Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Human immunodeficiency virus type 1 (HIV-1) infection involves the central nervous system (CNS) beginning during primary viremia and continuing over the course of untreated infection. Although the majority of patients with HIV infection do not present with neurologic symptoms, HIV disease has protean manifestations in the CNS determined largely by host characteristics such as immune status, treatment history, and access and adherence to antiretroviral therapy (ART).
Nearly four decades after the report of five unexplained cases of Pneumocystis carinii pneumonia in men suffering from what would eventually become recognized as the acquired immunodeficiency syndrome (AIDS), HIV infection remains the subject of intense biochemical, molecular, clinical, and epidemiologic investigation. HIV infection is a bloodborne and sexually transmitted disease that impacts both individuals and public health and has disproportionately affected vulnerable and marginalized populations including the poor and underserved, injection drug users, commercial sex workers, and men who have sex with men. The epidemic has also changed in the last decades. What once was largely a disease of young men who have sex with men in urban centers has expanded to affect all populations; the highest route of overall transmission of HIV is currently through heterosexual contact, often in rural areas, underserved and marginalized populations, and individuals unaware of their risk of HIV acquisition.
It is estimated that there are currently 38 million people living with HIV worldwide and 1.7 million new infections annually. Around one million individuals die from the disease and its sequelae each year. Although the greatest number of new infections and the worst outcomes occur in the lowest-resource settings in sub-Saharan Africa and Southeast Asia, nearly 40,000 individuals are newly infected in the United States each year.
A better understanding of the virus’s characteristics, including its pathogenesis and transmission patterns, has led to both prophylactic and therapeutic interventions, but many questions about the pathogenesis of HIV infection remain unanswered. An increased focus on the effects of HIV infection beyond the immune system has emerged, including its end-organ effects on the nervous system.
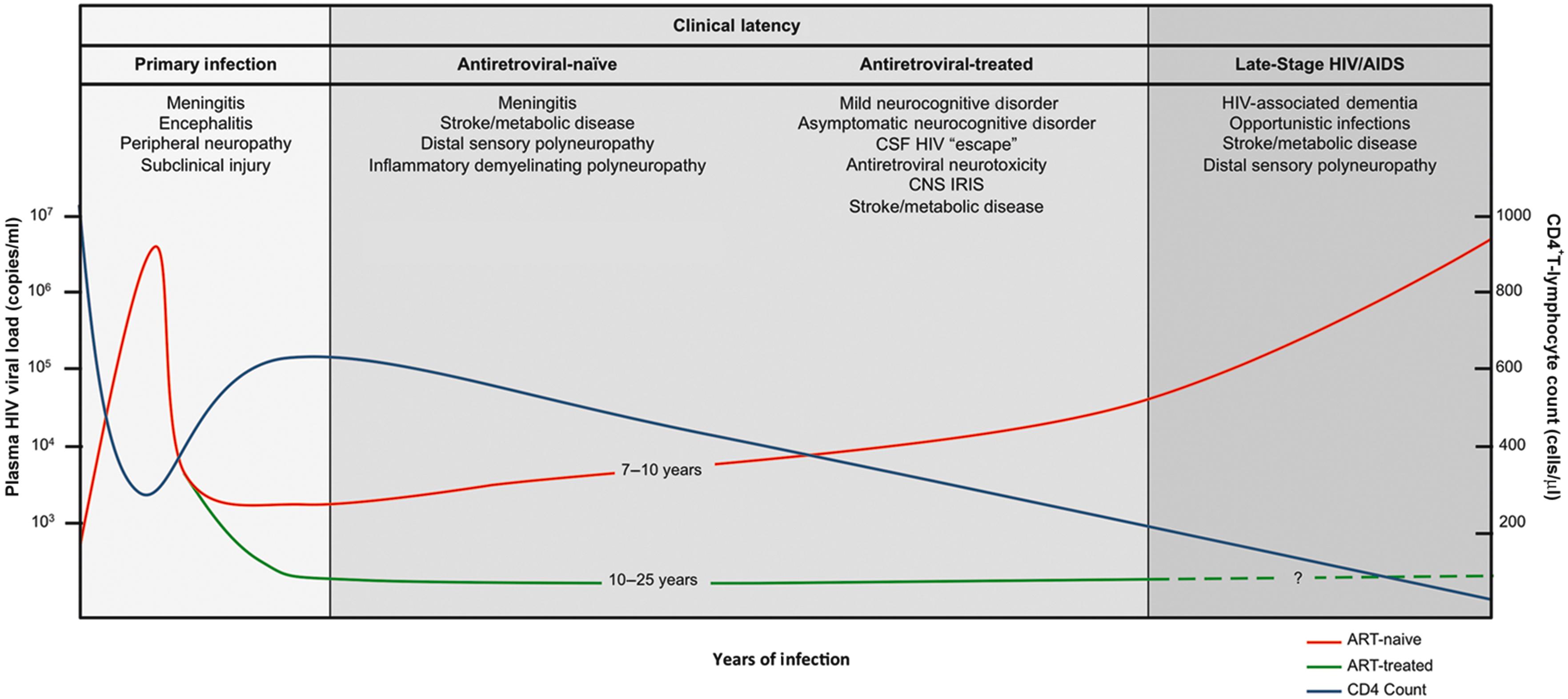
HIV is a single-stranded, positive-sense retrovirus of the genus Lentivirus . Once transmitted to a new host, the virus uses a reverse transcriptase to transcribe viral RNA into DNA. This DNA is, in turn, integrated into the genome of the host. The stages of HIV infection are divided into the acute phase that immediately follows transmission and includes the CD4 + T-cell nadir; the asymptomatic latent phase characterized by a slow decline in CD4 + T cells; and the symptomatic phase of chronic AIDS. In antiretroviral-naïve patients, it takes approximately 10 years to develop AIDS, although this time may be extended indefinitely with combination ART ( Fig. 43-1 ). Following the development of AIDS, median survival is typically between 1 and 4 years.
It is increasingly recognized that the body’s reaction to HIV infection can be as damaging as the activity of the virus itself, and this is particularly true in the nervous system. The inflammatory milieu that is induced by the activity of HIV in invading cells and that triggers an immune response has important implications throughout the time course of infection. It is thought that HIV pathogenesis within the CNS is mediated primarily by inflammation induced by both systemic immune activation as well as HIV infection of macrophages, microglia, and astrocytes. It is hypothesized that an indirect toxic injury to neurons results from a cascade of chronic and persistent neuroinflammation, in part fostered by cytokines produced by infected or activated cells within the brain. In the earliest stages of infection, the virus crosses the blood–brain barrier for the first time and initiates cytokine release that contributes to breakdown of that barrier. Other effects include a cerebrospinal fluid (CSF) pleocytosis, macrophage and lymphocyte activation, interference with neuronal synthesis and maintenance pathways, and ultimately neuronal injury that can be detected through biomarkers and neuroimaging. Although control of the virus through the initiation of ART can decrease the viral load and suppress the immune response, the CNS remains particularly vulnerable to further insult and may not normalize as well as systemic compartments.
Recent studies of the end-organ effects of HIV beyond the immune system have led to the recognition of a biologic compartmentalization that allows for infection and injury of target tissue, independent evolution of the virus from its counterparts in the plasma, and protection of the virus from systemic therapy. The existence of these distinct biologic compartments, such as those in the breast and genital tract, facilitates viral replication, complicates viral eradication, and leads to organ-specific effects. Chronic HIV infection is associated with the establishment of a CNS reservoir of infection, as evidenced by the detection of HIV DNA in perivascular brain macrophages, microglial cells, and astrocytes, compartmentalization of HIV quasi-species in CNS tissues, and clinical cases of isolated CNS “escape” from ART. Targeting treatments and strategies toward CNS reservoirs of HIV has become an important aspect of recent efforts to achieve a cure for HIV infection.
The introduction of ART in the mid-1990s fundamentally altered the landscape of both systemic and neurologic HIV disease. The profound immunodeficiency associated with HIV/AIDS that acted as the substrate for the “classic” neurologic manifestations of the disease itself and the opportunistic infections with which it was associated can now be significantly delayed or prevented, transforming the disease from one that was uniformly fatal into a manageable chronic illness.
In general, ART suppresses both plasma and CSF viral levels and improves neurologic outcomes in patients with HIV infection. Typically, plasma HIV RNA suppression is paralleled by suppression in the CSF, and the initiation of ART also limits the extent of immune activation in the CSF, as measured by white blood cell count and immunologic markers. With systemic control of the virus and improved immune status has come a striking decline in the occurrence of neurologic opportunistic infections over the last three decades, while the attenuation of viral replication and immune activation in the CNS has resulted in a decline in the incidence of the most dramatic forms of HIV-associated neurologic disease. Nevertheless, even individuals with well-controlled HIV infection continue to experience neurologic dysfunction which, although often less pronounced than the illnesses experienced 40 years ago, has the potential to seriously impact productivity and quality of life.
What was previously defined in its most severe form as the AIDS–dementia complex is now represented by a spectrum of disorders reflecting the variability in presentation, outcome, and impact of neurologic disease.
HIV-associated neurocognitive disorder (HAND) comprises a diverse set of neurocognitive diseases, ranging from clinically asymptomatic impairment to severe dementia. HAND is a clinical diagnosis defined by abnormalities identified through neuropsychologic testing and is subdivided into three categories of increasing severity: asymptomatic neurocognitive impairment, mild neurocognitive disorder, and HIV-associated dementia (HAD) ( Table 43-1 ). These diagnoses require the administration of a specific neuropsychologic test battery assessing language, attention, executive functioning, memory (learning and recall), processing speed, visuospatial abilities, and motor skills. While the incidence of the most severe manifestations of HAND has decreased in the setting of widespread access to ART, mild-to-moderate HAND has persisted and has become the most prevalent primary CNS complication of HIV infection.
| Asymptomatic Neurocognitive Impairment (ANI) | Mild Neurocognitive Disorder (MND) | HIV-Associated Dementia (HAD) | |
|---|---|---|---|
| Neuropsychologic Testing Abnormalities | One standard deviation below the age- and education-adjusted mean on two cognitive domains | One standard deviation below the age- and education-adjusted mean on two cognitive domains | Two standard deviations below the age- and education-adjusted mean on two cognitive domains, especially learning, information processing, and attention/concentration |
| Effect on Everyday Function | None | Mild | Marked |
| May be noted by self or others, including impairments in mental acuity, work efficiency, maintenance of home life, and social functioning | May be noted by self or others, manifesting as severe difficulty in work, independent maintenance of home life, and social functioning | ||
| Dementia | Absent | Absent | Present |
| Other causes such as opportunistic infection, vascular dementia, severe active drug use must be absent |
The most benign and most common manifestation of HAND is asymptomatic neurocognitive impairment, which has been identified in approximately one-third of people living with HIV. It is characterized as a subclinical cognitive decline with decreased performance on neuropsychologic testing in two or more domains not attributable to comorbid conditions (e.g., mood disorders, substance abuse). It specifically requires that no negative impact on everyday functioning is present, distinguishing it from mild neurocognitive disorder.
It is unclear whether asymptomatic neurocognitive impairment predicts more severe neurologic impairment later in the course of HIV, whether it contributes to neuropathologic vulnerability, and whether initiation of ART before clinically evident neurocognitive impairment develops might prevent ongoing deterioration.
A form of mild cognitive impairment is being recognized increasingly in individuals treated with ART, who typically have a relatively reconstituted immune system characterized by high CD4 + T-cell counts and suppressed or undetectable viral loads. As the population of patients with chronic, well-controlled HIV infection continues to grow, so too does the overall prevalence of mild neurocognitive disorder, which approaches 12 percent in some studies.
The diagnosis of mild neurocognitive disorder depends on the detection of abnormalities in neuropsychologic testing in attention, processing speed, memory, and executive function domains. In contrast to those with asymptomatic neurocognitive impairment, patients typically are aware of a subtle impairment in cognitive ability and increased difficulty carrying out activities of daily living. Mild neurocognitive disorder can also affect both pyramidal and extrapyramidal motor systems, producing symptoms such as ataxia, tremor, and incoordination that may worsen over time. It can lead to behavioral symptoms that are independent of those associated with mood disorders concomitant with HIV infection.
The disorder initially described 25 years ago as the AIDS–dementia complex is now known as HAD, the most dramatic manifestation of HAND. The diagnosis remains a challenge, as there are no diagnostic studies or laboratory tests that are specific for HAD.
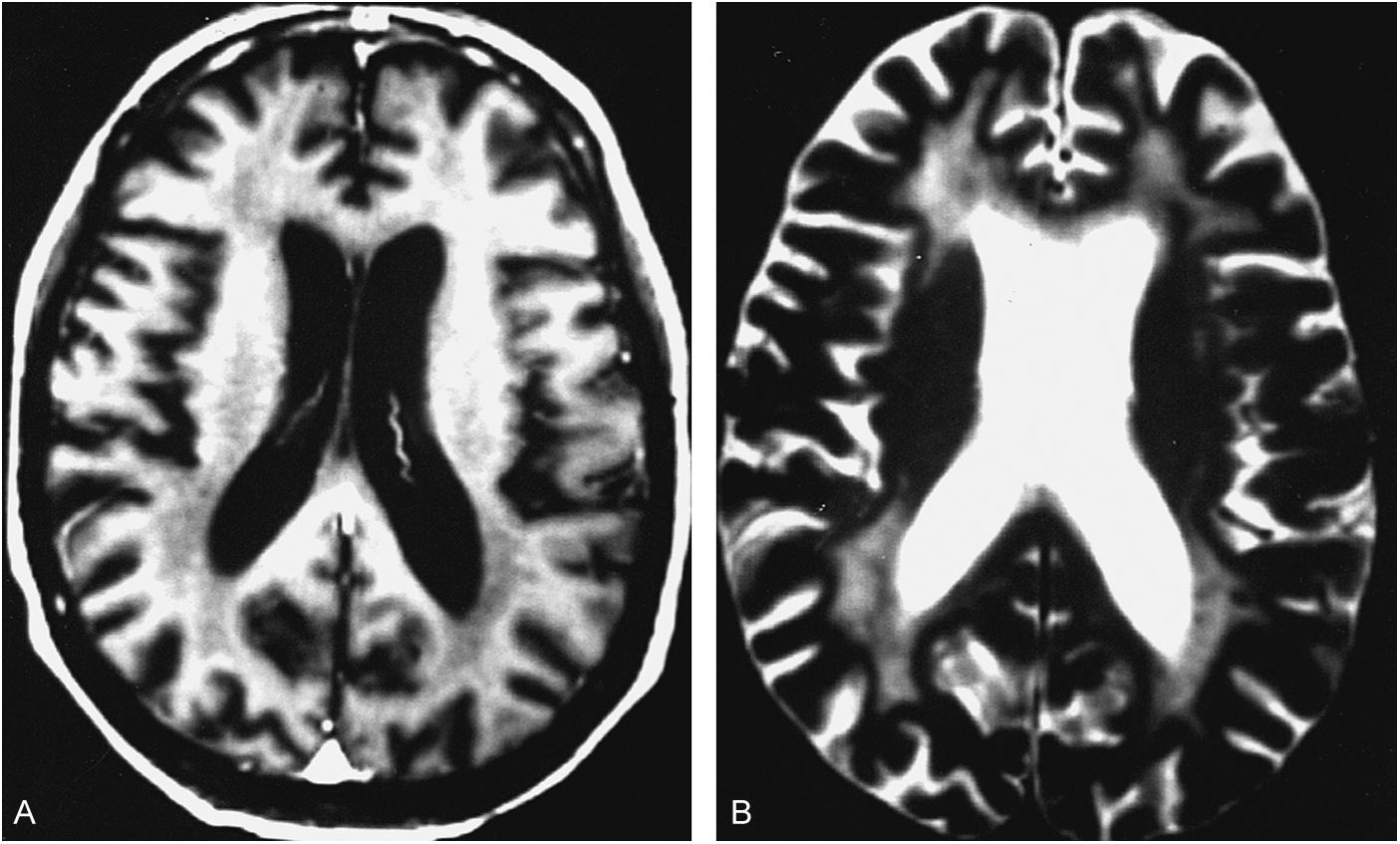
The diagnosis of HAD is based on progressive neurocognitive impairment and the exclusion of other conditions that can mimic these symptoms including CNS opportunistic infections and mass lesions such as tumors. HAD is still identified most commonly in patients not taking ART; the prevalence in treated patients may be as low as 2 percent. HAD most typically occurs in patients with slowed cognitive processing in the context of longstanding HIV infection and is often accompanied by motor abnormalities such as slowed movement and spastic gait along with hyperactive muscle stretch reflexes. Evaluation with computed tomography (CT) or magnetic resonance imaging (MRI) of the brain is used to exclude other AIDS-related neurologic conditions, including opportunistic infections and CNS lymphoma. With these diagnoses excluded, diffuse cerebral atrophy and subcortical or periventricular white matter changes are consistent with, although not specific for, HAD.
Although the biologic substrate of HAND in the setting of ART is unknown, one potential mechanism involves injury occurring in the earliest stages of HIV infection. Such injury would begin before treatment is initiated and would continue along a trajectory that may or may not be mitigated by initiation of ART. After several years, a combination of host susceptibility and disease factors may result in the development of symptomatic neurologic disease.
Another possibility is that, due to the compartment-specific nature of CNS HIV infection, neurologic injury is incurred despite the initiation and continuation of systemically suppressive treatment. Even in individuals with no overt signs or symptoms of neurocognitive impairment, the presence of HIV in the CNS may result in constant low-level inflammation and immune activation that may lead to ongoing brain injury. CSF immune activation, brain inflammation detected by magnetic resonance spectroscopy, and microglial activation persist in patients taking long-term suppressive ART.
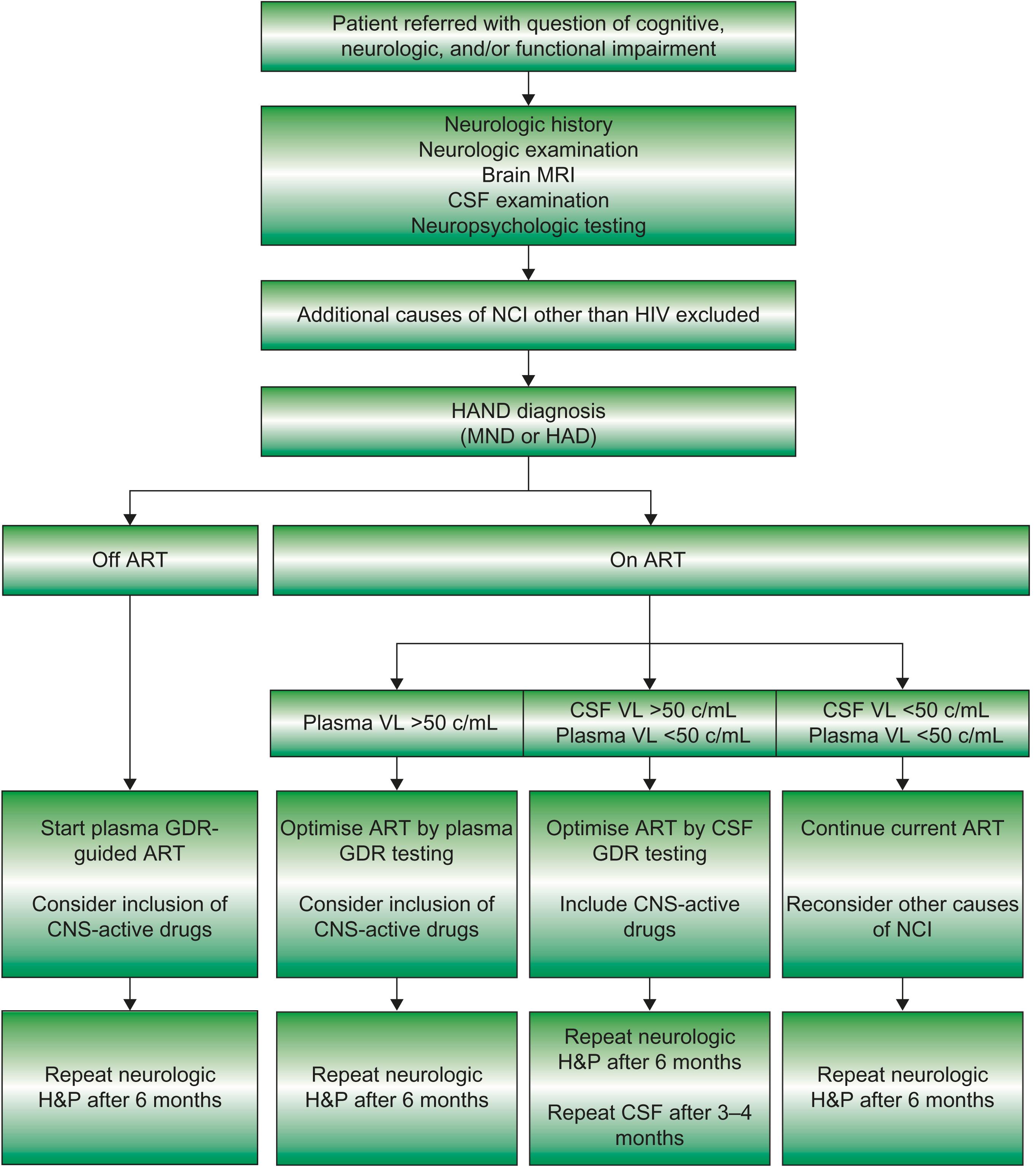
Because HAND has no specific diagnostic markers, it is necessary to exclude CNS opportunistic infections, delirium, toxic-metabolic disorders, psychiatric disease, and other neurodegenerative conditions before making the diagnosis. Although traditional neuroimaging is useful in excluding other HIV-associated disease processes, including CNS lymphoma, infections, and inflammatory processes, there are no findings on standard imaging that are specific for HAND ( Fig. 43-2 ). Efforts to use more advanced neuroimaging techniques to identify mild HAND have included brain mapping, high-field structural imaging, functional MRI, and magnetic resonance spectroscopy. The utility of these new modalities remains to be determined. Fig. 43-3 provides an algorithm for the identification and management of HAND.
Performance on neuropsychologic tests in severe forms of HAND improves with the initiation of ART. The introduction of ART has been associated with a decreased incidence and prevalence of HAD despite increases in the prevalence of milder forms of HAND. While initiation of ART significantly improves cognitive performance and neurologic function in antiretroviral-naïve patients with HAD, this improvement is frequently incomplete. Furthermore, many patients already taking ART also have HAND.
Adjunctive therapies that target the CNS have been studied in an attempt to attenuate the inflammatory events that are postulated in the pathogenesis of HAND. Adjunctive therapies other than antiretroviral agents, including memantine, selegiline, and nimodipine, have not been successful. Valproic acid and lithium have been hypothesized to decrease HIV replication and neuroinflammation through their glycogen synthase kinase-3β activity, as have serotonin reuptake inhibitors such as citalopram and paroxetine through an unknown mechanism; no clear improvement in HAND has been demonstrated with these agents. Due to its anti-inflammatory and antiviral effects, the antibiotic minocycline has also been suggested as a potential therapy, but randomized trials have been unsuccessful. Statins, which decrease progression of vascular disease in people living with HIV and have widespread anti-inflammatory effects, are under study. Methylphenidate successfully treats fatigue and psychomotor slowing in patients with HAND, but does not alter the course of the disease. An alternate treatment strategy using antiretroviral drugs that penetrate into the CNS is discussed later.
By separating the CNS from the systemic circulation, the blood–brain and blood–CSF barriers affect the ability of antiretroviral agents to access the CNS compartment. In addition, local spontaneous replication of HIV within this viral sanctuary may allow for independent mutations of HIV virions. While the response of CSF HIV RNA levels to ART parallels that in the plasma, the rate of decay in the CSF may be more gradual in some patients, suggesting a compartmentalization characterized by slower cell turnover, extended macrophage release, or attenuated drug entry.
Because of the blood–brain barrier, HIV in the CNS may be protected from the full effect of antiretroviral agents, especially those drugs that are large or hydrophilic. The CNS penetration-effectiveness (CPE) index represents an effort to estimate quantitatively the relative ability of each antiretroviral agent to penetrate the CNS and interfere with CSF HIV replication. Each agent is assigned a CPE score based on presumed CNS exposure and efficacy of the drug in relation to others in its class ( Table 43-2 ).
| CNS Penetration-Effectiveness Score | ||||
|---|---|---|---|---|
| Drug Class | 4 | 3 | 2 | 1 |
| NRT inhibitor | Zidovudine |
|
|
|
| NNRT inhibitor | Nevirapine |
|
Etravirine | – |
| Protease inhibitor | Indinavir/r |
|
|
|
| Entry inhibitor | Vicriviroc | Maraviroc | – | Enfurvirtide |
| Integrase inhibitor | – | Raltegravir | – | – |
Some studies have shown that antiretroviral regimens with higher CPE scores tend to be more successful at achieving HIV RNA suppression in the CNS. While more potent HIV RNA suppression in this compartment might be expected to lead to better neurocognitive outcomes, results have been mixed, with some studies showing that regimens with higher CPE scores actually lead to poorer neurocognitive performance or only benefit those patients treated with more than the standard ART regimen of three drugs.
The inability of ART to control the potential reservoir of HIV that exists in monocytes or other myeloid lineage cells has been proposed as one possible explanation for continued neurocognitive impairment in the setting of ART. As a result, a monocyte efficacy (ME) score has been proposed as an alternative means of quantifying the ability of antiretroviral agents to affect neurologic outcomes. Preliminary work has suggested that the ME score correlates with neurocognitive performance even when the CPE score does not.
Several new clinical syndromes have been recognized in patients taking systemically suppressive ART, often after a long period of apparently successful treatment.
CSF escape is a phenomenon of CSF/plasma discordance. In an individual with suppressed plasma HIV RNA, CSF escape occurs when the CSF HIV RNA is detectable despite plasma HIV suppression. In an individual with detectable plasma HIV RNA, CSF escape occurs when the level of HIV RNA in the CSF exceeds that in the plasma. A subset of individuals with CSF escape become symptomatic. Patients with symptomatic CSF escape include a broad spectrum of individuals with ART-managed HIV, including those persistently suppressed or well-controlled for many years, as well as those with recent spikes in viral load and those beginning to achieve viral control.
The pathogenesis of symptomatic CSF escape is related to the failure of ART to suppress local CNS infection despite peripheral CD4 + T-cell reconstitution. Although detectable CSF HIV RNA in the context of ART is required, it is not sufficient, since asymptomatic CSF escape has been identified as a frequent finding (up to 18%) in asymptomatic individuals undergoing lumbar puncture in the context of HIV research studies. A moderately reconstituted immune system may actually contribute to the symptomatic syndrome by eliciting a symptomatic inflammatory response and providing a substrate for ongoing discordant HIV replication within the CNS. This process may lie on the spectrum of the immune reconstitution inflammatory syndrome (IRIS), but may represent a stable state of antigen and immune response within the CNS, rather than the exaggerated response upon immune reconstitution that occurs in IRIS. Low CD4 + T-cell nadir (i.e., <250 cells/mm 3 ) may be a risk factor for CSF “escape,” suggesting that a history of advanced immunosuppression may confer increased risk of the syndrome. In addition to resistance and poor penetration, poor medication adherence might also contribute by leading to insufficient drug concentrations in the CSF and thus to the selection of resistant virus within this compartment.
The clinical features of symptomatic CSF escape include a variety of subacute to acute progressive neurologic symptoms including cognitive, sensory, and motor impairment resulting in substantial disability. The diagnosis is achieved through the recognition of plasma/CSF discordance and the exclusion of opportunistic infections including progressive multifocal leukoencephalopathy (PML). Imaging may show white matter hyperintensities on T2-weighted MRI sequences; these are often periventricular. Resistance genotyping of CSF HIV may demonstrate mutations in the CSF viral subpopulation. The CSF typically shows elevated protein levels and white blood cell counts consistent with an inflammatory response beyond that which is seen in healthy controls without HIV infection and neurologically asymptomatic subjects with HIV who are taking ART. Elevated CSF neopterin and pronounced inflammation with CD8 + T-lymphocyte infiltration is seen on brain biopsy. Some patients with CSF escape demonstrate improvement when the ART regimen is optimized based upon the results of genotyping and the CPE score of the current ART regimen.
Notably, a secondary CSF escape syndrome can also occur in the setting of a documented non–HIV-related CNS infection or inflammatory condition. It is possible that each type of CSF escape is associated with a different pathophysiologic process.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here