Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Primary hepatic malignancies consist of a diverse spectrum of tumors that arise from an equally diverse population of cells that constitute this complex organ. In addition to hepatocytes, the liver is made of cholangiocytes, neuroendocrine cells, hepatic progenitors, myofibroblastic mesenchymal cells, and vascular endothelial cells. Hepatocellular carcinoma (HCC) represents the vast majority (80% to 90%) of primary liver cancers. All other primary liver neoplasms, including cholangiocarcinoma, account for 10% to 20%. Table 133.1 outlines the primary hepatic malignancies with their corresponding cellular progenitors. This chapter focuses on nonhepatocellular primary malignancies of the liver. While less common than HCC, these tumor types can be aggressive and are being diagnosed with increasing frequency. The clinician should have a better understanding in the recognition and management of these malignancies.
| Cellular Phenotype | Primary Hepatic Tumor |
|---|---|
| Epithelial | |
| Hepatocellular | Hepatocellular carcinoma |
| Hepatic progenitor | Hepatoblastoma |
| Cholangiocellular | Intrahepatic cholangiocarcinoma |
| Hepatic cystadenocarcinoma | |
| Mixed | Mixed cholangiohepatocellular carcinoma |
| Other | Primary squamous cell carcinoma |
| Mesenchymal | |
| Muscular | Leiomyosarcoma |
| Rhabdomyosarcoma | |
| Fibroblastic | Fibrosarcoma |
| Adipose | Liposarcoma |
| Neural | Schwannoma |
| Vascular | Angiosarcoma |
| Epithelioid hemangioendothelioma |
Intrahepatic cholangiocarcinoma (ICC) is the second most common primary liver malignancy, accounting for up to 15% of all liver cancers. ICC is defined as cholangiocarcinoma proximal to the second-degree bile ducts and it consists of approximately 10% of all cholangiocarcinomas. The incidence of ICC appears to be increasing, in contrast to the incidence of extrahepatic (hilar and distal) cholangiocarcinoma, which has decreased both in the United States and worldwide. The increasing incidence of ICC could be partially attributed to our ability to more accurately diagnose these tumors, both radiographically and pathologically. In the past, many patients with ICC were erroneously diagnosed with liver metastasis, frequently called adenocarcinoma of unknown origin. However, there is epidemiologic evidence that the true incidence of ICC is also rising.
Recently, cirrhosis and hepatitis B and C have been identified as risk factors for the development of ICC. Much as with HCC, it has been hypothesized that the inflammatory cytokines and cell death associated with the aforementioned conditions promote oncogenesis and the development of ICC. In a recent meta-analysis of epidemiologic studies on ICC risk factors, cirrhosis had an odds ratio (OR) of 22.92 (95% confidence interval [CI], 18.24 to 28.79), hepatitis B: OR, 5.10 (CI, 2.91 to 8.95), and hepatitis C: OR, 4.84 (CI, 2.41 to 9.71). Hepatolithiasis has also been associated with the development of ICC. However, the vast majority of ICC develops de novo without any associated risk factors.
New studies have begun to elucidate the genetic profile of ICC and the mechanics driving tumorigenesis and tumor survival. These findings have also begun to identify potential targets for biologic therapies in patients with ICC. Sia et al. performed gene expression and mutation analysis on 149 ICC samples. They identified two classes of ICC, one in which inflammatory signals predominate (38% of ICC in their study) and another one in which proliferation pathways predominate (62%). In the inflammation class, cytokines were overexpressed and signal transducer and activator of transcription 3 (STAT3) was activated. In the proliferation class, the RAS/MAPK/MET signaling pathways were activated and KRAS and BRAF were frequently mutated. In another study, Andersen et al. profiled the transcriptome of 104 ICC and discovered frequent deregulation of the HER2 network and frequent overexpression of epidermal growth factor receptor (EGFR). Jiao et al. sequenced the exome of 32 ICCs and discovered frequent inactivating mutations in chromatin remodeling genes (ARID1A, BAP1, and PBRM1). They also discovered frequent mutations in the IDH1 and IDH2 genes that encode the metabolic enzymes isocitrate dehydrogenase. Mutations of IDH1 and IDH2 in ICC have been identified in several other studies as well. IDH1 and IDH2 mutations lead to hypermethylation of DNA and histones, which can result in altered gene expression and carcinogenesis. In addition, novel fibroblast growth factor receptor 2 (FGFR2) gene fusions have also been identified in ICC. Finally, notch signaling has also been studied and implicated in ICC. This pathway has been found to be important for cell differentiation and bile duct formation. In two preclinical models, mature hepatocytes with activated Notch converted into ICC precursors. Sia et al. have also demonstrated that the transcriptome of ICC has similarities to HCC. These studies challenge the theory that ICC arises from cholangiocytes and raises the question of which is the true cell of origin of ICC.
Our novel understanding of the genetic heterogeneity of ICC has important clinical implications. Zhu et al. analyzed 200 resected ICCs from seven centers and discovered that most somatic mutations were prevalent in low frequency. In fact, the majority (61.5%) of patients harbored no mutations. In the remaining minority of patients with mutations, only two mutations, KRAS and IDH, were identified with a frequency greater than 5%. These data suggest that applying targeted therapies in ICC is unlikely to benefit all patients. It calls for a personalized approach with sequencing of individual tumors and identification of mutations that would confer sensitivity to biologic therapies. Fortunately, all the commonly altered cancer driver pathways in ICC (IDH, FGFR2, EGFR, mammalian target of rapamycin [mTOR], Notch, chromatin remodelers, and MET) have “actionable” targets.
Histologically, ICC can be classified as mass-forming, periductal-infiltrating, intraductal, superficial spreading, and undefined categories. The mass-forming subtype is defined as a mass located in the liver parenchyma, invading through the portal venous system and is the most common form of ICC. The periductal-infiltrating subtype extends longitudinally along the bile duct and can cause peripheral biliary dilation. It often displays lymphatic invasion. The intraductal subtype proliferates into the bile duct, forming an intraductal tumor thrombus. Historically, superficial spreading and intraductal ICC have been associated with better prognosis, while periductal and mass-forming have had worse outcomes. ICC has a distinct immunohistochemistry profile that can help differentiate it from adenocarcinoma liver metastases and HCC. ICC stains strongly positive for CK7 and negative or weakly positive for CK20. In contrast to HCC, ICC does not express hepatocyte paraffin antibody 1 (HepPar1). Expression of N-cadherin can further differentiate ICC from extrahepatic cholangiocarcinoma, as the combination of CK7 and N-cadherin positivity has a specificity of 98% for ICC.
ICC is most often identified on cross-sectional imaging, computed tomography (CT), or magnetic resonance imaging (MRI) as a mass lesion ( Fig. 133.1A ). In most cases, ICC has imaging characteristics similar to adenocarcinoma metastases, including peripheral venous enhancement and central necrosis. Less commonly, ICC can appear with arterial enhancement or intravascular thrombus, mimicking radiologic findings of HCC. Contrast-enhanced MRI can help differentiate between ICC and other tumor types, including mixed cholangiohepatocellular carcinoma. More commonly, extracellular gadolinium contrast media is used in liver MRI. However, newer contrast agents such as gadoxetic acid (Eovist) and gadobenate dimeglumine (Multihance) have been shown to have improved ability to characterize ICC. Fluorodeoxyglucose positron emission tomography (FDG-PET) can also be useful as an adjunctive imaging modality. In particular, FDG-PET can be useful to detect nodal involvement or unsuspected distant metastases.
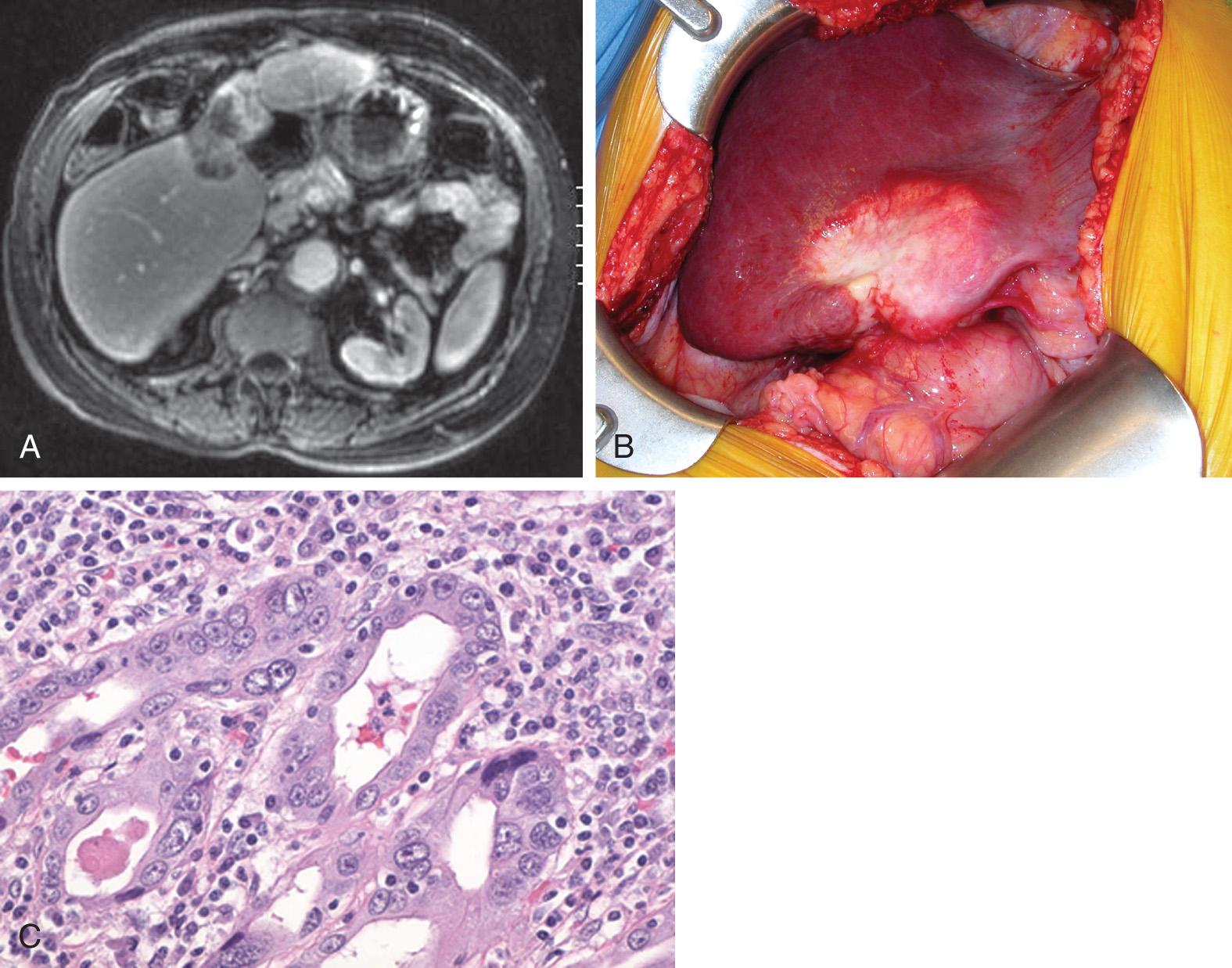
In some cases when a definitive diagnosis needs to be established to plan management, a percutaneous needle biopsy may be indicated. When a biopsy of a suspicious liver tumor reveals adenocarcinoma, it is important not to mistake the diagnosis of ICC from that of a hepatic metastasis from an extrahepatic origin. In such cases, a history of a previous malignancy, identification of other risk factors, and scrutiny of tumor markers are helpful. Moreover, multifocal lesions within the liver increase the likelihood that these are metastases. Staging of the chest and abdomen using CT scanning is useful to help rule out other potential primary tumors as well as metastatic disease. Upper and lower endoscopy should be considered to rule out an intestinal primary malignancy and, in women, mammography and gynecologic screening should be considered.
In ambiguous cases, serum tumor markers such as CA 19-9 can also aid in establishing the diagnosis of ICC. Bergquist et al. using the National Cancer Data Base (NCDB) reported that CA 19-9 was elevated in 66.7% of ICC patients. Additionally, elevated CA 19-9 was an independent predictor of mortality, similar to node positivity and positive resection margins. More recently, other biomarkers have been studied. Ferrone et al. found that tumor staining for albumin RNA using in situ hybridization was highly predictive of primary liver tumor origin rather than metastasis.
ICC is staged based on primary tumor characteristics, lymph nodal involvement, and distant metastasis per the American Joint Commission on Cancer (AJCC) 7th edition Cancer Staging Manual ( Table 133.2 ). This was a departure from previous staging systems that classified ICC with criteria similar to HCC. Notably, tumor size is no longer part of the T classification. Conversely, micro- or major vascular invasion, the number of total lesions, invasion of adjacent structures, and periductal invasion convey important prognostic information. The presence of involved regional lymph nodes (hilar, periduodenal, or peripancreatic) is classified as stage IV disease, as lymph nodal involvement has been shown to negatively affect survival in large multiinstitutional studies. The 7th edition AJCC staging system for ICC has been independently validated and found to distribute patients more equally among stages and to predict survival more accurately than older systems.
| PRIMARY TUMOR (T) | |||
| Tx | Primary tumor cannot be assessed | ||
| T0 | No evidence of primary tumor | ||
| Tis | Carcinoma in situ (Intraductal tumor) | ||
| T1 | Solitary tumor without vascular invasion | ||
| T2a | Solitary tumor with vascular invasion | ||
| T2b | Multiple tumors, with or without vascular invasion | ||
| T3 | Tumor perforating the visceral peritoneum or with direct invasion of local extrahepatic structures | ||
| T4 | Tumor with periductal invasion | ||
| REGIONAL LYMPH NODES (N) | |||
| Nx | Regional lymph nodes cannot be assessed | ||
| N0 | No regional lymph node metastasis | ||
| N1 | Regional lymph node metastasis present | ||
| DISTANT METASTASIS (M) | |||
| M0 | No distant metastasis | ||
| M1 | Distant metastasis present | ||
| ANATOMIC STAGE | |||
| Stage 0 | Tis | N0 | M0 |
| Stage I | T1 | N0 | M0 |
| Stage II | T2 | N0 | M0 |
| Stage III | T3 | N0 | M0 |
| Stage IVa | T4 | N0 | M0 |
| Stage IVb | Any T | N1 | M0 |
| Any T | Any N | M1 | |
When possible, surgical resection is the treatment of choice for ICC. The criteria for resectability of ICC are similar to other malignancies. Unlike HCC, where the presence of cirrhosis can impact resectability, ICC is typically associated with normal liver function. Staging laparoscopy prior to laparotomy for ICC is controversial and more commonly only recommended in high-risk circumstances (e.g., multicentric disease, high CA 19-9, radiologic findings suspicious for major vascular invasion or peritoneal disease). In two prospective studies evaluating operations for various hepatobiliary malignancies, staging laparoscopy avoided open exploration in approximately one third of patients due to the discovery of occult metastatic disease. However, neither study specifically examined ICC.
Intraoperatively, ICC can appear much like other hepatic malignancies, often with normal appearing surrounding liver (see Fig. 133.1B ). As with most liver surgery, intraoperative ultrasonography is important to determine resectability and rule out occult multifocal disease. The goal of resection is complete extirpation of all gross disease with negative margins while maintaining adequate remnant portal and arterial inflow, hepatic venous outflow, intact biliary drainage, and sufficient uninvolved remnant liver volume. Inclusion of a resection of the extrahepatic biliary tree with reconstruction or inclusion of major vascular (portal vein or inferior vena cava) resection may be required in some circumstances. In one series, 12% of hepatectomies were combined with major vascular resection with no difference in short- and long-term outcomes. In another multiinstitutional Italian series, biliary and major vascular resection were required in 19% and 5% of cases, respectively. With this aggressive surgical approach, negative-margin (R0) resection can be achieved in up to 80% of patients.
Complete resection is associated with improved long-term survival in patients with ICC, with reported 5-year overall survival rates of 30% to 40%. A meta-analysis of all published series of surgically resected ICC identified the following factors to be associated with worse survival: age, tumor size, multifocal tumors, lymph node metastasis, vascular invasion, and poor tumor differentiation. Hyder et al. studied 514 patients who underwent resection for ICC in United States, Europe, and Asia and used proportional hazards regression modeling to identify factors associated with survival and construct a nomogram. They also identified age at diagnosis, tumor size, multiple tumors, cirrhosis, lymph node metastasis, and macrovascular invasion to negatively impact survival. Fortunately, recent reports suggest improving outcomes in patients undergoing resection for ICC. Endo et al. reported improved disease-free survival when comparing patients resected between 2000 and 2006 compared with those resected between 1990 and 1999. Similarly, Nathan et al. found in a review of ICC outcomes in the United States using the Surveillance Epidemiology and End Results (SEER) database a trend toward better outcome following resection, despite overall poorer outcome in patients with ICC.
ICC can be associated with regional (periportal, periduodenal, or peripancreatic) nodal involvement. Moreover, involved lymph nodes are an important predictor of poor long-term outcome following resection. De Jong et al. found a 30% rate of lymph nodal metastasis among patients receiving lymphadenectomy in a multiinstitutional database of 449 patients. However, as node dissection was performed only selectively in this series, this is likely an overestimation of node positivity in resected ICC. In this study, positive nodes were associated with worse survival (median 24 vs. 30 months). Based on these data, some have questioned the value of resection in patients with ICC and lymph node involvement. Uenishi et al. found that while patients with negative nodes had better survival, those with lymph nodes still had favorable outcomes, with a 5-year survival rate of 26%. Similarly, Vitale et al. identified a survival benefit in patients who underwent therapeutic lymphadenectomy (defined as removal of more than three lymph nodes) in a propensity scores analysis using the SEER database. While still controversial, surgical resection can reasonably be offered to patients even with preoperative evidence of nodal involvement, provided all disease can be completely removed. Many experts now recommend routine hilar and periportal lymphadenectomy in all patients undergoing surgical resection for ICC
In patients with unresectable disease confined to the liver, a variety of locoregional therapies have the potential of “downstaging” initially unresectable ICC, allowing curative intent resection in selected patients. Intraarterial liver therapies, such as bland transarterial embolization (TAE), transarterial chemoembolization (TACE), hepatic arterial infusion therapy (HAI), and yttrium-90 radioembolization have been reported. Among the arterial therapies, TACE historically has been most often used. A recent meta-analysis on TACE for ICC revealed a pooled partial and complete radiologic response of 22% and 10%. In some selected series, TACE can achieve 40% to 70% response rates and median survival times of 12 to 29 months. Continuous HAI therapy with an implanted catheter and pump has been promoted by some groups. While reports are limited to highly selected unresectable ICC patients, response rates of up to 50% when combined with systemic chemotherapy have been reported. More recently, radioembolization with yttrium-90 microspheres is being used with increasing frequency as liver-directed therapy for unresectable ICC. While data are limited, the results appear promising and comparable to other intraarterial therapies, with reported median survival of 9 to 22 months. Alternatively, external beam stereotactic body radiotherapy (SBRT) is currently being investigated for unresectable ICC. A phase I/II study of 26 patients with either HCC or ICC treated with SBRT demonstrated a response rate of 42% with a 1-year survival of 45%. Local ablative therapies, radiofrequency ablation (RFA), and microwave ablation (MWA) have been reported in small series of patients with ICC. Because ablation is typically used only in small tumors, it is rarely recommended in unresectable ICC. However, in selected cases, ablation can be used in a patient with a small tumor who is not a candidate for surgery, during surgery as an adjunct to liver resection, or with recurrent disease following prior resection.
Unlike for HCC, the role of liver transplantation for cholangiocarcinoma is limited. Historically, 5-year survival after transplant for cholangiocarcinoma (combining hilar and intrahepatic tumors) was below 20%, leading many centers to consider cholangiocarcinoma as a contraindication for transplantation. More recently, a retrospective cohort multicenter study from Spain showed a 5-year actuarial survival rate of 51% after liver transplantation for ICC. Others have reported encouraging long-term survival in patients with incidentally discovered, early-stage cholangiocarcinomas in the explanted livers of patients with cirrhosis. In addition, some centers such as the Mayo Clinic have found favorable outcomes in patients with cholangiocarcinoma treated with transplant combined with neoadjuvant chemotherapy and chemoradiation. However, these patients had hilar cholangiocarcinoma, many with primary biliary cirrhosis, and not ICC. While encouraging, there is no established role for liver transplant in patients with ICC at this time outside of a clinical trial.
Most patients diagnosed with ICC have advanced disease at the time of diagnosis resulting in a median survival of less than 1 year. Randomized, phase III clinical trials examining chemotherapy have been difficult to conduct for ICC due to the small number of patients and the heterogeneous nature of biliary tract malignancies. Historically, 5-fluorouracil was offered as a single agent in unresectable ICC with only a 10% response rate. More recently, combination chemotherapy including platinum-containing regimens have shown increased efficacy. Specifically, Valle et al. reported in a large randomized phase III trial (ABC trial) that patients with advanced cholangiocarcinoma had significantly improved survival with gemcitabine plus cisplatin compared with gemcitabine alone (11.7 vs. 8.1 months). Ongoing trials are examining regimens to improve on these outcomes, including the addition of biologics. Advances in genomic sequencing may allow identification of potential actionable targets that can be exploited therapeutically. For example, studies are underway in ICC using targeting of FGFR2 fusions and IDH1/2 mutations.
There are currently limited data on the use of chemotherapy in the adjuvant setting for ICC following resection. The presence of high risk factors for recurrence, including lymphovascular and perineural invasion, lymph node metastasis, and positive surgical margins, increases enthusiasm for recommending adjuvant therapy. However, there are no controlled trials demonstrating benefit of adjuvant therapy for ICC. Patients with complete resection (R0) and absence of the above factors should be followed with observation alone. If a decision to administer adjuvant therapy is made, options include fluoropyrimidine chemoradiation, or fluoropyrimidine-based or gemcitabine-based chemotherapy. These recommendations are based on phase II trials. Participation in ongoing clinical trials should also be considered.
Our recently increased understanding of the bipotential nature of hepatic progenitor cells has revealed that they can differentiate between hepatocyte and biliary lineages. Therefore it is not surprising that tumors can develop with heterogeneous cellularity, termed as combined hepatocellular carcinoma-cholangiocarcinoma (cHCC-CC). These tumors are rare, with an estimated incidence of 1.3%. However, their true incidence might be higher, given the significant potential for misdiagnosis. Based upon the concomitant cytokeratin profile consistent with a biliary lineage, albumin expression consistent with a hepatocytic lineage, as well as carcinoembryonic antigen (CEA) and alpha-fetoprotein (AFP) levels, indeed, mixed cholangiohepatocellular carcinomas should be distinguishable from ICC and HCC. Much like HCC, these tumors are commonly associated with chronic viral hepatitis and cirrhosis, suggesting that chronic injury and expansion of hepatic progenitors may be earlier events in tumor progression. Yet, similar to ICC, these mixed tumors may occur sporadically without any risk factors. Histologically, cHCC-CC is defined as a tumor containing intimately mixed elements of both HCC and ICC. The presence of “transition zones,” which contain cells with intermediate morphology, is particularly important for the diagnosis. These transition zones distinguish “true” cHCC-CC from “collision tumors,” where the HCC and ICC components are clearly separated. Imaging characteristics, such as HCC, typically demonstrate arterial enhancement and heterogeneous necrosis on contrast CT or MRI ( Fig. 133.2 ).
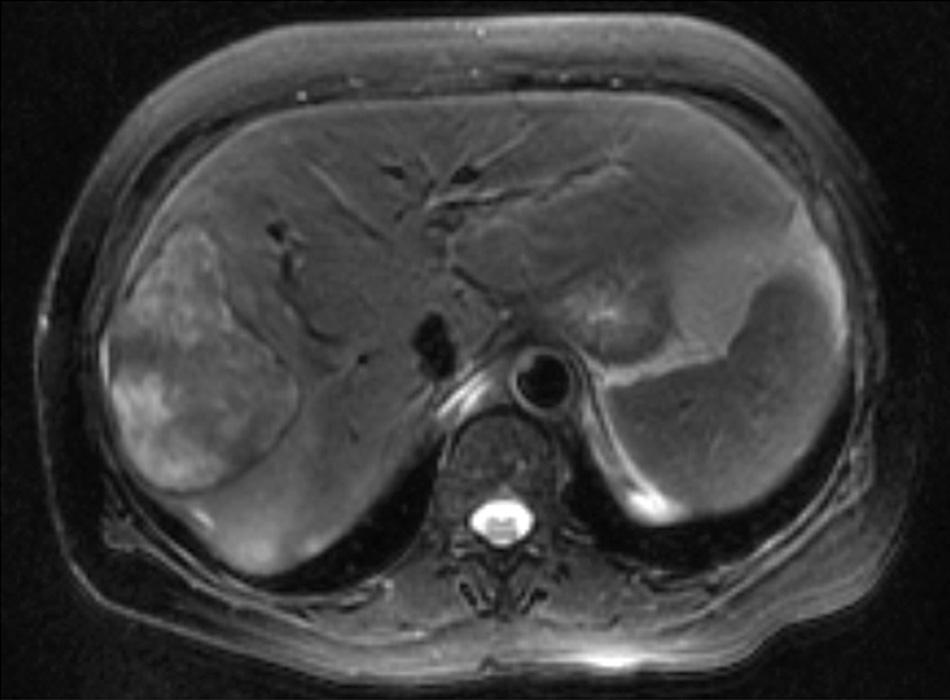
Management of cHCC-CC should include liver resection when possible. Some experts, extrapolating from the data on ICC, have recommended the addition of lymphadenectomy to resection. In cirrhotic patients, liver transplantation can be considered based on similar criteria to that of HCC. However, studies thus far have produced mixed results as to the outcomes of liver transplantation for cHCC-CC. Given the infrequency of thistumor type, no reliable histopathologic prognostic factors have been identified. In cases of advanced disease, it remains unclear whether systemic treatment strategies directed toward HCC or ICC are more effective in these tumors.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here