Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Hepatic tumors in children are relatively rare. The most common malignant hepatic neoplasms are not primary tumors but rather metastatic lesions such as Wilms tumor, lymphoma, and neuroblastoma (NB). Primary liver tumors make up 1–4% of all solid tumors in children. Malignant hepatic tumors occur at a rate of about 1 to 1.5 per million children per year. Ten primary liver masses occur with some frequency in the pediatric age group, and the age distribution for hepatic masses is distinctive ( Table 66.1 ). In the first 2 years of life, hepatoblastoma (HB) and infantile hepatic hemangioma (IHH) occur most commonly, and hepatocellular carcinoma (HCC) and focal nodular hyperplasia (FNH) are found most commonly after age 5 years ( Table 66.2 ).
| Type of Tumor | Number | % |
|---|---|---|
| Hepatoblastoma | 124 | 43.5 |
| Infantile hemangioendothelioma | 103 | 36.1 |
| Mesenchymal hamartoma | 38 | 13.3 |
| Nodular regenerative hyperplasia | 6 | 2.1 |
| Hepatocellular carcinoma | 4 | 1.4 |
| Angiosarcoma | 4 | 1.4 |
| Focal nodular hyperplasia | 3 | 1.1 |
| Undifferentiated embryonal sarcoma | 3 | 1.1 |
| Hepatocellular adenoma | 0 | 0 |
| Embryonal rhabdomyosarcoma | 0 | 0 |
| TOTAL | 285 | 100.0 |
| Type of Tumor | Number | % |
|---|---|---|
| Hepatocellular carcinoma | 96 | 36.6 |
| Focal nodular hyperplasia | 40 | 15.3 |
| Undifferentiated embryonal sarcoma | 39 | 14.9 |
| Nodular regenerative hyperplasia | 26 | 9.9 |
| Hepatocellular adenoma | 22 | 8.4 |
| Hepatoblastoma | 22 | 8.4 |
| Angiosarcoma | 6 | 2.3 |
| Mesenchymal hamartoma | 5 | 1.9 |
| Infantile hemangioendothelioma | 4 | 1.5 |
| Embryonal rhabdomyosarcoma | 2 | 0.8 |
| TOTAL | 262 | 100.0 |
Couinaud’s elegant description of the segmental anatomy of the liver has allowed hepatic operations to evolve to a level at which they can be performed with an acceptable morbidity and mortality ( Fig. 66.1 ). The cumulative experience with hepatic resection and hepatic transplantation has allowed the development of techniques for both subsegmental and multisegmental resections of the liver in children. With the continued expansion of knowledge about these tumors, reasonable surgical and medical management plans can be devised.
IHH is the most common benign solid hepatic tumor in childhood, accounting for about 16% of all pediatric liver tumors. It also is the most common liver tumor in the first year of life, and female infants are three times more likely to develop IHH than males. Almost all children with IHH are initially seen before the age of 6 months, and the majority are encountered in the first 2 months of life. Most IHHs are not present at birth but become apparent days to a few weeks after birth. They tend to have a rapid proliferation phase in the first 6–10 months, followed by slow involution that can take up to 10 years. At-risk populations include premature infants, females, and Caucasians.
IHHs can be either a single lesion that can expand to a massive size or a multinodular infiltrative mass. Hepatic hemangiomas can be associated with several congenital syndromes and defects, such as Osler–Weber–Rendu, Klippel–Trénaunay–Weber, Ehlers–Danlos, Beckwith–Wiedemann, diaphragmatic hernia, trisomy 21, transposition of the great arteries, and extranumerary digits.
Occasionally these lesions are either asymptomatic or present relatively innocuously as an abdominal mass or abdominal distention. However, these lesions can manifest with significant symptoms, including hepatomegaly, high-output congestive heart failure (CHF), respiratory distress, hypothyroidism, and anemia. Rarely, Kasabach–Merritt syndrome occurs, which is characterized by acute thrombocytopenia, microangiopathic hemolytic anemia, and consumptive coagulopathy. This syndrome can be life threatening and requires aggressive supportive treatment, as well as treatment of the hemangioma itself. Fortunately, Kasabach–Merritt syndrome occurs infrequently and is usually associated with IHH that has rapid growth to a diameter of 5 cm or more. No cases have been reported in association with smaller tumors.
Children who present with multiple cutaneous hemangiomas (>5) should also be screened for IHH, which can be found in up to 25% of this population. All types of IHH can be found on visceral screening, but the most common are the multifocal and diffuse types. Extrahepatic hemangiomas can also occur at other sites, including lung (10%), pancreas, lymph nodes, and bone.
IHHs are classified as focal, multifocal, or diffuse ( Table 66.3 ). Multifocal IHH has multiple lesions throughout the liver, and the individual lesions are separated by normal intervening liver parenchyma in contrast to patients with diffuse IHH in whom there is minimal to no detectable normal parenchyma between lesions and the liver is usually markedly enlarged.
| Type | Characteristics | Imaging | Associated Complications | Clinical Course | Outcome |
|---|---|---|---|---|---|
| Focal | • Solitary lesion • Glut 1 negative |
• MRI: Hypointense on T1 and hyperintense on T2 images • Gadolinium: Rapid filling, peripheral enhancement, variable central enhancement |
• Kasabach–Merritt syndrome: Occurs with IHH >5 cm • Can have high-flow shunts |
• Tend to be asymptomatic • Fully grown at birth and rapidly involute by 12–14 mo (similar to RICH) |
Excellent |
| Multifocal | • Multiple lesions separated by normal liver • Glut 1 positive |
• Same as focal but multiple | • Need brain and chest imaging looking for other lesions • Hypothyroidism • Can be seen with FNH and mesenchymal hamartoma |
• Frequently associate with high-output CHF • Starts after birth, growth for 9–12 mo, involution for 5–7 y • Progression needs treatment |
High mortality with CHF |
| Diffuse | • No detectable liver between lesions • Liver markedly enlarged • Glut 1 positive |
• Same as focal but multiple | • Massive hepatomegaly • Hypothyroidism • Abdominal compartment syndrome • Respiratory compromise |
• Starts after birth, growth for 9–12 mo, involution for 5–7 y | High mortality with abdominal compartment syndrome |
Focal IHHs tend to be asymptomatic, are less frequently associated with cutaneous hemangiomas, can be identified on prenatal ultrasound (US), are fully grown at birth, and usually rapidly involute after birth (similar to a cutaneous rapidly involuting congenital hemangioma [RICH]). Focal lesions can have high-flow shunts and have an associated low-grade anemia or thrombocytopenia.
Multifocal lesions also can be asymptomatic. However, they frequently are associated with high-output cardiac failure from either intralesional or portovenous shunting. Asymptomatic patients with multifocal hemangiomas should undergo imaging studies of the brain and chest to make sure there are no associated intracranial or pulmonary lesions.
The diffuse lesions manifest with near total replacement of the hepatic parenchyma with multiple hemangiomas, which can result in massive hepatomegaly, abdominal compartment syndrome, high-output cardiac failure, or significant respiratory compromise.
Both multifocal and diffuse lesions are associated with severe hypothyroidism due to increased degradation of thyroid hormones by type III iodothyronine deiodinase that is upregulated in the IHH. The hypothyroidism can be severe enough to cause low-output cardiac failure or significant mental retardation. Correction of the hypothyroidism can require very large doses of thyroid hormone replacement, and the hypothyroidism resolves with the involution of the IHH.
Other symptoms that can occur in patients with IHH include jaundice, failure to thrive, respiratory difficulties, or poor feeding. Historically, as many as 50–60% of infants with IHH have symptoms of CHF that seem to be age-related. Neonates with a focal hemangioma can present with high-output CHF at birth, whereas infants with multifocal lesions tend to present with CHF between the ages of 1 and 16 weeks. In patients with IHH, the hepatic transaminase levels and occasionally the α-fetoprotein (AFP) level can be elevated. The source of the AFP is not the tumor, but rather the hepatocytes that are surrounding the IHH. Moreover, an elevated AFP level in neonates must be interpreted with some caution. The AFP level is normally elevated in neonates and does not decrease to adult levels until about 6 months of age. When there is a significant elevation of the AFP level, however, HB must be excluded by either imaging studies or biopsy of the lesion.
A significant incidence of placental abnormalities has recently been reported in infants with very low birth weight (<1500 g) who present with infantile hemangiomas. These researchers discovered a variety of anomalies that could potentially lead to the shedding of placental cells into the fetal circulation. If fetal hypoxic stress is also present, these placental cells could be exposed to an increased level of angiogenic factors that could then lead to increased endothelial cell or placental cell proliferation in the postnatal period with the subsequent development of an IHH. Another theory is that IHH is a developmental anomaly of hemogenic endothelium that is derived from the primitive mesoderm and has a neural crest cell phenotype. The proliferation of this endothelium appears to be governed by the renin-angiotensin system, which may suggest that angiotensin-converting enzyme (ACE) inhibitors may be an additional therapeutic option.
In summary, workup of a hepatic mass that could be an IHH should include liver function tests, coagulation studies, thyroid functions (thyroid-stimulating hormone [TSH], triiodothyronine [T 3 ], thyroxine [T 4 ]), AFP level, radiologic studies (US, computed tomography [CT], magnetic resonance imaging [MRI]), and, if there is still question about the diagnosis, a biopsy of the lesion.
The US picture of IHH can be highly variable. Solitary lesions can have a heterogeneous echogenicity, and the Doppler spectral analysis can show a variety of flow patterns. Multifocal lesions, however, tend to be more uniform in their appearance and are seen as echolucent nodules associated with a high-flow vessel.
On CT with intravenous contrast, IHH will either enhance diffusely or show rim enhancement that is followed by gradual filling of the center of the lesion ( Fig. 66.2 ). In one series the CT enhancement pattern was correlated to the size of the lesions. Lesions less than 1 cm enhanced homogeneously in the arterial phase. Lesions greater than 2 cm demonstrated peripheral rim enhancement, and those between 1 and 2 cm showed a mixed enhancement pattern in the arterial phase. Unfortunately, these “classic” enhancement patterns are not always present in IHH, which can make the radiologic diagnosis more difficult.
MRI is emerging as the single most useful modality to show both the location of the hemangioma and its flow pattern and structure. The addition of intravenous gadolinium and a gradient-recalled-echo sequence to the MRI enhances its utility.
Focal lesions on MRI are well defined and hypointense to the liver on T1 images and hyperintense on T2-weighted images. Gadolinium demonstrates rapid filling with slow excretion as well as peripheral enhancement with variable central enhancement (related to the amount of hemorrhage or thrombosis present). Multifocal and diffuse lesions tend to show similar MRI findings except that there are multiple lesions present ( Fig. 66.3 ). Recently, however, there has been a case report of a hypervascular HB that had an MRI pattern identical to that of an IHH. Therefore, again, radiology alone cannot always reliably distinguish between benign and malignant lesions.
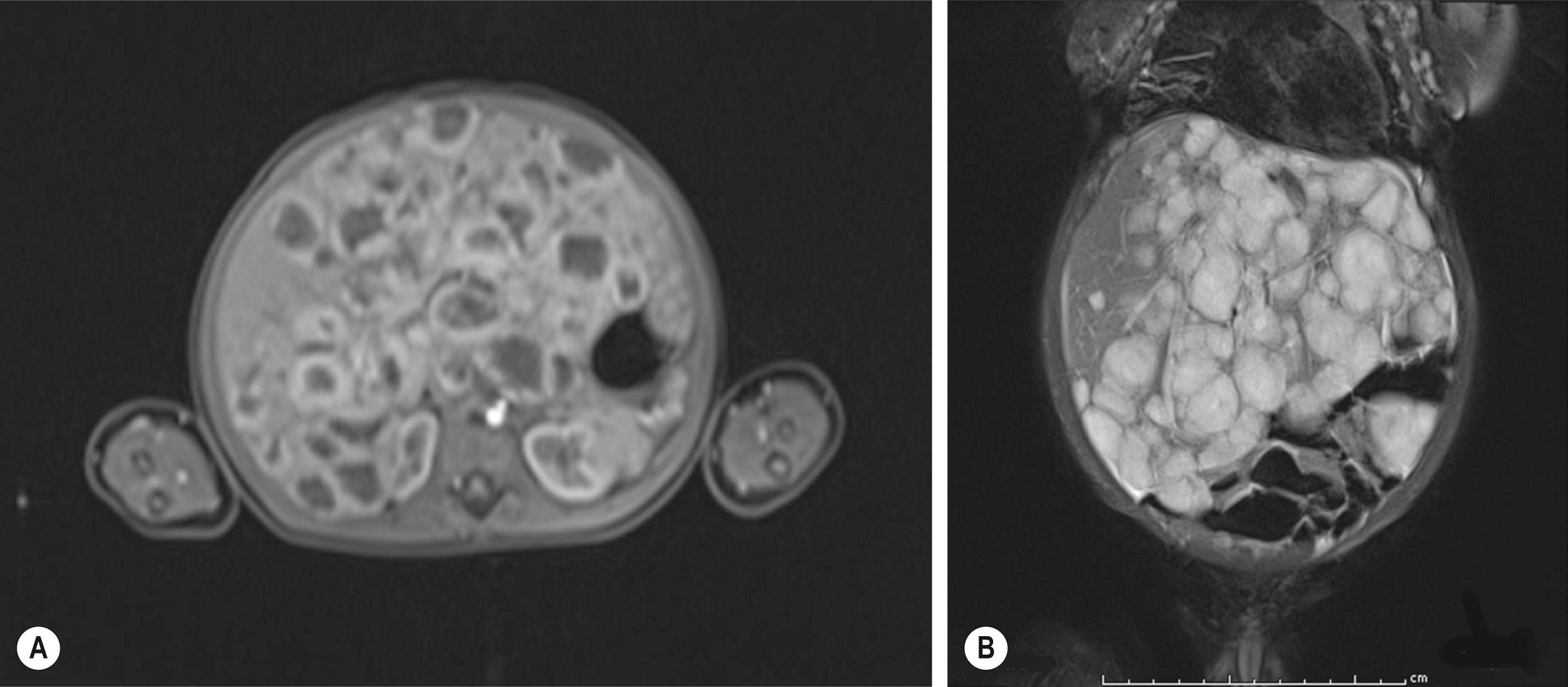
IHH also has been seen in conjunction with both FNH and hepatic mesenchymal hamartoma. These associations are important to remember during the radiologic evaluation of a child with multiple hepatic masses.
Microscopically, the histologic pattern has been divided into type 1 and type 2 lesions. A type 1 lesion consists of a single layer or, occasionally, several layers of flat endothelial cells on a supporting fibrous stroma. In a type 2 lesion (20% of cases), the endothelial cells are pleomorphic, larger, and more hyperchromatic than those seen in type 1 tumors. Also, there are poorly formed vascular spaces that often show tufting or branching. It is thought that the histologic picture of the type 2 lesion is more characteristic of a rapidly proliferating process. The histologic differentiation between a type 2 lesion and an angiosarcoma can be difficult. The addition of a histologic stain for the marker Glut 1 can be helpful in making the diagnosis of IHH. Focal lesions do not stain positive for Glut 1, whereas multifocal lesions will be positive for this marker. Well-preserved bile ducts can frequently be seen near the periphery of a type 1 lesion, whereas bile ducts are absent in a type 2 lesion.
The life cycle of infantile cutaneous hemangiomas is divided into three stages. The first starts usually after birth and is a characterized by a rapid growth phase that lasts about 9–12 months. In the second phase, the hemangioma begins the process of involution. This process can take anywhere from 5–7 years. The last phase is the involuted phase, in which the hemangioma is permanently replaced by fibrofatty tissue. Multifocal and diffuse hepatic hemangiomas tend to follow a similar life cycle. Focal IHHs, however, follow a unique clinical course that is similar to that of a cutaneous RICH lesion, in which involution is seen by 12–14 months of age.
Therapy for IHH depends on the severity of the presenting symptoms and the size of the mass(es) ( Table 66.4 ). Asymptomatic lesions are usually monitored, and no specific therapy is instituted until symptoms develop. However, some reports suggest that multifocal IHH and diffuse IHH are part of a continuous spectrum. If there is evidence that a multifocal IHH may be progressing, the current advice is to start treatment even though the patient may be asymptomatic in order to try to prevent progression to the diffuse type. In addition, all patients with multifocal/diffuse lesions should be screened for hypothyroidism. Asymptomatic patients must be carefully followed with serial US or MRI. This is especially true in children with multifocal lesions. Infants who present with CHF, coagulopathy, or respiratory compromise require intervention.
| Asymptomatic | Asymptomatic: Monitor serial MRI (all types) Asymptomatic multifocal that is growing requires treatment |
| Symptomatic | CHF: Digoxin and diuretics Coagulopathy: Factor replacement Abdominal compartment syndrome: Decompressive laparotomy Medical and/or interventional management |
| Prednisone | Prednisone: 2–3 mg/kg/day and about 45% effective |
| Hypothyroidism | Requires very-high-dose thyroid replacement Dose according to T3/T4 levels Will improve with effective treatment |
| Vincristine | 1–2 mg/m 2 wkly for 2 wk |
| Propranolol | Initially: ECG and ECHO 1–3 mg/kg/day with dose titrated up and can use divided dosing Side effects are rare: Hypotension, hypoglycemia, bradycardia, bronchial reactivity Treat until lesion is fully involuted or until at least 1 y of age |
| Resection | Single lobe involvement Can do in the face of congestive heart failure Rarely needed if pharmacotherapy and embolization are used first |
| Embolization | Must embolize both arterial and portal blood supply Occlude as distal as possible Successful treatment: Rapid clinical improvement without change in size of lesion Consider early in infants with congestive heart failure Not useful in diffuse disease: Does not have significant shunting |
| Liver transplant | Has been successfully performed Usually in patients with diffuse IHH subtype Infants with rapidly deteriorating course with congestive heart failure: High mortality after transplant |
Mortality decreased from a historical 17% to 90% to 9% in multifocal patients and 38% in patients with diffuse disease in one study. Risk factors for death include jaundice and multiple tumor nodules. However, the highest risk factor was the presence of CHF. The children at the greatest risk for death are those with diffuse infantile hemangiomas in whom there is near total replacement of the liver parenchyma with innumerable tumors. These children can develop abdominal compartment syndrome, respiratory distress, heart failure, profound hypothyroidism, and multiorgan failure. Of these risk factors, abdominal compartment syndrome is the greatest prognosticator of death. Aggressive medical therapy along with a decompressive laparotomy should be utilized in these children.
In patients with CHF, stabilization is initiated with digoxin and diuretics. Supportive measures may be necessary in patients with respiratory compromise from either high-output cardiac failure or restriction of diaphragmatic movement by the abdominal mass. Coagulation factors can be administered if a coagulopathy is present.
If there is hemodynamic instability, respiratory compromise, or coagulopathy, therapy directed toward the hemangioma is needed. Since focal IHHs are biologically different than the multifocal/diffuse form, it is not clear that medical management will be helpful. Shunting, with associated cardiac compromise, may best be treated in these children with direct vascular embolization of the IHH using interventional techniques.
The usual initial treatment is prednisone or prednisolone at a dose of 2–3 mg/kg/day. The symptomatic response rate to corticosteroids is reported to be about 45%. If steroids are ineffective, some IHHs have shown a response to vincristine (VCR) at a weekly dose of 1–2 mg/m 2 for 2 weeks.
Propranolol, however, is rapidly becoming the mainstay of treatment with an excellent safety profile. Its proposed mechanisms of action include vasoconstriction, inhibition of angiogenesis, and induction of apoptosis. Léauté-Labrèze et al. reported on the use of propranolol (2 mg/kg/day) for the treatment of severe infantile capillary hemangiomas with excellent results. Marsciani et al. have also successfully used the combination of propranolol (1 mg/kg/day followed by 2 mg/kg/day) and steroids in the treatment of a 2-month-old child with diffuse IHH and noted a dramatic decrease in liver size in only 1 week. Eight infants (multifocal and diffuse) and 13 children (focal, 3; multifocal, 2; diffuse, 8) were also treated with propranolol (2–3 mg/kg/day) with an overall 80% response rate. Treatment typically lasts several months (stopped when the lesion is fully involuted) or until at least 1 year of age before the propranolol is weaned. It also has been reported that during the course of propranolol treatment hypothyroidism will potentially reverse. Complications of propranolol treatment are unusual, but they can include hypotension, hypoglycemia, bronchial reactivity, and bradycardia. Prior to starting therapy, an echocardiogram and an electrocardiogram should be obtained. In order to minimize complications, the propranolol dose should be started low and slowly titrated, using either two or three times a day dosing to minimize any effects on the blood pressure.
Another agent that has been reported to be possibly effective in IHH is curcumin, which is derived from the spice turmeric. Curcumin has antiangiogenic activity and has been tested with some success in a variety of tumors. Hassell and Roanh described its apparent successful use in a 6-month-old patient with a multifocal IHH.
Resection should be considered if the hemangioma is confined to a single lobe. In this situation, a survival rate of 92% has been reported, even if the clinical situation is complicated by CHF. However, some authors think resection is rarely needed if both pharmacotherapy and embolization are aggressively utilized.
Embolization is an important part of the therapy of symptomatic hemangiomas. In order to decrease the chance of the IHH recurring after embolization, it is important that both the arterial and portal vascular supply to the IHH are occluded as distally as possible. After successful embolization, a rapid improvement in the clinical course usually occurs within 5 days despite the fact that there may not be any change in the size of the hemangioma itself. Embolization should be considered early in the treatment course in infants who are in significant cardiac failure. Unfortunately, embolization has not been helpful in infants with diffuse disease because of the absence of significant shunting.
Finally, in infants in whom other modes of treatment have failed, liver transplantation has been used successfully for severe CHF or unremitting coagulopathy (or both). The infants who are most commonly considered for a liver transplant will be those who have the diffuse IHH subtype. However, infants who experience rapid clinical deterioration secondary to CHF are very unstable and therefore have a high mortality after liver transplantation. Careful consideration needs to be given to these children’s transplant candidacy since many patients may be too ill to survive a transplant.
The Vascular Anomalies Center at Boston Children’s Hospital has suggested a treatment algorithm for IHH. All asymptomatic lesions should be monitored with US until resolution. Symptomatic lesions should be treated with steroids. If there is no response, the hemangioma should be embolized. Infants with diffuse disease should have TSH checked and be started on steroids. If abdominal compartment syndrome is present, the infant needs to be evaluated for a liver transplant in case there is no response to steroids.
Malignant transformation of an IHH to an angiosarcoma is rare, but has been reported in older children. For this reason, patients who are asymptomatic or who become asymptomatic after therapy must be monitored for complete anatomic resolution of their hemangioma. Resection of any residual lesion should be strongly considered. In addition, the possibility of malignant transformation should be considered in a multifocal or diffuse IHH patient who is unresponsive to treatment.
Mesenchymal hamartoma is reported to be the third most common hepatic tumor and the second most common benign tumor in children. Of all benign hepatic lesions, mesenchymal hamartomas account for 18–30% of these tumors. It affects males more than females, 80% are diagnosed in the first 2 years of life, and 95% are diagnosed before the age of 5 years.
The reasons for the development of a mesenchymal hamartoma are unclear. One theory is that it results from abnormal development of the primitive mesenchyme, which appears to occur at the level of the hepatic ductal plate, causing abnormal bile ducts. This concept is supported by the histologic finding of a combination of cystic, anaplastic, and proliferating bile ducts, as well as the presence of multiple portal vein branches, within the tumor. It is postulated that the tumor then develops a cystic component as a result of obstruction with dilatation of lymphatics or from occluded bile ducts (or both). The tumor enlarges during infancy as the cystic areas increase in size. Most of the proliferative growth appears to occur before or just after birth because no observable mesenchymal mitotic activity is visible on histologic sections of the tumor.
A second theory is that the lesions are reactive rather than developmental. It is hypothesized that an abnormal blood supply to an otherwise normal hepatic parenchyma causes ischemic necrosis, leading to reactive cystic changes within that portion of the liver. This theory is supported by the findings that hamartomas frequently have a necrotic center, are often attached to the liver by only a thin pedicle, and rarely are found centrally in the liver.
The third theory suggests that a mesenchymal hamartoma is a proliferative lesion. This theory is supported by several findings. Increased fibroblast growth factor-2 (FGF-2) staining has been noted in the proliferating hepatic stellate cells adjacent to the mesenchymal hamartoma. Both the stellate cells in the liver and the spindle cells in the mesenchymal hamartoma tissue strongly express molecules of the FGF-receptor family. It is speculated that a local increase of FGF-2 secretion could stimulate the growth of the spindle cells to form the mesenchymal hamartoma. FGF-2 is also a potent angiogenic factor that could contribute to the intense vascularization seen within some of these lesions. A recent study has identified that the spindle cells that make up the predominant cell line in a mesenchymal hamartoma appear to be derived from hepatic stellate cells.
A possible genetic cause for these abnormalities has been proposed as well. Cytogenetic studies of these tumors have documented several abnormalities, including translocation/deletion of chromosome19q13.4, the loss of heterozygosity due to multiple monosomies, and aneuploidy. Other studies, however, have shown a complex karyotyping picture including possible alterations of the MALATI gene. These findings raise the question as to whether mesenchymal hamartoma is a true neoplasm rather than a developmental lesion. The resulting dysregulated imprinting may be an additional cause for a mesenchymal hamartoma.
The widespread use of prenatal imaging has led to the detection of hepatic masses before birth. Cases of hepatic mesenchymal hamartoma that were diagnosed prenatally have been described. One of the unique characteristics of a prenatal mesenchymal hamartoma is that it can be solid as well as cystic. Unfortunately, the prognosis for these antenatally diagnosed neonates is often poor, with a reported mortality rate of 30% in one study. This mortality is related to (1) the development of CHF from compression of the inferior vena cava (IVC) or the umbilical vein by the tumor mass or (2) the development of hydrops that can occur from fluid losses into cysts or decreased albumin production by the liver. Thus, it may be best to deliver these fetuses before fetal hydrops develops. Another series reported the use of repeated intrauterine aspiration of the cysts until the infant can be delivered and definitive treatment initiated. Although this therapy is appealing, there currently is insufficient evidence to recommend its widespread use.
In the neonate, these lesions can have a varying presentation. High-output cardiac failure, pulmonary hypertension, and disseminated intravascular coagulopathy have been reported in neonates with highly vascular mesenchymal hamartomas. Respiratory distress secondary to a large hepatic mass impinging on the diaphragm also has been described. Unfortunately, the prognosis for infants with a perinatal diagnosis is also poor, with a reported mortality of 35%.
The presentation of a mesenchymal hamartoma in the older child is usually that of progressive abdominal distention or an abdominal mass (or both). There is a significant right-sided predilection for these masses, and they tend to be somewhat more common in males. Occasionally, the associated symptoms of nausea and vomiting can occur secondary to the compression of the stomach and intestine by the expanding mass.
On physical examination, abdominal distention or a palpable abdominal mass is most common. The mass tends to be nontender and fixed. Laboratory studies, including liver function studies, are almost always normal. The AFP level can be moderately elevated, and it returns to normal after resection. However, it is important to remember that patients have been treated with chemotherapy for HB until a tumor biopsy was performed and a mesenchymal hamartoma was subsequently diagnosed.
CT, US, and MRI have all been used for diagnosis. On CT and US, a multiseptated, multicystic, anechoic mass is usually located either in the periphery or scattered throughout the liver. Occasionally the mass is pedunculated ( Fig. 66.4 ). Calcification within the tumor is unusual, but has been reported, so the presence of calcifications within a tumor does not exclude a mesenchymal hamartoma. MR angiography has proven useful in both diagnosis and planning of resection. The finding of a small round hyperechoic parietal nodule on US is usually highly sensitive for making the diagnosis.
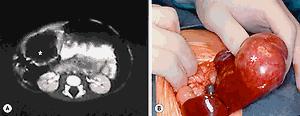
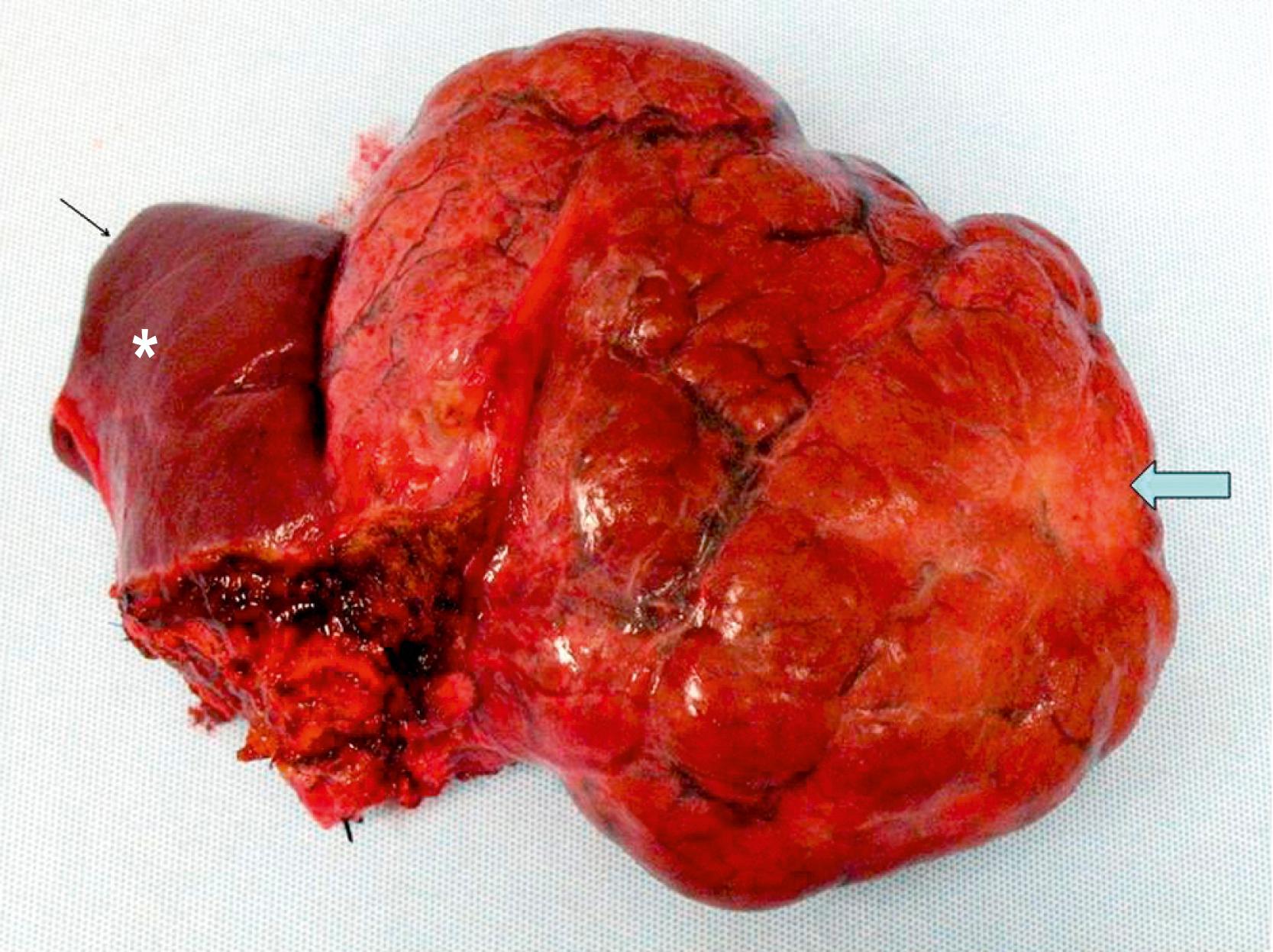
Mesenchymal hamartomas typically are large, well-circumscribed tumors that measure at least 8–10 cm in diameter. Of these tumors, 75% occur in the right lobe of the liver and only 3% are seen in both lobes of the liver. On cut section, multiple cysts can measure from a few millimeters to 15 cm in diameter. These cysts are filled with either serous or viscous fluid separated by loose fibrous and myxoid tissue ( Fig. 66.5 ). The surrounding tissue is yellow-tan to brown and is loose to moderately dense.
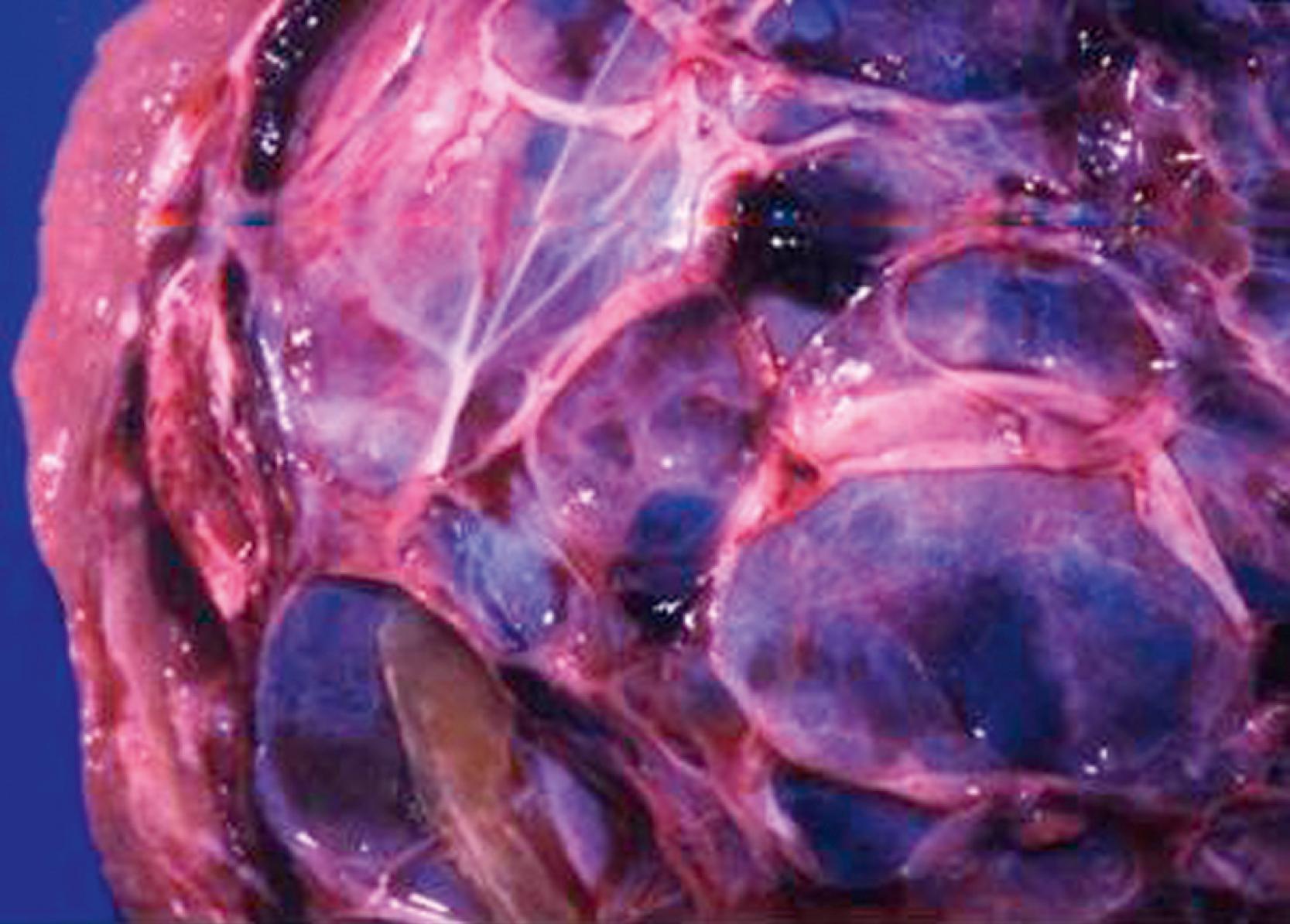
Microscopically, the tissue consists of a mixture of bile ducts, liver cell cysts, and mesenchyme. The cysts may be dilated bile ducts, dilated lymphatics, or amorphous cysts surrounded by mesenchyme. In older patients, the cysts may be lined with cuboidal epithelium ( Fig. 66.6 ). Elongated or tortuous bile ducts surrounded by connective tissue are unevenly distributed throughout the mesenchyme. Typically the hepatocytes appear normal, and they are not a predominant part of the pathologic process. The bile ducts in the periphery of the lesion seem to be undergoing active proliferation.
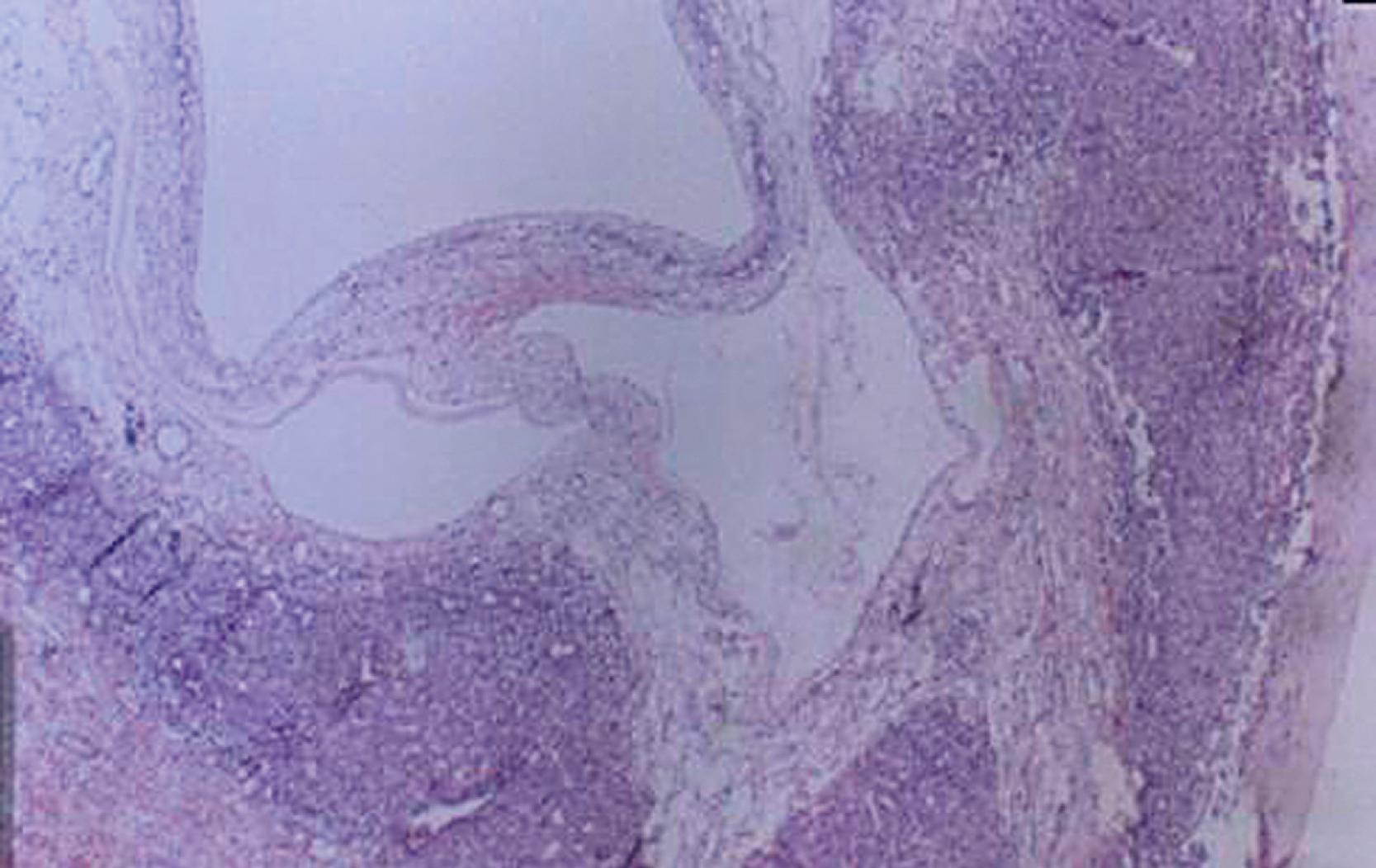
Despite the fact that the majority of these tumors are localized, there have been reports of these tumors being multifocal. This may account for the occasional recurrences that are seen after resection of the primary tumor.
Various management strategies have been used for these lesions, including enucleation for small lesions and marsupialization into the peritoneal cavity (significant recurrence rate) for larger lesions. However, complete excision of the lesion with a margin of normal liver is curative (including the use of liver transplantation for large, bilobar lesions) and is the recommended therapy because of the potential for a coexistent undifferentiated embryonal sarcoma. When massive, multiple cysts are present, one group reported the use of US-guided, intraoperative aspiration of the cysts, which substantially reduced the size of the mass and made the resection much easier. Spontaneous involution of these lesions has been reported but is unusual.
There is now strong evidence that an undifferentiated embryonal sarcoma of the liver can develop within a preexisting mesenchymal hamartoma. This association has occurred both synchronously and metachronously. The evidence for a direct link between a mesenchymal hamartoma and an undifferentiated embryonal hepatic sarcoma comes from the simultaneous finding of both tumors arising within the same mass. Moreover, aneuploidy and similar chromosomal abnormalities involving chromosome 19q13 have been reported in both a hepatic mesenchymal hamartoma and an undifferentiated embryonal sarcoma.
FNH accounts for about 10% of the hepatic tumors in children. The reported age range is 7 months to 16 years, with a mean of 7 years and a female predominance. These tumors are often discovered incidentally. However, 25–92% of patients are symptomatic at presentation. The most common symptom is abdominal pain, but some patients describe decreased appetite, an abdominal mass, weight loss, or a combination of symptoms. Hepatomegaly frequently occurs, and liver function abnormalities are often present.
FNH has been seen in association with a variety of different conditions and situations, including previous liver trauma, other liver tumors, hemochromatosis, Klinefelter syndrome, the use of itraconazole, after a successful Kasai procedure, after liver transplantation, and with cigarette smoking.
The etiology of FNH is not certain, but the evidence suggests it may be a congenital vascular abnormality. These lesions have a single feeding artery, and there is an absence of bile ducts or veins in the lesion. The large artery causes a hyperperfused area of the parenchyma with subsequent growth of the liver tissue around the artery. In addition, FNH has been associated with other vascular lesions, such as hemangiomas, arteriovenous malformations, and hereditary hemorrhagic telangiectasia. Further evidence that FNH is a reactive lesion secondary to vascular anomalies comes from a study in which an increase in the angiopoietin ratio (ANGPT1/ANGPT2) was seen. The ANGPT1 and ANGPT2 genes are necessary for normal vascular development. In FNH, an overexpression of the ANGPT1 gene and an absence of the antagonistic ANGPT2 gene could lead to uncontrolled and disorganized vascular development. This then could lead to hyperperfusion, which could cause a secondary hyperplastic/regenerative response. Although it is not clear that this is the exact pathogenesis of FNH, it certainly suggests that this genetic imbalance may play a causative role. FNH can also develop from an acquired abnormality of the hepatic vasculature. Thrombosis of either a hepatic artery or portal venous branch can initially cause ischemia of a portion of the liver followed by recannulation and reperfusion, which leads to hepatocyte proliferation.
Controversy exists about the relation between oral contraceptive use and the development of FNH. In a case-control study, it was noted that neither menstrual nor reproductive factors correlated with FNH risk. Since the use of oral contraceptives is associated with hepatocellular adenomas (HCAs), a history of oral contraceptive use does not help in distinguishing between these two entities. Even though there is no definite association between oral contraceptives and FNH, oral contraceptives should be stopped as part of any FNH treatment regimen.
In children, an association has been noted between the congenital absence of the portal vein (Abernathy syndrome) and FNH. These patients also have an increased incidence of other solid tumors such as HB, HCC, and HCA. FNH also has been seen, albeit less frequently than HCA, in patients with glycogen storage disease (GSD) type 1.
Over the last 6 years there has been an increasing awareness of the occurrence of FNH in oncologic patients who have completed their therapy. The reported incidence of FNH in the general pediatric population is 0.02% but is 0.45% in the postoncology treatment population. As opposed to nononcologic patients, these patients were usually asymptomatic (80%), and they frequently have multiple FNHs. The cause of these postoncologic FNHs is not known but is thought to be secondary to alterations in liver perfusion that occur as the result of the chemotherapy or radiation that these patients receive. The development of hepatic mass(es) in an oncology patient leads to a diagnostic dilemma between an FNH or a recurrent tumor. One of the higher risk groups for developing an FNH after therapy are patients who were treated for neuroblastoma. There also seems to be an increase in the development of FNHs in children who have received a hematopoietic stem cell transplant. Therefore, a liver lesion that occurs after treatment of a previous cancer does not always mean that there has been a recurrence of the primary tumor. A biopsy of the lesion should always be done before the initiation of further therapy.
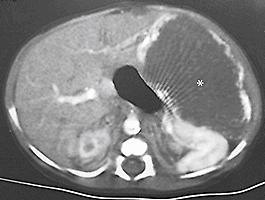
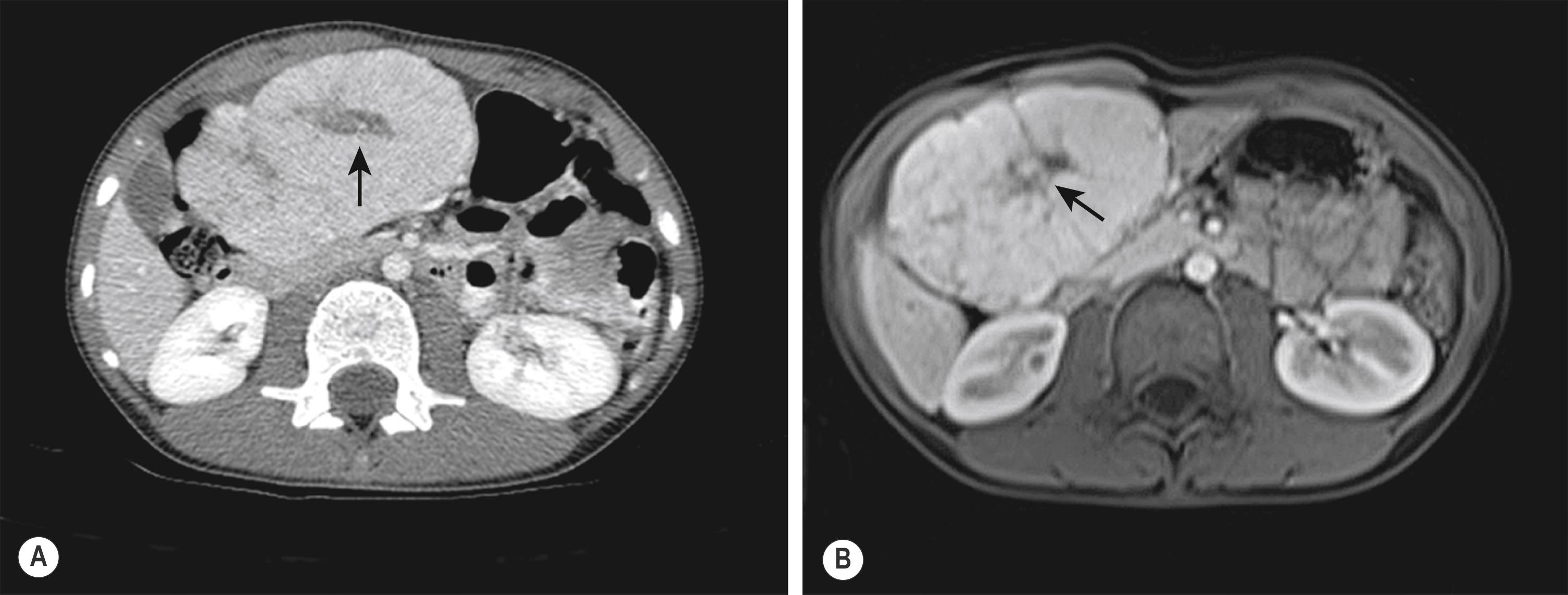
The diagnosis of FNH often requires the use of multiple different imaging modalities. On CT, the classic findings are early enhancement of the lesion and the presence of a central scar ( Fig. 66.7A ). Unfortunately, this pathognomonic association is not always seen. Additional imaging modalities include single-photon emission radionuclide scans with either radiolabeled sulfur colloid or hepatobiliary iminodiacetic acid (HIDA) imaging. These studies usually demonstrate hypervascularization, increased tumor tracer uptake, and a central cold area. Technetium-99m sulfur colloid scanning can be useful in distinguishing between FNH and a hepatic adenoma. FNH lesions take up the tracer because the Kupffer cells in the FNH take up the colloid, but hepatic adenomas do not take up tracer because they lack Kupffer cells. MRI also has been useful when coupled with either gadolinium enhancement or the use of liver-specific contrast agents such as gadoxetic acid (Eovist, Bayer Schering, Berlin, Germany) ( Fig. 66.7B ). During the arterial phase of dynamic contrast enhancement, an FNH will demonstrate distinct hypervascularity; during the hepatobiliary phase the FNH will be hyperintense; and the central scar can be visualized in 60–80% of the cases.
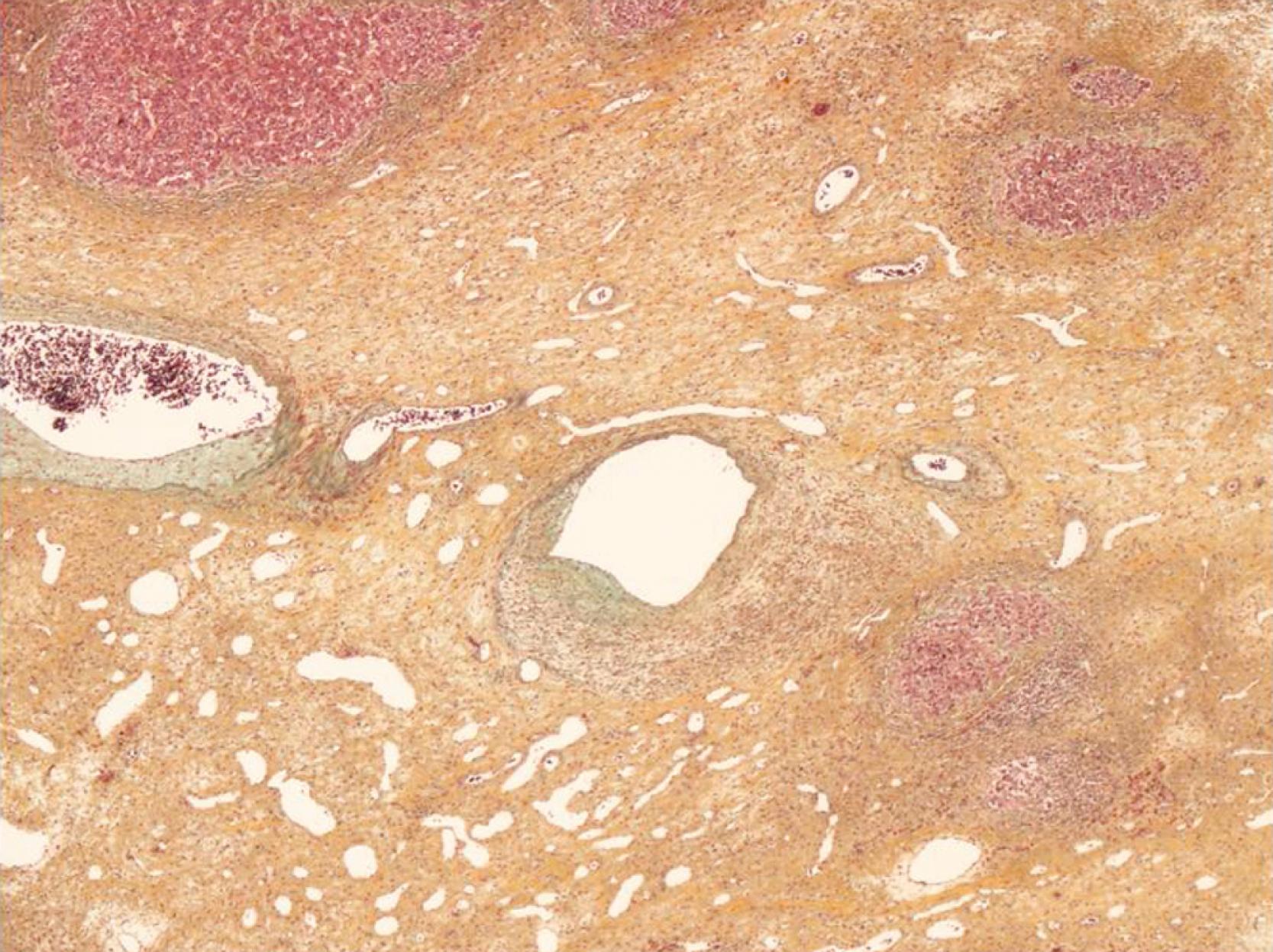
FNH classically is characterized by a nodular architecture, a central or eccentric scar containing malformed vessels that resemble an arteriovenous malformation, and a variable amount of bile duct proliferation ( Figs. 66.8 and 66.9 ). FNH always occurs in the setting of a noncirrhotic liver.
Histologically, the classic form of FNH with a central scar accounts for about 80% of the lesions. Twenty percent are nonclassic, in which the FNH lacks either the nodular architecture or the presence of the malformed blood vessels. These nonclassic lesions are subdivided into three histologic categories: the telangiectatic form, the mixed hyperplastic form, and the adenomatous form. These nonclassic categories always lack a macroscopic scar, and all three forms can be difficult to distinguish from and may be related to an HCA. Immunohistochemical staining for cytokeratins 7 and 19 along with staining for neuronal cell adhesion molecule (CD56) has shown to be very helpful in distinguishing between FNH and HCA. HCAs have been reported to be present in association with FNH in about 4% of the cases.
Recently, there has been a case report of what appears to be malignant transformation of an FNH to a fibrolamellar HCC. Also, FNH has been reported to occur in association with a well-differentiated fibrolamellar HCC. This observation is important to remember in patients who have multiple hepatic nodules such as occurs in children with type 1A GSD.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here