Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Hemostasis is an ordered process involving cellular and biochemical components that functions to limit blood loss secondary to vascular injury, maintain intravascular blood flow, and promote revascularization after thrombosis. Normal physiologic hemostasis is a constant balance between procoagulant pathways responsible for generating localized hemostatic clot and counter-regulatory mechanisms that inhibit uncontrolled thrombus propagation or premature thrombus degradation. Vascular endothelium, platelets, plasma coagulation proteins, and fibrinolytic enzymes play equally important roles in this process. Derangements in this delicate system because of acquired or congenital disease states in addition to medications can lead to excessive bleeding or pathologic thrombus formation.
Injury of the vascular endothelium results in platelet deposition at the injury site, a process often referred to as primary hemostasis. Although this initial platelet plug may prove adequate for a minor injury, control of more significant bleeding necessitates formation of a stable clot reinforced by crosslinked fibrin—a process mediated by activated plasma clotting factors and often referred to as secondary hemostasis ( Fig. 23.1 ). Advances in our understanding of the cellular and molecular processes underlying hemostasis suggest a far more complex and nonlinear interplay between vascular endothelium, platelets, and clotting factors than is reflected in this model ; however, the terms primary and secondary hemostasis remain relevant for descriptive and diagnostic purposes.
![Fig. 23.1, Normal Hemostasis. (A) Normal endothelium. Procoagulants (factors [F] VII, VIII, IX, X, XIII, prothrombin), fibrinogen, and platelets circulate in their inactive forms. Anticoagulants (tissue factor pathway inhibitor [TFPI], heparin, and tissue plasminogen activator [tPA]) actively prevent endothelial spontaneous thrombus formation. (B) Vascular injury, initial phase. Subendothelial tissue factor (TF) exposed to circulating FVII forms a TF:VII complex. TF:VII activates FIX and FX. FIXa binds to platelets. FXa activates FV (FVa) to form prothrombinase complex, which converts localized, small amounts of prothrombin to thrombin. (C) Vascular injury, role of thrombin. Thrombin (a) activates FX and FV to form prothrombinase complexes that generate the secondary thrombin burst, (b) activates platelets, (c) separates FVIII from von Willebrand factor (vWF) and activates FVIII, (d) converts fibrinogen to fibrin, (e) activates factor XI, and (f) activates FXIII, the stabilizer of crosslinked fibrin. A stable clot is formed. (D) Control of coagulation and fibrin clot dissolution. Antithrombin (AT) binds heparin and potently inhibits thrombin activity. TFPI binds to FXa to inhibit the TF:VIIa complex. Plasminogen is activated to plasmin by tPA and cleaves fibrin into soluble fibrin split products. Fig. 23.1, Normal Hemostasis. (A) Normal endothelium. Procoagulants (factors [F] VII, VIII, IX, X, XIII, prothrombin), fibrinogen, and platelets circulate in their inactive forms. Anticoagulants (tissue factor pathway inhibitor [TFPI], heparin, and tissue plasminogen activator [tPA]) actively prevent endothelial spontaneous thrombus formation. (B) Vascular injury, initial phase. Subendothelial tissue factor (TF) exposed to circulating FVII forms a TF:VII complex. TF:VII activates FIX and FX. FIXa binds to platelets. FXa activates FV (FVa) to form prothrombinase complex, which converts localized, small amounts of prothrombin to thrombin. (C) Vascular injury, role of thrombin. Thrombin (a) activates FX and FV to form prothrombinase complexes that generate the secondary thrombin burst, (b) activates platelets, (c) separates FVIII from von Willebrand factor (vWF) and activates FVIII, (d) converts fibrinogen to fibrin, (e) activates factor XI, and (f) activates FXIII, the stabilizer of crosslinked fibrin. A stable clot is formed. (D) Control of coagulation and fibrin clot dissolution. Antithrombin (AT) binds heparin and potently inhibits thrombin activity. TFPI binds to FXa to inhibit the TF:VIIa complex. Plasminogen is activated to plasmin by tPA and cleaves fibrin into soluble fibrin split products.](https://storage.googleapis.com/dl.dentistrykey.com/clinical/Hemostasis/0_3s20B9780323796774000231.jpg)
The vascular endothelium serves an important role in reducing unprovoked thrombosis. Healthy endothelial cells possess antiplatelet and anticoagulant activity that functions to inhibit clot formation. The negatively charged vascular endothelium repels platelets, and endothelial cells produce potent platelet inhibitors such as prostacyclin (prostaglandin I 2 ) and nitric oxide that prevent adhesion of quiescent platelets. An adenosine diphosphatase (CD39) expressed on the surface of vascular endothelial cells also serves to block platelet activation through degradation of adenosine diphosphate (ADP), a potent platelet activator. The vascular endothelium also plays a pivotal anticoagulant role by expressing several inhibitors of plasma-mediated hemostasis. Endothelial cells can increase activation of protein C, an anticoagulant, via surface expression of thrombomodulin (TM), which acts as a cofactor for thrombin-mediated activation of protein C, making its activation 1000 times faster. Endothelial cells also produce tissue factor pathway inhibitor (TFPI), which inhibits the procoagulant activity of factor Xa and the TF–VIIa complex. Finally, the vascular endothelium synthesizes tissue plasminogen activator (tPA), which is responsible for activating fibrinolysis, a primary counterregulatory mechanism limiting clot propagation.
Despite these natural defense mechanisms, upon injury the endothelium may shift the balance to promote platelet adhesion, activation, and aggregation. Damage to vascular endothelial cells exposes the underlying extracellular matrix (ECM), which contains collagen, von Willebrand factor (vWF), and other platelet-adhesive glycoproteins. Platelet receptors for vWF (glycoprotein Ib-IX-V complex) and collagen (integrin α 2 β 1 ) facilitate platelet adhesion to the site of vessel injury. Absence of either vWF (von Willebrand disease) or glycoprotein Ib-IX-V complex receptors (Bernard–Soulier syndrome) results in a clinically significant bleeding disorder.
In addition to promoting their adhesion to the vessel wall, the platelet interaction with collagen serves as a potent stimulus for the subsequent phase of platelet activation. During the activation phase, platelets secrete agonists such as thromboxane A 2 (TxA 2 ) and release granular contents, resulting in recruitment and activation of additional platelets and propagation of plasma-mediated coagulation. Platelets contain two specific types of storage granules: α-granules and dense bodies. α-Granules contain numerous proteins essential to hemostasis and wound repair, including fibrinogen, coagulation factors V and VIII, vWF, platelet-derived growth factor, and others. Dense bodies contain the adenine nucleotides ADP and adenosine triphosphate (ATP), in addition to calcium, serotonin, histamine, and epinephrine. Redistribution of platelet membrane phospholipids during activation exposes newly activated platelet surface receptors and binding sites for calcium and coagulation factor activation complexes, which is critical to propagation of plasma-mediated hemostasis. During activation, platelets also undergo structural changes to develop pseudopod-like membrane extensions and to release physiologically active microparticles, which serve to dramatically increase the platelet membrane surface area.
During the final phase, platelet aggregation, activators released during the activation phase recruit additional platelets to the site of injury. Newly active glycoprotein IIb/IIIa receptors on the platelet surface gain higher affinity for fibrinogen, thereby promoting crosslinking and aggregation with adjacent platelets. The importance of these receptors is reflected by the bleeding disorder associated with their hereditary deficiency, Glanzmann thrombasthenia.
Plasma-mediated hemostasis has been described as a cascade or waterfall sequence of steps involving the serial activation of proenzymes (zymogens) to enzymes and cofactors to accelerate and amplify fibrin generation by thrombin. Traditionally, the coagulation cascade has been depicted as extrinsic and intrinsic pathways, both of which culminate in a common pathway in which fibrin generation occurs ( Fig. 23.2 ). This cascade model has proven to be an oversimplification, as it does not fully reflect in vivo hemostasis; however, it remains a useful descriptive tool for organizing discussions of plasma-mediated hemostasis and interpreting in vitro coagulation tests.
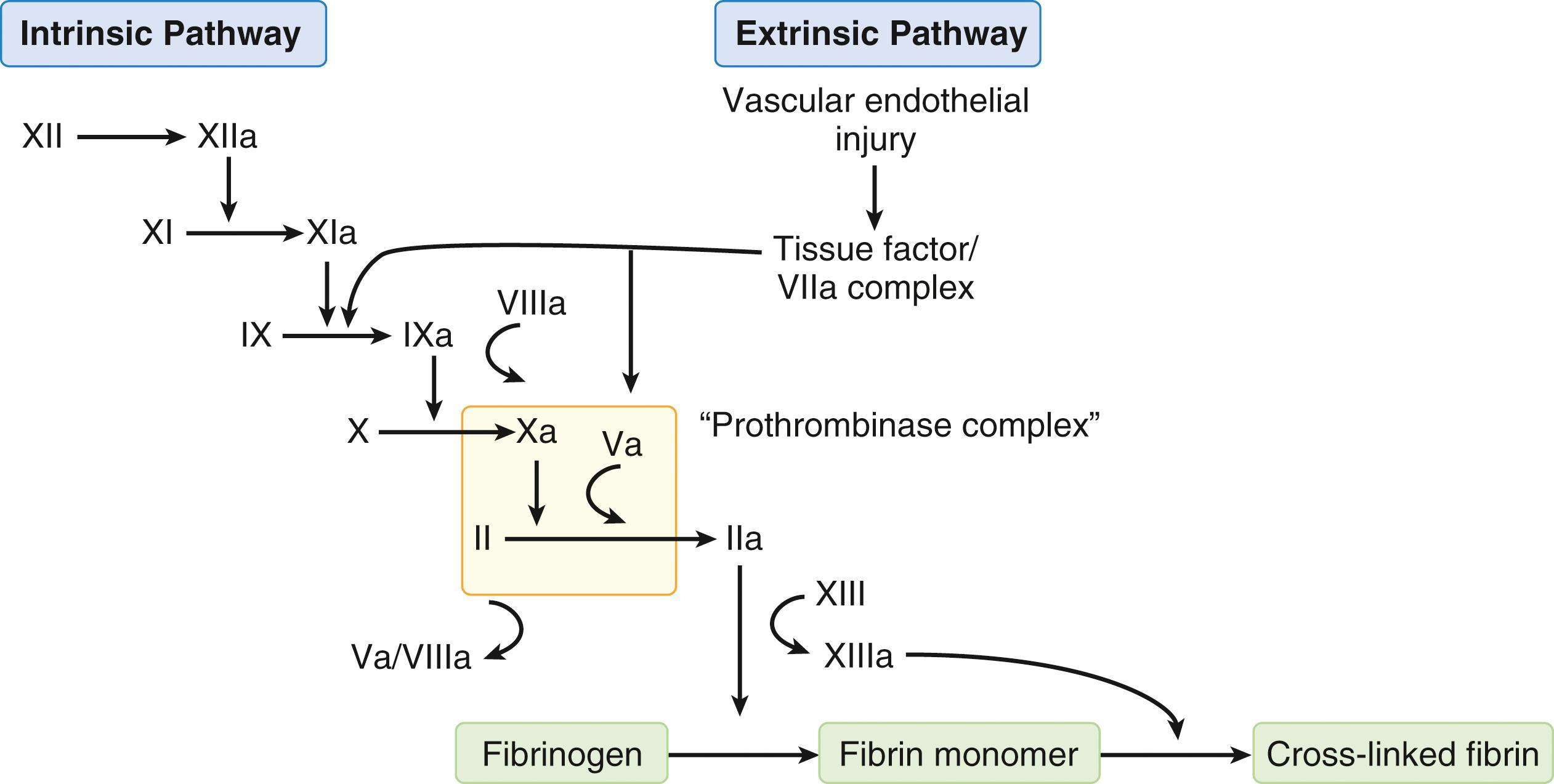
The extrinsic pathway of coagulation is now understood to represent the initiation phase of plasma-mediated hemostasis and begins with exposure of blood plasma to tissue factor (TF). TF is prevalent in subendothelial tissues surrounding the vasculature; however, after vascular injury, small concentrations of factor VIIa circulating in plasma form phospholipid-bound activation complexes with TF, factor X, and calcium to promote conversion of factor X to Xa. Additionally, the TF/factor VIIa complex activates factor IX of the intrinsic pathway, further demonstrating the key role of TF in initiating hemostasis. Recent cell-based models of coagulation suggest that thrombin generation by way of the extrinsic pathway is limited by a natural inhibitor, TFPI; however, the small quantities of thrombin generated do activate factor XI and the intrinsic pathway. The intrinsic pathway then subsequently amplifies and propagates the hemostatic response to increase overall thrombin generation.
The final pathway, common to both extrinsic and intrinsic coagulation cascades, depicts thrombin generation and subsequent fibrin formation. Signal amplification results from activation of factor X by both intrinsic (FIXa, FVIIIa, Ca 2+ ) and extrinsic (TF, FVIIa, Ca 2+ ) tenase complexes. The tenase complexes in turn facilitate formation of the prothrombinase complex (FXa, FII [prothrombin], FVa [cofactor], and Ca 2+ ), which mediates a surge in thrombin generation from prothrombin. Thrombin proteolytically cleaves fibrinogen molecules to generate fibrin monomers, which polymerize into fibrin strands to form a clot. Finally, factor XIII is activated by thrombin and acts to covalently crosslink fibrin strands, producing an insoluble fibrin clot resistant to fibrinolytic degradation.
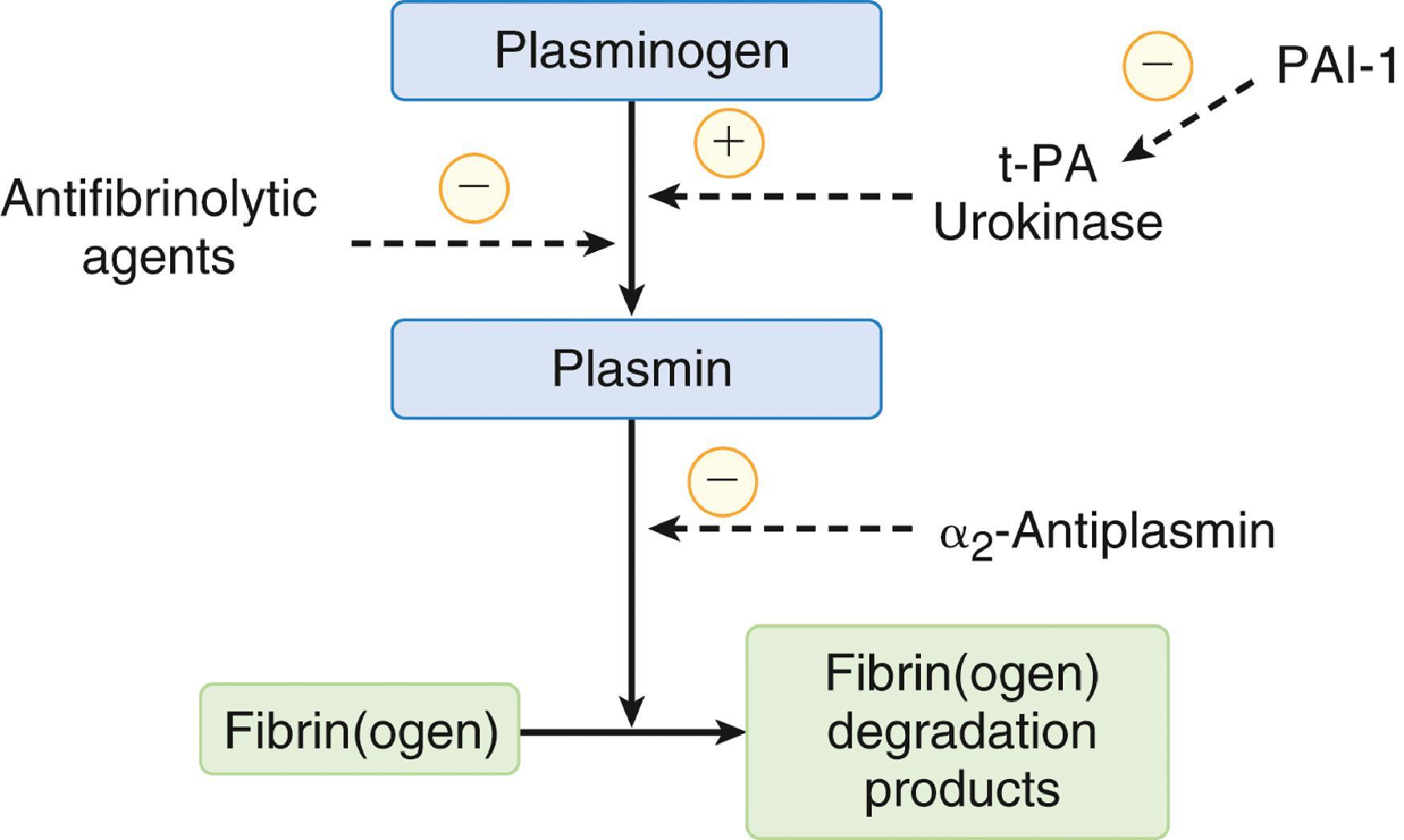
Once activated, regulation of hemostasis proves essential to limit clot propagation beyond the injury site. Four major counterregulatory pathways have been identified that appear particularly important for downregulating hemostasis: fibrinolysis, TFPI, the protein C system, and serine protease inhibitors (SERPINs). The fibrinolytic system comprises a series of amplifying reactions that ultimately convert plasminogen to plasmin, a serine protease, which is responsible for the degradation of fibrin and fibrinogen ( Fig. 23.3 ). In vivo, plasmin generation is most often accomplished by release of tPA or urokinase from the vascular endothelium. Activity of tPA and urokinase is accelerated in the presence of fibrin, thereby limiting fibrinolysis to areas of clot formation. In addition to enzymatic degradation of fibrin and fibrinogen, plasmin inhibits coagulation by degrading essential cofactors V and VIII and reducing platelet glycoprotein surface receptors essential to adhesion and aggregation. As the fibrin clot is broken down, fibrinolytic activity is reduced by the rapid inhibition of free plasmin. Furthermore, excessive fibrinolysis is prevented by the function of two key SERPINs, namely plasmin-activator inhibitor-1 (PAI-1) and α 2 ‐antiplasmin. PAI-1 serves as the primary inhibitor of tPA and urokinase, thereby decreasing plasmin generation, whereas α 2 ‐antiplasmin directly inactivates circulating plasmin.
TFPI binds and inhibits factor Xa through the formation of membrane-bound complexes. These factor Xa–TFPI complexes also act to inhibit TF/factor VIIa complexes, thereby downregulating the extrinsic coagulation pathway. The protein C system proves particularly important in regulating coagulation through inhibition of thrombin and the essential cofactors Va and VIIIa. After binding to thrombomodulin on the surface of the endothelial cell, thrombin's procoagulant function decreases and instead its ability to activate protein C is augmented. Activated protein C (APC), complexed with the cofactor protein S, degrades both factors Va and VIIIa. Loss of these critical cofactors limits formation of tenase and prothrombinase complexes essential to formation of factor Xa and thrombin, respectively. One of the most significant SERPINs regulating hemostasis is antithrombin (AT, formerly antithrombin III). AT inhibits thrombin in addition to factors IXa, Xa, XIa, and XIIa. Heparin binds AT, causing a conformational change that accelerates AT-mediated inhibition of targeted enzymes by over 100-fold.
Certain hereditary or acquired disorders, systemic diseases, and environmental conditions can predispose a patient to excessive bleeding after tissue injury, including surgery. Given the complexity of the hemostatic system, bleeding can result from defects in or deficiency of coagulation factors, platelets, fibrinolysis, and vascular integrity. Patients with less than 20% to 30% of normal coagulation factor activity or platelet counts of less than 50,000/μL are more likely than patients with normal values to have uncontrolled intraoperative bleeding. Bleeding diatheses vary in clinical presentation depending on what component of the hemostatic system is affected.
Inherited disorders of hemostasis include those involving platelet quantity and function, coagulation factor deficiencies, or disorders of fibrinolytic pathways. Among these inherited bleeding disorders, von Willebrand disease (vWD) is the most common, affecting up to 1% of the population. vWD is categorized into three main types (type 1, 2, and 3), with most cases demonstrating an autosomal dominant inheritance pattern ( Table 23.1 ). vWF is synthesized by megakaryocytes and endothelial cells and once released from these cells, it circulates as a series of multimers formed from a basic dimer subunit. Under normal conditions vWF plays a critical role in platelet adhesion to the ECM and prevents degradation of factor VIII by serving as a carrier molecule. vWD is characterized by quantitative or qualitative deficiencies of vWF resulting in defective platelet adhesion and aggregation. Classically, patients with vWD describe a history of easy bruising, recurrent epistaxis, and menorrhagia, which are characteristic of defects in platelet-mediated hemostasis. In more severe cases (i.e., type 3 vWD), concomitant reductions in factor VIII may lead to serious spontaneous hemorrhage, including hemarthroses.
| Type | Characteristic | Inheritance | Prevalence | Diagnosis | Treatment |
|---|---|---|---|---|---|
| 1 | Partial quantitative deficiency of VWF | AD | Up to 1% | ↓ vWF:Ag ↓ vWF:RCo |
DDAVP FVIII/vWF concentrate |
| 2 | Qualitative defect of vWF (subtypes below) | ||||
| 2A | ↓ binding of vWF to platelets ↓ large multimers |
AD | Uncommon | ↓ vWF:Ag ↓↓↓ vWF:RCo ↓ large multimers |
DDAVP FVIII/vWF concentrate |
| 2B | ↑ binding of vWF to platelets ↓ large multimers |
AD | Uncommon | ↑↑↑ LD-RIPA | FVIII/vWF concentrate |
| 2M | ↓ vWF function despite normal large multimers | AD | Uncommon | ↓ vWF:Ag ↓↓ vWF:RCo ↓ RIPA |
DDAVP FVIII/vWF concentrate |
| 2N | ↓ binding of VWF to FVIII | AR | Uncommon | ↓↓ FVIII | DDAVP FVIII/vWF concentrate |
| 3 | Absent vWF | AR | Rare (1:250,000–1:1,000,000) | Absent vWF:Ag | FVIII/vWF concentrate Factor VIII concentrate Platelet transfusion |
Routine coagulation studies are generally not helpful in the diagnosis of vWD, as the platelet count and prothrombin time (PT) will be normal in most patients and the activated partial thromboplastin time (aPTT) may demonstrate mild-to-moderate prolongation depending on the level of factor VIII reduction. Initial screening tests for vWD involve measurement of vWF levels (vWF antigen) and vWF platelet binding activity in the presence of the ristocetin cofactor, which leads to platelet agglutination. Mild cases of vWD often respond to desmopressin acetate (DDAVP), which results in the release of vWF from endothelial cells. Use of vWF:factor VIII concentrates (e.g., Humate-P) may be indicated in the perioperative period if there is a significant bleeding history.
Hemophilia A (factor VIII deficiency) and hemophilia B (factor IX deficiency) are both X-linked inherited bleeding disorders most frequently presenting in childhood as spontaneous hemorrhage involving joints and/or deep muscles. Hemophilia A is more common with an incidence of 1:5000 males, whereas hemophilia B occurs in 1:30,000 males. The severity of the disease is dependent on an individual's baseline factor activity level. Severe disease, defined by less than 1% of coagulation factor activity, occurs in approximately two-thirds of patients with hemophilia A and one half of patients with hemophilia B. Classically, laboratory testing in patients with hemophilia reveals prolongation of the aPTT, whereas the PT, bleeding time, and platelet count remain within normal limits. However, a normal aPTT may also be seen in mild forms of hemophilia, and it is important to exclude vWD as a cause of factor VIII deficiency.
An increasingly common complication of hemophilia is the development of alloantibodies to factors VIII or IX, which block exogenous factor activity. This can occur in up to 30% of patients with severe hemophilia A and 3% to 5% of those with severe hemophilia B. Several approaches to reduce bleeding in these patients include substitution of porcine factor VIII, administration of activated factor VIII bypass activity (FEIBA) or nonactivated prothrombin complex concentrates (PCCs), or treatment with recombinant factor VIIa (rFVIIa).
Less common inherited factor deficiencies include deficiencies of factors VII, XI, XII, and XIII. Factor VII deficiency (prevalence: 1 in 500,000) most commonly presents with excessive bleeding after invasive procedures; heavy menstrual bleeding; or mucosal tract, joint, and muscle bleeding. Bleeding is uncommon with factor VII activity levels >10% and can be managed with rFVIIa. Factor XI deficiency, known as hemophilia C or Rosenthal syndrome (prevalence: 1 in 1,000,000), is characterized by isolated prolongation in aPTT and variable bleeding severity. Factor XI activity levels, however, do not correlate well with bleeding risk. Most individuals do not experience spontaneous bleeding, hemarthrosis, or muscle hematomas, though bleeding episodes can occur under situations of hemostatic challenge such as trauma, surgery, or childbirth. Factor XII deficiency (prevalence: 1 in 1,000,000) can cause marked prolongation of aPTT but is associated with clotting rather than bleeding. Factor XIII is involved in stabilizing the fibrin clot. Factor XIII deficiency (prevalence: 1 in 2,000,000) presents with delayed bleeding after hemostasis, impaired wound healing, and, occasionally, pregnancy loss. Laboratory evaluation in these patients will demonstrate normal aPTT and PT, but the diagnosis can be confirmed by measurement of factor XIII activity levels.
Medications represent the most significant cause of acquired coagulopathy in perioperative patients. In addition to anticoagulants such as heparin and warfarin, the increasing number of direct oral anticoagulants (DOACs) and antiplatelet drugs has further complicated perioperative management. Additionally, several classes of medications may unintentionally increase bleeding risk because of side effects, primarily through inhibition of platelet aggregation. These drugs include β-lactam antibiotics, nitroprusside, nitroglycerin, nitric oxide, and selective serotonin reuptake inhibitors (SSRIs), all of which can result in clinically significant bleeding in individuals with higher baseline risk. These medications should be considered in patients with an otherwise unexplained coagulopathy.
Vitamin K is an essential fat-soluble vitamin that is required for the carboxylation of factors II, VII, IX, and X and proteins C and S. Without carboxylation, these factors cannot bind to the phospholipid membrane of platelets and participate in hemostasis. Vitamin K is present in dietary sources (leafy greens) and also synthesized by bacteria in the gastrointestinal tract. Patients who are fasting, have poor dietary intake, or are receiving total parenteral nutrition and those with fat malabsorption (obstructive jaundice, intestinal ileus or obstruction, or bowel resection) are prone to vitamin K deficiency. As gut microbiota are a source of vitamin K, newborns, who have not yet developed normal microbiota, and patients undergoing oral antibiotic therapy are also susceptible to vitamin K deficiency.
The liver is the primary site for production of procoagulant factors, including fibrinogen; prothrombin (factor II); factors V, VII, IX, X, XI, and XII; the anticoagulants protein C and S; and AT. Severe liver disease impairs synthesis of coagulation factors, produces quantitative and qualitative platelet dysfunction, and impedes clearance of activated clotting and fibrinolytic proteins. Laboratory findings commonly associated with liver disease include a prolonged PT and possible prolongation of the aPTT, suggesting that these individuals are at increased risk of bleeding. However, these abnormal values only reflect decreases in procoagulant factors and do not account for parallel decreases in anticoagulant factors (protein C, protein S, and AT). As a result, patients with chronic liver disease are thought to have a rebalanced hemostasis and actually generate amounts of thrombin equivalent to healthy individuals.
Similarly, thrombocytopenia from platelet sequestration in the spleen is often observed in patients with liver disease and portal hypertension. However, levels of the plasma metalloprotease ADAMTS13, responsible for cleaving vWF multimers, are also decreased in chronic liver disease and result in high circulating levels of large vWF multimers, which promote platelet aggregation. Consequently, this increase in vWF may partially correct for thrombocytopenia and platelet dysfunction but can also result in a prothrombotic state and increase clotting risk.
Fibrinolysis of a formed clot is also aberrant in patients with liver disease. Excessive fibrinolysis is prevented by thrombin-activatable fibrinolysis inhibitor (TAFI), which blocks activation of plasmin from plasminogen. TAFI is synthesized by the liver, and because levels are decreased in patients with chronic liver disease, it was believed that such individuals are at increased bleeding risk because of hyperfibrinolysis. However, levels of PAI-1, an inhibitor of tPA and urokinase, are also increased in liver disease and may serve to normalize fibrinolytic activity. Thus, in patients with chronic liver disease, hemostatic mechanisms are rebalanced, but decreases in procoagulant and anticoagulant factors create a tenuous equilibrium that is easily disrupted. As a result, these patients are at risk for both bleeding and inappropriate clotting.
Platelet dysfunction commonly occurs in association with chronic renal failure and uremia and has primarily been attributed to decreased platelet aggregation and adhesion to injured vessel walls. Impaired adhesion is likely the result of defects of glycoprotein IIb/IIIa, which facilitates platelet binding of fibrinogen and vWF. Additionally, accumulation of guanidinosuccinic acid and the resulting increase in endothelial nitric oxide synthesis further decrease platelet responsiveness. Red blood cell (RBC) concentration has also been suggested to contribute to impaired platelet activity, as correction of anemia shortens bleeding times. This is thought to be the result of the increased RBC mass displacing platelets from the center of the vessel and bringing them into close proximity of the endothelium, thereby promoting adhesion.
Disseminated intravascular coagulation (DIC) is a pathologic hemostatic response to TF/factor VIIa complex that leads to excessive activation of the extrinsic pathway, which overwhelms natural anticoagulant mechanisms and generates intravascular thrombin. Numerous underlying disorders may precipitate DIC, including sepsis, trauma, amniotic fluid embolus, malignancy, or incompatible blood transfusions ( Table 23.2 ). Most often, DIC presents clinically as a diffuse bleeding disorder associated with consumption of coagulation factors and platelets during widespread microvascular thrombotic activity resulting in multiorgan dysfunction. Laboratory findings typical of DIC include reductions in platelet count; prolongation of the PT, aPTT, and thrombin time (TT); and elevated concentrations of soluble fibrin and fibrin degradation products (D-dimers). However, DIC is both a clinical and laboratory diagnosis, so laboratory data alone do not provide sufficient sensitivity or specificity to confirm a diagnosis.
| Category | Conditions |
|---|---|
| Infections | Bacterial (gram-negative bacilli, gram-positive cocci) Viral (CMV, EBV, HIV, VZV, hepatitis) Fungal (histoplasma) Parasites (malaria) |
| Malignancy | Hematologic (AML) Solid tumors (prostate cancer, pancreatic cancer) Malignant tumors (mucin-secreting adenocarcinoma) |
| Obstetric causes | Amniotic fluid embolism Preeclampsia/eclampsia Placental abruption Acute fatty liver of pregnancy Intrauterine fetal demise |
| Massive inflammation | Severe trauma Burns Traumatic brain injury Crush injury Severe pancreatitis |
| Toxic/immunologic | Snake envenomation Massive transfusion ABO blood type incompatibility Graft versus host disease |
| Other | Liver disease/fulminant hepatic failure Vascular disease (aortic aneurysms, giant hemangiomas) Ventricular assist devices |
Institution of cardiopulmonary bypass (CPB) by directing blood flow through an extracorporeal circuit causes significant perturbations to the hemostatic system. Initial priming of the bypass circuit results in hemodilution and thrombocytopenia. Adhesion of platelets to the synthetic surfaces of the bypass circuit further decreases platelet counts and contributes to platelet dysfunction. During CPB, expression of platelet surface receptors important for adhesion and aggregation (GPIb, GPIIb/IIIa) are downregulated and the number of vWF-containing α-granules are decreased, thereby impairing platelet function. Furthermore, induced hypothermia during CPB results in reduced platelet aggregation and plasma-mediated coagulation by decreasing clotting factor production and enzymatic activity. Increased plasmin generation may also occur during CPB, a process that accelerates clot lysis. Thus, antifibrinolytic drugs are often administered to decrease intraoperative blood loss.
Uncontrolled hemorrhage is a frequent cause of trauma-related deaths. Coagulopathy in this setting may be the result of acidosis, hypothermia, and hemodilution from resuscitation; however, an independent trauma-induced coagulopathy (TIC) is also experienced by these individuals. The anticoagulant effect of APC is thought to play a primary role in TIC by decreasing thrombin generation via inhibition of factor Va and VIIIa and promoting fibrinolysis through inhibition of PAI-1. Hypoperfusion subsequent to traumatic injury is thought to be the stimulus for APC activation. Additionally, tissue damage results in shedding of the endothelial glycocalyx (EG), a gel-like matrix with anticoagulant properties that lines the vascular endothelium. The EG contains proteoglycans such as syndecan-1, hyaluronic acid, heparan sulfate, and chondroitin sulfate, that when shed during endothelial injury, result in an “autoheparinization” phenomenon that contributes to TIC. Impaired platelet responsiveness also contributes to increased bleeding in TIC. Although platelet counts appear to be normal, response to various agonists, including ADP, arachidonic acid (AA), and collagen, is reduced and thought to be the result of “platelet exhaustion” resulting from activation caused by widespread release of ADP from injured tissues. This diffuse activation renders platelets unresponsive to subsequent stimulation. Platelet insensitivity to ADP is also associated with increased susceptibility of clots to tPA-mediated fibrinolysis. The importance of early treatment to reduce hyperfibrinolysis in trauma is supported by the findings of the Clinical Randomisation of an Antifibrinolytic in Significant Haemorrhage 2 (CRASH-2) trial, which demonstrated a mortality benefit from early administration of the antifibrinolytic tranexamic acid.
Mild cases of vWD often respond to DDAVP, which causes the release of vWF from endothelial cells. One dose of DDAVP (0.3 μg/kg IV) will produce a complete or near-complete response in the majority of patients with type 1 vWD. DDAVP is contraindicated in type 2B vWD, as it may precipitate thrombocytopenia, which could worsen bleeding. In the setting of more significant surgical bleeding, use of plasma-derived vWF:factor VIII concentrate (Humate-P) or recombinant vWF (Vonvendi) is indicated. If vWF concentrates are not available, cryoprecipitate, which contains high levels of vWF, can be used, but its use is considered second line, as most cryoprecipitate has not undergone the pathogen inactivation processes used in preparing vWF concentrates.
In most cases, perioperative management of patients with hemophilia A or B necessitates consultation with a hematologist and administration of recombinant or purified factor VIII or factor IX concentrates, respectively. Mild cases of hemophilia A may be treated with desmopressin. An increasingly common complication of hemophilia, particularly in the case of hemophilia A, has been the development of alloantibodies directed against the factor VIII protein. Administration of factor VIII concentrates will fail to control bleeding in patients with high-titer antibodies. Several approaches to reduce bleeding in these patients include substitution of porcine factor VIII, administration of activated FEIBA or nonactivated PCCs, or treatment with rFVIIa.
Management of DIC requires correction of the underlying condition precipitating hemostatic activation. Otherwise, treatment is mostly supportive and includes selective blood component transfusions to replete coagulation factors and platelets consumed in the process. The use of anticoagulants such as heparin remains controversial with recommendations that its use be limited to conditions with the highest thrombotic risk. Antifibrinolytic therapy generally is contraindicated in DIC owing to the potential for catastrophic thrombotic complications.
In the nonbleeding patient, treatment of thrombocytopenia in the form of platelet transfusion is usually withheld until the platelet count is less than 10,000/μL. In the patient who is actively bleeding or requires surgical intervention, platelet transfusion is recommended to a goal of 50,000/μL, or in some cases, such as intracranial hemorrhage or neurosurgery, 100,000/μL. Individuals who receive repeated platelet transfusions are at increased risk of forming antibodies to human leukocyte antigens (HLAs) or human platelet antigens. If such platelet alloimmunization develops, transfusion of standard platelet units may not increase platelet counts appropriately and use of HLA-matched units may be required.
Treatment of platelet dysfunction related to chronic renal disease includes administration of DDAVP (0.3 μg/kg IV), which stimulates release of vWF from endothelial cells. Additionally, conjugated estrogens (0.6 mg/kg/day intravenously for 5 days) have been demonstrated to shorten bleeding times, perhaps via decreased generation of nitric oxide. Transfusion of cryoprecipitate (rich in vWF and fibrinogen) may also be used to correct uremic platelet dysfunction; however, its use is often limited to patients with life-threatening bleeding because of the risks associated with transfusion of allogeneic blood products.
Thrombophilia, a propensity for thrombotic events, commonly manifests clinically in the form of venous thrombosis (deep venous thrombosis [DVT] or pulmonary embolus [PE]). The pathogenesis of thrombosis is thought to be caused by the Virchow triad (blood stasis, endothelial injury, and hypercoagulability). In the majority of cases, a risk factor or precipitating event is identified; however, a single factor generally does not result in clinically significant thrombosis. Instead, multiple factors act synergistically to increase risk. For example, thrombotic complications often occur after surgery or during pregnancy in association with an inherited thrombophilia; underlying malignancy; or other clinical risk factors such as obesity, smoking, or oral contraceptive use.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here