Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Human immunodeficiency virus (HIV) infection is associated with a number of hematologic complications throughout the course of the disease. To a certain extent, these complications depend on the stage of the disease and how well controlled HIV infection is but to a certain extent it does not. In untreated infection, the major hematologic complications were cytopenias, either isolated or multiple simultaneously. As combination antiretroviral therapy (ART) became standard of care in developed countries, cytopenias were observed far less frequently, and other complications were seen as a function of a longer life span. In particular, these are lymphomas and venous thromboembolic events; Kaposi sarcoma is not covered in this chapter because it is not a hematologic malignancy. Therefore, this review will focus on lymphoma (both Hodgkin and non-Hodgkin including acquired immunodeficiency syndrome [AIDS]-defining lymphomas) and on thromboembolic events with a section on cytopenias ( Table 152.1 ). The introduction will be a state of the art description of HIV itself and will focus on adults.
| Cytopenias including human immunodeficiency virus (HIV)-immune thrombocytopenia, anemia, and other cytopenias |
| Lymphoma |
|
| Thromboembolic events |
|
An estimated 38 million people are living with HIV worldwide, of which approximately 25.4 million are on ART. Most cases currently come from sub-Saharan Africa, followed by the Caribbean. In 2019, there were 1.7 million new infections documented and 690,000 AIDS-related deaths recorded, with 23% attributed to men who have sex with men (MSM), 19% to clients of sex workers and their partners, 8% to sex workers themselves, 10% to drug use, 2% to transgender individuals, and the remaining 38% were unknown in the report (other factors include sexual assault of women and vertical transmission). Compared to 2010, 2019 witnessed a 23% reduction in the incidence of HIV and a 39% reduction in AIDS-related mortality. Furthermore, when looking at patients from 2015 to 2019, 59% of patients had an undetectable viral load. Despite substantial global strides in achieving control of HIV, there are fears expressed by the Joint United Nations Programme on HIV/AIDS (UNAIDS) driven by adverse consequences of COVID-19 on HIV, as a result of which 20% of individuals were unable to access therapy for longer than 6 months. As a result, UNAIDS projects an increase of 110,000 AIDS-related deaths in the upcoming 2 years.
In the United States, the incidence of HIV between 2014 and 2018 was 36,400 per year diagnosed with HIV due to any cause. Of the newly diagnosed cases, 66% were attributed to MSM, 24% to heterosexual contact, 7% to drug use, and 4% to a combination of MSM and drug use. When stratified by race/ethnicity, new HIV infections were found in 43% Black/African American, 26% Hispanic/Latino, 26% White, and 6% Other. This highlights a racial/ethnic disparity in healthcare, as Blacks/African Americans account for only 13% of the general population. Furthermore, there were evident disparities in geographical locations within the United States, where 52% of all the cases were in the South, 15% in the Northeast, 13% in the Midwest, and 20% in the West of the United States. The geographical disparity of cases in the US may be related to poverty, lack of access to health care, lack of appropriate sex education, or other (unknown) factors. These are all issues that pertain to the application of management strategies in the future.
AIDS is caused by two lentiviruses, HIV types 1 and 2 (HIV-1 and HIV-2) were first documented in the United States in 1981 when clusters of Kaposi sarcoma and pneumocystis jiroveci pneumonia in homosexual men were identified. HIV-1, a single-stranded RNA retrovirus with multiple names, was identified as the culprit. It is believed to have been zoonotically transmitted to humans from African primates of chimpanzee origin in central Africa, harboring simian immunodeficiency virus (SIV) (40% to 60% DNA homology to HIV-1). Although there are no confirmed direct cases of such transmissions, HIV’s cross-over to humans is attributed to the exposure of blood from hunting bushmeat, similar to other zoonoses (i.e., Ebolavirus and SARS-CoV-2). HIV-1 consists of four distinct lineages, termed groups M, N, O, and P, each of which resulted from independent cross-species transmission events. The majority of HIV infections are caused by the M group, which is further subdivided into 11 clades: A to K. Subtype B is the predominant type in the Western world, whereas subtype C is dominant in southern Africa. The lesser-known virus, HIV-2, also from the same family and often not accounted for when speaking about “HIV,” is mostly found in West Africa and is theorized to have been zoonotically transmitted from sooty mangabey monkeys. It has six phylogenetic lineages, A to F. HIV-2 is characterized by low virulence, lower viral loads (in the thousands rather than millions), and individuals that remain as “long term non-progressors” to AIDS. When individuals do progress, the process mimics HIV-1 in pathology. One needs to be aware of HIV-2, especially when immunodeficiency is suspected in an individual from West Africa, as the fourth generation immunoassay for HIV detection in the United States only tests for HIV-1; the earlier tests identified both.
As mentioned above, most HIV is transmitted sexually. In the US, it is mostly found in MSM, whereas in sub-Saharan Africa, it is transmitted mostly via heterosexual penile-vaginal sex. Exposure to infected blood via injected drug use is another common way to acquire HIV. In the past, hemophiliacs and thalassemics also acquired HIV through contaminated blood transfusions in the 1970s and 1980s before the virus was identified, and other transfused individuals receiving multiple single donor products also were infected. Currently, screening of donors and viral eradication in plasma products has made this mode of viral acquisition very rare to essentially non-existent. Mothers can also transmit HIV either perinatally (maternal-fetal hemorrhage of blood or swallowed blood during delivery) or to neonates via breastmilk. Individuals are at an increased risk of contracting HIV if the exposure involves a high viral load (in the acute phase of the infection) or having damage to the mucosa thus providing access to the virus; this can be seen in co-infections with other sexually transmitted infections (STIs) or in unprotected receptive anal sex. Factors that are protective against acquiring HIV include the use of protection during intercourse and male circumcision since foreskin harbors a concentration of Langerhans cells and T cells that are HIV targets. Protective factors preventing the host from transmitting HIV include achieving an undetectable viral load by the host through compliance with ART. This includes the use of ART during pregnancy and delivery to prevent vertical transmission to the neonate, reducing risk from 25.5% to 8.3% in the first study, with further reductions seen in subsequent studies. Sexual partners of HIV-positive patients can also reduce their risk by taking pre-exposure prophylaxis (PrEP) consisting of tenofovir disoproxil with emtricitabine or lamivudine. If an individual is exposed to a high-risk sexual encounter or a needle-stick injury, that is, healthcare workers, then post-exposure prophylaxis (PEP) of 28 days duration with tenofovir + emtricitabine with raltegravir is indicated. The efficacy of PEP has been observed in retrospective studies, where a Cochrane systematic review concluded that HIV-positive individuals were less likely to have been taking PEP (OR 0.19, 95% CI 0.06 to 0.52).
HIV is composed of a viral capsule constructed of glycoproteins gp120 and gp41 that form a lipid bilayer. These glycoproteins facilitate entry into cells by binding to the CD4 receptor and chemokine co-receptors CXCR4 and/or CCR5. Within the viral capsule, there are two positive-sense, single-stranded RNA strands that carry nine genes. Of those, three are integral to viral replication and survival: gag, pol, and env genes transcribe core proteins p24 and p17, reverse transcriptase and integrase, and the glycoproteins respectively. In the cytoplasm, the enzyme reverse transcriptase generates a single strand of DNA from which a double DNA strand (dsDNA) is transcribed. dsDNA enters the nucleus, and viral integrase integrates the viral DNA into the host cell’s DNA. From then on, the virus uses the host’s machinery to replicate. The viral protease enzyme is vital in the final step of cleaving gag and gag-pol precursor proteins into the functional units known as gp120 and gp41.
HIV not only infects T cells and myeloid cells (monocyte/macrophages) but also glial cells in the central nervous system (CNS). Furthermore, there are resting memory CD4 T cells that contain HIV but do not contribute to viral replication, making up a “latent HIV reservoir” posing important challenges to the eradication of the virus despite the ability to achieve an undetectable viral load with ART. This latter can be very important in that CNS virus and/or HIV in other extra-vascular locations may not be eradicated by highly active antiretroviral therapy (HAART) and contributes to viral resurgence and ongoing inflammation. Targeting viral entry as a therapeutic strategy gained traction when a CCR5-delta 32 mutation, found in populations of European descent, conferred resistance to acquiring HIV infection in homozygotes by inhibiting viral entry. Entry inhibitors have been developed to target the CCR5. Further, this was proven in vivo in the only two cases cured of HIV after receiving hematopoietic stem cell transplantation selected for the CCr5-delta 32 mutation as part of the treatment for acute myeloid leukemia and Hodgkin lymphoma. In 2018, a physicist in China used CRISPR-Cas9 gene-editing technology to genetically engineer twin girls with knockout CCR5 to prevent them from contracting HIV from their father. In addition to the questionable clinical reasoning and the unethical nature of his actions, one could speculate that the twin girls may be at increased risk of autoimmune diseases, as CCr5-delta 32 mutation has been associated with MS and lupus nephritis. Nonetheless, these cases demonstrate the utility of targeting viral entry as a therapeutic modality although once an infection has occurred, the previously described latent cells may vitiate this strategy.
In the United States, the 4th generation enzyme immunoassay is the standard HIV screening test used to detect HIV in the acute setting, where it is able to detect HIV IgM, IgG antibodies, and p24 antigen 15 to 20 days after exposure in comparison to the first-generation enzyme immunoassay, which only detected IgG after 35 to 45 days. The gold standard confirmatory testing for HIV remains the western blot, which has a window of 45 to 60 days. The presence of neutralizing antibodies has raised the question of a possible vaccine, which to this day remains a very active but puzzlingly unsolved area of research. One of the first vaccines developed in 1998 targeted neutralizing antibodies against gp120. Despite achieving high titers in the serum, the vaccine did not work as the targeted region V3 was shielded within a trimeric structure rendering it inaccessible. Surprisingly, in vivo animal experiments demonstrated that neutralizing gp120 antibodies were able to prevent HIV infection in pig-tailed macaques receiving IgG from HIV-1 infected chimpanzees. Other explanations for the lack of neutralizing antibodies in humans include the mutation rate of the HIV reverse transcriptase, giving HIV the ability to escape the host’s immune system. Furthermore, there are many clades and subtypes of the virus, another factor making a universal vaccine even more challenging.
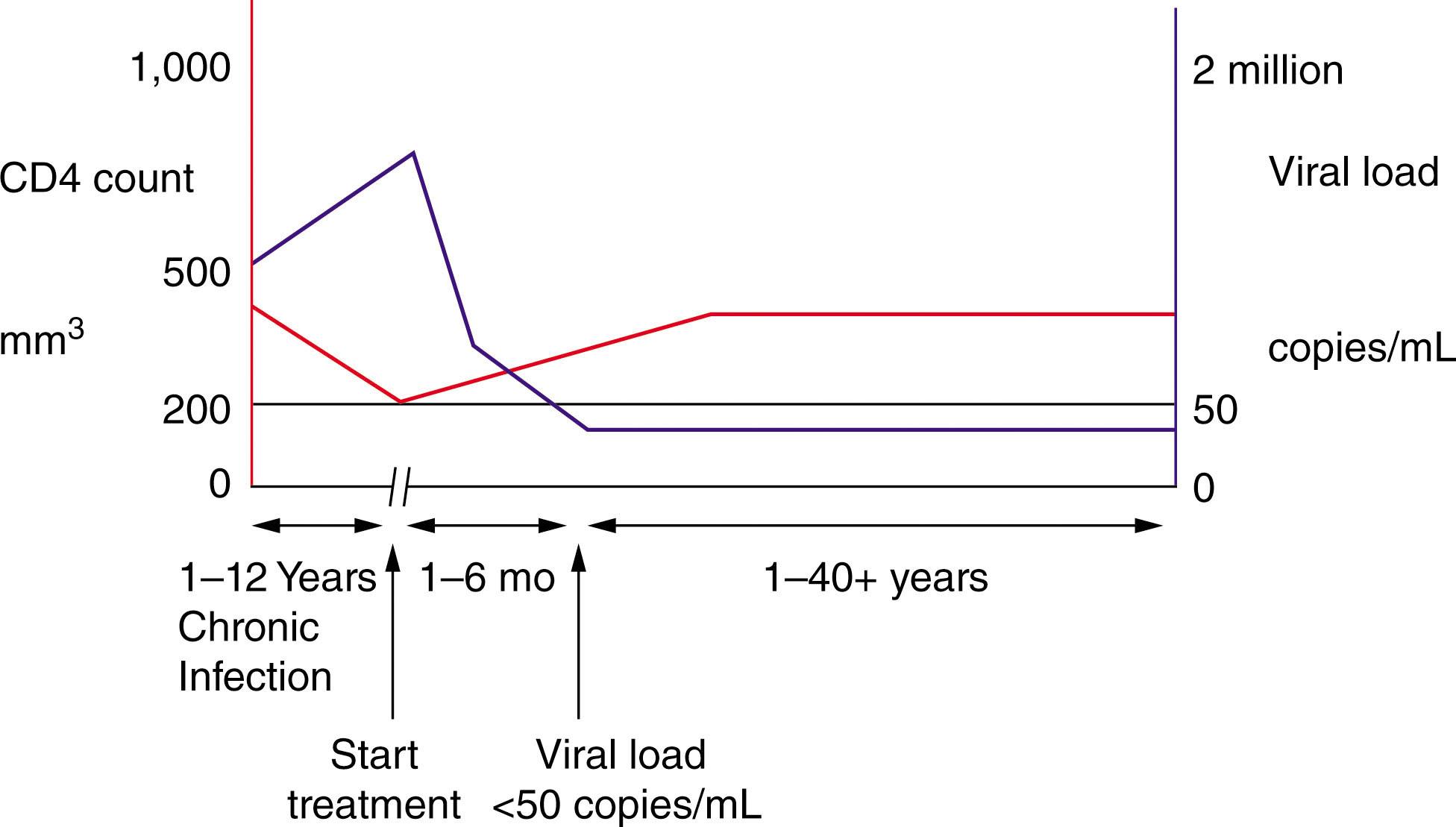
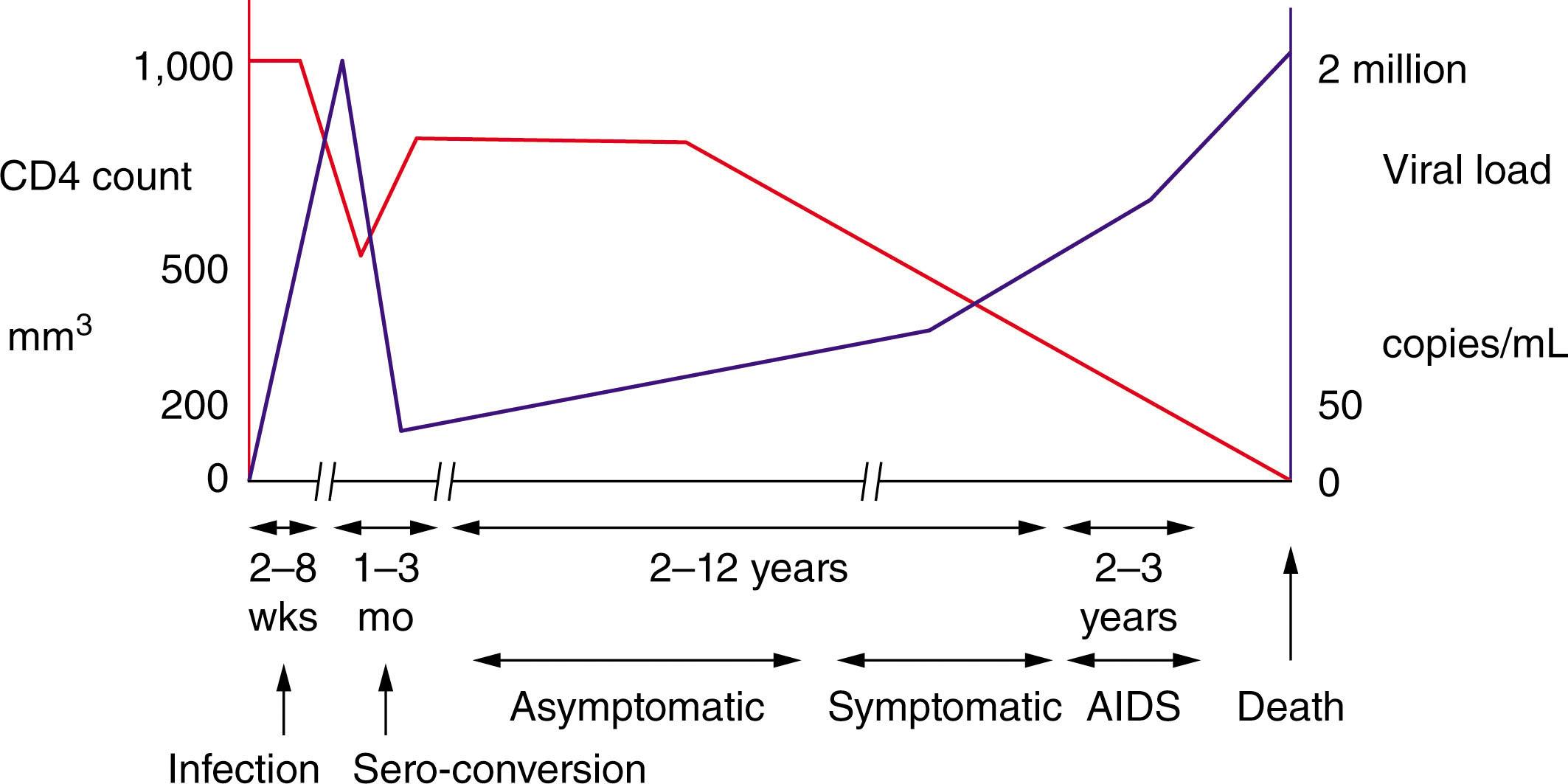
The highest viral load is seen in the acute infection phase, accompanied by a drop in the CD4 count. The viral load starts to decline after approximately 4 weeks because of the cytotoxic T-cell immune response. The CD4 count then starts to increase but does not completely recover to what it was prior to the infection, referred to as an immunological set point. This is followed by the asymptomatic phase, which can last for a decade. Without anti-HIV treatment, the virus continues to replicate, and the CD4 T cell count continues to decline until an AIDS-defining opportunistic infection (OI) or a malignancy causes death. AIDS, by definition, occurs either when the CD4 T cell count is below 200 or by having one of the AIDS-defining illnesses irrespective of the CD4 T cell count. This “natural history” of untreated HIV infection is mitigated by starting antiretroviral therapy, where the CD4 T cell count starts to climb up and the HIV plasma viral load declines, potentially to undetectable levels using commercial assays with sensitivity typically down to 50 HIV viral copies/mL, thus reducing morbidity and mortality drastically (see Fig. 152.2 ).
Currently, HIV is treated with ART targeting the replication process at various stages from entry to maturity. There are nucleoside/nucleotide reverse transcriptase inhibitors (NRTI), non-nucleoside reverse transcriptase inhibitors (NNRTI), integrase inhibitors (INSTI), and protease inhibitors (PI). Additional ART includes entry inhibitors that prevent the virus from entering the targeted cells. These include enfuvirtide (Fuzeon) and maraviroc (Selzentry). Most HIV-infected persons are on 2 NRTI and 1 INSTI. In the past, a person with HIV took 13 or more pills a day, which in addition to side effects, added challenges to medication compliance. Nowadays, patients with HIV take 1 to 4 pills/day depending on the presence of co-infections with hepatitis B or C and the genotypic profile of HIV. Newer medications also offer an improved side effect profile, particularly for cytopenic effects, and there are single pills formulated to contain all of the antiretroviral therapy ( Table 152.2 ). Very recently, long-acting ART was approved, involving a monthly injection.
| Single Tablet Regimens | Indication | Components | Side Effects/Contraindications | Mechanism of Action for Class/List of Drugs |
|---|---|---|---|---|
| Biktarvy | 1st line initial therapy for most people (2 NRTIs +INSTI) |
|
Nausea, headache, and diarrhea. Cannot be taken with hepatitis B medication | INSTI: inhibits HIV integrase from integrating viral genome into the DNA of a host cell
NRTI: competitive inhibition of HIV reverse transcriptase by incorporating into viral DNA and inducing chain termination
NNRTI: noncompetitive inhibition of HIV reverse transcriptase via binding of the enzyme’s active site
PI: preventing the cleavage of gag and gag-pol precursors by HIV protease, which is required for the formation of the viral capsid
Booster: Inhibits CYP3A, which is responsible for metabolizing darunavir or elvitegravir thus serving as a pharmacoenhancer
|
| Triumeq |
|
Insomnia, headache, and fatigue. Cannot be used in HLA-B*5701 given hypersensitivity to abacavir | ||
| Dovato |
|
Headache, nausea, diarrhea, fatigue. Reversible increase in serum Cr without affecting kidney function. Cannot be used if VL > 50,000 or HBV co-infection | ||
| Atripla |
|
|
Suicidal ideation and QTc prolongation (efavirenz). Should not be taken with medication treating hepatitis B. Efavirenz should be avoided in pregnancy due to increased risk of neural tube defects | |
| Complera |
|
Insomnia, headache, and depressive disorders | ||
| Delstrigo |
|
Dizziness, nausea, abnormal dreams, and headaches, depression, and suicidal ideation. | ||
| Genvoya |
|
Weight gain, nausea, and vomiting. Cobicistat can cause a reversible increase in serum creatinine without affecting actual kidney function | ||
| Odefsey |
|
|
||
| Stribild |
|
Nausea, diarrhea, abnormal dreams, weight gain, depression, suicidal ideation | ||
| Symfi |
|
Headache, body pain, fever, diarrhea, depression, and rash | ||
| Symtuza |
|
Contraindicated in patients taking steroids, direct oral anticoagulants, and high dose statin. Relative contraindication to co-administration with Colchicine if renal or hepatic impairment present | ||
| Triumeq |
|
Weight gain, suicidal ideation, reversible Cr increase that does not affect kidney function. Cannot be used in HLA-B*5701 given hypersensitivity to abacavir |
Antiretroviral therapy protects against opportunistic infections by restoring the CD4 T cell count and suppressing viral replication. The increase in CD4 T cell counts when starting ART can, in a minority of patients (~20%), sometimes lead to a paradoxical effect known as the immune reconstitution inflammatory syndrome (IRIS), especially if the individual had a low CD4 count or a high viral load. IRIS is a strong immune response characterized by cytokine release leading to an array of clinical syndromes. IRIS can resemble an opportunistic infection when the target is an occult infection that has now become unmasked in the setting of increased immune surveillance, that is, TB leading to constitutional symptoms or untreated hepatitis B leading to liver failure; or an autoimmune phenomenon, where the recovery of the immune system aggravates a pre-existing autoimmune disease, that is, thyroiditis, similar to the use of checkpoint inhibition. Depending on the cause, its management can target treatment of the inciting infection, which sometimes necessitates interrupting ART until treatment is completed if concurrent treatment is not possible. IRIS can resolve on its own if it arose from a previously recently adequately treated opportunistic infection, where the immune system precipitously recognizes remnants of it. To this end, IRIS remains a diagnosis of exclusion, with the main diagnostic clue being its timing.
In assessing a patient, one should consider the following three factors: the absolute T-cell count (CD4 count), followed by whether they take antiretroviral therapy, and whether the peripheral blood viral load (VL) is undetectable. These three factors determine the risk of imminent mortality ( Figs. 152.1 and 152.2 ). The classic analogy is to think of a train traveling on railroad tracks approaching the edge of a cliff. The speed of the train is the viral load, the distance to the cliff is the absolute T-cell count, and the cliff represents AIDS, where an individual is now at risk of an opportunistic infection or dying from complications of HIV.
Currently, in well-managed care settings, the great majority of HIV-infected patients are taking effective antiretroviral therapy that often involves 1 pill a day covering three agents. Testing for resistance is possible if a compliant patient nonetheless develops a high viral load, and the relatively large number of agents usually makes it possible to identify an effective combination. Isolated cytopenias are infrequent and generally seen in settings without available anti-HIV treatment. As will be discussed, thromboembolic events and malignancies persist, albeit at a lower rate.
Cytopenias occur frequently in HIV infection. The effect of HIV on blood counts is pervasive and multifactorial. In the United States when HIV infections became prevalent and recognized (i.e., in the first half of the 1980s), it became clear that leukopenia, especially lymphopenia, anemia, and thrombocytopenia, all occurred but often for different reasons. At one end of the spectrum was uncontrolled HIV infection itself, causing low platelets for multiple reasons. This could be anti-platelet antibodies to amino acids 49 to 66 on glycoprotein IIIa as described in one seminal study by the Karpatkin group at New York University (NYU). Soon thereafter, infection of megakaryocytes by HIV was described as the creation of an inflammatory milieu of the bone marrow, all of which contributed to lower blood counts.
Confirmation of the role of active viral infection in the decrease in the platelet count has come from the use of antiretroviral treatments, which bring up the platelet count when the viral load goes to zero. Early on, there were several examples. First, when the initial randomized azidothymidine (AZT) study was performed, a subsequent letter showed that the patients who entered the study with counts between 100 and 150,000/μL (entry with counts below 100,000/μL was not allowed) showed that platelet counts increased on AZT therapy. This was in marked contrast to the hemoglobin and leukocyte count, both of which decreased in patients on AZT. Another study from Switzerland used a crossover design. Patients were randomized to either receive AZT or placebo, and then after a number of weeks they crossed over to the opposite arm. The results were astonishingly clear that the platelet counts went up dramatically on AZT and did not rise or fall back to baseline on placebo. Finally, Oksenhendler, in a non-randomized study, demonstrated the efficacy of not only AZT but also splenectomy in increasing the count in patients with HIV-related thrombocytopenia. These findings demonstrated that the thrombocytopenia at least resembled immune thrombocytopenia (ITP) in certain ways and furthermore that it was clearly linked to the presence of the virus. The bottom line was that thrombocytopenia was of sufficient importance that Holtzman et al. published a study in 1994 suggesting that at least 40% of patients with HIV would eventually develop thrombocytopenia and also suggested that worse thrombocytopenia was linked to greater mortality. However, this description is reflective of past history and applies only to a situation in which ART is not readily available.
Currently, thrombocytopenia is very infrequent in patients with HIV who are generally well controlled on ART. Furthermore, the viral load suddenly increasing either from resistance or non-compliance is not typically heralded by thrombocytopenia. In high-income countries, thrombocytopenia is generally not an issue in patients on ART, and neither is platelet dysfunction in general. In lower-income countries, which do not have a wide availability of testing or multiple up-to-date treatments of HIV, there is a substantial incidence of thrombocytopenia in these patients as there was initially in the United States. Thus there are still numerous reports from lower-income countries, for example, Ethiopia, in which thrombocytopenia is relatively frequent. Other causes could be co-infections. The best-described co-infection with HIV has been hepatitis C, in which thrombocytopenia, in particular, occurs relative to liver disease and hypersplenism. Liver disease can be a cause of unexplained thrombocytopenia, as seen in a study of non-HIV-infected ITP patients published by Rajan in 2006. Another co-infection that would cause cytopenias is CMV which is certainly not a rare infection in these or any group of hospital-bound patients.
Leukopenia is the least well-defined cytopenia in HIV-infected patients. Some of it is due to lymphopenia as the virus affects patients with infection, as described in the Introduction. Some of it may be related to other medications, inflammation, and/or infections.
Alternatively, anemia seems to be a result of low-grade inflammation in the marrow. It could also be from poor nutrition, but that is not very common and, if present, is typically very mild. Thrombocytopenia, leukopenia, and anemia, frequently present in the past, were often a result of an overt infection that was rampant and resulted in pancytopenia, which could be substantial and was often a sign of impending mortality. However, this fortunately is quite uncommon in the current climate. Similarly, true autoimmune disease is relatively uncommon in the current climate as well. This means that “ITP” and autoimmune hemolytic anemia are no longer commonly found in HIV-infected patients.
Much of anemia, leukopenia, and thrombocytopenia in the past involved not only viral loads but also the toxicity of medications for HIV such as AZT and medications used as prophylaxis or treatment of associated infections such as Bactrim for Pneumocystis pneumonia (PCP) or valganciclovir for cytomegalovirus (CMV). AZT compliance could be measured not so much by identifying a mild degree of anemia but rather by seeing that the mean corpuscular volume (MCV) had substantially increased. However, newer drugs have been designed so that they do not cause cytopenias and therefore these are not much of a problem in the setting of ART. Similarly, the use of Bactrim or trimethoprim sulfa 3 days a week in prophylaxis rarely causes anemia. In the differential diagnosis of anemia, all of the standard causes would need to be considered, for example, GI bleeding, iron deficiency, or B12 deficiency.
Treatments of cytopenias typically depend on the etiology. Management of an underlying disease, including but not limited to HIV itself, is almost always useful. For HIV-related ITP, steroids are generally useful and surprisingly safe if not administered for long periods of time. PCP is certainly a risk during discontinuation and fungal infection, especially candida, is probably the most common if prophylaxis is not given. Two studies from our center demonstrated the utility of a now-infrequently used ITP treatment, IV anti-D (Winrho SD), in the HIV-related ITP. Surprisingly, in this population, a small, randomized crossover study suggested that it was more effective than IVIG. Anemia was not a particular problem in these patients despite the hemolysis from anti-D and the predisposition to anemia. Oksenhendler demonstrated the utility of splenectomy. Other treatments now commonly used for classical ITP, such as rituximab, are either not used (the former) or supported by anecdotal reports (the latter). All of these treatments were developed in the pre-ART era and are now infrequently used.
In summary, in well-treated HIV infection, at the present time, cytopenias are relatively uncommon and have not been a serious problem. In patients that are not well treated either for compliance, resistance, or unavailability of ART medications, cytopenias continue to be seen as they were in the original cohorts in the 1980s before effective, nontoxic, antiretroviral treatments were developed. Again, the better shape a patient is in from the point of view of nutrition, undetectable viral load, inflammation, and not requiring large amounts of concomitant medications, the less likely they are to have cytopenias or other problems.
Lymphomas of all types are increased in people living with HIV. This increased occurrence continues even in well-treated patients with low to undetectable viral cultures although the incidence is lower. The explanation for this is complicated and remains not fully worked out. Theories include: (1) failure of adequate immune surveillance resulting in failure to eradicate early lesions, (2) chronic inflammation characterizing HIV may stimulate lymphocytes such that they may then become malignant, and (3) increased co-infections, especially failure to regulate EBV. These explanations tend to be consistent with finding B cell non-Hodgkin lymphoma but do not explain equally well the increased risk of Hodgkin disease.
The link between immunosuppression and cancers has been well established, whether it is related to inherited immune deficiencies, medications (e.g., steroids or chemotherapy), or uncontrolled HIV with decreased immune surveillance. Polymorphic lymphoproliferative disorders are reported in both HIV-positive individuals and solid organ transplant recipients. This overlap is likely related to the state of immunosuppression. Also, in congenital immunodeficiencies, failure of immunoregulation may likely be abnormal and lead to uncontrolled proliferation including the development of lymphoma. Most lymphomas arise from B-lymphocytes; however, HIV does not directly infect B cells, and various theories have been proposed to explain this phenomenon.
The classic theory is that the state of immunosuppression leads to decreased immune surveillance, along with co-infection with oncogenic viruses: herpesviruses, Epstein-Barr virus (EBV), and Kaposi sarcoma-associated herpesvirus (KSHV), leading to a loss of immunoregulatory mechanisms. HIV infection is also often accompanied by a chronic pro-inflammatory state resulting in cytokine overproduction (i.e., IL-6 and IL-10). In turn, this leads to uncontrolled stimulation of B cells. Dysregulated B-cell stimulation aids in the emergence of monoclonal B cells; these can be seen without indicating the presence of malignancy. Certain genetic alterations may then take place, which leads to the emergence of lymphomas. Furthermore, there are unique characteristics of lymphomas that present within the HIV population.
The introduction of HAART, through the suppression of the HIV viral load and normalizing the CD4 count leading to greater immunocompetence, played a significant role in the reduction of cases of NHL in the HIV population. However, non-Hodgkin lymphoma (NHL) still comprises the great majority of lymphoma cases in HIV-positive individuals. There are non-AIDS defining NHL subtypes, i.e., DLBCL and Burkitt lymphoma, that remain higher in prevalence in the HIV-positive population despite viral control with antiretroviral therapy. Some lymphomas, such as Hodgkin lymphoma, tend to present at later stages with extra nodal disease, are more aggressive, and are more likely to have EBV co-infection when compared to those seen in HIV-negative patients described in greater detail below. Prior to HAART, most patients with HIV succumbed to opportunistic infections or one of the aforementioned AIDS-defining malignancies, including Kaposi sarcoma (see Table 152.2 ). HAART made the prognosis of HIV not dismal and restored the lifespan of patients to more normal. Other malignancies, beyond the AIDS-defining ones, have surfaced and have been likened in prevalence to those who underwent solid organ transplantation and are taking lifelong immunosuppressive therapy, where they were noted to occur at a higher rate when compared to the general population. Of all AIDS-defining cancers, lymphomas make up 50% of cases.
A metanalysis by Grulich et al. looked at the incidence of cancers in HIV/AIDS individuals and compared it to immunosuppressed transplant recipients; seven papers were included for the HIV group and six for the transplant group. The incidence of non-Hodgkin lymphoma was 70-fold more in HIV patients when compared to the general population before the introduction of ART. The metanalysis did not consider the CD4 count of the HIV patients and the degree of the immunosuppression in their findings and did not further stratify the results for HIV versus AIDS. Both groups in the metanalysis had an increased incidence of both Hodgkin lymphoma and non-Hodgkin lymphoma, with a substantially higher meta-analysis standardized incidence ratio (SIR) in the HIV population for both conditions. For Hodgkin lymphoma, the metanalysis SIR for HIV/AIDS was 11.03 (8.43 to 14.4, CI 95%) versus 3.89 (2.42 to 6.26, CI 95%) in transplant recipients. For non-Hodgkin lymphoma in HIV/AIDS, it was 76.67 (39.4 to 149, CI 95%) versus 8.07 (6.40 to 10.2, CI 95%) in transplant recipients. Robbins et al. examined the incidence of cancers in the HIV population over time, obtaining data from US HIV AIDS Cancer Match Study, and included 275,975 HIV-positive patients. They found that the incidence of Hodgkin lymphoma declined by 4%, whereas the incidence of non-Hodgkin lymphoma declined by 15% over the first seven years, followed by a 5.5% decline subsequently. Despite this decline after the introduction of ART, the HIV population still has a higher incidence of both non-Hodgkin lymphoma and Hodgkin lymphoma than the general population (10-fold and 11-fold more respectively).
Non-Hodgkin lymphoma is characterized by multiple subtypes. Many but not all of these are increased in HIV infection, and the subtypes that are increased can be further distinguished between those that are related to the state of the HIV and those that are independent of it .
Diffuse large B-cell lymphoma (DLBCL), an aggressive lymphoma, is the most common type of NHL in the HIV population, constituting 50% of lymphomas in HIV. Unlike Hodgkin lymphoma and Burkitt lymphoma, DLBCL is associated with low CD4 T cell counts. Traditionally, DLBCL had been further sub-classified cytologically into centroblasts (multilobulated vesicular nuclei with numerous peripheral nucleoli), immunoblasts (round vesicular nucleus with one central nucleoli), and anaplastic cells (bizarre pleomorphic nucleus). This classification is no longer used when thinking of DLBCL given the lack of both inter-observer and intra-observer reproducibility, advances in molecular genetics that aid in better prognostication of disease, and heterogeneity of DLBCL per se in the general population. However, this classification is not entirely useless in the HIV population, as cytology can still confer information, and the utility of the new World Health Organization (WHO) 2016 classification of DLCBL for HIV remains unclear. DLBCL with centroblastic features is 70% EBV-negative, whereas 80% to 90% of immunoblastic DLBCL are EBV-positive and express latent membrane protein 1 (LMP-1), a viral oncoprotein resembling CD40 that activates nuclear factor kappa B (NF-κB) leading to unregulated B-cell proliferation.
In patients with HIV, centroblastic DLBCL occurs in individuals with almost normal CD4 counts, whereas immunoblastic DLBCL occurs late in the course of HIV infection and in those who are immunosuppressed, not surprising given the role of EBV primarily in the latter. Diamond et al. examined data from 1988 to 2000 from the San Diego County AIDS and Cancer registries and found that immunoblastic DLBCL decreased from 26% to 7% in the population of patients with AIDS and cancer after the introduction of ART. Despite increases in both overall CD4 T cell count and survival, centroblastic DLBCL increased from 21% to 44% in the same population.
As of 2016, the WHO classifies DLBCL by cell of origin derived from a micro-array gene expression profile (GEP). There are three categories: germinal center B-cell (GC), activated B-cell (ABC), and unclassified (accounting for 20% of DLBCL). In the general population, this classification predicts response to R-CHOP (rituximab-cyclophosphamide-hydroxydaunorubicin-Oncovin-prednisone) therapy, to which GC has a more favorable 5-year survival response when compared to ABC (60% vs. 35%). The majority of GC are centroblastic, whereas the majority of ABC are immunoblastic.
Unfortunately, GEP is expensive and not readily available. So instead, the Hans algorithm using three immunohistochemical markers, CD10, Bcl-6, and MUM1, is used. With this algorithm, DLBCL is classified as GC if CD10 is positive or if MUM-1 is negative with a positive BCL-6. All other DLBCL cases are classified as non-GC.
HIV-negative individuals with GC DLBCL have a better prognosis when compared to non-GC DLBCL, but this does not apply to HIV-positive patients. A study by Chadburn et al. examined tissue from 81 DLBCL patients with AIDS enrolled in AIDS-malignancy consortium (AMC) trials comparing the effects of R-CHOP versus CHOP (AMC010) and concurrent R+EPOCH versus sequential EPOCH followed by Rituximab. Hans algorithm was used to classify the samples into GC and non-GC. No difference in overall survival between the two groups was observed ( P = 0.859). The presence of EBV did not correlate with event-free survival. Also, there was no correlation between CD4 counts and the presence of EBV ( P = 0.47). However, immunohistochemical staining with Ki-67, a marker of cell proliferation, predicted overall survival ( P = 0.05), with a high proliferation index associated with a better clinical outcome, possibly providing a target for chemotherapeutic agents.
Madan et al. examined differences between HIV-positive and HIV-negative individuals by describing 39 de novo DLBCL in 12 AIDS DLBCL and 27 non-AIDS DLBCL Classically, GC is associated with CD10, BCL-6, Cyclin H, and non-GC with MUM1, BCL-2, CD38, PAL1, and CD44. Each of the immunohistochemical stains was designated a number on the basis of the positivity of the samples. Furthermore, the different markers for GC and non-GC were combined to give a score, which was plotted on a two-dimensional contour plot. For the HIV-negative DLBCL samples, the plot had two separate clusters delineating the two subtypes. When the HIV-positive samples were plotted, the two distinct clusters were not appreciated, instead, an intermediate state of both GC and non-GC was appreciated with overlap with both GC and non-GC from the HIV-negative samples. This alludes to a possibly different underlying mechanism for lymphomagenesis, which may imply a need to consider different targets for therapy.
Another adverse prognostic factor termed the “double-expressor” phenotype exists where MYC and BCL-2 are both overexpressed without genetic re-arrangement. This is distinct from “double-hit” or “triple-hit” lymphomas, a term designated for high-grade B-cell lymphoma (HGBL), which morphologically resemble DLBCL. They are considered a separate entity in the 2016 WHO classification, which denotes that re-arrangements in MYC and BCL2 and/or BCL6 are implicated in a worse prognosis.
The “Double-Expresser” phenotype is also associated with poorer outcomes, where the 5-year overall survival is around 40% with R-CHOP therapy. Dodero et al. compared clinical outcomes in double-expresser DLBCL treated with R-CHOP versus dose-adjusted EPOCH-R (etoposide + prednisone + vincristine + cyclophosphamide + doxorubicin + rituximab). One hundred fourteen patients with the double-expresser DLBCL phenotype were identified in three Italian institutions. Fifty-one patients received dose-adjusted EPOCH-R between 2013 and 2017, and 63 patients received R-CHOP between 2009 and 2017. The two primary outcomes were overall survival (OS) and progress-free survival (PFS). In the dose-adjusted EPOCH-R group, dose-escalation to level 3 was possible in 73% of patients under the age of 65 compared to many fewer patients above 65, where the majority received dose-adjusted EPOCH-R at level 1 because of either comorbidities or therapy-associated toxicities. There were no statistical differences in OS and PFS in patients receiving R-CHOP versus dose-adjusted EPOCH-R; however, when adjusting for age, patients below the age of 65 exhibited favorable clinical outcomes in the dose-adjusted EPOCH-R group when compared to patients receiving R-CHOP, where PFS was 82% versus 43% ( P = 0.02) and OS 90% vs. 63% ( P = 0.042). The clinically significant difference after age adjustment likely emerged because younger patients were able to better tolerate higher levels of dose-adjusted EPOCH-R therapy.
Other mutations have been observed in HIV-related DLBCL: over 90% of cases were noted to harbor somatic hypermutations in the immunoglobulin variable genes responsible for extracellular antigen binding. Specifically, there was overexpression of the IGHV4–34 gene in DLBCL, which is normally expressed on the surface of naïve B cells but not expressed by mature activated or memory B cells. This aberrant overexpression in HIV-related DLBCL implies that there may be an antigen-driven process that indirectly stimulates B cells leading to lymphomagenesis. Similar to HIV-negative lymphomas, other mutations in proto-oncogenes PIM-1 and RhoH/TTF (signal transducers) and PAX-5 and c-MYC (transcription factors) have been observed in HIV-related DLBCL; these are typical features of aggressive lymphomas. Furthermore, over 60% of HIV-related DLBCL harbor mutations in BCL-6, a transcription repressor protein-encoding gene required for germinal center formation, where it normally suppresses p53, a tumor suppressor gene that regulates the cell cycle.
Primary CNS lymphoma (PCNSL) is an AIDS-defining malignancy. It involves the brain, leptomeninges, or spinal cord, is considered a subtype of DLBCL in over 90% of cases and is classified under the immunoblastic variant in which almost all cases are EBV driven and express LMP-1. It is associated with CD4 counts below 50 and thus occurs late rather than early in the course of HIV. Post-transplant patients can also present with PCNSL, comprising around 0.9% of diagnosed lymphomas in these patients. Immunocompetent HIV-negative patients can also get PCNSL; however, it is exceedingly rare with an incidence of 7 per million in the United States, presents in much older individuals (median age of 67 vs. 37 in HIV vs. 52 in transplant patients), and is not EBV driven. In the early 1990s, over 60% of PCNSL cases were found in HIV-positive individuals. In 2007–2011, only 12% of PCNSL cases were attributed to HIV, which correlates with better immunocompetence in ART-treated HIV-positive individuals.
Plasmablastic lymphoma (PBL), a subtype of DLBCL comprising 3% of lymphoma cases in HIV, is also considered an AIDS-defining lymphoma. Half of the cases involve the oral cavity and jaw, with the other half mostly involving the GI tract or any other site, i.e., skin, bones, or sinonasal cavity. This lymphoma is morphologically categorized into a monomorphic and a plasmacytic variant. The former resembles DLBCL, where it appears immunoblastic or centroblastic in morphology (described above), and the latter variant resembles plasma cells. More than half of the cases express EBV with LMP-1, and the remainder expresses HHV8 alone, similar to primary effusion lymphoma (described in greater detail below). The immunohistochemical profile stains CD38+, CD138+, IRF4/MUM1, CD79a, BLIMP-1 and is negative for mature B-cell markers such as CD19, CD20, and Pax 5. Some PBLs are weakly positive for CD45 which confers a positive prognostic factor along with EBV positive cases. It carries an extremely poor prognosis with a median survival between 3 and 5 months.
Burkitt lymphoma (BL), also an aggressive NHL, histologically identified by the starry sky appearance is typically seen in children; however, it is the second most common lymphoma subtype in adults with HIV after DLBCL, representing 30% of NHL. BL is classified into three subtypes. Endemic BL typically involves the jaw and is EBV-driven in most cases. Geographically, it is found in tropical Africa and Papua New Guinea, correlating nicely with the incidence of malaria, where the malaria infection is thought to decrease one’s resistance to EBV. Sporadic BL is not confined geographically nor has the predilection for EBV (20% of cases) and usually presents in the ileocecal region in the abdomen in children and adolescents in the United States and Western Europe with a predilection for persons of European descent. The third form is immunodeficiency-associated BL, found mainly in HIV patients typically with CD4 counts above 200. It behaves in many ways like the sporadic type, where they both have similarities in staging and organ involvement. Furthermore, this subtype is also observed to a lesser extent in post-transplant patients and also in rare case reports of congenital immunodeficiencies, that is, Common variable immunodeficiency (CVID), Bloom syndrome, and Wiskott-Aldrich syndrome. All three subtypes have translocations leading to constitutive expression of the MYC proto-oncogene on chromosome 8. Eighty percent of BL have a t(8;14), where chromosome 14 encodes the heavy immunoglobulin chain. The remainder of BL translocations involve the immunoglobulin light chain, where 15% are with the kappa chain t(2;8) and lambda chain t(8;22). Immunohistochemically, BL stains for CD20, CD79a, CD10, and BCL-6 and is negative for BCL-2 and TdT. Interestingly, EBV-associated BL does not express LMP-1; instead, it expresses EBNA1, a viral protein responsible for DNA replication in EBV’s latent phase, which is thought to contribute to the survival of B cells by protecting them from apoptosis induced by the c-myc gene. For unclear reasons, it was observed that BL increased from 4% to 9% after the introduction of ART in the San Diego County AIDS and Cancer registry from 1988 to 2000.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here