Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Peripheral artery disease (PAD) is a process characterised by the formation of atheromatous plaque and calcification within the arteries of the lower extremity. Typically, this leads to luminal stenosis or occlusions of both large and small arteries. Patients who suffer from the condition may either be asymptomatic or complain of a broad variety of clinical symptoms ranging from calf claudication to ischaemic tissue loss. Interventional therapy for PAD has traditionally been reliant upon open vascular surgery, such as endarterectomy and bypass to achieve definitive revascularisation. However, since the advent of peripheral angioplasty in the mid-1960s, there has been a large increase in its use, driven in large part by technological advances in what we now know as the field of endovascular surgery. Herein we seek to outline the contemporary literature and recommend an evidence-based approach to revascularisation of patients with chronic lower extremity ischaemia.
The spectrum of symptoms that may occur when lower extremity PAD becomes symptomatic range from intermittent claudication (IC), through to ischaemic rest pain, ulceration and gangrene.
IC is muscular discomfort in a particular compartment of the lower limb that occurs reproducibly upon exercise and is relieved after a short period of rest. It occurs when muscular oxygen demand increases above its limited metabolic supply. It most often affects the calf muscles but can also affect more proximal muscle groups such as the thigh, hip and buttock. It is typically described as cramp, ache, fatigue or a combination of the three, and is graded according to severity ( Table 4.1 ). The decision to intervene is based upon an individualised patient assessment that considers severity of symptoms, quality of life (QoL), risk of intervention and predicted durability of treatment.
| Fontaine’s classification | Rutherford-Becker classification | ||
|---|---|---|---|
| Stage | Symptom | Category | Symptom |
| I | Asymptomatic | 0 | Asymptomatic |
| II | Intermittent claudication | 1 | Mild claudication |
| 2 | Moderate claudication | ||
| 3 | Severe claudication | ||
| III | Ischaemic rest pain | 4 | Ischaemic rest pain |
| IV | Ulceration or gangrene | 5 | Minor tissue loss |
| 6 | Major tissue loss | ||
| Trial | Year | Study design | Patients ( n ) | Mean lesion length (mm) | Endpoints | |
|---|---|---|---|---|---|---|
| YUKON-BTK (Rastan et al. ) |
2011 and 2012 | RCT | Sirolimus-eluting stent (polymer free) vs. Bare-metal stent |
161 (75 CLI; 86 IC) |
31 | Primary patency (12 month); 81% vs. 56% ( P = 0.004) Secondary patency (12 month); 92% vs. 71% ( P = 0.005) TLR (12 month); 9.7% vs. 17.5% ( P = 0.29) |
| Limb salvage in CLI (2.8 y); 97.4% vs. 87.1% ( P = 0.10) Freedom from amputation, TVR, AMI and death (2.8 y) 65.8 vs. 44.6% ( P = 0.02) |
||||||
| DESTINY (Bosiers et al. ) |
2012 | RCT | Everolimus-eluting stent vs. Bare-metal stent |
140 (all CLI) |
17 | Primary patency (12 month); 85% vs. 54% ( P = 0.0001) LLL (12 month); 0.78 mm vs. 1.41 mm ( P = 0.001) TLR (12 month); 8% vs. 35% ( P = 0.005) Limb salvage (12 month); 99% vs. 97% (NS) Death (12 month); 18% vs. 16% ( P = 0.06) |
| ACHILLES (Scheinert et al. ) |
2012 | RCT | Sirolimus-eluting stent vs. Angioplasty |
200 (CLI and IC) |
27 | Primary patency (12 month); 78 vs. 58% ( P = 0.019) TLR (12 month); 10% vs. 17% ( P = 0.257) Limb salvage (12 month); 86% vs. 80% ( P = 0.3) Death; 10% vs. 12% ( P = 0.82) |
| IDEAS (Siablis et al. ) |
2014 | RCT | Zotarolimus-, sirolimus- or everolimus-eluting stents vs. Paclitaxel-coated balloon | 50 (CLI and IC) |
148/127 | Binary restenosis (6 month); 28% vs. 57.9% ( P = 0.0457) TLR (6 month); 7.7% vs. 13.6% ( P = 0.65) |
Chronic limb-threatening ischaemia (CLTI) is the chronic manifestation of an ischaemic process (>2 weeks) with symptoms that include rest pain, ulceration or gangrene. Whilst ulceration may be triggered by a non-ischaemic process such as trauma or neuropathy, a non-healing ulcer perpetuated by ischaemia is also considered CLTI. Ischaemic rest pain may be constant or nocturnal. It is typically present in the distal portion of the foot and often relieved by hanging the leg dependent. It should not be confused with neuropathic symptoms, which are common in diabetes mellitus. When gangrene occurs, it typically affects digits and pressure areas such as the heel. Often forming an eschar, it may remain dry or develop an underlying suppurative infection.
In 2019 the European Society of Vascular Surgery, Society for Vascular Surgery (SVS) and the World Federation of Vascular Societies published guidelines to address the growing prevalence of CLTI, with its associated morbidity, mortality, increased healthcare costs and variability of practice (further information in Chapter 2 ). Key components of that document were the promotion of the term CLTI to replace critical limb ischaemia (CLI), as the authors felt the term CLI ‘…connotes specific hemodynamic thresholds and fails to recognise the full spectrum and inter-relatedness of components beyond ischaemia that contribute to major limb amputation…’. They also incorporated the SVS Lower Extremity Threatened Limb Classification System known as WIfI ( Wound Ischaemia foot Infection ) as part of disease staging, and the Global Limb Anatomic Staging System (GLASS) as a new anatomical system to facilitate clinical decision making in CLTI.
The WIfI classification system was designed as a pathophysiological grading tool, analogous to the TNM staging system for malignancy. With four different grades for each of wound, infection and ischaemia there are 64 combinations and individual classes. Each class is placed into a particular clinical stage. To gauge the significance of those grades, an expert Delphi consensus panel was asked two questions regarding each combination ( Fig. 4.1 ). The first question was ‘what is the perceived risk of amputation, assuming medical therapy alone’, then ‘what is the perceived benefit from revascularisation’. Each response was allocated to one of four predictive clinical stages (very low risk– Clinical Stage 1, low risk-2, moderate risk-3 and high risk-4). A meta-analysis of predominantly retrospective and observational studies evaluated the prognostic value of WIfI, finding the likelihood of amputation in patients with CLTI increased with higher WIfI stage, recommending a more rigorous prospective evaluation.
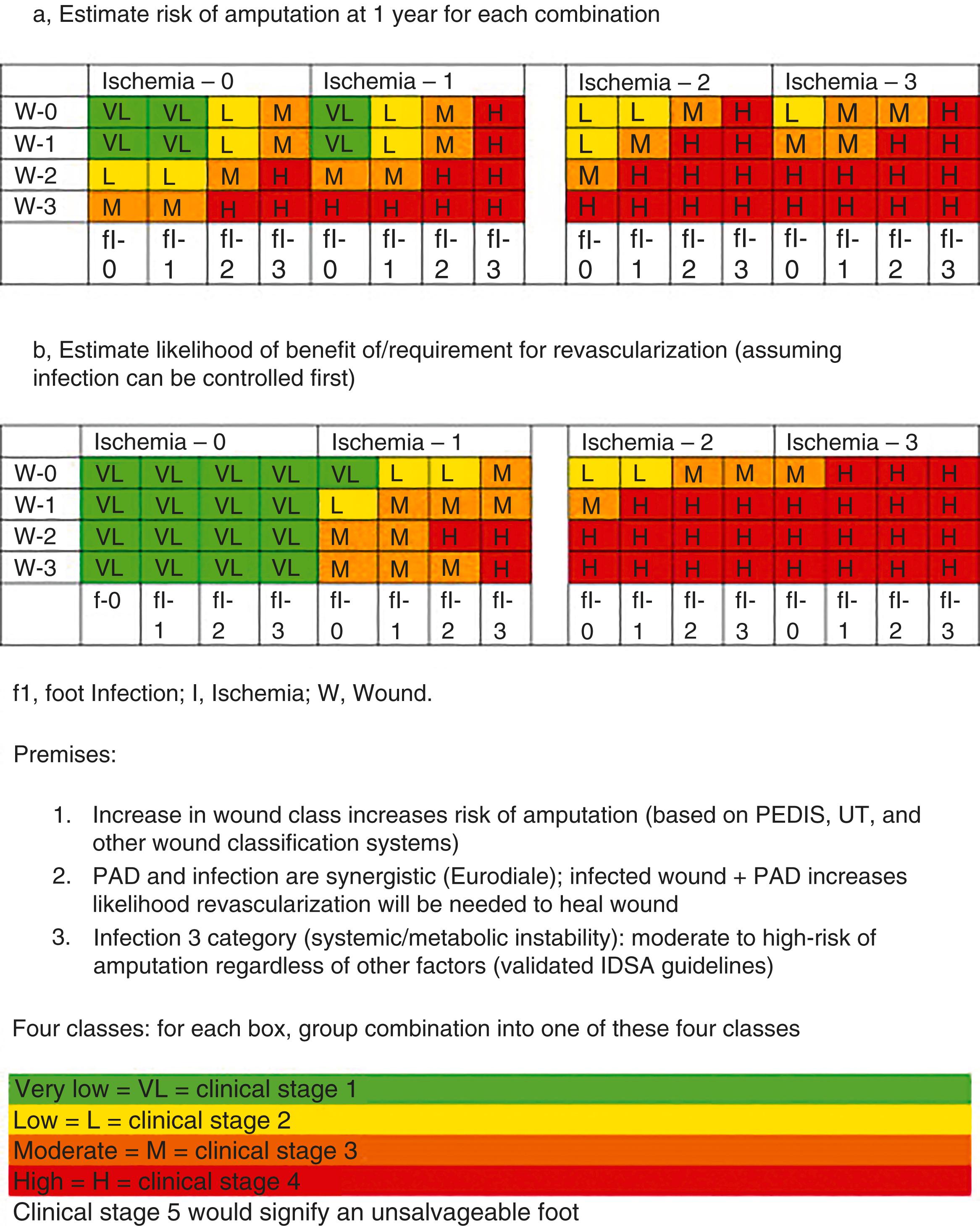
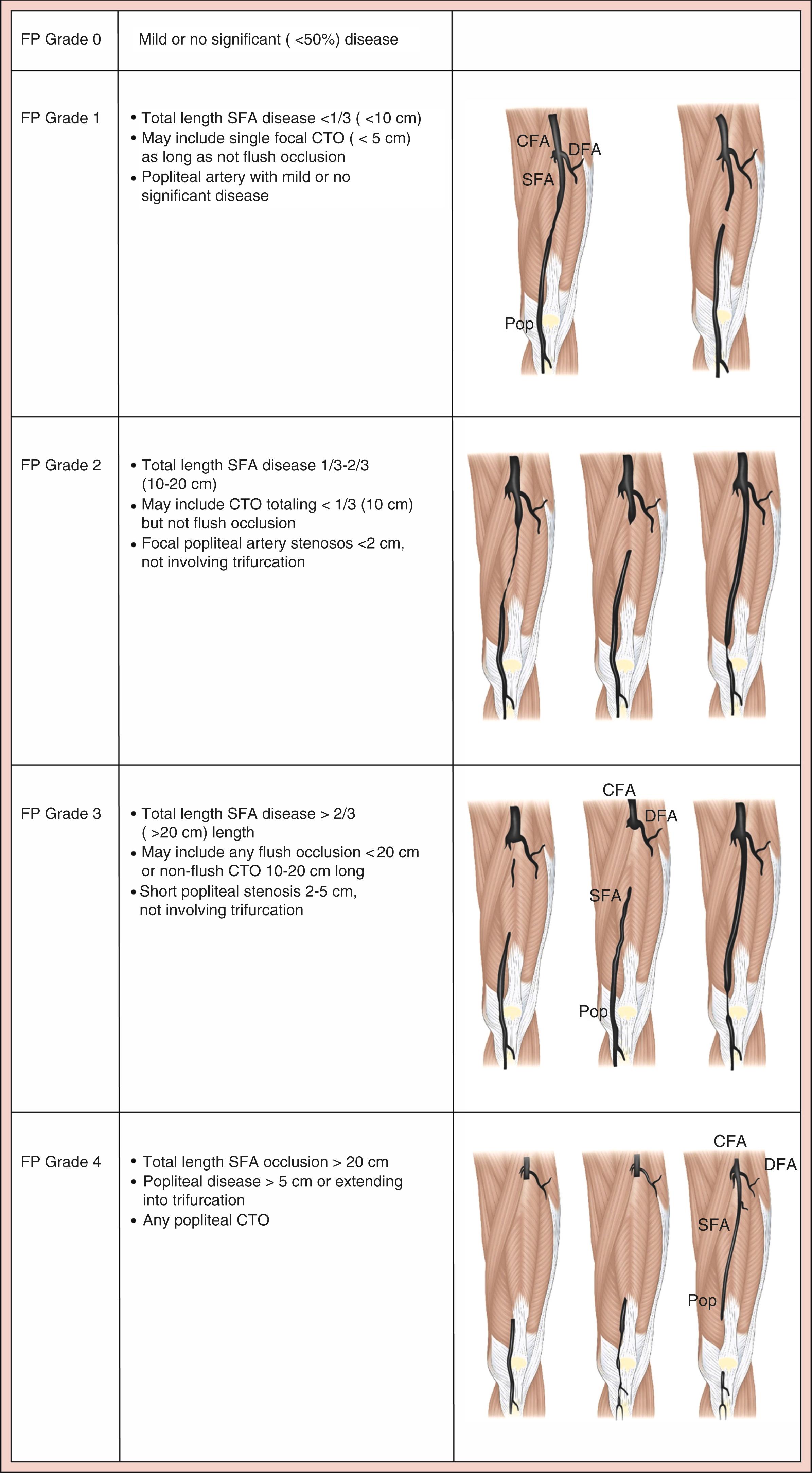
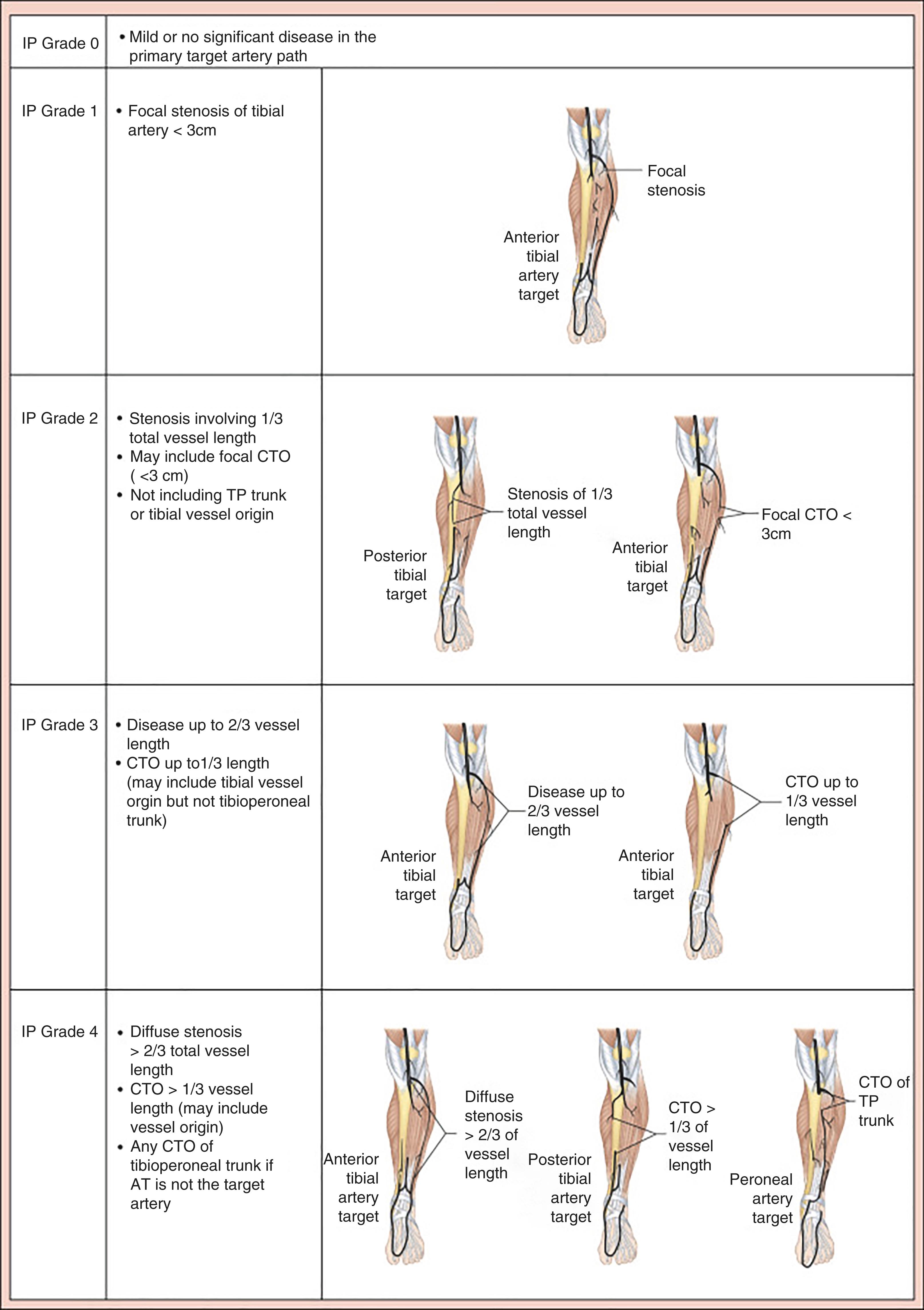
Current anatomical staging systems for PAD may be vague, lesion focussed and lack detail beyond the concept of ‘in-line pulsatile flow to the foot’. GLASS attempts to stage the disease by incorporating a full spectrum of anatomies and calcification with the goal of facilitating evidence-based revascularisation outcomes. Described within the Global Vascular Guidelines after consensus was sought from an expert panel of that document’s authors, it incorporates two novel concepts: The Target Arterial Path, defined as the physician preferred route for endovascular therapy and the estimated Limb-Based Patency (LBP). It focusses on infra-inguinal disease, with the aorto-iliac segment, common and deep femoral arteries considered the inflow. It is assumed that inflow disease is corrected before, or at the same time as, the infra-inguinal revascularisation. Infra-inguinal assessment is based on length of disease and extent of complete occlusions. There is a dichotomous calcification scale, whereby if there is >50% circumference of calcification, diffuse, bulky calcification or ‘coral reef’ plaque, the within-segment grade is increased by one numerical value. Once the grade for femoropopliteal ( Fig. 4.2 ) and infra-popliteal (IP) ( Fig 4.3 ) disease is determined (0–4) then staging can be performed based on the matrix grid provided (1–3). Staging allows for estimation of the peripheral endovascular intervention outcomes to be predicted, Immediate Technical Failure and 1-year LBP. Like WIfI, GLASS has also been validated and shows an association with treatment outcomes for CLTI.
![]() There is a body of evidence to support the effectiveness of supervised exercise therapy (SET) for the resolution of IC symptoms and multiple guidelines recommend its use as first-line therapy for IC.
There is a body of evidence to support the effectiveness of supervised exercise therapy (SET) for the resolution of IC symptoms and multiple guidelines recommend its use as first-line therapy for IC.
Such a programme must be supervised walking, 30–60-minute treadmill walks per day, be associated with risk factor modification and best medical therapy (BMT) to be most effective. The patient is instructed to walk until they experience moderate claudication, stop until symptoms resolve, then repeat until the predetermined time period has elapsed. The speed, inclination and treadmill walking time are incrementally increased throughout the programme.
The CLEVER (Claudication: Exercise Versus Endoluminal Revascularization) study was a randomised, multicentre, clinical trial. One-hundred and eleven participants with aorto-iliac occlusive disease were randomised to SET with BMT, endovascular revascularisation (ER) with BMT or general advice to exercise and BMT. SET consisted of a 26-week programme of 60-minute treadmill walking followed by telephone counselling to encourage ongoing exercise participation. After 18 months of follow-up, functional walking improvement and QoL were persistently improved in both the SET + BMT and ER + BMT groups. Those two groups could not be separated in their effectiveness, attesting to the value of each; and both of those strategies were superior to BMT with general advice to exercise.
In the CETAC study, 151 patients were randomised to either SET or ER for PAD of the aorto-iliac or femoropopliteal arterial segments and both groups received BMT. SET consisted of a 24-week programme whereby participants walked for 30 minutes on a treadmill twice a week, were encouraged to walk for an additional 30 minutes three times per week at home and continue walking for at least 1 hour per day after the programme was complete. After a mean follow-up period of 7 years, both groups had significantly improved maximum walking distance, pain-free walking distance and QoL. There was no difference between the two groups in any of those outcome measures. This trial demonstrated the long-term effectiveness of SET as an alternative therapy, equivalent to ER.
Despite this compelling evidence to support the use of SET, ER is widely used in the treatment of patients with claudication. The reasons for this are multifactorial. First, ER offers a fast treatment with almost instant relief of symptoms. It is less labour-intensive than SET, which requires effort and persistence. Moreover, SET is frequently unavailable or not reimbursed in countries with insurance-based health care, a fact that is disappointing given the additional benefits of exercise in obesity reduction, blood pressure control, lipid lowering and glucose control, over-and-above that of claudication treatment.
All patients with macroscopic arterial disease benefit from risk factor management. Optimal medical therapy should be instigated in conjunction with interventional therapy and continued long term to prevent secondary cardiovascular morbidity. Medical treatment for PAD, and risk factor modification have been comprehensively described in Chapter 3 .
There are more than 200 million people living with PAD around the world and the incidence is increasing. , In high-income countries, there has been an increase in the proportion of patients undergoing ER procedures, with decreasing open-surgical revascularisation procedures, in-hospital mortality and major amputation. Factors that may be driving this trend include the association of endovascular procedures with reduced in-hospital mortality, length-of-stay and cost when compared to surgical revascularisation. , However, a causal relationship linking the endovascular approach to reduced amputation and improved outcomes remains to be demonstrated.
The aorto-iliac segment is a frequently diseased vascular territory, which may be found both in isolation (claudication) and in conjunction with multilevel, occlusive disease in CLTI. Isolated aorto-iliac revascularisation may be sufficient to address symptoms in patients who also have concomitant infra-inguinal disease, and it is often necessary to maximise inflow to maintain infra-inguinal revascularisation strategies. It is therefore recommended that all vascular specialists have a thorough knowledge of treatment options for aorto-iliac occlusive disease.
The open-surgery, gold-standard for the treatment of aorto-iliac occlusive disease is aorto-(bi)-femoral (ABF) or bi-iliac bypass, which has durable patency that remains unchallenged by all other peripheral bypass operations. However, whilst those patency rates are excellent, one must also consider the risk of major complications, which may occur in up to a quarter of patients, sexual dysfunction in up to 40% and perioperative mortality rates as high as 5%. , It is this burden of risk, which has led some to question whether a first-line, endovascular approach is more appropriate for most patients with aorto-iliac disease.
Percutaneous interventions have gained increasing popularity, driven by patient preference, enhanced safety and the increasingly complex patient comorbidities encountered in an ageing population. Moreover, minimally invasive procedures have demonstrated reduced length-of-stay requirements and intensive care utilisation, making them extremely attractive.
Whilst in the past it was common for endovascular therapies to face insurmountable technical challenges to wire passage and stent delivery during the treatment of extensive aorto-iliac disease, dedicated chronic-total-occlusion wires and catheters, bi-directional techniques and re-entry devices have contributed to very high technical success in contemporary experience. , This has made possible the percutaneous treatment of even the most extensive aorto-iliac occlusion for the skilled interventionist.
Several open surgical options exist to revascularise the lower limbs in the presence of aorto-iliac occlusive disease. They include extra-anatomical bypass (femoro-femoral crossover and axillo-(bi)-femoral bypass), aortic endarterectomy and ABF bypass.
ABF bypass grafting has been performed since the advent of the synthetic aortic graft in the early1950s. The aorta may be approached from a trans- or retro-peritoneal route, and the bypass may be configured with either a proximal anastomosis that is end-to-end, or end-to-side. The latter configuration has the advantage of preserving flow to the distal inferior mesenteric, aberrant renal or internal iliac arteries; however, no patency advantage has been demonstrated with either technique and the decision as to which method is best remains contentious amongst vascular surgeons. An individual patient approach appears reasonable, dependent upon patient anatomy and disease morphology.
Patency rates with ABF are considered the gold standard for the treatment of aorto-iliac occlusive disease. A meta-analysis by De Vries et al. analysed 23 studies published between 1970 and 1996. Primary patency at 5 and 10 years was 91% and 86.8%, respectively, in those with claudication. For those with CLI, the rates were slightly lower at 86.8% and 81.8%. However, despite those excellent surgical results, current practice in most units is to favour an endovascular-first approach for the majority of patients because of the relatively high perioperative risk. De Vries et al. found a combined 30-day mortality rate of 4.4%, which was higher in the older series (4.6% in studies published before 1975; 3.3% 1975–1996; P = 0.01). Furthermore, major morbidity rates were 19.7% (12.1% systemic complications; 7.6% local) and in series that looked specifically at sexual dysfunction, morbidity was noted in as many as 40%, with a combination of retrograde ejaculation and iatrogenic impotency. ,
Extra-anatomic bypass is a less morbid operation than ABF, but it suffers from lower 5-year primary patency rates of 51% (range 44–79%) for axillo-uni-femoral, 71% (range 50–76%) for axillo-bi-femoral and 75% (range 55–92%) for femoro-femoral bypass. For that reason, most vascular surgeons are likely to reserve those options for patients with severe comorbidities or a hostile abdomen where transperitoneal surgery is best avoided.
It should be noted that many of the studies included in the meta-analyses, which have evaluated open-surgical techniques for aorto-iliac occlusive disease are from an era when open surgery was used in the majority. Although outcomes from vascular surgery have generally improved over the years, care must be taken in their interpretation to consider that they are likely to have included patients with relatively minor disease who would be treated with angioplasty or stenting using current standards of practice. The inclusion of those relatively simple cases means that the patency and morbidity results from these studies may be skewed towards longevity and safety relative to the real-world application of ABF using present-day treatment algorithms.
Patients often prefer minimally invasive therapeutic options, as they experience less pain, less operative risk and a faster return to normal activities. The TASC II document pooled results of 2222 iliac artery angioplasty procedures for both claudication (76%) and critical ischaemia (24%). They found a high technical success rate of 96% (range 90–99), as well as 1-, 3- and 5-year primary patency rates of 86% (range 81–94%), 82% (72–90%) and 71%(64–75%), respectively, demonstrating both the technical feasibility and durability of percutaneous transluminal angioplasty (PTA) for aorto-iliac occlusive disease. In other contemporary findings from a large, single-centre study describing an experience with 505 aorto-iliac lesions treated with angioplasty ± stenting over a 9-year period, the authors found a technical success rate of 98%. Eight-year primary and primary-assisted patency rates were 74% and 81%, respectively and safety was demonstrated with a 30-day mortality of 0.5%, comparing favourably to open surgery.
New techniques and technology have allowed us to treat ever more extensive patterns of disease, including long infra-renal aortic occlusions that extend into the iliac arteries ( Fig. 4.4 ). These have traditionally been considered only suitable for open surgery. The most important technical aspect of treating advanced aorto-iliac occlusive disease proximal to the aortic bifurcation is the reliable passage of guidewire into the aortic true lumen from below. When the occlusion approaches the renal arteries, then accurate localisation of the true lumen entry point becomes particularly critical in order that stents can be deployed in the true lumen but as far distal to the renal arteries as possible. These two aspects of treatment are reliably achieved using a bi-directional approach (combined antegrade from an upper limb access and retrograde from a femoral access) and by using re-entry devices with fine, retractable needles that can accurately facilitate a point of re-entry.
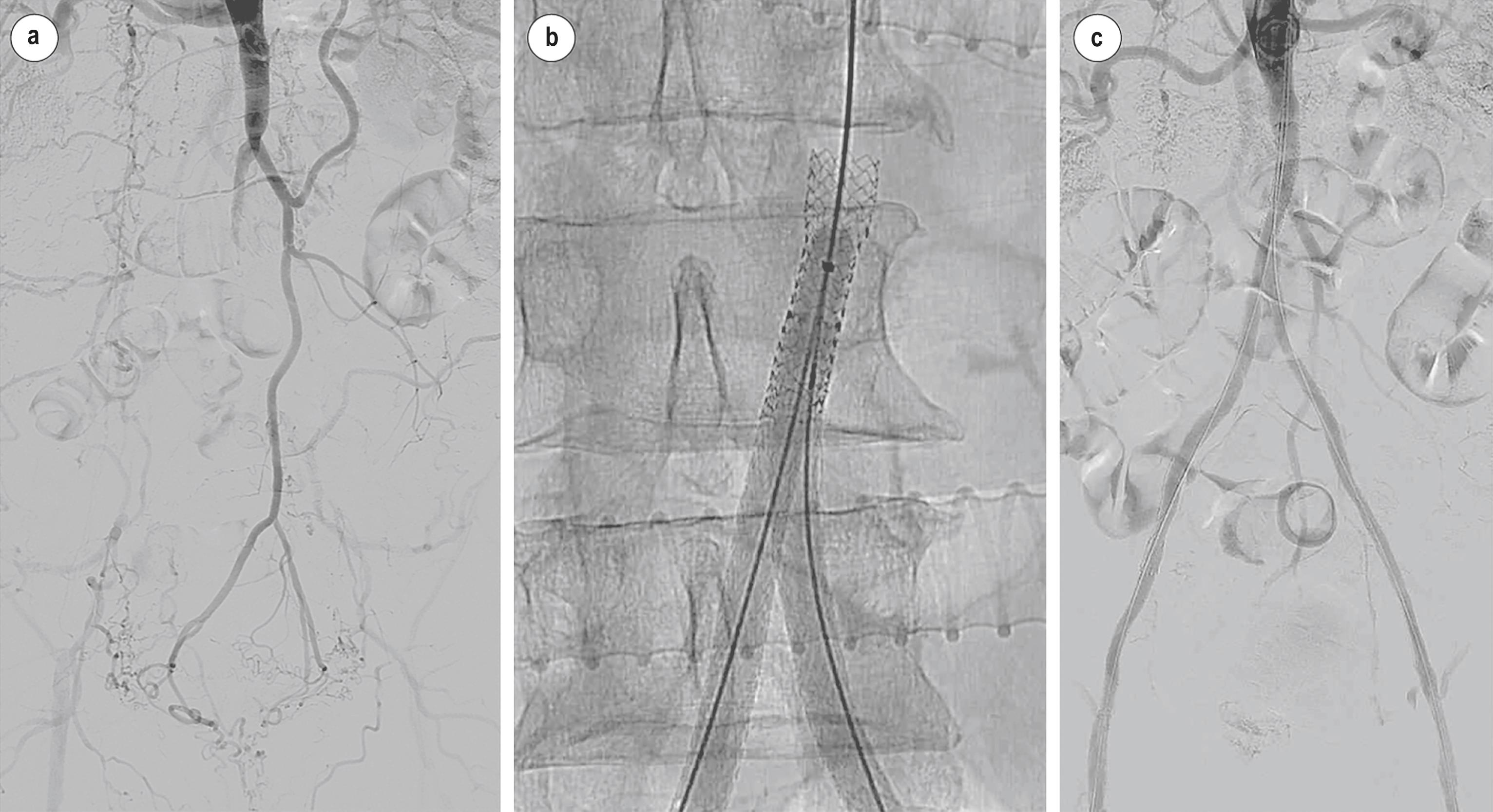
Whilst it is generally accepted that an endovascular-first approach should be the treatment of choice for those with less extensive disease (TASC A and B; for details of TASC classification see Chapter 2 ), there are few robust data with which to compare the two methods of revascularisation for more complex and extensive disease patterns. Current evidence is largely based on single-centre, retrospective studies with no large-scale, contemporary, randomised clinical trials to compare endovascular and open surgery for the treatment of TASC C and D aorto-iliac lesions. However, a contemporary meta-analysis has identified all studies that included TASC C and D disease between 1989 and 2010. Data were extracted for 1625 patients from all 28 studies that evaluated ER and 3733 patients from the 29 studies, which evaluated open bypass surgery. ER was found to be safer (complication rate 13.4% vs. 18.0%, P <0.001; 30-day mortality 0.7% vs. 2.6%, P <0.001) and incur a shorter length-of-stay (4 days vs. 13 days, P <0.001). The complications observed in the ER studies were more likely to be minor and self-limiting, such as haematoma and fever; whereas cardiac complications were frequent after bypass surgery, as were wound and graft infections. Findings favouring the more invasive procedure were that bypass surgery had increased primary patency rates at 1, 3 and 5 years of 94.8% versus 86.0%, 86.0% versus 80.0% and 82.7% versus 71.4%, respectively (all P <0.001), with a similar difference seen in secondary patency. It is apparent that ER is safer and less invasive, whilst surgery remains more durable. The published data reinforce the fact that both approaches are reasonable and should be considered a standard part of the armamentarium in the everyday clinical practice of patients with aorto-iliac disease. A reasonable approach is to apply the most appropriate form of treatment, matched to the individual patient’s needs, disease morphology and risk factors. That may favour the durability of overall survival for the young, fit patient with claudication or a minimally invasive strategy for the elderly patient with tissue loss. Some argue that an endovascular-first approach to all patients remains reasonable as long as that procedure does not impact future surgical options.
The Dutch Iliac Stent Trial randomised 279 patients with mostly stenotic disease to undergo either primary stent placement ( n = 143) or PTA ( n = 136) between 1993 and 1997. At a mean follow-up of 6.3 and 5.7 years for the two groups respectively, there was no difference between the group who ultimately received stents versus those who underwent uncomplicated PTA. The group that fared best were those that underwent PTA and had bailout stenting. Those patients had superior preservation of symptomatic relief and long-term quality of life. No difference was observed between ankle-brachial pressure index (ABPI) or patency between any of the groups. The authors concluded that PTA with selective stenting strategy was the preferred method of iliac revascularisation.
The STAG trial was a contemporary, British, multicentre randomised controlled trial (RCT), which recruited 118 patients with iliac artery occlusions to undergo either PTA ( n = 55) or primary iliac stenting ( n = 57) (six were excluded because of protocol violations). Primary stent placement resulted in increased technical success rates (98% vs. 84%; P = 0.007) and reduced major complication rates (5% vs. 20%; P = 0.010) compared with simple PTA. There was no difference in primary patency after a median follow-up period of 721 days and the authors concluded that primary stent placement was preferable to improve technical success and reduce complications.
Whilst there is no randomised trial that has compared self-expanding and balloon-expandable stents for the iliac artery, there are 24-month follow-up data from the BRAVISSIMO European prospective, multicentre registry. That study enrolled 325 patients with aorto-iliac occlusive disease ( n = 190, TASC A and B; n = 135 TASC C and D) and followed them with duplex ultrasound. They found similar primary patency rates of 88.0%, 88.5%, 91.9% and 84.8% for TASC A, B, C and D groups, respectively. There was a trend toward better patency for self-expanding stents (92.1%), compared with balloon expandable (85.2%) or a combination of both stent types (75.3%) ( P = 0.06). These results suggest that more advanced disease may also be treated with endovascular intervention and that both stent types are reasonable to use in the aorto-iliac segment. However, when stenting the iliac arteries, it is worthwhile to consider the common and external iliac segments as separate regions with individual characteristics that favour different stent types. The common iliac artery is well suited to a balloon-expandable stent, which has placement accuracy and increased radial strength to overcome the calcification that is common in this region. Typically, disease involving the aortic bifurcation will be treated with bilateral stents placed in a ‘kissing’ configuration to reduce the risk of plaque-shift compromising the contralateral flow lumen. The external iliac artery is exposed to increased range of movement and mechanical strain upon hip flexion, which may benefit from a more flexible, self-expanding stent. The external iliac is also prone to rupture during dilatation, which may lead to sudden, catastrophic blood loss. This should always be considered, and a range of covered stents kept available to expeditiously deal with such events.
Covered stents may also be used as primary therapy in the iliac arteries because of their known property of in-stent restenosis reduction. COBEST (Covered versus Balloon Expandable Stent Trial) was an Australian, multicentre trial, which enrolled 125 patients with iliac occlusive disease (TASC B–D) and randomised them to treatment with a PTFE-covered (Advanta V12; Atrium) or a bare-metal stent (BMS; both self-expanding and balloon-expandable varieties were used) chosen at the operator’s discretion. The long-term results showed a significantly higher patency for covered stents compared to bare metal stents at 5 years (74.7% vs. 62.5%; P = 0.01). Subgroup analysis after 5 years found better patency of the TASC C and D lesions treated with covered stents (hazard ratio [HR], 8.64; 95% confidence interval [CI], 54.25–75.75; P = 0.003) along with a survival benefit compared to BMS. There was no such patency difference observed in the TASC B subgroup.
![]() The COBEST trial results suggested that covered stents may confer protection from restenosis and should be considered in more severe patterns of aorto-iliac occlusive disease.
The COBEST trial results suggested that covered stents may confer protection from restenosis and should be considered in more severe patterns of aorto-iliac occlusive disease.
Common femoral artery (CFA) disease has continued to be treated predominantly by open surgery. This is partly because of the nature of the disease as well as the technical difficulties with using stents in this region.
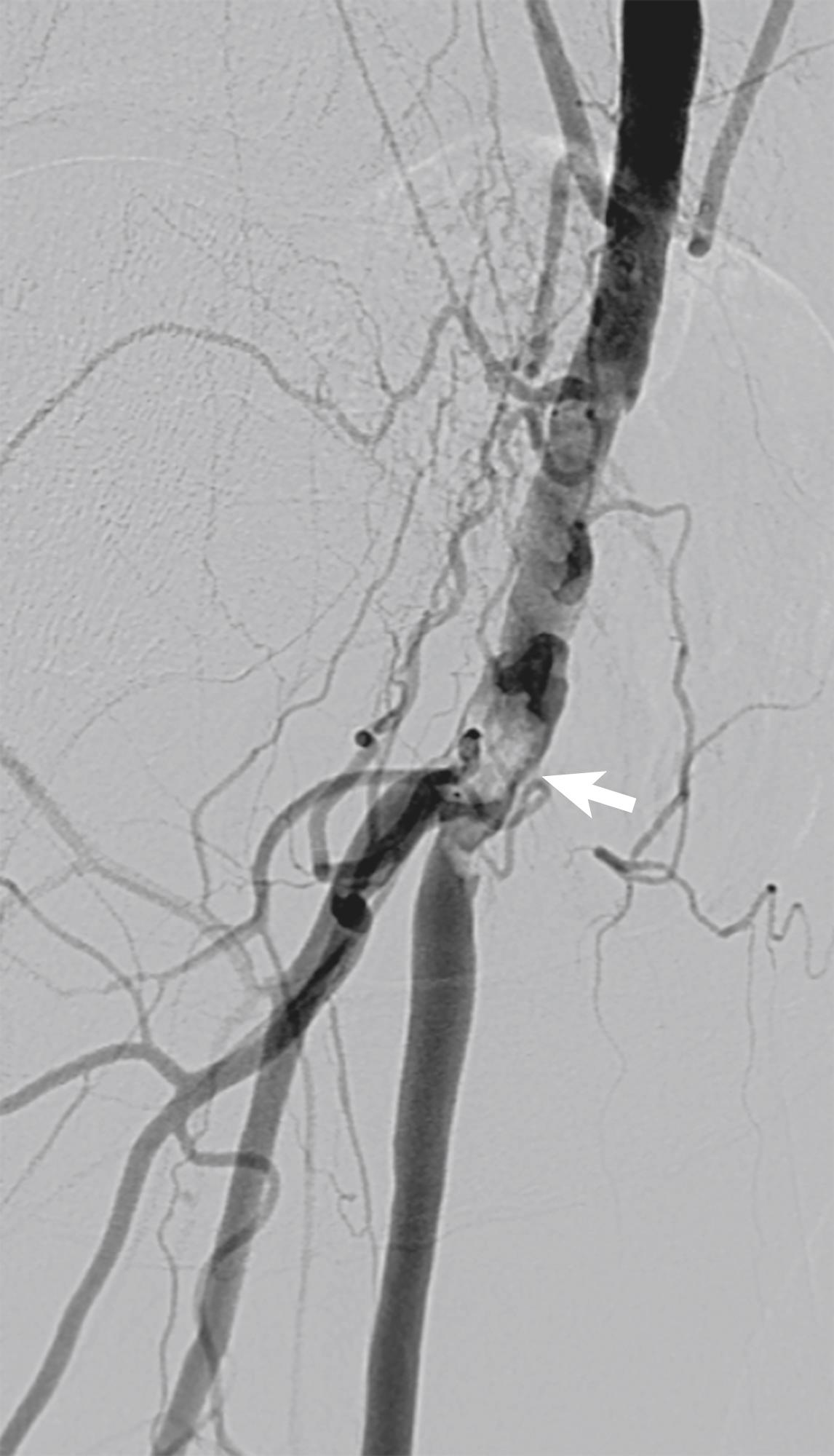
The CFA is prone to the development of eccentric and calcified atheromatous plaque with a predilection for the posterior artery wall ( Fig. 4.5 ). Upon handling, the plaque is often friable and it frequently involves the bifurcation, proximal superficial femoral (SFA) and profunda femoris arteries. Whilst PTA can be effective in treating CFA disease, there is a risk of embolisation and if stents are required, there is not a good solution for dealing with the bifurcation. Furthermore, it is a commonly held belief that the CFA is a flexion point that puts stents at risk of fracture, and that following implantation, the blood vessel cannot be used as an access site for future arterial access. Whilst neither of these points are entirely true, with the majority of vessel flexion taking place through the distal external iliac artery and sheath insertion through well-incorporated self-expanding stents, a simple, safe manoeuvre, it helps explain the general reluctance to use endovascular techniques in this short arterial segment.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here