Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Checkpoint inhibitors revolutionized cancer therapy and the field of oncology when the first drug of this class was introduced for melanoma treatment. But their therapeutic benefit comes at a cost of immunotherapy-related adverse events (IrAEs).
Checkpoint inhibitors are immunomodulatory antibodies that are used to activate the immune system to detect and destroy malignant cells. Several immune checkpoint receptors are targeted by different immunotherapy agents, such as cytotoxic T lymphocyte-associated antigen 4 (CTLA-4), programmed cell death protein 1 (PD-1), and programmed cell death protein ligand 1 (PD-L1). Different mechanisms of action explain diverse toxicity profiles: patients who are treated with anti-CTLA-4 therapy generally have more severe IrAEs than PD-1 inhibitors, which inhibit T cells in peripheral tissues at later stages of the immune response. The pathophysiological mechanisms of immunotherapy-related toxicity is still unclear, but translational studies suggest that T cells, antibodies, and cytokine responses may be involved.
Immunotherapy agents are currently widely used in patients with hematologic malignancies. Toxicity profiles for these drugs are generally well studied and include dermatologic, gastrointestinal, hepatic, endocrine, hematologic, and other less common complications ( Fig. 97.1 ). It is important to note that at the moment there are no prospective trials on IrAEs management, and all available data are based on clinical experience and retrospective analysis.
Although rare, severe and sometimes fatal toxicities may occur with checkpoint inhibitors. Therefore, there is a strong need for predictive biomarkers for IrAEs. Such biomarkers can potentially identify the group of patients who will tolerate immunotherapy. Biomarkers that are being studied include serum proteins, factors in the tumor microenvironment, as well as genomic predictors of toxicity.
One of the most common IrAEs is fatigue. According to different studies it accounts for 16% to 24% of all IrAEs. In general, fatigue is relatively mild and does not have serious negative impact on daily activity and quality of life. Therefore, for most patients, no specific treatment is needed. Severe fatigue is rare for immunotherapy agents. If it does occur it is rational to exclude other fatigue-related causes like cancer progression or endocrine IrAEs.
Systemic toxicities such as fever and chills can be caused by cytokine release syndrome. Usually it is mild and can be treated by anti-inflammatory agents and acetaminophen. Management of an infusion reaction depends on its grade and requires the use of antihistamines and/or corticosteroids, as well as a dose reduction or infusion discontinuation. Life-threatening consequences of this IrAE occur in less than 2% of patients.
Rash or pruritus appears in approximately 40% of patients receiving PD-1 inhibitors. Usually it develops early in the course of treatment. Rash can present in various forms such as spongiotic dermatitis-like eczema described as maculopapular, faintly erythematous, and pruritic vitiligo. Less common manifestations include lichenoid, eczematous, follicular, or psoriasiform, dermatitides, and bullous pemphigoid. A clinician must rule out other etiologies like infection, an autoimmune condition, or side effect of a different drug. Diagnostic work-up includes patient history and physical examination, blood cell count, liver and kidney tests. In some situations, skin biopsy should be considered. Management of this IrAE depends on the type, affected body surface area (BSA), signs, response to initial treatment, etc. Diagnostic and treatment approaches are well summarized in the American Society of Clinical Oncology (ASCO) guidelines.
Grade 1 implies close weekly monitoring without treatment discontinuation. Topical emollients, oral antihistamines, and/or mild-strength topical corticosteroids can be used.
Grade 2 also does not require interruption of immunotherapy, but close monitoring is necessary, and therapy should be stopped if symptoms are not resolving to grade 1. Therapeutic strategies include topical emollients, oral antihistamines, and median-to-high-strength topical steroids.
Grade 3 requires immediate pause in immunotherapy until the symptoms resolve to grade 1. Management includes the use of emollients, oral antihistamines, and high-potency topical corticosteroids. Initiating methylprednisolone (or equivalent) 1 to 2 mg/kg should be considered, depending on the severity of the symptoms.
Grade 4 demands discontinuation of immune checkpoint inhibitors, immediate hospital admission, and dermatologist consultation (with infectious disease specialist if necessary), as well as intravenous corticosteroids.
Oral mucositis associated with immunotherapy can be a therapeutic dilemma especially in patients who received oral corticosteroids for management of other IrAEs. Oral candidiasis remains an important consideration in the differential diagnosis. In some patients improvement can be achieved with oral corticosteroid rinses and lidocaine.
Diarrhea and colitis (symptoms include abdominal pain, fever, nausea, vomiting, bloody stool) or radiographic findings of bowel inflammation are well-documented toxicities of immunotherapy agents. These toxicities are more common and better described for anti-CTLA-4 antibodies than for anti-PD-1 agents. Severe colitis occurs with anti-CTLA-4 drugs in up to 7% of patients compared to 1.8% of those receiving anti-PD-1 therapy. Diagnostic work-up includes testing stool sample for Clostridium difficile , Salmonella, and other bacterial enteropathogens. Other causes of colitis may include viral infection like cytomegalovirus. Patients with severe, stable, or progressive course should undergo computed tomography. In case of uncertain diagnosis colonoscopy with biopsy should be considered.
Treatment of immunotherapy-related diarrhea and colitis depends on the grade of toxicity.
Grade 1 with mild symptoms that last for less than one week can be managed symptomatically: antidiarrheals, bowel rest, adequate fluid, and electrolyte replacement. Short delay in immune checkpoints inhibitors therapy could be considered in individual cases.
Grade 2 (4 to 6 liquid stools per day over baseline or abdominal pain, or blood in stool, or nausea, or nocturnal episodes) usually does not require hospital admission, but sometimes is the reason to stop therapy, until the symptoms resolve to grade 1.
For severe forms (grade 3 or 4) described as more than 7 liquid stools per day or a life-threatening condition, therapy interruption, hospitalization, gastroenterologist consultation, and corticosteroids 1 to 2 mg/kg are mandatory. The tumor necrosis factor inhibitor infliximab can be used in patients who do not respond to intravenous corticosteroids within 48 to 72 hours.
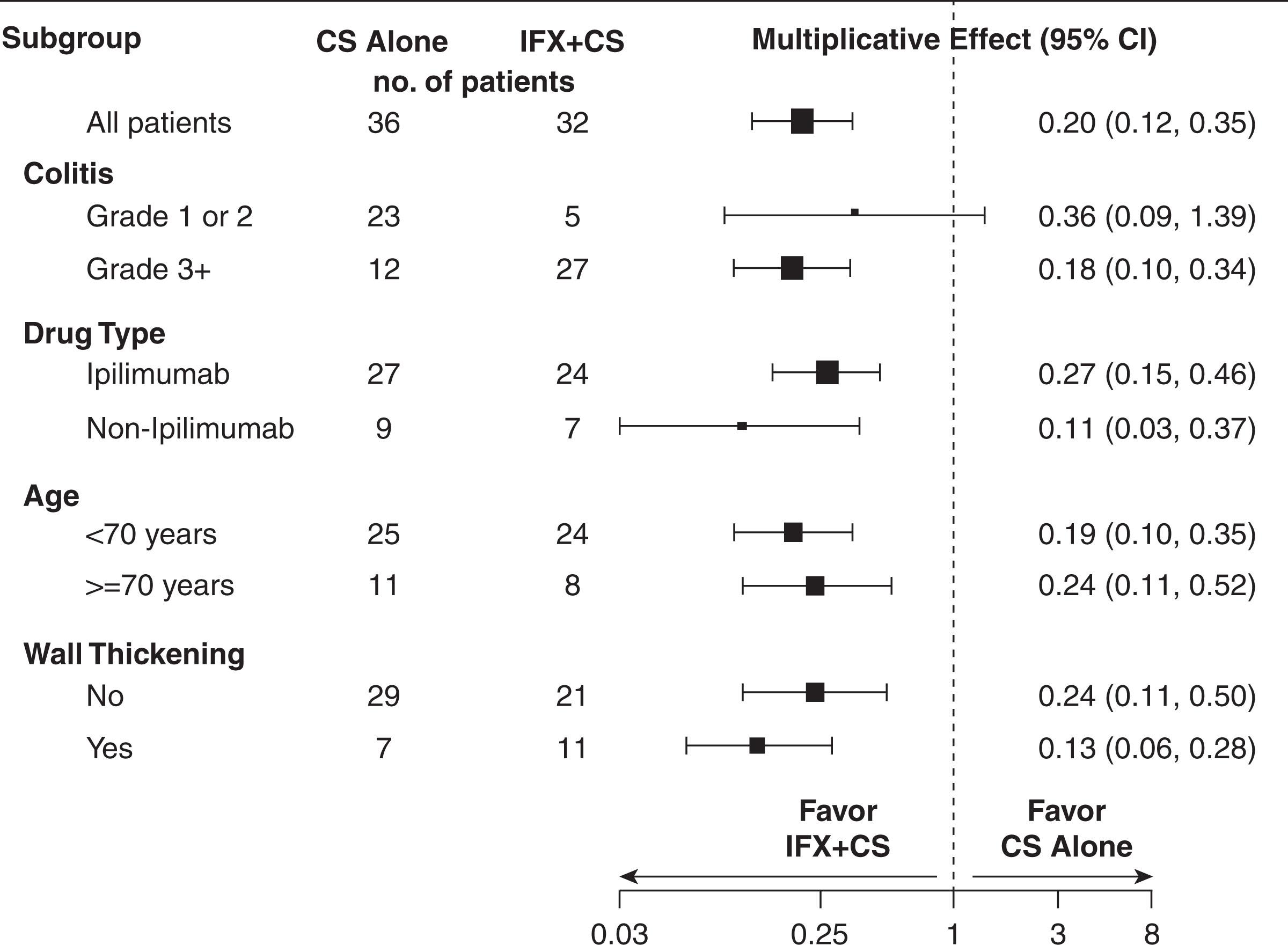
In a D.H. Johnson et al. analysis, treatment with infliximab was associated with faster symptom resolution compared with corticosteroids alone. In the infliximab group higher-grade colitis were observed (grade 3/4: 86% vs. 34%; P < .001), but despite that, median time to diarrhea resolution (3 vs. 9 days; P < .001), steroid titration (4 vs. 13 days; P < .001), and total steroid duration (median 35 vs. 51 days; P =.150) were shorter than in the corticosteroid group ( Fig. 97.2 ).
Generally, hepatitis occurs within 8 to 12 weeks after the therapy initiation. According to different reports, immunotherapy-related hepatitis is more common for CTLA-4 than for PD-1 inhibitors (10% vs. 5% respectively). It presents with asymptomatic elevation of aspartate aminotransferase, alanine aminotransferase (AST/ALT). An increase in bilirubin levels and fever are less common. The grade of toxicity depends on AST/ALT or total bilirubin elevation. Diagnostic work-up includes complete blood count, comprehensive metabolic panel, and stool sampling (culture, C. difficile assay, ova and parasite exam). Testing for lactoferrin and calprotectin is optional. The differential diagnosis includes viral hepatitis, alcohol use, medication toxicities, and metastatic liver disease which should be ruled out by appropriate testing which may include computed tomography and liver biopsy. Endoscopic evaluation is highly recommended for Grade 2 cases or higher to stratify patients for early treatment with infliximab based on the endoscopic findings and to determine the safety of resuming anti-PD-1/PD-L1 therapy.
Treatment options for grade 2 hepatotoxicity include withholding immunotherapy until symptoms resolution to grade 1, repeated liver function tests after 1 week, and, if rising, starting prednisone at a dose of 1 mg/kg. In the case of grade 3 toxicity with immune checkpoint inhibitors, therapy should be discontinued, with possible resuming if symptoms resolve to grade 1. If symptoms persist ≥3 to 5 days on oral corticosteroids, consider administering intravenous steroids. Monitoring of AST/ALT and total bilirubin should be done every 48 hours, switching to weekly testing following improvement, until they normalize. Patients refractory to intravenous corticosteroids are rare and they should be treated with mycophenolate mofetil (500 mg every 12 hours). Infliximab should be avoided in patients with elevated AST/ALT since infliximab can worsen hepatotoxicity.
Pneumonitis is a rare but potentially fatal complication of checkpoint inhibitors. It is usually a therapeutic dilemma, because drug-induced pneumonitis is a diagnosis of exclusion, and other diagnoses, like infection and tumor growth, need to be ruled out. This IrAE is reported more frequently with anti-PD-1 agents than with anti-CTLA-4 therapy. The median time of symptom manifestation is almost 3 months from initiation of therapy. Clinical presentation is diverse: patients can be asymptomatic or have progressive dyspnea, cough, wheezing, or even be in need of supplemental oxygen. In the diagnostic algorithm, computed tomography is preferred over chest radiography. In moderate and life-threating cases, bronchoscopy should be considered for diagnostic evaluation.
Treatment of immunotherapy-related pneumonitis depends on severity:
Mild pneumonitis may only require withholding of immune checkpoint inhibitors and subsequent computed tomography in 2 to 4 weeks.
Moderate cases demand immunotherapy interruption and prescription of corticosteroids (0.5 to 1 mg/kg daily).
Grade 4 requires complete discontinuation of immunotherapy, intravenous corticosteroids, and possible additional immunosuppression with infliximab, mycophenolate mofetil, intravenous immunoglobulin, or cyclophosphamide. Up to 12% of patients with such severe IrAE are resistant to the treatment despite immunosuppression.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here