Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
For the purpose of this chapter, the term vascular lesion or vascular anomaly is used to define a variety of cutaneous neoplasms and malformations originating from blood vessels and related structures. Owing to major differences in prognosis and treatment strategies, it is imperative to accurately differentiate between these various abnormalities. Vascular anomalies can be divided into two main groups: vascular tumors and vascular malformations. Overall, vascular anomalies affect approximately 8% to 10% of births worldwide, with nearly 400,000 new cases per year in the United States alone.
Infantile hemangiomas, the most common vascular anomaly, are also the most common benign tumor occurring in infancy and childhood and are present in about 2% of neonates. They are true tumors, exhibiting the features of all neoplasms such as increased mitosis and hyperplasia. Although up to 30% of these tumors may be present at birth, the majority become apparent in the first weeks of life. Infantile hemangiomas have a unique and characteristic natural history. , During the first few months of life, they increase in size by proliferation (hyperplasia) and involve skin, mucosa, and subcutaneous tissues to variable degrees. Proliferation typically ends within the first 4 to 6 months and is followed by a quiescent plateau before regression begins. Rarely, growth may continue for 12 to 14 months after birth ( Fig. 28.1 ).
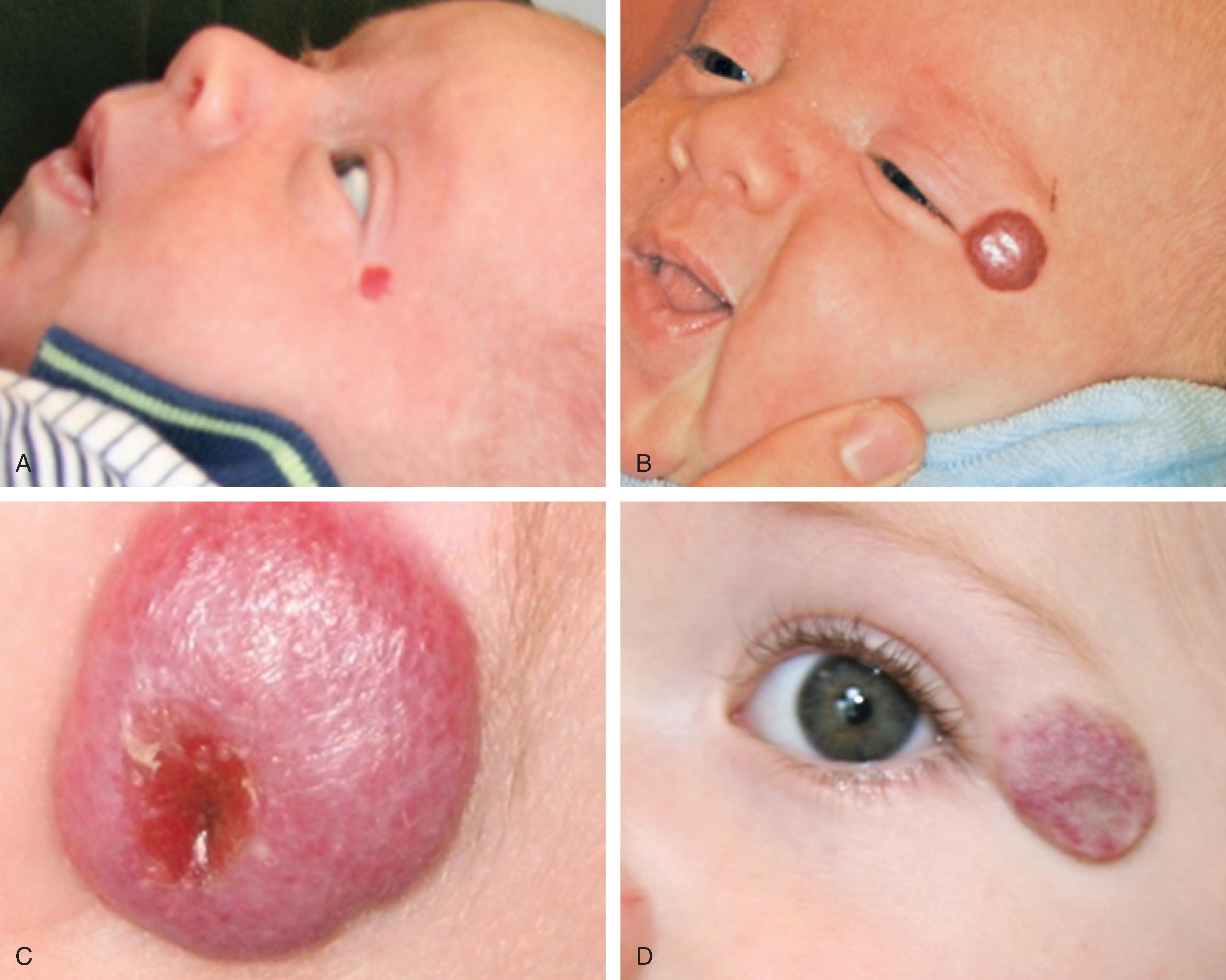
The end of the proliferative phase marks the beginning of the involutional phase. During this phase, which may last for years, the hemangioma undergoes varying amounts of regression in size and replacement with fibrofatty tissue. The majority of hemangiomas will not require treatment because there are so many small, favorably located lesions. Nevertheless, approximately 30% to 40% of infantile hemangiomas will not involute to a cosmetically and functionally acceptable point. Necessary intervention is sought by the remaining majority of these patients. Cutaneous hemangiomas may involve the upper dermis (superficial), the deeper dermis and/or subcutaneous tissue (deep), or both full-thickness skin and subcutaneous tissue (compound). They are most commonly focal, well-defined growths or may be segmental, involving diffuse segments of skin. There are sites of predilection on the face (midface, nose, lips) for the development of hemangiomas.
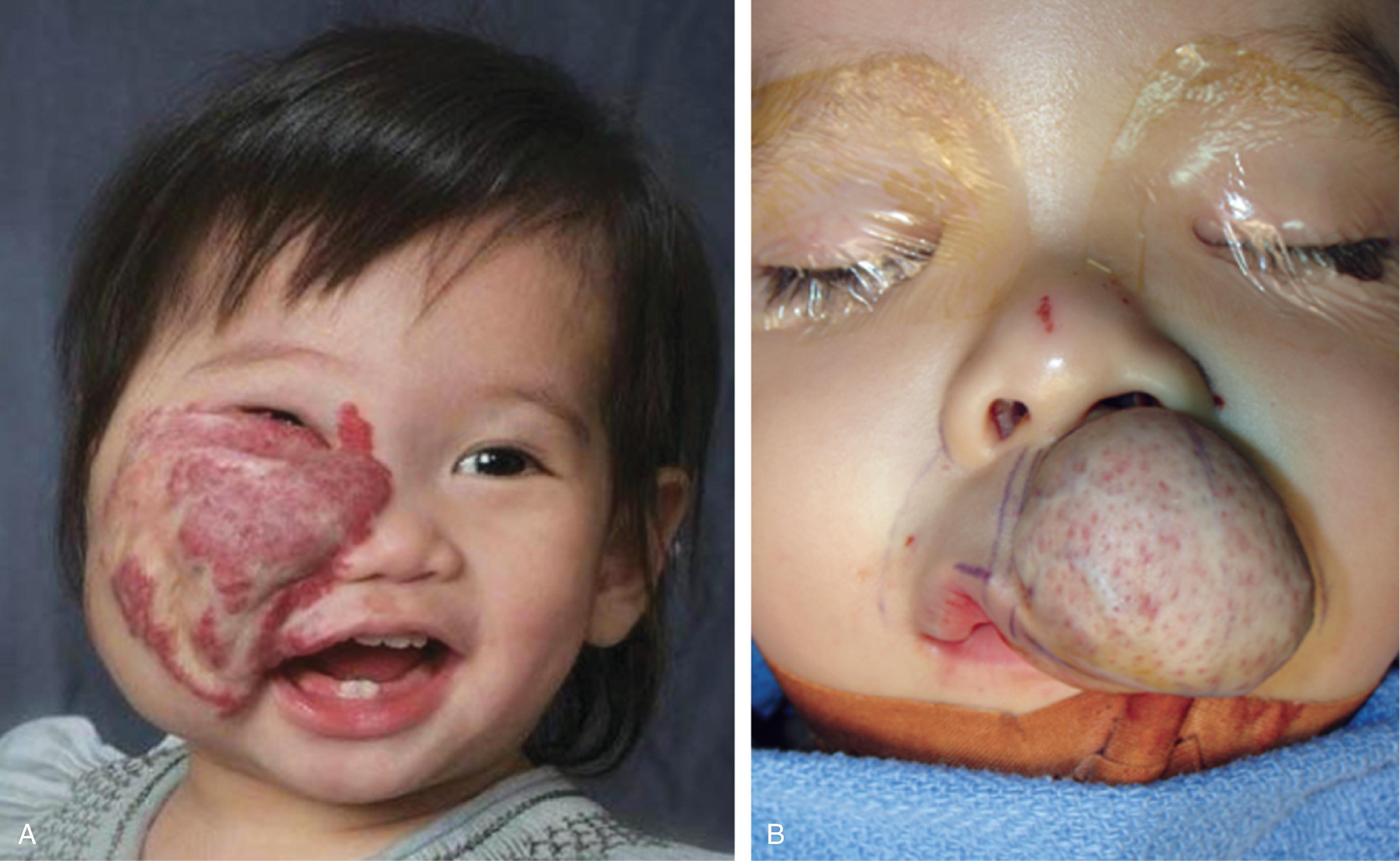
After the involution of the hemangiomas is complete, superficial hemangiomas leave atrophic redundant skin manifesting variable degrees of telangiectasia, whereas deep hemangiomas leave a residual mass of fibrofatty tissue. Involuting compound hemangiomas show varying degrees of all these features ( Fig. 28.2 ).
Accurate diagnosis and treatment planning of infantile hemangiomas are entirely dependent on clinical history and physical examination. Imaging studies are rarely required and are of limited importance in general management. Treatment of infantile hemangiomas is dependent on multiple factors including the degree of skin involvement, the stage of its natural history, the size and anatomic location of the tumor, and the age of the child. Congenital hemangiomas are completely formed and present at birth and are very different from infantile hemangiomas in their natural history and prognosis. Rapidly involuting congenital hemangiomas appear fully formed at birth, do not proliferate, and may regress within the first year of life; those that do not regress are called non–rapidly involuting hemangiomas . There are other, rarer vascular tumors that are beyond the scope of this chapter. Terms such as strawberry or capillary angioma and cavernous hemangioma are of historic and folkloric interest and should not be used in discussing these abnormalities.
More than 60% of infantile hemangiomas occur on the head and neck, predominantly in white children and somewhat less commonly in darker skin types. For unclear reasons, females are more likely to be affected than males in a 3 to 5: 1 ratio. Infantile hemangiomas are more common in premature infants, the incidence in which correlates with decreasing gestational age and birth weight. ,
A plethora of new knowledge is tantalizing regarding the possible etiology of hemangiomas, but to date none of the information has proven causative. Many of the characteristic molecular markers of infantile hemangioma blood vessels are expressed uniquely by normal fetal micro vessels. In particular, the glucose transporter 1 (GLUT-1) protein is almost exclusively found in infantile hemangiomas and placenta tissue but not in other vascular anomalies or other normal tissues. There is also a striking similarity between infantile hemangioma mRNA transcriptome and that found in the placenta. An early unproven hypothesis explaining this relationship is that fetal placental or precursor cells metastasize from the placenta to the fetus and become implanted in areas of high blood flow, such as the head and neck. A more recent unproven theory is that a mutation in the vascular endothelial growth factor (VEGF) receptor leads to increased levels of circulating VEGF and an increase in hypoxia inducible factor 1 alpha (HIFA). Little is known about the mechanism of the process; however, upregulation at these sites of HIFA-responsive chemokines, such as VEGF, is known to promote the recruitment and proliferation of endothelial progenitor cells and may be an etiologic factor in developing hemangiomas. ,
In contrast to infantile hemangiomas, vascular malformations ( Fig. 28.3 ) are always present at birth, although they may not be apparent. They enlarge by hypertrophy, never proliferate, and never involute. They are true developmental anomalies, not neoplasms. Their rate of hypertrophy and hence their functional and cosmetic significance are extremely variable. Vascular malformations may originate from capillaries, veins, venules, lymphatics, arterioles, or any combination of these structures. Vascular malformations may involve skin, subcutaneous tissues, and mucosa and thus may be superficial, deep, compound, focal, or diffuse.
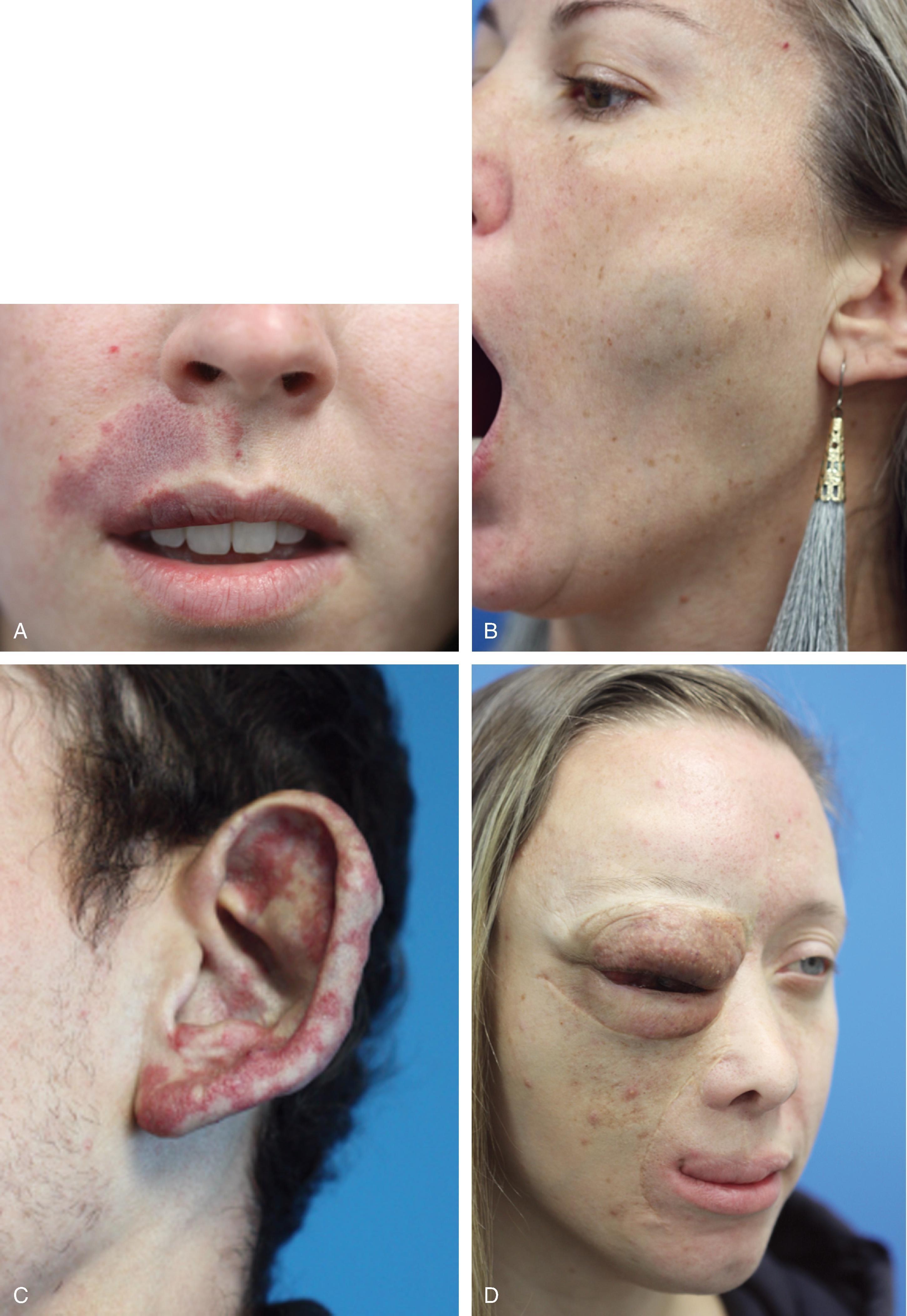
Capillary malformations of infancy (nevus flammeus) are superficial, pink macules folklorically known as a salmon patch , angel’s kiss , or stork bite . They most commonly involve the midline of the nape of the neck or the forehead. Although they are classified as vascular malformations, these lesions typically fade with advancing age and are of passing significance.
Current data support that capillary malformations (port-wine stains) should be patho-anatomically defined as resulting from differentiation-impaired endothelial cells with a progressive dilatation of immature venule-like structures. Dysregulation of vascular mitogen-activated protein kinase (MAPK) and/or phosphoinositide 3-kinase (PI3K) signaling during human embryonic development plays a part in the pathogenesis and progression of these lesions. Sporadic low-frequency somatic mutations, such as GNAQ , contribute to the development of these lesions and are proving to be present in lesions previously incorrectly characterized as tumors. Although they may manifest as flat pink macules at the beginning of life, they usually darken and thicken with advancing age, forming a cobblestone appearance as the dermal vessels continue to dilate under constant hydrostatic pressure. Their distribution patterns often correspond to dermatomes, and the presumed etiology is deficient or ineffective postcapillary venule innervation. Although this is the presumed mechanism, no definitive evidence exists to prove it.
Venous malformations are composed of ectatic veins. They commonly involve the lips, tongue, floor of mouth, and buccal space. Patients suffering from venous malformations frequently complain of swelling with dependency, pain, limitations in the function of the affected region, and cosmetic deformity. Superficial venous malformations are visible as purple masses. Deeper lesions are manifested as bluish or colorless subcutaneous masses.
Arteriovenous malformations are rare vascular lesions originating from arteriovenous channels that failed to regress during fetal development. A palpable mass with an obvious well-developed arterial supply and dilated tortuous veins are typical features. A murmur may be heard or a thrill may be palpated over the mass. Malformations must be differentiated from arteriovenous fistulas, which are usually precipitated by trauma.
Lymphatic malformations are dilated lymphatic channels arising from congenital blockage or arrest of the normal development of the primordial lymphatic plexus. Although they grow at a slow and steady rate, a sudden increase in size may be seen with infection, trauma, or hormonal changes. More than 80% of lymphatic malformations of the head and neck are located in the cervical region. They may also involve the oral cavity, orbit, supraclavicular area, and parotid gland. Some lymphatic malformations tend to be well defined, with macrocystic features (>2 cm 3 ). Others tend to be interstitial, infiltrating, and poorly defined microcystic lesions (<2 cm 3 ). Still others demonstrate both macrocystic and microcystic features. Most lymphatic malformations are diagnosed in infancy; 90% are clinically apparent by 2 years of age. Magnetic resonance imaging (MRI) is the tool of choice to confirm the diagnosis of lymphatic malformations.
In contrast to dealing with infantile hemangiomas, imaging studies are important in assisting with the establishment of an accurate diagnosis and treatment planning for vascular malformations. MRI, angiography, and ultrasonography are important diagnostic tools, and the particulars of differentiating the different types of vascular malformations by radiographic studies have been well described.
There is no accepted consensus on the treatment of infantile hemangiomas, although the prevailing trend is to intervene rather than to follow the old dictum of benign neglect (“leave it alone, it will go away”). This is supported by the fact that a significant number of hemangiomas incompletely involute regardless of how long patients are willing to wait. Incomplete involution often results in a cosmetic deformity necessitating treatment because of the high social impact of facial differences. In addition, the psychology literature has well-documented the impact of physical appearance on the development of self-image in children between the ages of 2 and 5 years. , Therefore early treatment of hemangiomas is justified. There are numerous treatment options available. The goal of treatment is to restore to the child as much normal appearance and function as possible. This ideally should be accomplished by the age of 3 years and no later than elementary school age. Treatment of hemangiomas depends on the anatomic location and functional and cosmetic impairment the neoplasm is causing. The depth of tissue involvement; the presence of complicating factors (ulceration, visual axis impingement); and the degree of proliferation or involution also influence the approach to treatment.
An important decision to make in treatment of hemangiomas is whether to intervene and what treatment modality to use. This decision is best made by asking: Can the surgeon obtain a result with available treatment modalities that is as effective as allowing the hemangioma to follow its natural course? If the answer is yes, intervention is justified. If the answer is no, then observation is continued until a predetermined point of re-evaluation. Serial observation is an active treatment option and is very different from telling parents of children afflicted with hemangiomas to wait an indeterminate number of years for the hemangioma to “go away.” In addition to observation, during the proliferative period, treatment may include medical therapy, laser treatment, surgical excision, or a combination of these treatments.
Superficial cutaneous hemangiomas may be treated with a yellow light laser, such as the flashlamp-pumped pulsed dye laser (PDL; 595-nm wavelength). The achievable goal is complete resolution of superficial lesions thinner than 1.5 mm. With use of the PDL, lightening and retardation of growth may be accomplished with thicker lesions in preparation for other treatment modalities. If the lesion is compound, the superficial component may be treated with the laser first in preparation for surgical excision of the deep component. The PDL is very useful in assisting the healing of ulcerating hemangiomas and preventing further scarring from tissue destruction while waiting for other interventions.
Rapidly proliferating hemangiomas that pose a serious functional or cosmetic concern may be treated with medical therapy. Before 2008, corticosteroids were the first line of medical treatment. Since then, propranolol has become the first drug of choice. , Like corticosteroids, propranolol seems to be most useful during the proliferative phase. It may also be useful during involution as well. Whereas steroids exerted their effect through blunting of neo-vasculogenesis, propranolol seems to have an effect by vasoconstriction, inhibiting vasculogenesis and promoting apoptosis. Continued experience with the drug is defining the exact role for propranolol in the treatment algorithm.
Steroid injections of infantile hemangiomas are useful for small, well-defined, deep hemangiomas when the treatment might be definitive. Select cases of hemangioma involving the parotid gland, eyelid, and mid cheek are treated by direct injection of a mixture of triamcinolone (40 mg/mL) and betamethasone (6 mg/mL). Auricular and nasal tip hemangiomas are not injected with steroids because it is associated with weakening of the cartilage in the area of the injection. In addition, the deposited steroid causes a white plaque to be visible through the thin skin covering these structures. The goal of depot steroid injections is to slow proliferation of the deep component of the hemangioma. Great care must be taken with use of the PDL to treat focal or diffuse compound hemangiomas that have been injected with steroids because there is a higher risk for skin ulceration. This is particularly true in the malar area. Hemangiomas that are life threatening or do not respond to propranolol or steroid therapy can be treated with other medications.
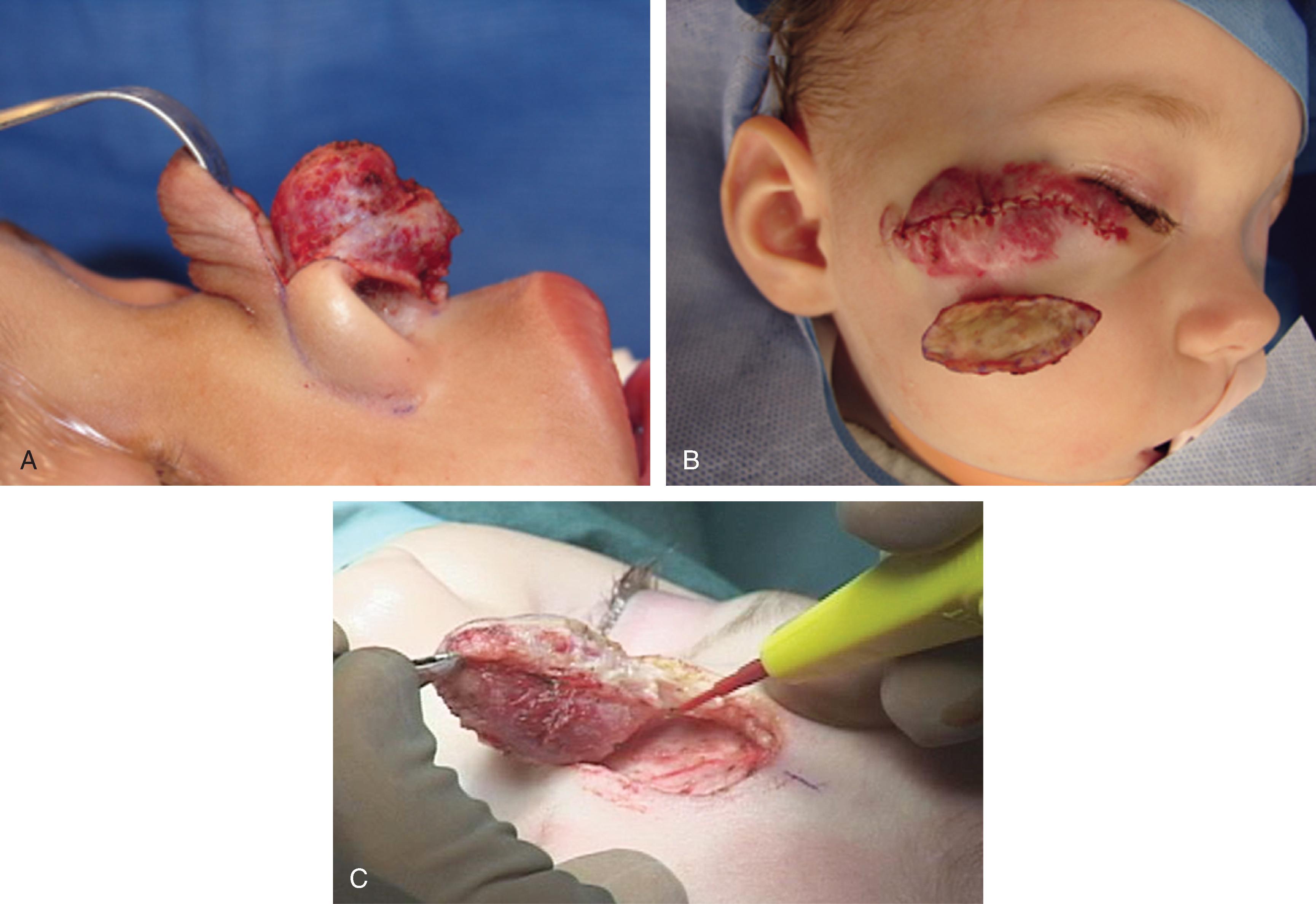
Surgical management is integral to the overall treatment algorithm of infantile hemangioma. , Historical misgivings and misconceptions about the operability of hemangiomas have been supplanted by experience and a better understanding of these tumors. Surgical planes exist between hemangiomas and surrounding structures and can be created between the superficial and deep components or within the deep component of hemangiomas ( Fig. 28.4 ). Hemangiomas are solid tumors with few isolated feeding vessels. Therefore meticulous technique and the routine use of unipolar and bipolar microneedle electrocautery devices make dissection virtually bloodless. Conservatism is critical in resecting facial tissue involved with hemangiomas in children. The goal is to resect sufficient tissue and still be able to achieve primary closure of the wound. When this is not possible because of the size or location of the tumor, serial excision has proved helpful as with other benign tumors. The senior author avoids the use of flaps and grafts in small children except in the most severe, complicated cases in which additional scars resulting from the use of sophisticated techniques are justified. To preserve contour or set the stage for an additional resection, subtotal excision of the deep component of a hemangioma is commonly performed. To minimize the potential psychological sequelae on a child afflicted with a hemangioma, every effort is made to obtain the best possible functional and cosmetic result before the child reaches school age. The author’s threshold for excision of nasal tip and periorbital hemangiomas is lower than for other sites because of the obvious potential for severe functional and cosmetic impairment. Recently, the early treatment of nasal hemangiomas has been supported by the best available evidence. Treatment is geared toward obtaining the best possible result by the time of the social and developmental milestones previously described. The superficial component of compound hemangiomas is treated with photocoagulation if it cannot be entirely resected together with the deep portion ( Fig. 28.5 ).
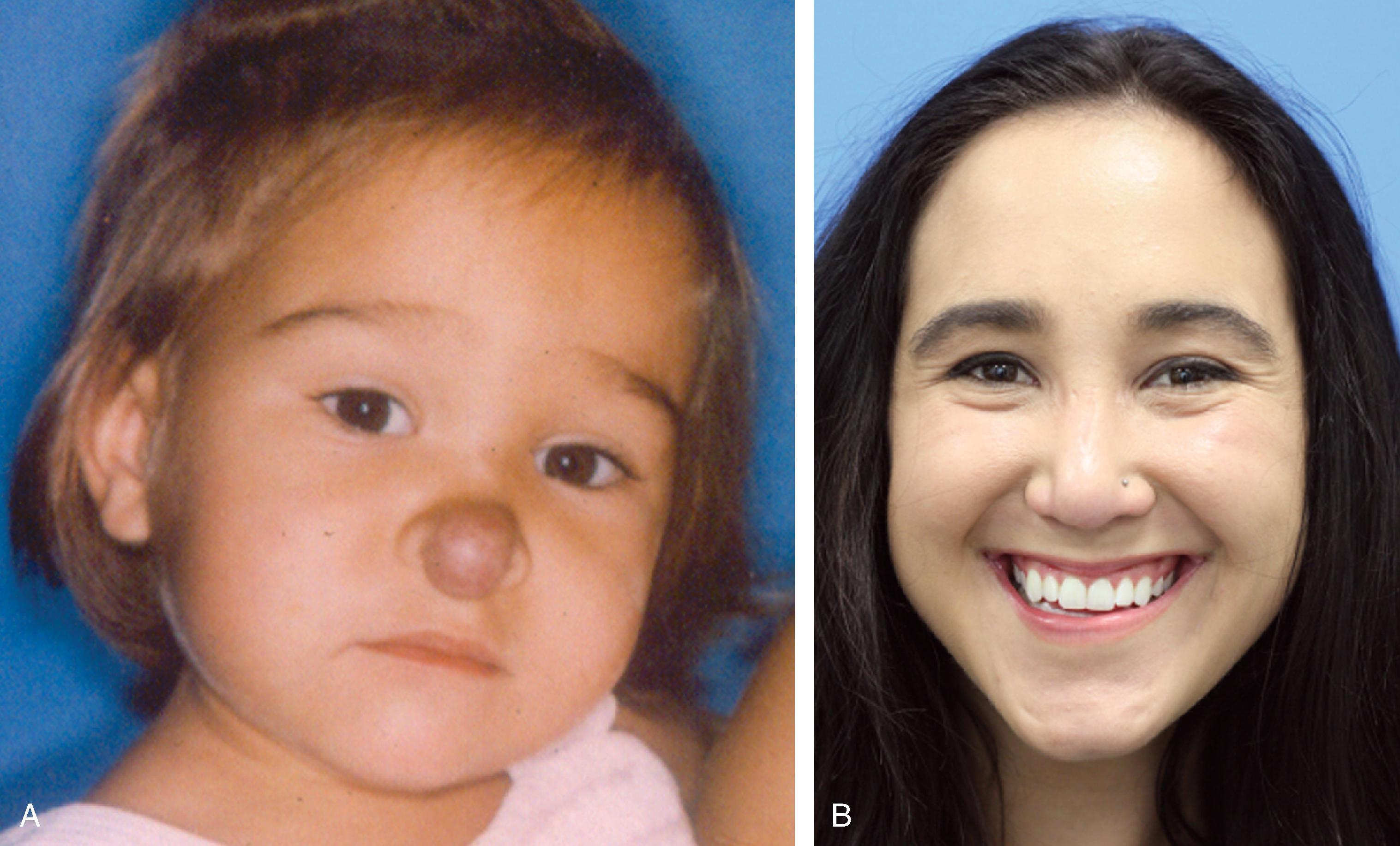
When a hemangioma completes involution, the surgeon’s focus is directed toward treatment of residual atrophic tissue, telangiectasias, and residual excess subcutaneous fibrofatty tissue. Atrophic tissue may be treated with carbon dioxide or erbium laser skin resurfacing. Telangiectasias respond well to photocoagulation with the PDL. Residual subcutaneous fibrofatty tissue is excised and sculpted to obtain a natural contour. This can be done through traditional approaches (direct excision) or remotely through minimal incisions under endoscopic visualization and ultrasonic aspiration ( Fig. 28.6 ).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here