Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The identification of luminal chemosensors in the gastrointestinal (GI) tract has emerged in part from the de-orphanization and characterization of nutrient-sensing G protein-coupled receptors (GPCRs) in the most recent decade. Since luminal chemical substances including nutrients or meal components, or endogenously secreted molecules such as gastric acid, digestive enzymes, and bile acids release gut hormones or neurotransmitters, gut luminal chemosensing is implicated not only in local or remote regulation of gut function, but also in the systemic regulation of metabolism, energy balance, and food intake.
Gut physiological processes such as secretion, digestion, absorption, and motility are affected by these luminal substances, in addition to central nervous control via vagal nerves initiated at the cephalic phase of food intake. The first discovery of gut hormone release in response to a luminal chemical species was secretin release in response to luminal hydrogen ion (H + ) concentration. To date, ~ 20 gut hormones, principally localized in enteroendocrine cells or in myenteric neurons in the GI tract, have been identified. Extensive studies have clarified the contributions of these gut hormones in the regulation of gut function via GPCRs as well as via food intake control through vagal afferent signals. Nevertheless, the mechanism underlying food-evoked gut hormone release has been poorly understood.
Duodenal mucosal chemosensing can be systematized into three pathways ( Fig. 30.1 ). The first model is based on the assumption that luminal chemical substances diffuse or are transported through the GI mucosa into the subepithelial space, where they activate receptors expressed on subepithelial afferent nerves, followed by mucosal responses ( Fig. 30.1 A). This pattern includes luminal H + /CO 2 and capsaicin sensing. The second pathway is based on the hypothesis that luminal chemical substances, interacting with brush border ectoenzymes, activate receptors expressed on the brush border membrane of epithelial cells, generating cellular signaling cascades such as luminal purinergic signaling ( Fig. 30.1 B). The third is that luminal chemical substances activate apical membrane receptors expressed on enteroendocrine cells, followed by the release of gut hormones or mediators, which exert local and remote responses ( Fig. 30.1 C). Most nutrient-sensing pathways follow this latter pattern. These three pathways are incorporated into the discussion below.
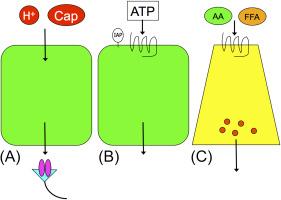
Duodenal defense factors from luminal H + include
and mucus secretion (preepithelial), intracellular pH (pH i ) regulation with ion transporters and ecto- and cytosolic enzyme activities (epithelial), and blood-flow response regulated via afferent nerves and mediator releases (subepithelial). Rapid changes in these defense factors in response to topical application of luminal chemical substances imply the presence of mucosal recognition of luminal chemical substances via the pathways depicted in Fig. 30.1 . We have assessed duodenal mucosal defense factors using an in vivo microscopic system, where the integrated regulation of mucosal defense factors is studied such as mucosal blood flow, mucus secretion, and enterocyte pH i in response to luminal chemical substances, as well as by measuring the rate of
secretion using a duodenal loop perfusion system. These approach the observation of a rapid response to luminal compounds in order to identify the mechanisms underlying these responses using pharmacological or genetic tools.
Luminal H + /CO 2 and natural pungent compounds contained in spices are sensed by subepithelial afferent nerves as shown in Fig. 30.1 A. We will describe the mechanisms underlying the sensing of these stimuli in the following sections.
Luminal acid sensing in the duodenum is essential to the protection of the mucosa from acid-induced injury. Rapid shifts of luminal pH and the high P CO 2 environment in the duodenal lumen suggest that rapid CO 2 absorption by the duodenal mucosa occurs prior to acid sensing. Epithelial cytosolic and membrane-bound carbonic anhydrase (CA) activities are essential to this process. We have reported that luminal CO 2 is absorbed by the duodenal epithelium dependent on stimulated
secretion and CA activity, accompanied by portal venous acidification. Furthermore, CO 2 -induced intracellular acidification of epithelial cells is also CA dependent and accompanied by hyperemia of the mesenteric vasculature due to the activation of transient receptor potential vanilloid 1 (TRPV1) expressed on capsaicin-sensitive afferent nerves. These results suggest that luminal H + is actively absorbed into the epithelium as CO 2 , which is converted into H + and
, facilitated by epithelial CAs. Intracellular H + is extruded from the epithelial cells via basolateral NHE-1, with the extruded H + -activating TRPV1 expressed on afferent nerves. Activated afferents release vasoactive mediators such as calcitonin-gene-related peptide (CGRP) and nitric oxide (NO). Finally, mucosal blood flow and mucus secretion are increased, followed by cyclooxygenase (COX)-dependent mucus and
secretion. These results demonstrate that the duodenal mucosa senses luminal acidity using epithelial CA activities, ion transporters, and neuronal acid sensors.
The duodenum absorbs luminal H + secreted by the stomach in order to maintain the acid-base balance between the stomach and duodenum. Acid-base balance between the stomach and duodenum is clinically important, since loss of gastric content by vomiting in patients with pyloric obstruction induces acute metabolic alkalosis and hypochloremia.
High P CO 2 -induced
secretion has also been confirmed by Sasaki et al., reporting that CO 2 -induced
secretion is differentially regulated in the stomach and the duodenum. Acid-induced intracellular acidification of the duodenal epithelial cells has also been reported by Sjöblom et al., confirming the importance of CA activities in the acid-sensing pathway of the duodenum.
H + -sensitive GPCRs are acid sensor candidates. Up to now, 4 GPCRs, including GPR4, OGR1, TDAG8, and G2A are confirmed H + -sensitive GPCRs, implicated in the detection of tissue or blood acidosis. These GPCRs are also activated by phospholipids, whereas low pH-induced activation of these GPCRs is inhibited by phospholipids. H + -sensitive GPCRs are expressed in the afferent neurons as well as the vasculature and in immune cells, suggesting that H + -sensitive GPCRs may be involved in mucosal defense mechanisms in the upper GI tract, in addition to acidosis and inflammation.
Ingested pungent compounds contained in spices such as peppers of the genus Capsicum also activate subepithelial afferent nerves, that in turn enhance GI mucosal defenses such as epithelial
secretion. TRPV1 was originally identified as a capsaicin receptor, representing a thermal and pain sensor expressed on afferent nerves, but it also acts as an acid sensor, since low pH activates TRPV1. Since capsaicin enhances mucosal defenses in the stomach and duodenum, and since low pH activates TRPV1, acid sensing in the upper GI mucosa is plausibly mediated by TRPV1. Indeed, luminal acid-induced mucosal responses are mediated by TRPV1 in the esophagus, stomach, and duodenum in a fashion similar to capsaicin-induced responses, although differential activation pathways between acid and capsaicin are present in the stomach. In both cases, TRPV1 expressed on mucosal afferent nerves ultimately detects luminal acidity in order to trigger the protective mucosal responses.
A member of the TRP ankyrin family, TRPA1, is activated by a variety of pungent compounds derived from flavoring substances in foods including allyl isothiocyanate (AITC) from wasabi , horseradish, and mustard oil, allicin from garlic, cinnamaldehyde from cinnamon, and gingerol from ginger. TRPA1 agonists such as AITC stimulate anion secretion from the colonic mucosa, and increase duodenal blood flow, suggesting that, similar to capsaicin, TRPA1 acts as a spice sensor to enhance mucosal defense mechanisms. Furthermore, TRPA1 agonists stimulate 5-hydroxytryptamine (5-HT) release from intestinal enterochromaffin (EC) cells, and delay gastric emptying via 5-HT 3 receptors.
Since TRPV1 and TRPA1 are expressed on vagal afferent nerves, whose stimulation is associated with increased body oxygen consumption, the stimulation of these GI mucosal TRP channels with luminal spices may influence energy balance and systemic metabolism, thus serving as a potential target for obesity therapy.
Similar to duodenal acid sensing, the esophageal mucosa also senses luminal acidity in the form of the permeant gas CO 2 facilitated by epithelial and neuronal CA activities and acid sensors, TRPV1 and acid-sensing ion channels (ASICs). This complex acid-sensing mechanism maintains the interstitial pH of the esophageal epithelial cells at a physiological level through the regulation of the hyperemic response to luminal H + /CO 2. The concept that CA activity and acid sensors are the essential components of esophageal H + /CO 2 sensing is supported by the observation that the taste of carbonation on the tongue is mediated and sensed by CA activity coupled to an acid sensor.
Gastric acid sensing is accomplished by a mechanism distinct from those described for the esophagus. Physiological gastric acid sensing may be attributed to the calcium-sensing receptor (CaSR) expressed on gastric endocrine cells (see a review in Ref. ). This hypothesis is supported by the observations that extracellular Ca 2 + stimulates gastrin release from antral G cells, that CaSR is pH-sensitive and stimulated by L-type amino acids, and finally that the stomach of CaSR knockout mice does not release gastrin in response to the ‘classic’ stimuli of luminal alkalinity, Ca 2 + or peptone.
Once pathological acid back diffuses into the subepithelium or the submucosa, acid may activate acid sensors such as TRPV1 or ASICs expressed on afferent nerves with subsequent COX activation, implicated in not only protective afferent responses, but also noxious sensations conducted to the central nervous system.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here