Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Malignant disease resulting in proximal or high bile duct obstruction ( Fig. 52.1 )—that is, in close proximity to or involving the biliary confluence—may arise from a variety of cancer types and is a common clinical problem. Historically, the proximal biliary tree was defined as above the level of the cystic duct insertion into the common hepatic duct (CHD). Given the wide anatomic variability of the cystic duct/CHD confluence (see Chapter 2 ), this definition is not accurate, and high bile duct obstruction is best thought of as obstruction involving the hepatic confluence and the 2 to 4 cm of CHD distal to the confluence.
Although frequently seen with hilar cholangiocarcinoma and intraductal tumor (see Chapters 50 and 51 ), hilar obstruction can result from other common malignancies, such as colorectal, breast, and pancreatic cancers (see Chapters 62 and Chapter 90, Chapter 91, Chapter 92 ). Significant technical progress has occurred in both endoscopic (see Chapter 30 ) and percutaneous approaches to biliary drainage (see Chapter 31 ), allowing for safer palliative treatment of patients with such obstructions. Because these patients may be asymptomatic at presentation, the goals of treatment should be clearly defined before the physician commits the patient to any biliary drainage procedure. The most important question concerns resectability. With the exception of clearly palliative situations, these patients are ideally discussed in a multidisciplinary group with hepatobiliary surgeons, interventional radiologists, oncologists, and gastroenterologists to outline a plan of treatment. A thorough understanding of this plan (specifically involving details of the potential surgical approaches), and of the patient’s prognosis, facilitates concomitant development of a strategy for drainage, when indicated. Accepted indications for palliative biliary drainage include intractable pruritus, cholangitis, the need to restore liver function to allow for administration of chemotherapeutic agents with biliary metabolism/excretion, access for intraluminal brachytherapy, and diversion for bile leak. Given the availability of high-quality magnetic resonance cholangiopancreatography (MRCP; see Chapter 16 ), direct cholangiography as a diagnostic tool is rarely warranted (see Chapter 20 ). ,
Neither isolated hyperbilirubinemia nor imaging findings of bile duct dilation are an indication for biliary drainage. Pruritus, cholangitis, and the need to lower the bilirubin to administer certain chemotherapeutic agents, on the other hand, are all accepted indications for biliary drainage. Postoperative bile leaks that require drainage for diversion may develop in patients who have undergone biliary-enteric bypass as part of curative resection for a benign or malignant lesion (see Chapter 28 ). In some cases, access to the biliary tree may be undertaken as a method of delivering local treatment for primary bile duct cancer, such as brachytherapy or photodynamic therapy. Many physicians have the impression that patients feel better and have improved performance status after relief of jaundice, but this has never been definitively demonstrated in clinical studies. Indeed, in a prospective trial, Robson and colleagues showed that percutaneous drainage of high biliary obstruction does not significantly improve the quality of life (QOL) of patients with malignant biliary obstruction, except for the minority with associated pruritus. Controversy remains with regard to the role of biliary drainage before surgery. Preoperative drainage of the future remnant liver is often performed for jaundiced patients that require major hepatectomy , or before preoperative portal vein embolization (see Chapters 51B , 102C , and 119B ). When preoperative drainage is necessary for relief of symptoms (pruritus, cholangitis), if neoadjuvant therapy is planned, or when surgery will be delayed, endoscopic methods are preferred for low bile duct obstruction because of the lower complication rate, whereas high obstruction is treated with carefully targeted percutaneous methods to reduce the risk of cholangitis in undrained segments. Patients with Bismuth type II and higher levels of obstruction are at high risk for requiring multiple drainage procedures (see Chapter 42 ).
Patients with low bile duct obstruction are typically treated endoscopically. Patients with high bile duct obstruction, particularly when the obstruction extends above the hilus, have traditionally been treated percutaneously. The success rate of percutaneous drainage is higher, and the complication rate lower, when compared with endoscopic methods. , This perspective is evolving while technologic advances in endoscope design occur and endoscopists become better trained and more experienced in wire-guided procedures, with access to better drainage devices and accessories. Currently, however, high bile duct obstruction, with rare exception, should be approached percutaneously. This has been called into question with a randomized trial of percutaneous versus endoscopic preoperative drainage in patients with hilar cholangiocarcinoma that was stopped prematurely because of high overall mortality in the percutaneous group. The reported 11% mortality in the percutaneous group is vastly higher than any recent report on percutaneous biliary drainage. It is also well outside the 1.7% mortality threshold established by the Society of Interventional Radiology in 2010, calling into question the experience and expertise of those involved in performing the trial.
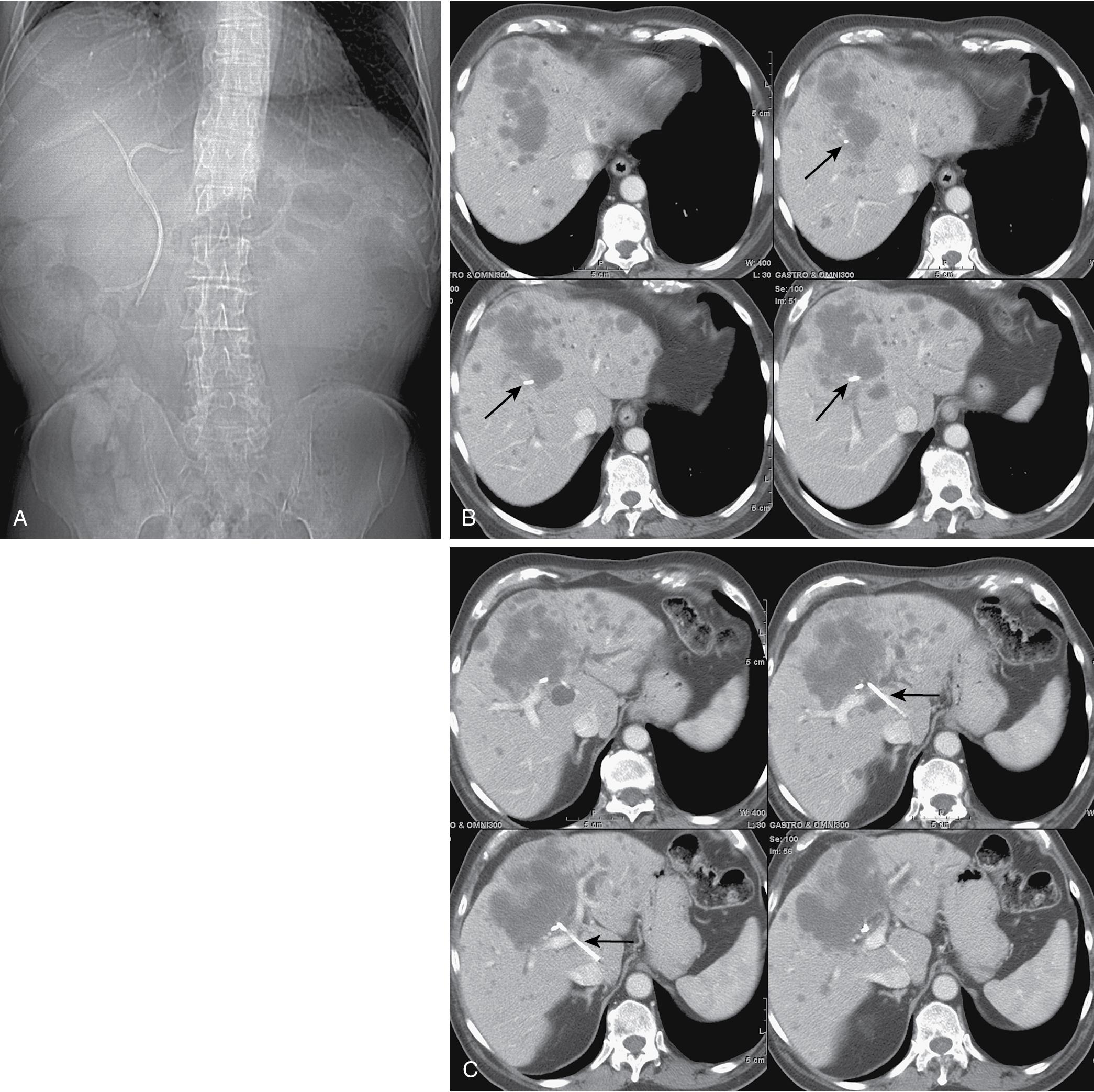
Two major drawbacks to endoscopic drainage for high bile duct obstruction are the lack of ability to reliably target a specific area of the liver for drainage and the risk of enteric contamination, by retrograde injection of contrast, of parts of the biliary tree that will not be drained. In a patient thought to have undergone successful endoscopic stenting of the right and left liver, repeat imaging may reveal that this is not the case at all ( Fig. 52.2 ). Despite improvements in endoscopic techniques, a paper from Wiggers et al. in 2015 reported a high risk of inadequate preoperative endoscopic drainage, with 108 of 288 (38%) patients with perihilar cholangiocarcinoma requiring additional percutaneous procedures subsequent to endoscopic retrograde cholangiopancreatography (ERCP).
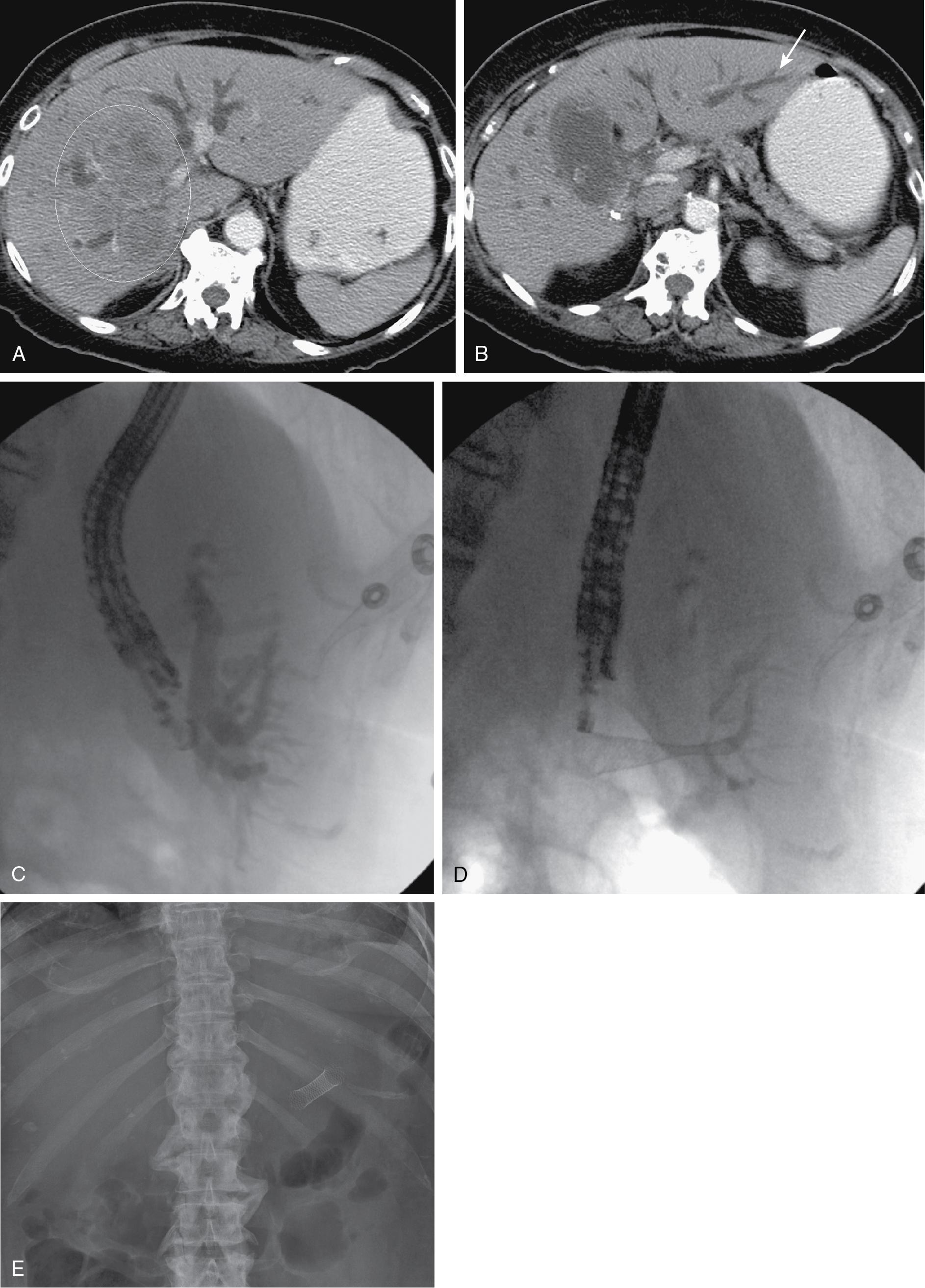
Development of the linear-array echoendoscope has allowed for the expansion of endoscopic ultrasound–guided biliary drainage procedures, including choledochoduodenostomy and hepaticogastrostomy. Hepaticogastrostomy may be considered when a patient is best treated by draining the left lateral segment, anatomy is favorable, and the percutaneous approach is technically difficult, or when the risk of contaminating the right-sided bile ducts is thought to be prohibitive. This is particularly the case when draining the right liver would be of no clinical benefit, either because of replacement of the liver by tumor or occlusion of the right portal vein ( Fig. 52.3 ). High technical success rates with few complications are reported by some authors, , whereas others report high technical success with less favorable complication and stent patency rates. Most operators are early on the learning curve at present. When technically feasible, and in the hands of highly trained endoscopists, this procedure can be gratifying for the patient, with relief of pruritus and lowering of bilirubin without the added burden of a percutaneous catheter.
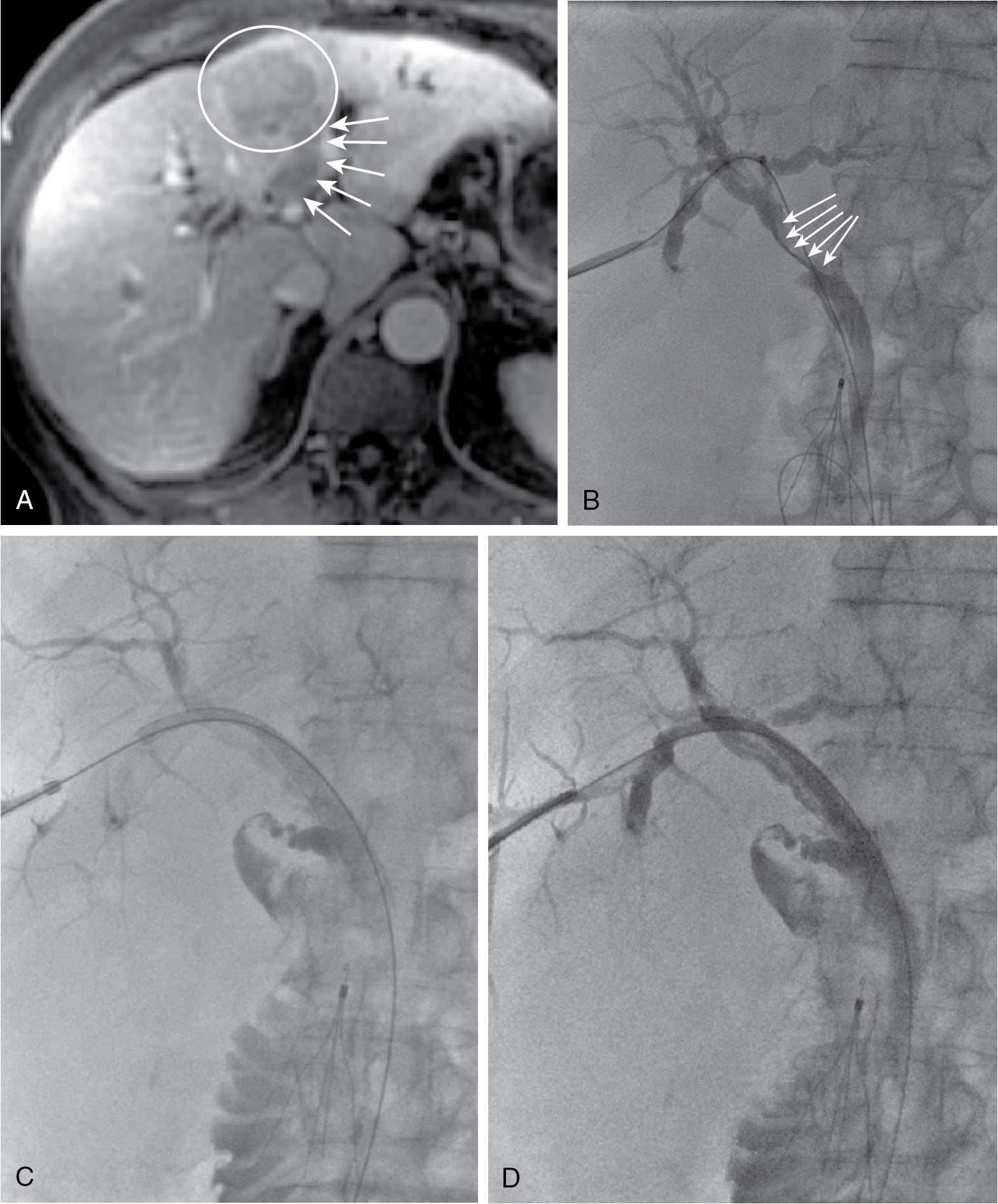
Endoscopic drainage is rarely indicated in patients with papillary intraductal tumor because of inevitable tumor ingrowth into the metallic stent with early stent occlusion. Intraductal tumor can arise from cancers other than papillary cholangiocarcinoma, including colorectal metastases, gallbladder cancer, and hepatocellular carcinoma. When intraductal tumor occurs in the setting of metastatic colorectal cancer or hepatocellular cancer, it is usually the direct extension of a parenchymal metastasis into the duct ( Fig. 52.4 A). Patients with intraductal tumor often require permanent indwelling catheter drainage, with an indwelling, multi-sidehole catheter allowing bile to drain around the intraductal tumor, but they may be stented when the intraductal tumor can be effectively excluded (see Fig. 52.4 B–D).
A realistic idea of what is feasible endoscopically and percutaneously is important when the initial decision is made regarding who will treat the patient. This depends on a thorough understanding of the goal of treatment as well as knowledge of the skill level of the interventional radiologists and endoscopists available to care for the patient.
The importance of excellent preprocedure imaging in patients with bile duct obstruction cannot be overemphasized. This imaging should include, at a minimum, a contrast-enhanced computed tomography (CT) scan of the abdomen (see Chapter 16 ). Ultrasound is often used to establish the presence of dilated bile ducts, identify the level of obstruction, evaluate portal vein patency, and demonstrate intraductal tumor, but ultrasound is not adequate for drainage planning. Although MRCP provides a detailed three-dimensional (3D) rendering of the obstructed bile ducts and is effective at delineating intraductal abnormalities and depicting isolated bile ducts ( Fig. 52.5 ), it excludes certain details that can serve as targeting aides for percutaneous drainage. These shortcomings of MRCP include poor visualization of surgical clips, dystrophic calcifications, and bony landmarks (see Chapter 16 ). In addition, if the patient is unable to breath-hold during image acquisition, MRCP will be suboptimal. CT scans performed on multi-slice scanners in a picture-archiving and communication system (PACS) environment allow a 3D rendering of the anatomy, while identifying relevant landmarks that can be used for targeting and demonstration of other structures that should be avoided, such as liver tumors and bowel. Also, as with magnetic resonance imaging (MRI), one may identify ancillary important findings, such as ascites, hepatic lobar atrophy, and portal vein occlusion or encasement. All this information is critical to make prognostic assessments and determine the best approach for drainage.
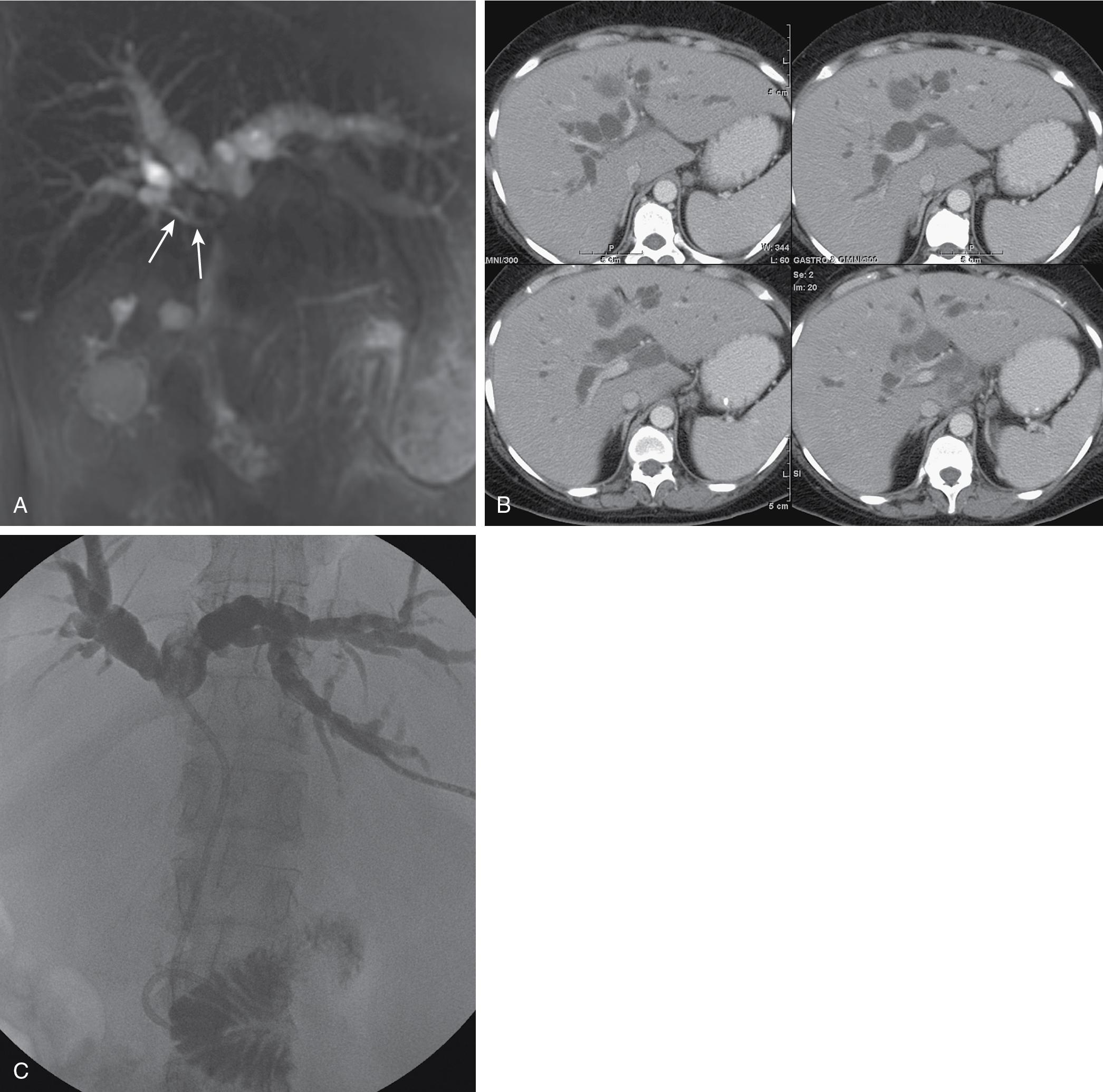
The amount of functional parenchyma that can be drained should be estimated. Functional parenchyma is that part of the liver that is not replaced by tumor and that has an intact portal venous supply and hepatic venous drainage. If 75% of the hepatic parenchyma is replaced by tumor, even if it were possible to drain the entire biliary tree, it is unlikely that normal hepatic function or normalization of serum bilirubin level would result. The portal vein provides the trophic blood supply to the liver, and occlusion results in atrophy of the affected segment(s), particularly when the ipsilateral bile duct is also occluded (see Chapter 5 ). Atrophy may be recognized by the diminutive size of the involved part of the liver, accompanied by crowding of the bile ducts ( Fig. 52.6 ; see Chapters 16 and 51A ). When portal vein occlusion causes atrophy, drainage of the atrophied liver does not improve liver function. In addition, it is frequently difficult to manipulate catheters through the obstructed/atrophied portion of the liver into the central biliary tree. The end result is an external drainage catheter that provides no clinical benefit to the patient and only serves to risk complications and degrade QOL.
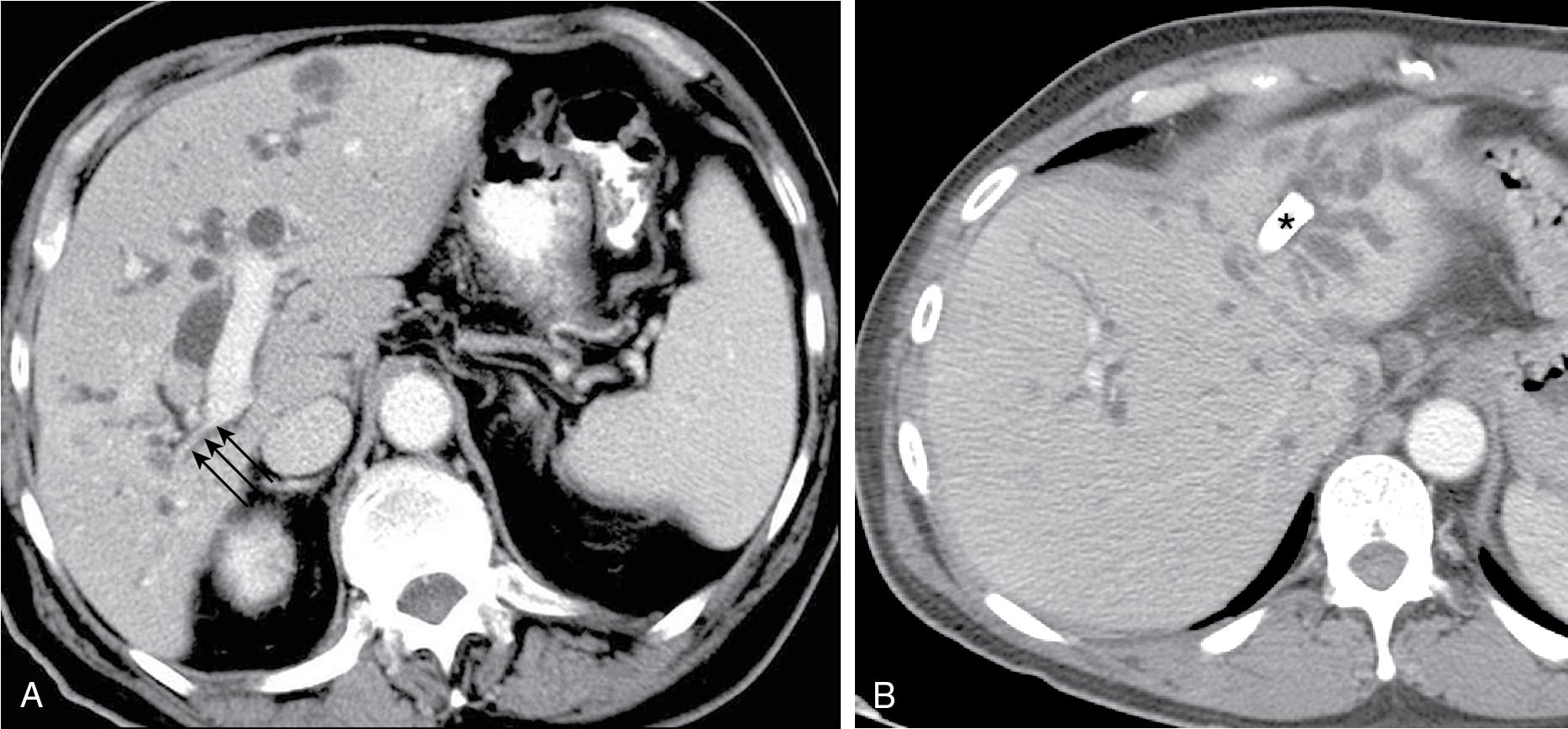
Central tumors frequently obstruct the right and the left biliary tree, isolating them from one another. Obstruction may extend even higher, isolating the ductal system at the sectoral, segmental, or subsegmental level. When drainage is undertaken for relief of pruritus, draining even a segment of the liver may result in relief of symptoms. , When isolation is present, and drainage is undertaken to lower the serum bilirubin, as mentioned previously, it is important to estimate how much functional parenchyma can be drained with one catheter. Drainage of at least 75% of the liver is associated with a significantly increased probability of returning the serum bilirubin to normal or near-normal levels. In some cases, this goal may be impossible to achieve, unless more than one drainage catheter or stent is placed. Recognition of this allows for informing both the patient and the referring physician about the potential need for more than one catheter or stent.
Cholangitis is rarely the primary indication for drainage in patients with malignant bile duct obstruction, except in patients who have undergone prior biliary instrumentation and in those who have a contaminated biliary tree related to previous biliary-enteric bypass, cross-ampullary stenting, or sphincterotomy. Drainage of an isolated region of the liver, either percutaneously or endoscopically, may contaminate other functionally “isolated” parts of the biliary tree. In this case, subsequent cholangitis may drive the placement of additional catheters to relieve infection. Widespread contamination of a severely isolated biliary tree with drainage of only part of the obstructed system can result in chronic recurrent cholangitis. Whether induced by endoscopic or percutaneous means, this situation can be difficult to manage and should be anticipated and avoided if possible.
Preoperative labs for biliary drainage include a complete blood count, hepatic function panel, and coagulation studies. The serum bilirubin can change rapidly when patients become obstructed. Because decisions regarding the need for further drainage in high obstruction with isolated bile ducts depend, in part, on the serum bilirubin, it is important to draw this reference laboratory value within 24 hours of drainage. The bilirubin is typically rising at the time of drainage and the serum bilirubin may be higher the morning after the drainage procedure than the morning of the procedure. Serum bilirubin values drawn 48 hours after drainage more accurately predict effectiveness of drainage. The lack of bile salts in the intestine impairs vitamin K absorption and may result in elevation of the prothrombin time (PT), international normalized ratio (INR), and the partial thromboplastin time (PTT); therefore these values should also be checked within a few days of the procedure. Coagulation abnormalities may be exacerbated if there is also poor hepatic synthetic function. This can be corrected by the administration of vitamin K or fresh frozen plasma, depending on the urgency of drainage.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here