Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The fetus and the newborn are susceptible to multiple infections: bacterial, viral, and fungal. This susceptibility is multifactorial and stems from maternal risk factors, obstetrical complications, the postnatal environment, prematurity, and the immature host defenses of the newborn. Throughout this chapter, we will review common pathogens to which the neonate may be exposed, as well as briefly discuss newer congenitally-acquired infections.
Neonatal sepsis is defined as a systemic inflammatory response syndrome secondary to infection. The age of onset of sepsis reflects the likely mode of acquisition, microbiology, and clinical presentation with consequent implications on morbidity and mortality. Worldwide, it is estimated that more than 1.4 million neonatal deaths per year are caused by invasive infections. Epidemiologically, neonatal sepsis is divided into the following categories: early-onset sepsis (EOS), late-onset sepsis, and very late–onset sepsis.
EOS is a systemic, multiorgan disease that presents in the first week of life and usually within the first 72 hours of life. Infection is most often acquired before delivery. Perinatal factors contribute to the development of infection and include rupture of membranes prematurely (before onset of labor) or a prolonged period (>18 hours) before delivery, chorioamnionitis, maternal fever, maternal urinary tract infection, and infant prematurity or low birth weight. These infants have a fulminant onset of respiratory symptoms, usually due to pneumonia, poor perfusion, temperature instability, and acidosis. The microbiologic features of EOS reflect maternal genitourinary and gastrointestinal colonization.
In the Active Bacterial Core Surveillance in four states from 2005 to 2008, as assessed by the Centers for Disease Control and Prevention (CDC), the overall rate of EOS in the United States was estimated at 0.77 per 1000 live births with a case fatality rate of 10.9%. Before the adoption of intrapartum antibiotic prophylaxis (IAP) against group B Streptococcus (GBS), this pathogen caused the overwhelming majority of EOS. Today, GBS still causes most cases of EOS; however, enteric bacilli such as Escherichia coli have become more prevalent in term infants and are as likely as GBS to cause EOS in very low-birth-weight premature infants. GBS is estimated to be responsible for 0.29 infections per 1000 live births with case fatality rate of 7%, and E. coli is responsible for 0.19 infections per 1000 live births with a case fatality rate of 25%. Although GBS a enteric bacilli cause the preponderance of EOS, a third pathogen, Listeria monocytogenes, can cause EOS, although decreasing in incidence. Unlike GBS and the gram-negative pathogens, which usually are acquired through asymptomatic maternal colonization, L. monocytogenes generally causes a flulike or gastrointestinal illness in the mother. This organism is mostly acquired from animal products: unpasteurized milk, cheese, delicatessen meats, and hot dogs. The importance of this organism will become clear in the discussion of empiric antibiotic therapy.
Late-onset sepsis is defined as the infections that occur beyond the first week of life but before 30 days of life. Very late–onset sepsis occurs beyond 30 days of life. Although intrapartum complications may be identified, these are not typical. Late-onset disease is more likely to reflect infection with gram-positive organisms acquired in the nursery: coagulase-positive staphylococci, Staphylococcus aureus , and enterococci. Invasive candidiasis is also an emerging cause of late-onset diseases. Very late–onset disease includes infections caused by GBS, gram-negative bacilli, and Streptococcus pneumoniae.
The high incidence of gram-positive infection in the hospitalized infant usually reflects the combination of lower gestational age and low birth weight and the consequent need for the insertion of central venous catheters for supportive care. Although many of these infants will manifest poor feeding, temperature instability, and lethargy, they are more likely to have localized disease: urinary tract infection, osteoarthritis, or soft tissue infection. Meningitis is common. Presentation may be slowly progressive or fulminant. Mortality is lower than with EOS but may still be 20% to 40%.
The widespread use of IAP in the United States has been shown to have decreased the incidence of EOS by 70% to 0.44 cases per 1000 live births, an incidence equivalent to that of late-onset sepsis. Of importance is that the improved survival of very low-birth-weight infants has put them at increased risk of systemic nosocomial infection.
The most important risk factors for the development of neonatal sepsis are low birth weight and prematurity. The incidence of sepsis is inversely proportional to the gestational age or birth weight of the infant. Sepsis is the most common cause of death in infants under 1500 g. Other independent risk factors for sepsis include immature immune function, exposure to invasive procedures, hypoxia, metabolic acidosis, hypothermia, and low socioeconomic status; all aforementioned factors are associated with low birth weight and prematurity. In a multicenter survey of GBS disease carried out by the CDC, 13.5 cases per 1000 live births were diagnosed among black infants compared with 4.5 cases among 1000 white infants, and EOS was twice as common among black infants as among white infants. Although males have a higher incidence of sepsis, once respiratory distress syndrome is accounted for, they are not at a significantly higher risk of sepsis, contrary to the results of older studies. It is generally felt that sepsis is more common among the firstborn of twins. Infants with galactosemia are more likely to become infected with gram-negative organisms, in particular, E. coli. The administration of iron for anemia appears to increase risk because iron may be a growth factor for a number of bacteria. Finally, the widespread use of broad-spectrum antibiotics may cause a shift in the nursery to a higher prevalence of resistant bacteria that are also more invasive, as well as yeast. This phenomenon informs the need for better antimicrobial stewardship. Prevention of healthcare-associated infections through antimicrobial stewardship, limiting use of invasive devices and standardization of catheter procedures, and improved hand hygiene are important ways to decrease late-onset disease.
Definitive diagnosis of bacterial infection is predicated on the recovery of a pathogen from a normally sterile body site such as blood, urine, or cerebrospinal fluid (CSF). Although many indirect indices of infection have been identified and studied, including total white blood cell count, absolute neutrophil count, C-reactive protein level, procalcitonin level, and levels of a variety of inflammatory cytokines, taken individually, these tests are nonspecific and are not adequately sensitive to confirm or exclude systemic infection.
In infants with suspected EOS, cultures of blood should be drawn, and, if the infant is in a hemodynamically stable condition, spinal fluid should be obtained, and the infant should be started on intravenous antibiotics. The need for lumbar puncture in the first 24 to 72 hours of life has been a topic of some controversy. Data suggesting that lumbar puncture is unnecessary in these infants comes primarily from retrospective studies of asymptomatic infants. The poor correlation between the results of neonatal blood cultures and CSF cultures underscores the need for lumbar puncture. Several studies report that bacterial meningitis would be missed in approximately one-third of very low-birth-weight neonates on the basis of blood culture results alone. Antibiotic regimens should cover GBS, gram-negative bacilli, and L. monocytogenes. The most commonly used regimens are ampicillin and cefotaxime or ampicillin and gentamicin. Both regimens are quite effective against GBS. Unfortunately, E. coli has increasingly become resistant to ampicillin. In many institutions, more than half of the E. coli isolates are resistant to ampicillin. A search of the Cochrane database for evidence suggesting that one regimen is superior to another does not yield a conclusion. Regardless of the regimen used, ampicillin should be included because the cephalosporins have no activity against L. monocytogenes, and gentamicin monotherapy would be ineffective.
The data for empirical therapy in late- and very late–onset disease are not definitively in favor of any one regimen. Given the prevalence of staphylococcal species, many clinicians would include vancomycin in the empiric treatment of a hospitalized neonate with signs of sepsis beyond the seventh day of life. If the infant is being admitted from the community, the regimen should include coverage for GBS, E. coli, and S. pneumoniae. Commonly cited guidelines for the evaluation of febrile children without a focus of infection who are between 30 and 60 days of life include obtaining blood and urine samples for culture and performing a lumbar puncture before administration of antibiotics.
Streptococcus agalactiae, or GBS, is the most common cause of vertically transmitted neonatal sepsis. It is a significant cause of maternal bacteriuria and endometritis, and a major cause of serious bacterial infection in infants up to 3 months of age. Ten serotypes of GBS have been identified on the basis of differing polysaccharide capsules: Ia, Ib, II, and III through IX. Epidemiologically, the serotypes responsible for neonatal disease shifted significantly in the 1990s. Type Ia, Ib, III, and V cause 95% of cases in infants in the United States. Type III still causes the majority of late-onset disease and neonatal meningitis. Antibodies against specific serotypes of GBS are protective but not cross-reactive.
Although GBS can cross the placenta, the primary mode of transmission is after rupture of membranes and during passage through the birth canal. Approximately 20% to 40% of women are colonized in their genital tract, but the primary reservoir of GBS is the lower gastrointestinal tract. High genital inoculum at delivery increases the likelihood of transmission and the consequent rate of early-onset GBS (EOGBS). Half of infants born to colonized women will themselves be colonized with GBS. Of these, 98% will be asymptomatic, whereas 2% will have evidence of invasive disease. Before the use of IAP targeted against GBS, the incidence of EOGBS ranged from 1 to 3 cases per 1000 live births. By definition, EOGBS presents in the first 6 days of life, and close to 90% of cases present within 24 hours of life. The vast majority of these infants demonstrate systemic illness by 12 hours.
In 1986, Boyer and Gotoff published the first randomized, controlled trial showing the effectiveness of IAP in reducing neonatal colonization and EOGBS. In 1996, the CDC published the first set of guidelines for the prevention of perinatal GBS disease. The guidelines endorsed two approaches to IAP: (1) women with vaginal or rectal cultures positive for GBS should receive IAP; or (2) women with any of the following risk factors—delivery before 37 weeks’ gestation, membrane rupture 18 hours or longer before delivery, or maternal fever of 38 C or higher—should receive IAP. In addition, any woman who had a history of GBS bacteriuria or who had previously delivered an infant with EOGBS was to receive IAP. In addition to the administration of IAP, the guidelines provided for the evaluation of the infant after delivery. These strategies reduced the incidence to 0.34–0.37 cases per 1000 live births in 2010. Ongoing active surveillance of GBS demonstrated that the screening-based approach was superior to the risk-based approach in preventing EOGBS. In 2010, the CDC published revised guidelines that promoted the universal screening of all pregnant women between 35 and 37 weeks’ gestation using rectovaginal cultures, and recommended that all women with positive culture results receive IAP. The guidelines also recommended IAP for mothers who had any history of GBS bacteriuria during the pregnancy, who had suspected chorioamnionitis, or who had previously delivered an infant with EOGBS. These guidelines also clarified the antibiotic dosages for IAP and provided alternatives for mothers with penicillin allergy, and the management of an exposed newborn ( Fig. 13.1 ).
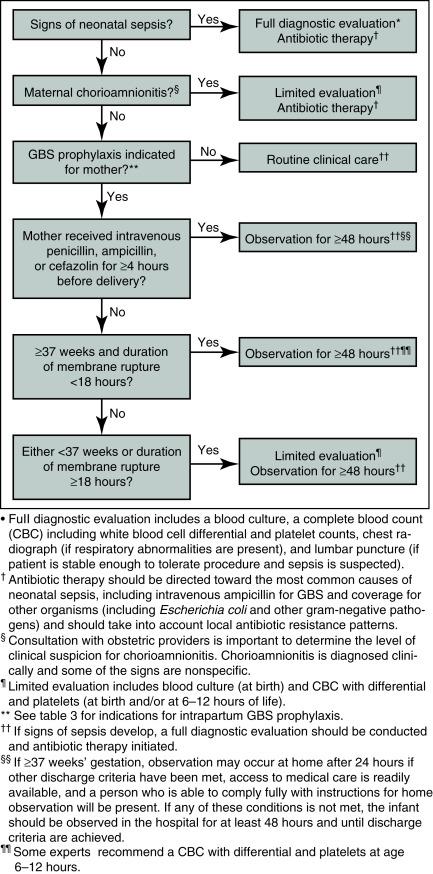
As of 2003, the incidence of EOGBS was down to 0.3 cases per 1000 live births. Although effective, the screening-based approach incurs the costs of testing, IAP, and management of the exposed infant. Although to date, no studies have shown an association between penicillin and ampicillin IAP and the emergence of antibiotic resistance in other bacteria, this risk remains a concern. An immunization-based strategy targeting pregnant women has the potential to prevent EOGBS, late-onset disease, and some maternal disease and to be more cost-effective. A multivalent protein conjugate vaccine has proved effective in a murine model, and several human trials of individual serotype conjugate vaccines have shown promise.
For documented GBS infection, penicillin is the drug of choice and is the most narrow-spectrum agent. Ampicillin is an acceptable alternative agent. No penicillin resistance has been reported to date. The dosages and intervals depend on the postgestational age of the infant. The duration of therapy is 10 days for bacteremia without a focus, 14 days for uncomplicated meningitis, and up to 4 weeks for septic arthritis, endocarditis, or ventriculitis.
For several decades, coagulase-negative staphylococci have been the most common cause of nosocomial bloodstream infections in the neonatal intensive care unit and are responsible for the majority of cases of late-onset sepsis in preterm neonates. Infections with these gram-positive bacteria are most often associated with indwelling central venous catheters. These bacteria are part of normal human skin flora. Staphylococcus epidermidis is the most common species of coagulase-negative staphylococci recovered from human skin and mucous membranes. Most infants are colonized within the first week of life from passage through the birth canal and repeated exposure from colonized caregivers.
The major virulence factor for coagulase-negative staphylococci is its ability to adhere to plastic and other foreign bodies by producing a biofilm. The biofilm consists of multiple layers of bacteria surrounded by an exopolysaccharide matrix or slime. This biofilm protects the bacteria from host phagocytic cells and interferes with the ability of many antimicrobial agents to effectively eliminate infection. This affinity for plastic foreign bodies explains the high recovery rate of these organisms from infected catheters, ventricular shunts, endotracheal tubes, and artificial vascular grafts and cardiac valves.
Neonatal infections with coagulase-negative staphylococci typically present without localizing signs with fever, new-onset respiratory distress, or a deterioration in respiratory status. Other common nonspecific signs of coagulase-negative staphylococcus sepsis include apnea, bradycardia, poikilothermia, poor perfusion, poor feeding, irritability, and lethargy. Indolent infection is more common than fulminant disease, with mortality generally under 15%. Coagulase-negative staphylococci infections, however, are a major source of morbidity, leading to increased antibiotic exposure, length of stay, and hospital costs.
Treatment of coagulase-negative staphylococci often requires the use of vancomycin. More than 80% of strains acquired in the hospital are resistant to beta-lactam antibiotics. Resistance is typically attributable to altered penicillin-binding proteins and beta-lactamase production. Unfortunately, these types of resistance can be inducible and therefore may not be detected by routine microdilutional methods. If a strain is reported as penicillin sensitive, consultation with the hospital microbiologist is recommended to confirm testing for inducible resistance. More than 50% of coagulase-negative staphylococci are resistant to clindamycin, trimethoprim-sulfamethoxazole, gentamicin, and ciprofloxacin. Coagulase-negative staphylococci isolated from hospitalized patients show varying rates of resistance to the tetracyclines, chloramphenicol, rifampin, and newer-generation quinolone antibiotics. Some S. epidermidis isolates have been recovered that show resistance to vancomycin; however, these species have been susceptible to the newer agents for gram-positive organisms: linezolid, quinupristin–dalfopristin, and daptomycin. Pharmacokinetic (PK) data and clinical experience with these agents in neonates are limited, and these drugs should be used only in consultation with a physician with expertise in infectious diseases.
The most effective management of coagulase-negative staphylococci infections is the combination of systemic antimicrobial therapy and, whenever possible, the removal of the foreign body. When a foreign body cannot be feasibly removed, the combination of vancomycin with rifampin, and/or an aminoglycoside, may be used. In the case of ventricular shunt infections, antibiotics may be administered both systemically and intraventricularly. If an attempt is made to manage an infection without foreign body removal, consultation with an infectious disease expert would be advised to determine the best antimicrobial agents and duration of therapy.
Although many groups have proposed different strategies to prevent neonatal catheter infections, few studies have yielded promising results. Several groups have studied the use of prophylactic antibiotics in neonates with indwelling catheters. The Cochrane Neonatal Group found no evidence to support this practice for neonates with umbilical arterial or venous catheters, nor did they find evidence to support routine use of vancomycin in preterm infants to prevent nosocomial sepsis. The use of a vancomycin–heparin lock solution to prevent nosocomial bloodstream infection showed promise in a small randomized, controlled, double-blinded study in critically ill neonates with peripherally inserted central venous catheters ; however, larger studies are needed. The use of antibiotic- or silver-impregnated catheters has not been studied in neonates. In 2002, the CDC recommended against the routine use of antimicrobial prophylaxis for patients with central venous catheters. In 2006, the Cochrane Neonatal Group began a systematic review of the use of systemic antibiotics to reduce morbidity and mortality in neonates with central venous catheters. Although prophylactic systemic antibiotics reduced the rate of proven or suspected septicemia, there was no significant difference in overall mortality. There was a lack of data pertaining to selection of resistant organisms or long-term neurodevelopmental outcomes; thus this is not routinely recommended.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here