Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The lysosomal storage diseases encompass a group of more than 70 different inherited disorders, all sharing a defect in lysosomal function. Lysosomes are acidic, membrane-bound organelles that are located in the cytoplasm and contain enzymes that degrade macromolecules. Lysosomal storage disorders ensue when one or more of the hydrolytic enzymes are deficient or when essential lysosomal transporters, receptors, cofactors, or protective proteins are defective or lacking. Typically, complex macromolecules, including glycolipids, mucopolysaccharides, and glycoproteins, are delivered to the lysosome, where they undergo sequential modification by a series of hydrolases. An enzymatic deficiency becomes clinically important when macromolecules accumulate due to inadequate degradation. Different categories of defects result in the various forms of lysosomal dysfunction that are encountered in the lysosomal storage disorders ( Table 192-1 ).
| SPHINGOLIPIDOSES |
| Fabry disease (α-galactosidase) Farber disease (ceramidase) GM 1 gangliosidosis/ (β-galactosidase) GM 2 gangliosidosis/Tay-Sachs disease (β-hexosaminidase A) GM 2 gangliosidosis/Sandoff disease (β-hexosaminidase B) Gaucher disease (glucocerebrosidase) Niemann-Pick disease, types A and B (sphingomyelinase) Metachromatic leukodystrophy (arylsulfatase A) Globoid cell leukodystrophy/Krabbe disease (β-galactocerebrosidase) Metachromatic leukodystrophy (arylsulfatase A and prosaposin) Niemann-Pick disease type C (NPC1 protein, intracellular cholesterol transporter) |
| LIPID STORAGE DISORDERS |
| Wolman disease (lysosomal acid lipase) Ceroid-lipofuscinosis, adult type, Kufs/Parry (heterogeneous, with 13 enzymes involved in lysosomal, endoplasmic reticular, and cytoplasmic vesicular transmembrane proteins) |
| MUCOPOLYSACCHARIDOSES |
| Type I/Hurler disease (α- l -iduronidase) Type II/Hunter disease (iduronate-2-sulfatase) Type III/Sanfilippo disease (four different enzymes in the degradation of heparan sulfate defining types A-D) Type IV/Morquio disease ( N -acetylgalactosamine 6-sulfatase-type IVA, β-galactosidase-type IVB) Type VI/Maroteaux-Lamy disease (arylsulfatase B) Type VII/Sly disease (β-glucuronidase) |
| OLIGOSACCHARIDOSES |
| Aspartylglucosaminuria (aspartylglucosaminidase) Fucosidosis (α-fucosidase) α and β Mannosidosis (α-mannosidase or β-mannosidase) Schindler disease (α- N -acetylgalactosaminidase) Sialidosis I (sialidase) Sialidosis II/mucolipidosis I (sialidase) |
| MUCOLIPIDOSES |
| Mucolipidosis I/sialidosis (neuraminidase) Mucolipidosis II/I-cell disease ( N -acetylglucosaminylphosphotransferase) Mucolipidosis III/pseudo-Hurler ( N -acetylglucosaminylphosphotransferase) Mucolipidosis IV (mucolipin TRP cation channel 1/ MCOLN1 mutation) |
| LYSOSOMAL GLYCOGEN STORAGE DISORDERS |
| Glycogenosis type II/Pompe disease (α-1,4-glucosidase or acid maltase) |
| INTEGRAL MEMBRANE PROTEIN DISORDERS |
| Sialic acid storage disease/Salla disease (sialin/SLC17A5) Cystinosis (cystine transporter/CTNS) Niemann-Pick disease, type C (intracellular cholesterol transport) Danon disease (LAMP2) |
| MULTIPLE ENZYME DEFICIENCY DISORDERS |
| Galactosialidosis (β-galactosidase and sialidase) Multiple sulfatase deficiency/Austin disease (sulfatases) |
∗ Full chapters describing each of these disorders are available in Valle D, Beaudet AL, Vogelstein B, et al., eds. The Online Metabolic and Molecular Bases of Inherited Disease. http://www.ommbid.com/OMMBID .
Although most lysosomal storage disorders are rare, with the incidence of individual diseases ranging from 1 in 50,000 to 1 in 250,000 live births, as a group their frequency is estimated to be 1 per 7000 to 8000 live births. These percentages are undoubtedly an underestimate because milder or attenuated forms of these disorders are often not identified. Each of the disorders has an autosomal recessive pattern of inheritance, except for Fabry disease and Hunter syndrome (mucopolysaccharidosis II), which are X-linked recessive, and Danon disease, which is caused by deficiency of the lysosome-associated membrane protein 2 (LAMP-2) and is inherited in an X-linked dominant manner. All lysosomal storage disorders are characterized by a broad spectrum of oftentimes subtle manifestations, so they may evade diagnosis when the focus is on an individual organ system. Many of these disorders were traditionally classified into infantile, juvenile, and adult types, based on the patient’s age at the onset of manifestations, but such age-based distinctions can be misleading. Among the factors that contribute to this phenotypic diversity are the amount of residual enzyme activity, the cellular localization of the enzyme, the genotype, and the genetic background of the affected individual, as well as other environmental and epigenetic influences.
With the advent of new therapies for some of the lysosomal storage disorders, early establishment of the diagnosis is paramount. Suggestive clinical findings include coarse facial features; organomegaly; specific eye findings, including corneal clouding or a cherry red spot (of the macula); cardiac involvement; cytopenia; neurologic involvement, including psychiatric symptoms or delay or progressive loss of developmental skills; and skeletal abnormalities, notably dysostosis multiplex ( Table 192-2 ). The index of suspicion should be heightened whenever these features occur in concert, the findings are progressive, developmental milestones are missed, new psychiatric features appear, or the affected individual appears dissimilar to other family members.
| FINDING | DISORDERS |
|---|---|
| Hepatosplenomegaly | GM 1 gangliosidosis, Niemann-Pick disease, Gaucher disease, Wolman disease, Schindler disease, fucosidosis, Pompe disease, mannosidosis, galactosialidosis, multiple sulfatase deficiency, sialidosis, sialic acid storage disease, several mucopolysaccharidoses, aspartylglucosaminuria |
| Coarse facies | GM 1 gangliosidosis, fucosidosis, mannosidosis, sialidosis, galactosialidosis, multiple sulfatase deficiency, sialic acid storage disease, several mucopolysaccharidoses, mucolipidosis II and III, aspartylglucosaminuria |
| Skeletal findings | GM 1 gangliosidosis, Gaucher disease, fucosidosis, mannosidosis, sialidosis, galactosialidosis, several mucopolysaccharidoses, mucolipidosis II and III |
| Cherry red spot (retina) | Infantile forms of GM 1 gangliosidosis, Sandhoff disease, Tay-Sachs disease, Niemann-Pick disease, sialidosis, galactosialidosis, I-cell disease |
| Corneal clouding | GM 1 gangliosidosis, several mucopolysaccharidoses, mannosidosis, I-cell disease, mucolipidosis III and IV, multiple sulfatase deficiency, galactosialidosis |
| Brain involvement | GM 1 gangliosidosis, GM 2 gangliosidosis, Sandhoff disease, Tay-Sachs disease, Niemann-Pick disease, Gaucher disease type 2, Fabry disease, Schindler disease, fucosidosis, mannosidosis, multiple sulfatase deficiency, sialidosis, galactosialidosis, several mucopolysaccharidoses, sialic acid storage disease, aspartylglucosaminuria, I-cell disease, mucolipidosis III and IV, Krabbe disease, metachromatic leukodystrophy, neuronal ceroid lipofuscinosis, Danon disease |
| Peripheral nervous system findings | Metachromatic leukodystrophy, Fabry disease, Sandhoff disease |
| Cardiac | Mucopolysaccharidosis (I, II, IV, and VII), mucolipidosis I and II, Fabry disease, Farber disease, galactosialidosis, Pompe disease |
| Hematologic
Granulated or vacuolated white blood cells |
Anemia, thrombocytopenia in Gaucher disease; eosinophil abnormalities in multiple sulfatase deficiency, GM 1 gangliosidosis, Salla disease, foam cell in GM 1 gangliosidosis, Niemann-Pick disease, Gaucher disease, acid lipase deficiency, fucosidosis Several mucopolysaccharidoses, sialidosis, galactosialidosis, neuronal ceroid-lipofuscinosis, Niemann-Pick disease, Wolman disease, fucosidosis, mannosidosis, aspartylglucosaminuria, I-cell disease, mucolipidosis III, multiple sulfatase deficiency |
| Psychiatric or behavioral manifestations | Several mucopolysaccharidoses (especially Sanfilippo), sialidosis, galactosialidosis, Fabry disease, mannosidosis, neuronal ceroid-lipofuscinosis, metachromatic leukodystrophy, Tay-Sachs disease, Niemann-Pick disease (type C) |
| Newborn presentations | Gaucher disease (type 2), GM 1 gangliosidosis, Krabbe disease, Niemann-Pick disease, (types A and C), mucopolysaccharidosis (I, IVA, VII), Pompe disease, sialidosis (types I and II), mucolipidosis (types I and II), Schindler disease, Wolman disease, infantile sialic acid storage disease, sialuria, Salla disease, galactosialidosis, multiple sulfatase deficiency, prosaposin deficiency |
The diagnostic evaluation includes a careful history, with analysis of the family pedigree and assessment of developmental milestones in childhood and adolescence. A family history of consanguinity, other affected siblings, multiple miscarriages, or early deaths can aid in making the diagnosis. Ethnicity can be a helpful clue because some of the lysosomal disorders occur with increased incidence in specific populations, such as Ashkenazi Jews (Gaucher disease type 1, Tay-Sachs disease, mucolipidosis type IV) and Scandinavians (mannosidosis, sialic acid storage disease, aspartylglucosaminuria, Gaucher disease type 3).
On physical examination, special attention should be paid to head circumference (microcephaly or macrocephaly); facial appearance (coarse facies); enlargement of the tongue; hepatosplenomegaly; skeletal manifestations, including kyphosis, broadening of the long bones, and stiffness of the joints; and behavior and development. Evaluations by multiple specialists are often helpful to define related manifestations across all organ systems. Skin evaluation may reveal angiokeratoma, especially around the umbilicus and in skin creases, seen in Fabry disease, fucosidosis, sialidosis, galactosialidosis, and GM 1 gangliosidosis. The eye evaluation should include a funduscopic and slit lamp examination to screen for storage material within the retina (cherry red spots), seen in sialidosis, galactosialidosis, and GM 1 and GM 2 gangliosidosis, as well as an assessment for atypical eye movements, which may be pathognomonic for disorders such as neuronopathic Gaucher disease and Niemann-Pick disease type C. Unexplained cardiomyopathy and cryptogenic stroke may be the initial presentation of Fabry disease. A careful neurologic and cognitive evaluation can be fruitful, because the later presentations of some lysosomal disorders include dementia and psychiatric manifestations. Regression of developmental milestones can provide an early diagnostic clue in some disorders, whereas developmental delay occurs in others.
Physicians’ increased awareness of the range of manifestations and presentations of lysosomal disorders may lessen the lengthy delays in diagnosis that patients frequently experience. Preliminary clinical diagnostic studies include urine or blood chromatography screens for storage material, blood count with smear for vacuolated white blood cells, skeletal radiography, and ophthalmologic examination.
The diagnosis of a specific lysosomal storage disorder can be established by sequencing the specific gene or by an enzyme assay. DNA sequence analysis is typically more successful when a mutation has already been identified in a family or when specific mutations are known to be common in a given ethnic group. In addition, when clinical signs point to several different disorders, diagnostic testing based on DNA sequencing of panels of genes implicated in lysosomal storage disease is available. Next-generation sequencing at times can also reveal the diagnosis, although the identification of new variants of unknown significance can introduce uncertainty. The expanded use of next-generation DNA sequencing has led to the identification of lysosomal storage disorders in patients who do not have classic manifestations of their storage disease. Newborn screening in some states may also include specific lysosomal storage disorders to optimize early identification and treatment.
Enzymatic activity may be assayed in a blood sample using a lysosomal panel that evaluates the activities of multiple lysosomal enzymes from the same sample. A tissue biopsy, most often of the bone marrow or liver, can confirm the diagnosis but now is rarely indicated.
Biomarkers also can be suggestive of a specific disorder. For example, elevated levels of the lipid glucosylsphingosine (lysoGb1) are highly suggestive of Gaucher disease.
Patients with lysomal storage disorders should receive disease-specific care via specialized centers, where improved care and enzyme-specific therapeutic modalities ( Table 192-3 ) under expert supervision have transformed the natural history of several of these disorders. With patients’ increased longevity, diseases that were once encountered only by pediatricians are now seen by adult physicians. Moreover, many of the classic complications are now avoided by early therapeutic interventions. In some instances, however, prolonged survival has unmasked unanticipated clinical consequences such as myopathy in cystinosis and learning disabilities in Gaucher disease type 3.
| DISEASE | DEFICIENT ENZYME | ENZYME REPLACEMENT THERAPY | OTHER DISEASE-MODIFYING DRUGS (SUBSTRATE REDUCTION THERAPY) |
|---|---|---|---|
| Gaucher disease | Glucocerebrosidase | Imiglucerase, velaglucerase alfa, taliglucerase alfa | Miglustat, eliglustat |
| Fabry disease | α-Galactosidase | Agalsidase alfa, agalsidase beta | Migalastat |
| Pompe disease | α-1,4-Glucosidase | Pegunigalsidase alpha, alglucosidase alfa, avalglucosidase alfa | |
| Mucopolysaccharidosis type I | α- l -Iduronidase | Laronidase | |
| Mucopolysaccharidosis type II | Iduronate-2-sulfatase | α- l -Iduronidase, JR-141 | |
| Mucopolysaccharidosis type VI | Arylsulfatase B | Galsulfase | |
| Niemann-Pick C | Miglustat |
Gaucher disease is divided into three types based on the absence or presence and the rate of progression of neurologic involvement. Type 1, the non-neuronopathic form, is the most common type in the Western world and can manifest at any age. Type 2, the acute neuronopathic form, manifests before or shortly after birth and has a rapid and progressive course. Type 3 is the subacute neuronopathic form. The spectrum of manifestations encountered in this disorder ranges from asymptomatic octogenarians to infants who succumb in utero. Some patients defy classification into one of the three types.
Gaucher disease is a pan-ethnic disorder. Type 1 Gaucher disease is more frequent among Ashkenazi Jews, in whom the carrier frequency is about 1 in 16, whereas the carrier frequency in the general population is approximately 1 in 100.
Gaucher disease, the autosomal recessively inherited deficiency of the lysosomal enzyme glucocerebrosidase, is a disorder primarily of the reticuloendothelial system. Lysosomes within macrophages become engorged with the substrate glucocerebroside, thereby giving rise to the characteristic Gaucher cells, which have a wrinkled-paper appearance that results from the intracytoplasmic deposition of glucocerebroside. The accumulated glycolipids, glucocerebroside and glucosylsphingosine, result from the degradation of senescent leukocytes or erythrocyte membranes.
The gene encoding glucocerebrosidase, GBA1, is located on chromosome 1q21. More than 600 different mutations have been found, several of which are encountered with increased frequency in specific types. For example, mutation N370S (p.N409S), which is the most common allele among Ashkenazi Jews, is seen only in type 1 Gaucher disease. This specific mutation increases resistance to tuberculosis in homozygotes. Homozygosity for L444P (p.L483P) is most often associated with type 3 Gaucher disease. Oftentimes, however, identified mutations cannot be used to predict the clinical phenotype.
Commonly encountered symptoms in all types of Gaucher disease include easy bruisability, hepatomegaly, splenomegaly, chronic fatigue, and bone pain or pathologic fractures. Laboratory findings include anemia, thrombocytopenia, and elevations of ferritin, acid phosphatase, angiotensin-converting enzyme, and, at times, liver enzymes. Levels of lyso-Gb1 are massively elevated, and assays for this lipid are increasingly used as a biomarker of disease. About 50% of patients develop polyclonal gammopathy, and about 30% develop monoclonal gammopathy ( Chapter 173 ). Altered macrophage function, complement processing, elevated cytokine levels, and an increased risk for multiple myeloma ( Chapter 173 ) have been reported.
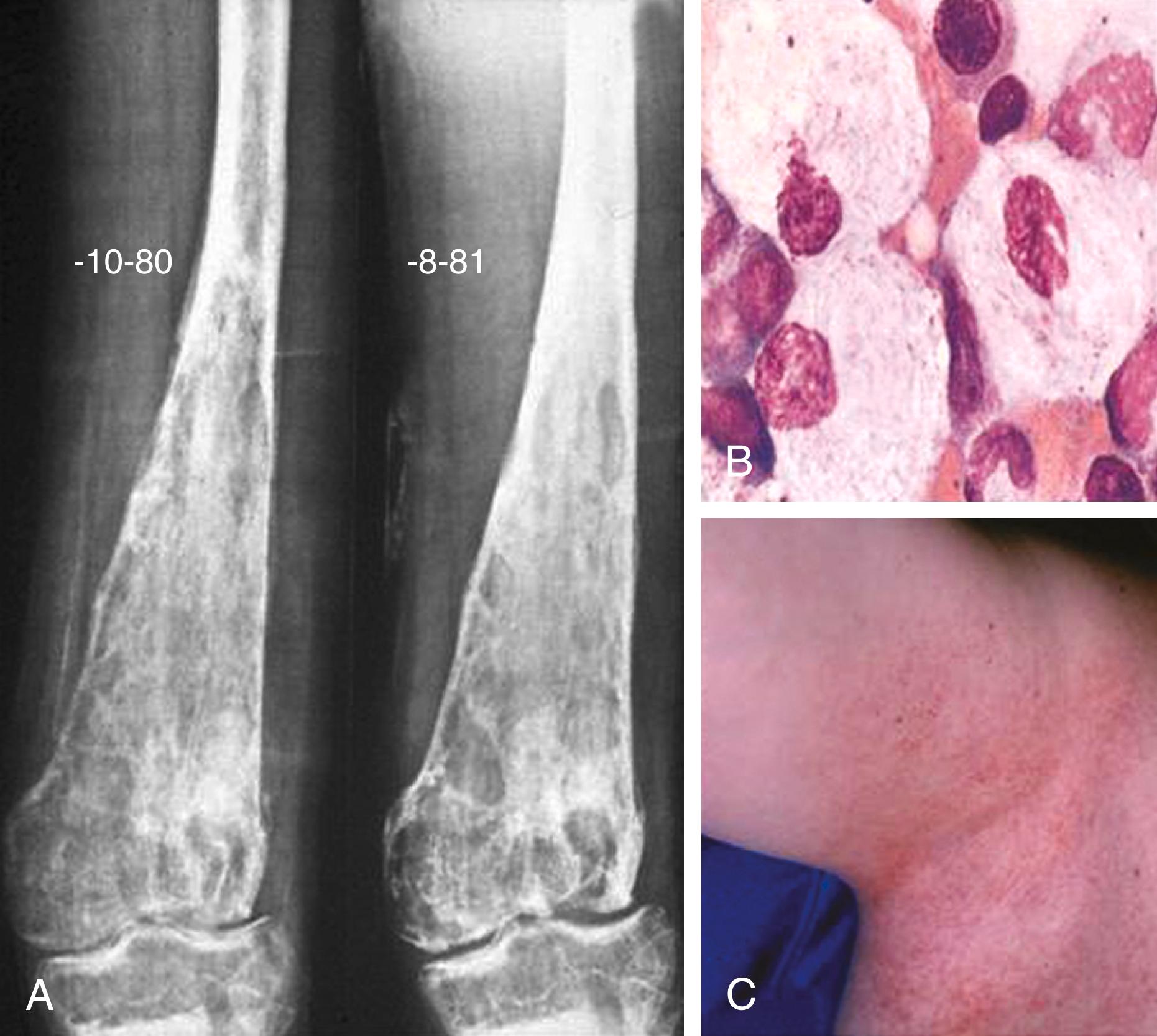
Painless splenomegaly is the most common presentation in patients with Gaucher disease type 1, and the spleen can be massively enlarged. Thrombocytopenia and/or anemia are frequent. Occasional patients have pulmonary involvement or pulmonary hypertension. Bone involvement, which is a significant cause of morbidity, can manifest with extreme bone pain or pathologic fractures. Most patients have radiologic evidence of skeletal involvement, including the classic Erlenmeyer flask deformity of the distal end of the femur and osteopenia ( Fig. 192-1A ). Pathologic fractures (especially of the hip, ribs, or spine), lytic bone lesions, and osteoporosis may occur. Painful bone crises, which are episodes of bone infarction, can persist for weeks and may require aggressive pain management.
An important association exists between the presence of pathologic variants in GBA1 and parkinsonism ( Chapter 378 ). Both patients with Gaucher disease as well as heterozygous carriers have a higher incidence of Parkinson disease and Lewy body disorders. Studies in large Parkinson cohorts around the world reveal a 5- to 10-fold increased frequency of GBA1 mutations, thereby rendering this the most common genetic risk factor for parkinsonism identified to date. However, the majority of patients with Gaucher disease and carriers of a GBA1 mutation do not develop Parkinson disease, and this reduced penetrance remains unexplained.
Type 2 Gaucher disease, which is rare, is characterized by a rapid neurodegenerative course with extensive visceral involvement, and it is associated with failure to thrive, laryngospasm, strabismus, and seizures. Death occurs within the first years of life.
Type 3 disease is clinically variable and is often noted in childhood. In addition to organomegaly and bone involvement, patients have abnormal horizontal eye movements, and some develop myoclonic epilepsy or neurodegeneration. A subgroup of patients have cardiac calcifications, hydrocephalus, and other atypical manifestations. Patients with this specific Gaucher phenotype (Gaucher disease type 3C) are homozygous for the GBA1 variant D409H (p.D448H).
Gaucher disease should be considered in the differential diagnosis when patients of any age have unexplained organomegaly, easy bruisability, or bone pain ( Table 192-4 ). Measurement of the biomarker LysoGbl, performed on dried blood spots or serum, is increasingly being used as a diagnostic screen. Elevated levels can then be confirmed by enzymatic or DNA analyses to establish the definitive diagnosis.
| GAUCHER DISEASE | FABRY DISEASE |
|---|---|
| Family member with Gaucher disease Hepatomegaly, splenomegaly (sometimes massive) Frequent epistaxis Easy bruising Abnormal saccadic eye movements Thrombocytopenia or anemia Painful bone crisis Erlenmeyer flask deformity of the distal femur, aseptic necrosis of the femoral heads Pathologic fractures, unexplained rib fracture Multiple myeloma Parkinsonism Elevated serum ferritin, glucosylsphingosine, angiotensin-converting enzyme, or tartrate-resistant acid phosphatase |
Family history of Fabry disease Cutaneous lesions of capillaries (angiokeratoma) Corneal and lenticular opacities Hypohidrosis or heat intolerance Intermittent severe extremity pain (acroparesthesias) Left ventricular hypertrophy of unknown etiology in young adulthood Stroke of unknown etiology in young adulthood Chronic kidney disease of unknown etiology in young adulthood Multiple renal sinus cysts Female carriers may have more variable and less severe symptoms with later onset |
The diagnosis can be made by demonstrating deficient glucocerebrosidase activity in leukocytes or cultured cells. In some populations, particularly Ashkenazi Jews, screening for specific mutations can be diagnostic because mutation N370S accounts for about 70% of mutant alleles. Generally, sequencing of the entire GBA1 gene is required for an accurate molecular diagnosis. Since the presence of a highly homologous pseudogene sequence nearby can complicate molecular analysis, diagnostic laboratories must be knowledgeable about the gene. Bone marrow and liver biopsies show pathologic changes (see Fig. 192-1B ) but are not indicated for diagnosis. Carrier identification is best achieved by DNA testing when the mutant allele is known. Prenatal diagnosis is possible by determining the enzymatic activity or specific mutations in chorionic villi or cultured amniotic fluid cells.
Two treatment approaches to reduce the pathologic accumulation of glucocerebroside are enzyme replacement therapy and substrate reduction therapy. Asymptomatic and mildly symptomatic adults do not always require treatment.
Enzyme replacement with recombinant glucocerebrosidase (see Table 192-2 ) is available for the treatment of symptomatic patients with types 1 and 3 Gaucher disease ( Table 192-5 ). Anemia, thrombocytopenia, and organomegaly are reversed within 12 to 36 months with enzyme doses between 15 and 60 IU/kg given every other week. The treatment is ongoing, administered intravenously, and extremely expensive. The enzyme does not cross the blood-brain barrier and does not alter the neurologic progression of patients who have neuronopathic forms of Gaucher disease, but it can still be useful in alleviating visceral manifestations.
| GAUCHER DISEASE | FABRY DISEASE |
|---|---|
| ERT is a costly but effective intravenous therapy, generally administered every other week for life. An oral substrate reduction drug eliglucerase also successfully reverses disease manifestations. Decreased splenic and hepatic volumes and increases in hemoglobin levels and platelet counts are expected in the first year of treatment. Asymptomatic and mildly symptomatic adults do not always require treatment. ERT and eliglustat do not cross the blood-brain barrier and do not correct neurologic features of neuronopathic forms of Gaucher disease. ERT will not prevent the development of parkinsonism. |
ERT is a costly but effective intravenous therapy, generally administered every other week for life. An oral pharmacologic chaperone (migalastat) can now be used instead of ERT in patients with amenable genetic variants. Hemizygous males with a low or undetectable level of α-galactosidase A should be treated with ERT (or migalastat) whether or not clinical features are present. Current trials suggest that female carriers and atypically affected males with clinical features of Fabry disease (renal, neurologic, cardiovascular) should be treated with ERT or, if appropriate, migalastat. In patients with end-stage renal disease due to Fabry disease, ERT should be considered on an individual basis, potentially to reduce other organ involvement. |
Substrate reduction therapy uses eliglustat, which is a ceramide analogue and a potent specific inhibitor of glucosylceramide synthase, which reduces glucosylceramide production to match its impaired rate of degradation. Eliglustat, which is administered orally under expert supervision, significantly improves spleen and liver volumes, hemoglobin levels, and platelet counts. Currently eliglustat is being used instead of enzyme replacement therapy in some adults who prefer an oral alternative. CYP2D6 status should be evaluated before use to determine both eligibility for the drug and the appropriate dosing. Ultra-rapid CYPRD6 metabolizers should not receive eliglustat, whereas normal or intermediate metabolizers should receive a dose of 84 mg twice daily, and poor metabolizers should be treated with 84 mg once daily.
Currently available therapies appear to have a positive effect on bone disease but do not prevent the development of parkinsonism. Efforts are ongoing to develop alternative therapies, including brain-penetrant enzyme replacement and substrate reduction therapies, chemical chaperones, and gene therapy.
Useful supportive therapies include bisphosphonates for osteoporosis, orthopedic surgery for bone fractures, and palliative therapy and hydration for bone crises. Total or partial splenectomy is now rarely indicated. Hematopoietic stem cell transplantation ( Chapter 163 ) can improve systemic but not neurologic manifestations; it has been performed in patients who have severe organomegaly and cytopenia in countries where other options are not available.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here