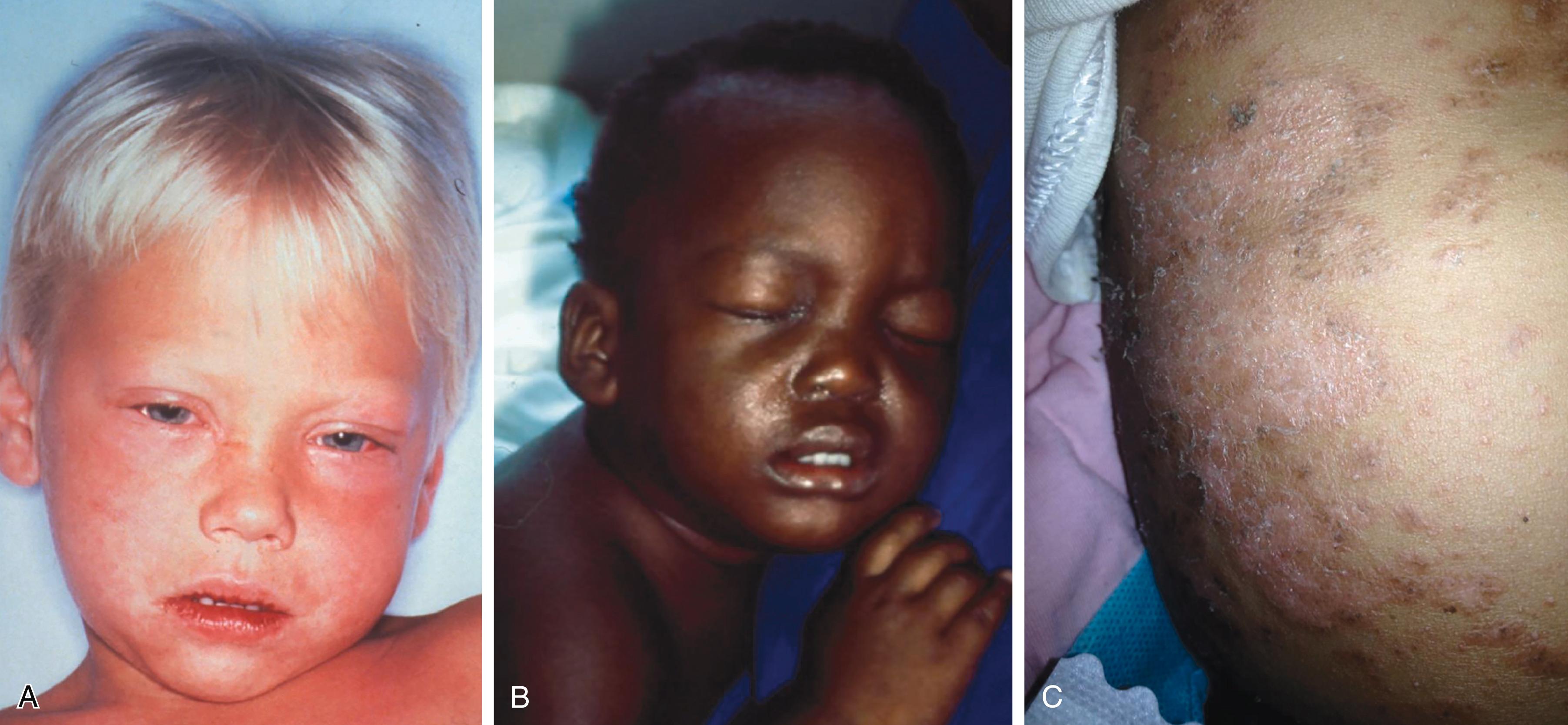
Measles should be suspected in the presence of fever and a maculopapular (nonvesicular) rash. Measles has a 7- to 21-day incubation period, with onset of fever and malaise (prodrome), as well as the more specific combination of a “croupy or brassy” cough, coryza, conjunctivitis, and photophobia beginning about 10 days after exposure ( Fig. 338-1 ). Koplik spots ( Fig. 338-2 ), which are raised bluish-white papules inside both cheeks, may appear at this time; though pathognomonic, they are often missed or misdiagnosed as thrush, and they fade as the rash emerges. The classic erythematous blanching maculopapular rash begins on the face, spreads down the body, and becomes confluent and darker in color over days ( Fig. 338-3A ). The rash is more subtle in dark-skinned patients ( Fig. 338-3B ), in whom the diagnosis may be delayed. In malnourished and immunosuppressed individuals, a prolonged desquamating dermatitis is commonly seen ( Fig. 338-3C ). Body temperature is high, 39° to 40.5° C, beginning with the prodrome and continuing at least 4 days into the rash. Patients are contagious to others from 4 days prior until 4 days after onset of the rash. As the rash darkens and fades, the skin will often flake and peel. In individuals with incomplete postimmunization protection, clinical symptoms may be milder, with less fever and catarrh, a later onset (12 to 16 days), and markedly reduced infectivity.

Approximately one fourth of the reported measles cases identified in the United States from 2002 to 2016 resulted in hospitalization. Of these 1018 patients, reported complications included dehydration (16%), hyponatremia (14%), diarrhea (10%), thrombocytopenia (10%), pancytopenia (3%), conjunctivitis (9%), septicemia (8%), bacterial pneumonia (8%), bronchitis (4%), pleurisy (4%), and viral pneumonia (1%). One of the most specific though rare complications was encephalitis (2%). By comparison, complications seen in 307 children admitted to a children’s hospital in Islamabad, Pakistan from 2013 through 2017 included pneumonia (63%) and encephalitis (9%).
Involvement of the CNS leads to several of the most serious complications of measles infection, with high rates of morbidity and mortality.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here