Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Intrahepatic cholangiocarcinoma (IHCC) is a primary adenocarcinoma of the liver. Like other primary adenocarcinomas of the upper gastrointestinal tract, IHCC often presents with symptoms due to advanced local disease or metastatic disease. IHCC is a biologically aggressive disease; complete resection, where possible, is the only known potentially curative therapy. In addition, there are few effective systemic therapies.
IHCC is also known as peripheral cholangiocarcinoma, cholangiolar carcinoma, or cholangiolocellular carcinoma, and these terms have previously been used interchangeably. The term cholangiolocellular carcinoma was first used in 1959 by Steiner and Higginson to describe a subtype of cholangiocarcinoma in which the glands are small and regular with inconspicuous lumens and resemble proliferating cholangioles. Recent evidence suggests that these tumors represent a distinct subtype of intrahepatic cholangiocarcinoma and develop from hepatic stem cells. Current nomenclature uses the term intrahepatic cholangiocarcinoma to define tumors arising from biliary epithelium in intrahepatic bile ducts above the level of the left main and right main ducts. , These tumors constitute 10% of primary hepatic malignancies and 20% of all cholangiocarcinomas. Although much is known about extrahepatic cholangiocarcinoma, IHCC is less well understood.
Hepatic resection for IHCC was infrequently described until recently. Foster and Berman reported only 13 cases in their summary of hepatic surgery in the United States, whereas they presented 112 resections for hepatocellular carcinoma and 47 cases of hepatoblastoma. This low number of resections likely represented the frequency with which advanced disease was diagnosed at presentation. In addition, the recognition of IHCC as a discrete primary liver tumor was slow to occur and many were historically diagnosed as metastatic lesions from unknown primary sites. However, with increased recognition of IHCC significant clinical effort has been directed at developing comprehensive treatment strategies incorporating surgery, systemic therapies, and regional therapies for patients with this malignancy.
IHCC is the second most common primary liver cancer after hepatocellular carcinoma and represents 3% of all gastrointestinal cancers. The geographic incidence of cholangiocarcinoma is highly variable ( Fig. 50.1 ), reflecting the influence of locally prevalent risk factors. The highest incidence rates are observed in Chile, Bolivia, South Korea, and Northern Thailand, where IHCC constitutes 89% of all primary liver cancers. Overall, the worldwide incidence of IHCC has been increasing both in areas of high and low incidence, while the worldwide incidence of extrahepatic cholangiocarcinoma has decreased. , IHCC is more commonly diagnosed in males and usually presents in the 7th or 8th decades of life. However, for individuals with specific risk factors (primary sclerosing cholangitis or untreated choledochal cysts), the average age of primary presentation falls to between 30 and 50 years of age.
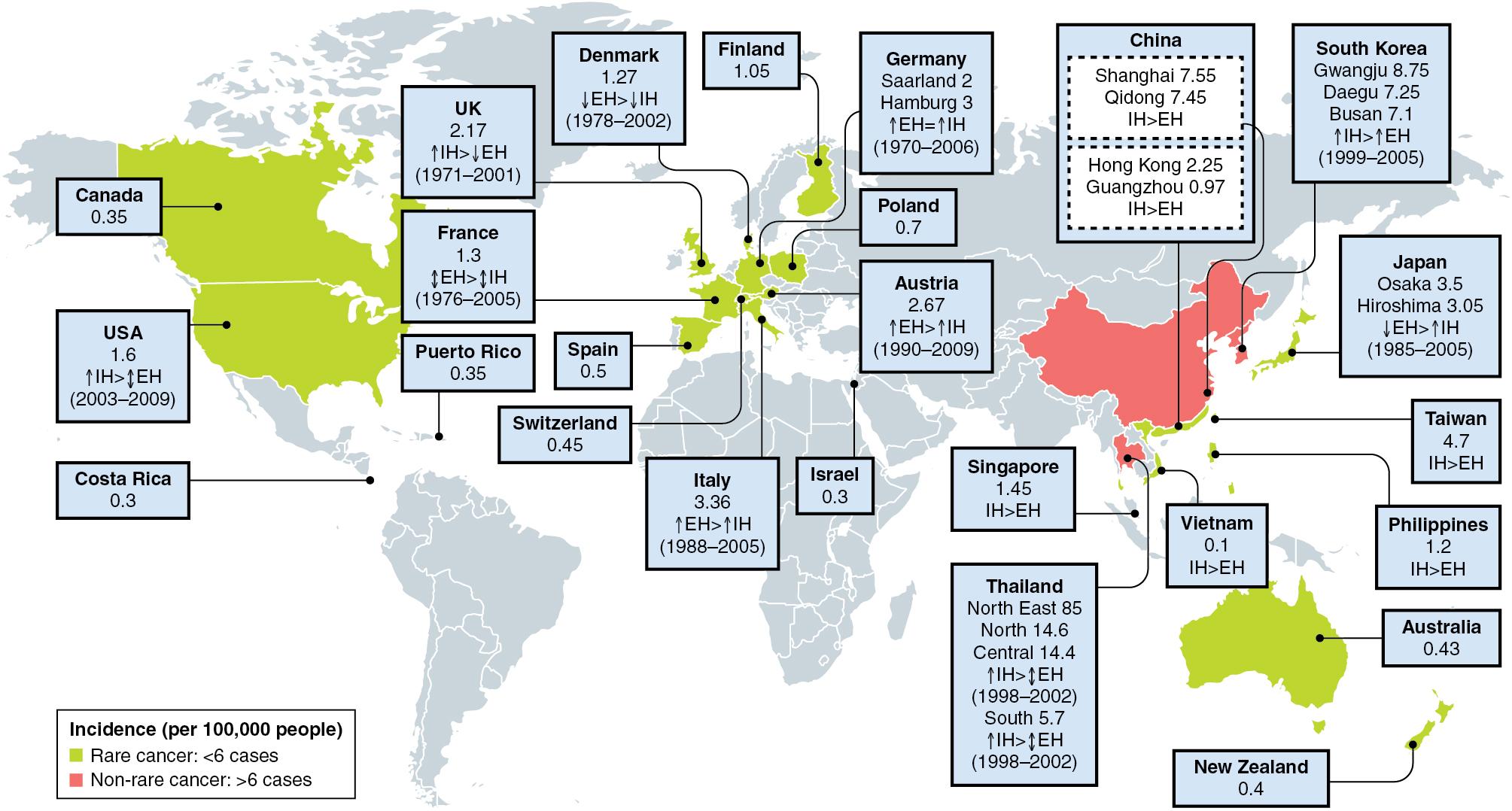
The incidence of IHCC also shows marked ethnic variation. In the United States approximately 5000 patients present annually with a diagnosis of cholangiocarcinoma. The age-adjusted incidence of IHCC is highest in Hispanic Americans, is lowest in African Americans, and shows an annual rate of increase of 5% per year for the last two decades that is most marked in males and in Hispanic Americans. In addition, Hispanics and African Americans have a poorer survival compared with Caucasians. There are significant variations in treatment related to patient socioeconomic status, demographics, and geographic region. ,
In Western countries over 90% of patients presenting with IHCC do not have a recognized risk factor. , However, a number of factors are understood to increase the risk of developing cholangiocarcinoma via sometimes overlapping effects of sustained, chronic inflammation of biliary epithelia, the presence of specific at-risk precursor lesions, and more general conditions that may intensify the effects of known carcinogens. Many of these risk factors contribute to the development of both intrahepatic and extrahepatic cholangiocarcinoma.
Primary sclerosing cholangitis (PSC) is the most common risk factor for cholangiocarcinoma in Western countries , (see Chapter 41 ). The cumulative annual risk of cholangiocarcinoma in patients with PSC is 1.5% per year, and the prevalence of cholangiocarcinoma in these patients is between 8% and 40%. The risk for PSC patients developing cholangiocarcinoma is increased in those with associated inflammatory bowel disease where the 10 and 20 year rates for cholangiocarcinoma is 14% and 31% respectively versus 2% and 2% in patients without inflammatory bowel disease. Cholangiocarcinoma often develops two to three decades earlier in patients with PSC than in those with sporadic tumors (30–50 years versus 60–70 years of age), , and PSC-associated tumors often present with advanced stage disease because of difficulties in detecting malignant change in inflammatory strictures. Surgical treatment can be difficult because of the presence of chronic liver disease. Patients are also often ineligible for orthotropic liver transplantation at presentation, because of the cancer diagnosis, and they consequently have a poor prognosis (see Chapter 105 ). Factors predictive of cholangiocarcinoma in patients with PSC are sudden development of jaundice, weight loss, marked biliary dilation proximal to a dominant stricture, a sudden rise in carbohydrate antigen 19-9 (CA 19-9), the presence of a hypovascular mass with late contrast enhancement on radiologic imaging, and cytologic evidence of dysplasia or malignancy obtained on brushings.
Chronic infection with the liver flukes ( Opisthorchis viverrini and Clinorchis sinensis ) is the most common risk factor for developing cholangiocarcinoma in Southeast Asia (see Chapter 45 ). The mechanism of carcinogenesis is unclear. However, mechanical irritation, excreted metabolic products, and the actions of proinflammatory cytokines, particularly those that stimulate the release of nitric oxide from activated white cells, are all important. , ,
A further parasitic hepatic infestation is caused by the Trematodes Fasciola hepatica or Fasciola gigantica (see Chapters 45 and 71 ). These parasites are widely spread throughout Asia, Africa, the Americas, and Oceania. Fasciola species migrate into the liver from the duodenum and cause hepatic fibrosis. There is no evidence that fascioliasis increases the risk of cholangiocarcinoma per se, although the radiologic and fibrotic pathologic changes accompanying an infection can be difficult to distinguish from carcinoma. ,
Recurrent pyogenic cholangiohepatitis (previously known as oriental cholangiohepatitis) is characterized by recurrent episodes of ascending cholangitis, hepatolithiasis, biliary strictures, and biliary dilation (see Chapters 39 and 44 ). The syndrome is present in one fifth of the population of Southeast Asia and up to 10% of these patients develop IHCC, possibly because of chronic bile stasis leading to chronic infection and inflammation with malignant transformation. Patients present with recurrent episodes of cholangitis and, on investigation, have significant hepatolithiasis and associated inflammatory biliary strictures. The mean interval between the treatment of hepatolithiasis and the development of cholangiocarcinoma is between 3 and 8 years, and patients remain at risk of developing IHCC following hepatectomy if affected parts of the liver remain. Infection with liver flukes may also occur concurrently in many patients, but recurrent pyogenic cholangitis is a separate condition and can develop in the absence of parasitic infection.
Untreated choledochal cysts and Caroli disease carry an increased risk of developing cholangiocarcinoma (see Chapter 46 ). The incidence of cholangiocarcinoma is estimated at between 10% and 20% if the cyst is not resected by a patient age of 20 years. , Correspondingly, patients who have had their cysts resected have a very low incidence of cholangiocarcinoma, although subsequent development of cholangiocarcinoma has been recorded following cyst excision. The mechanism of malignant transformation is not understood but many patients with choledochal cysts have an abnormally high union of the pancreatic and bile ducts, suggesting that biliary stasis and chronic reflux of pancreatic secretions may contribute to the development of chronic inflammation of biliary epithelium. In patients with pancreaticobiliary maljunction alone there is a 7% risk of cholangiocarcinoma.
The risk of developing IHCC is increased in patients with cirrhosis (10.7% versus 0.7% in the general population), , and 1% of explanted cirrhotic livers will harbor a previously unsuspected IHCC (see Chapters 9B , 9C , 68 , 74 , and 89 ). An association between hepatitis C infection and IHCC was postulated in 1991. A case control study from Japan demonstrated that the risk of developing IHCC is 3.5% at 10 years in patients with hepatitis C–related cirrhosis. A separate large cohort study found a significant association between IHCC and hepatitis C after adjusting for potential confounders, including the presence of cirrhosis with a relative risk of 2.55 (95% confidence intervals 1.3–4.95). In these patients carcinogenesis may be due to a direct viral effect since biliary dysplasia is more commonly observed in explanted livers of patients transplanted for hepatitis C–related cirrhosis than in other conditions. IHCC is also more frequent in patients with chronic hepatitis B viral infection (11.5% versus 5.5% in the general population), although the relative risk (RR) is less than for hepatitis C infection (RR 1.8, 95% confidence interval 1.4–2.4). IHCC developing in the setting of hepatitis B infection is more likely to present with a mass-forming growth pattern, which carries a more favorable prognosis following resection. It has been suggested the increasing incidence of IHCC in the West is related to the increasing prevalence of chronic liver disease and chronic viral infection. , Co-infection with both hepatitis B and Opisthorchis viverrini or Clinorchis sinensis is an important factor in the high incidence of IHCC observed in Asia. In addition there is evidence that nonalcoholic steatohepatitis is present in up to 20% of patients with IHCC, although this may reflect lifestyle rather than representing an etiologic factor. Diabetes and obesity have been associated with an increased risk of cholangiocarcinoma, although this association may be stronger for carcinoma of the gallbladder.
Human immunodeficiency virus (HIV) does not cause cirrhosis, but cholangiocarcinomas have been found in up to 0.5% of patients infected with the virus compared with 0.1% in controls, suggesting that it too is associated with an increased risk of biliary carcinogenesis.
The development of biliary cystadenocarcinomas from biliary cystadenomas is rare and, in general, occurs if a cystadenocarcinoma is untreated for many years. Biliary cystadenomas without ovarian stromal tissue appear to be at higher risk of malignant change and patients present with cystadenocarcinomas in the 6th or 7th decades of life, whereas cystadenomas present at an earlier age. IHCCs have also been reported developing in patients with biliary papillomata, , and three related pathologic precursor lesions are now recognized for IHCC—flat intraepithelial neoplasia (BillN), intraductal papillary mucinous neoplasm of the bile duct (IPNB; previously recognized as biliary papillomatosis), and intraductal tubulo-papillary neoplasm (ITNB). All three lesions are often seen in the context chronic biliary inflammation. Progression of bile duct adenoma to IHCC has been reported, and Von Meyenberg complex has also been suggested as a possible premalignant lesion for IHCC due to the occasional association of Von Meyenberg complexes and IHCC.
Thorotrast (thorium dioxide) was used as a radiologic contrast agent between 1928 and 1950. It is an alpha emitter with a biologic half-life of 400 years. Thorotrast accumulates in reticuloendothelial cells in the liver and spleen, and increases the risk of cholangiocarcinoma by 300 times in comparison to the general population. It is now no longer in use, although the latency period of 16 to 45 years means that patients will occasionally still present having received this agent during childhood radiologic examinations.
A number of other agents have been implicated in the development of cholangiocarcinoma. Associations have been shown for asbestos, vinyl chloride, nitrosamines, the antituberculosis agent isoniazid, and first-generation oral contraceptives.
Surgical biliary-enteric bypass and surgical sphincteroplasty increase the risk of developing cholangiocarcinoma. Tobacco smoking is a significant risk factor for the development of cholangiocarcinoma in patients with PSC, although the relationship is less marked in the general population. Finally, congenital hepatic fibrosis has also been associated with an increased risk of developing cholangiocarcinoma in later life.
IHCC develops within the hepatic biliary system in second-order and more proximal bile ducts (septal, interlobular bile ducts, and ductules). Hepatic bile ducts are lined with specialized cholangiocytes whose function centers around the modification of bile at the canalicular surface and detoxification of xenobiotics.
Within the liver the segmental and septal bile ducts are lined with cylindrical mucin-producing cholangiocytes. IHCC that develop from these cells form adenocarcinomas with significant mucus secretion. These tumors may invade along the biliary tree and morphologically form periductal infiltrating or large nodular mass-forming tumors. However, the interlobular bile ducts and ductules are lined with cuboidal cholangiocytes that do not secrete mucus. IHCCs developing from these cells are non–mucin-secreting adenocarcinomas and usually form mass lesions at the periphery of the liver. The ductules also contain hepatic progenitor stem cells, which can display varying degrees of hepatocyte or cholangiocytic differentiation. Cholangiolocellular carcinoma is considered to develop from these progenitor cells and also forms mass lesions at the periphery of the liver. ,
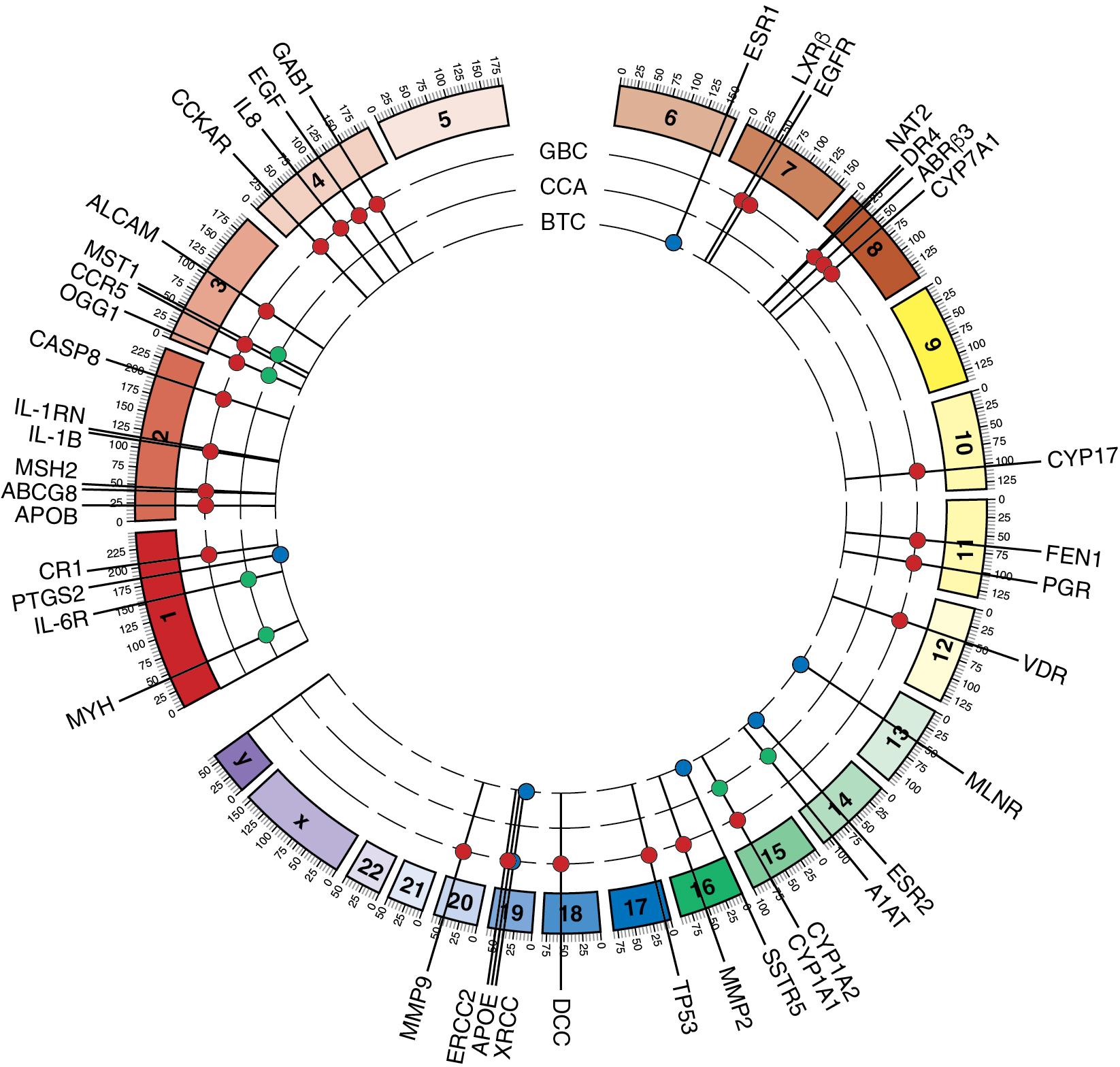
Recent investigations have defined chromosomal aberrations in patients with IHCC (see Chapter 9E ). Deleted genomic areas were defined in 1p, 3p, and 4q, and the main areas of amplification were present in 1q, 7p, 7q, and 8q. , Interestingly, these investigations suggest that hepatocellular carcinoma and IHCC may be closely related at a molecular level as they share chromosomal gains at 1q, 8q, and 17q, as well as chromosomal losses at 4q, 8p, and 17p ( Fig. 50.2 ).
Two distinct genomic classes have been characterized in IHCC (see Chapter 9E ): an inflammatory class with predominant activation of inflammatory pathways (38% of IHCCs) and a proliferation class (62% IHCCs) with predominant activation of oncogenic signaling pathways, the latter correlating with a worse prognosis ( Fig. 50.3 ). Within the inflammatory group of tumors STAT3 activation and overexpression of interleukin-6 (IL-6) is common. IL-6 stimulates cholangiocyte growth via activation of the MAPK pathway and epigenetic control of gene expression and apopotosis. In addition, inflammatory cytokines induce the expression of nitric oxide synthetase, which in turn enhances the expression of COX-2, a proliferative stimulus for cholangiocytes. Nitric oxide synthetase also increases tissue levels of nitric oxide, which promotes DNA damage and inhibits DNA repair mechanisms. ,
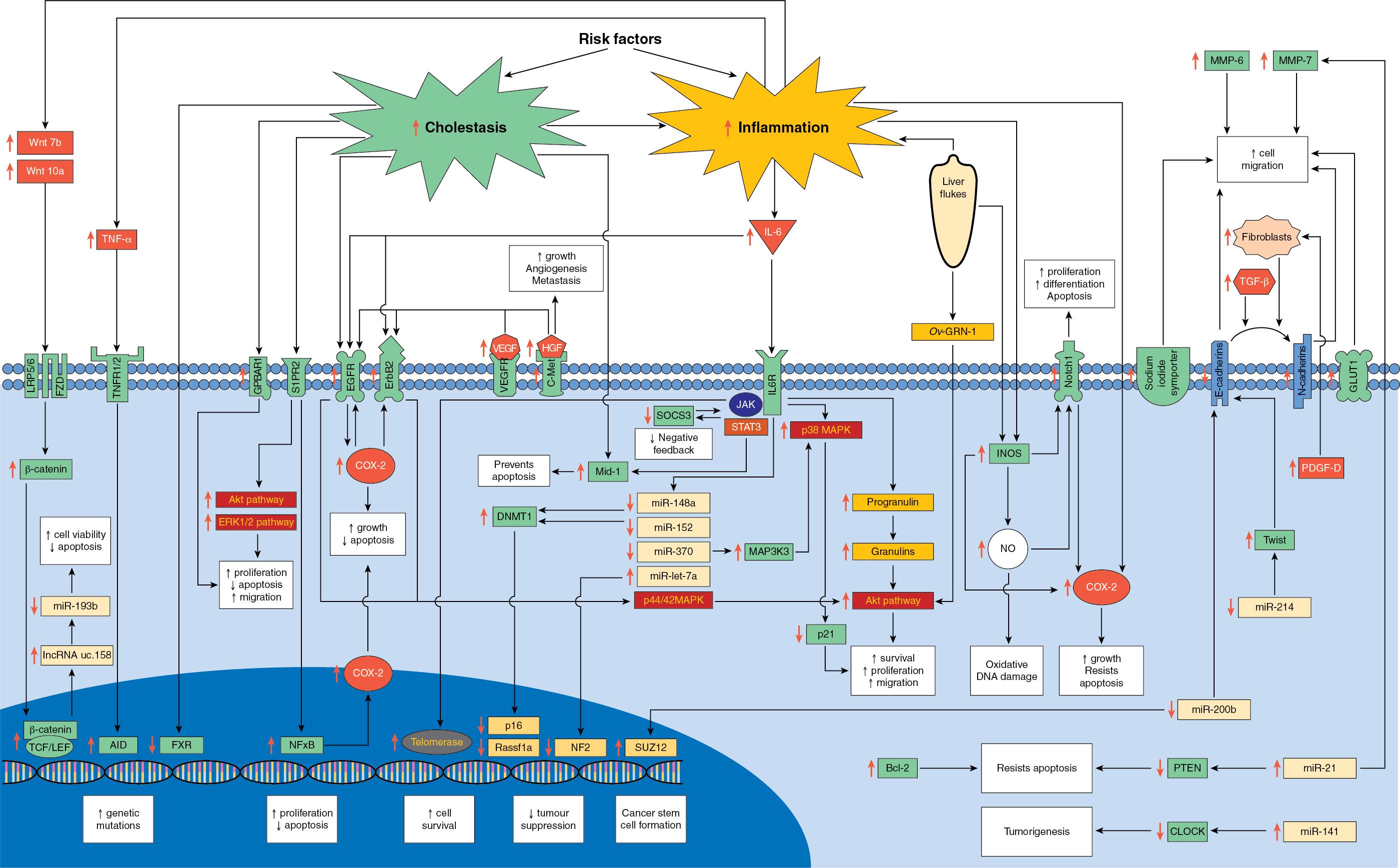
Within the proliferative class of tumors there are widespread alterations in pathways related to DNA repair (TP53), the WNT-CTNNB1 pathway, tyrosine kinase signaling (KRAS, BRAF, SMAD4, and FGFR2), , protein tyrosine phosphatase (PTPN3), epigenetic remodeling factors (IDH1, IDH2), , chromatin-remodeling factors (histone-lysine N-methyltransferase 2C), the SW1/SNF complex, and deregulated Notch signaling. , Variations have also been found in the genetic signaling for human telomerase reverse transcriptidase (TERT) in IHCC associated with chronic hepatitis.
The presence of numerous hormones (estrogens, secretin, gastric, cholecystokinin), bile acids, and growth factors (serotonin, dopamine, leptin, histamine, endothelin-1, opioids, and endocannabinoids) promote or retard proliferation and apoptosis in cholangiocytes, indicating that the development of a malignant phenotype is a complex, multistep process and confirming the clinical observation of significant biologic heterogeneity with IHCC. In addition, it has been suggested that specific cancer-associated fibroblasts, derived from activated hepatic stellate cells that form the characteristic desmoplastic stroma of IHCC, contribute to cholangiocyte proliferation, migration, and invasion. , The extent of neovascularization has also emerged as an important determinant of IHCC prognosis, with patients having a high tumor microvessel density having a poorer outcome following resection than those with low microvessel density.
IHCCs are firm, white sclerotic tumors often with associated satellite lesions nearby (see Chapter 47 ). The Liver Study Group of Japan has subdivided IHCC into four categories , :
Mass-forming (MF) type consisting of a well-delineated, firm, nonencapsulated mass with no discernable connection to a bile duct. This is the most common subtype and accounts for 65% of all IHCCs.
Periductal infiltrating (PI; 6% of IHCC) type characterized by growth spreading along portal tracts with biliary stenoses and proximal biliary dilation.
Intraductal growth (IG; 4% of IHCC) type, usually a polypoid or papillary lesion growing within the lumen of large bile duct.
Mixed growth pattern which demonstrates features of several of the other growth patterns and accounts for 25% of IHCC. This morphologic classification does have prognostic implications with the MF type having a better 5-year survival than the PI type and lymph node metastases being rare in the IG type compared with the other histotypes.
Intrahepatic metastases occur commonly with IHCC, usually on the basis of vascular invasion. Metastases to intraabdominal lymph nodes are present in up to 75% of cases at presentation, and up to two thirds of patients may have evidence of remote organ metastases, most commonly lung and bone, at presentation. , Nozaki et al. showed that there were significant differences in lymphatic spread between left lobar and right lobar tumors. Patients with right lobar tumors always had lymph node metastases in the hepaticoduodenal ligament; in patients with left lobar tumors, 50% of the nodal metastases were found distant from the hepaticoduodenal ligament in the cardia and around the lesser curvature of the stomach. Furthermore, in these patients, no lymph node metastases were present in the hepatoduodenal ligament.
Histologically, IHCCs are usually well-to-moderately differentiated adenocarcinomas with varying degrees of desmoplasia. Sempoux et al. proposed a detailed subclassification of IHCC ( Box 50.1 ). Of these, the pathologic classification of unconventional IHCC is the subject of significant investigation. These tumors most commonly develop on a background of chronic liver disease and cirrhosis and may mimic hepatocellular carcinoma in appearance (see Chapter 47 ).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here