Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Rhesus D (RhD) alloimmunization is the major cause of haemolytic disease of the fetus and newborn (HDFN). Over the past 40 years, the incidence of HDFN has fallen dramatically due to greater understanding of the pathophysiology of the disease and advances in management including the widespread use of prophylactic anti-D immunoglobulin. The reduction in stillbirths and neonatal deaths, although primarily attributable to the use of anti-D immunoglobulin, is also a result of significant advances in the management of affected fetuses and neonates. The use of high-resolution real-time ultrasound to screen for fetal anaemia to allow timely fetal blood transfusion means that pregnancies complicated by alloimmunization now have a favourable outcome.
The red cell membrane contains numerous surface antigens, including the RhD antigen ( Table 19-1 ). The RhD antigen is encoded by the RhD gene located on chromosome 1. The RhD-negative phenotype is due to the absence of the RhD antigen on the red cell membrane as a result of homozygous absence or partial deletion of the RhD gene. A small minority of individuals, known as RhD u individuals, are missing part of the RhD antigen and are therefore at risk of producing anti-D if exposed to RhD antigen. Growing evidence suggests that these women should receive anti-D immunoglobulin although current guidelines do not advocate this.
| No. | System Name | System Symbol | Gene Name(s) a | Chromosomal Location |
|---|---|---|---|---|
| 1 | ABO | ABO | ABO | 9q34.2 |
| 2 | MNS | MNS | GYPA, GYPB, GYPE | 4q31.21 |
| 3 | P | P1 | 22q11.2-qter | |
| 4 | Rh | RH | RHD, RHCE | 1p36.11 |
| 5 | Lutheran | LU | LU | 19q13.32 |
| 6 | Kell | KEL | KEL | 7q34 |
| 7 | Lewis | LE | FUT3 | 19p13.3 |
| 8 | Duffy | FY | FY | 1q23.2 |
| 9 | Kidd | JK | SLC14A1 | 18q12.3 |
| 10 | Diego | DI | SLC4A1 | 17q21.31 |
| 11 | Yt | YT | ACHE | 7q22.1 |
| 12 | Xg | XG | XG, MIC2 | Xp22.33, Yp11.3 |
| 13 | Scianna | SC | ERMAP | 1p34.2 |
| 14 | Dombrock | DO | DO | 12p12.3 |
| 15 | Colton | CO | AQP1 | 7p14.3 |
| 16 | Landsteiner–Wiener | LW | ICAM4 | 19p13.2 |
| 17 | Chido/Rodgers | CH/RG | C4A, C4B | 6p21.3 |
| 18 | H | H | FUT1 | 19q13.33 |
| 19 | Kx | XK | XK | Xp21.1 |
| 20 | Gerbich | GE | GYPC | 2q14.3 |
| 21 | Cromer | CROM | DAF | 1q32.2 |
| 22 | Knops | KN | CR1 | 1q32.2 |
| 23 | Indian | IN | CD44 | 11p13 |
| 24 | Ok | OK | BSG | 19p13.3 |
| 25 | Raph | RAPH | CD151 | 11p15.5 |
| 26 | John Milton Hagen | JMH | SEMA7A | 11p15.5 |
| 27 | I | I | GCNT2 | 6p24.2 |
| 28 | Globoside | GLOB | B3GALT3 | 3q26.1 |
| 29 | Gill | GIL | AQP3 | 9p13.3 |
a As recognized by the HUGO Gene Nomenclature Committee ( www.genenames.org/ ). From Eyre et al., 2006.
The pathophysiology of rhesus alloimmunization is well understood and involves the production of maternal IgG antibodies against the RhD antigen in a mother who is RhD-negative, as a result of exposure to foreign RhD-positive red cells. The exposure usually occurs due to transplacental haemorrhage from a rhesus D-positive fetus to the mother, but can occur after heterologous blood transfusion. Transplacental haemorrhage from the fetus to the mother occurs during sensitizing events, which include miscarriage, termination of pregnancy, invasive prenatal testing and childbirth. The dose of anti-D administered after a sensitizing event varies according to gestation and amount of feto-maternal haemorrhage (FMH), as assessed by Kleihauer testing. In general 250 IU is given before 12 weeks for cases of therapeutic termination and between 12 and 20 weeks after a sensitizing event. After 20 weeks a dose of 500 IU is sufficient for a 4 mL FMH. More anti-D may be required depending on the size of the FMH. Anti-D should be given within 72 hours of a sensitizing event. During the initial exposure to foreign fetal RhD antigen, the RhD-negative mother produces a primary immune response which involves the production of IgM antibodies against RhD antigen. These antibodies are unable to cross the placenta and have no direct effect on the fetus. However, on subsequent exposure to rhesus D antigens, often in the next pregnancy, the mother produces a more rapid immune response involving the production of IgG antibodies which are actively transported across the placenta. IgG antibodies bind to fetal red cells expressing the RhD antigen and cause red cell destruction by phagocytosis and cytolysis ( Figure 19-1 ). This produces fetal anaemia which may lead to hydrops fetalis and death. Fetal death or the development of hydrops does not occur before 16 weeks' gestation, presumably because the fetal reticuloendothelial system is too immature to destroy antibody-coated erythrocytes.
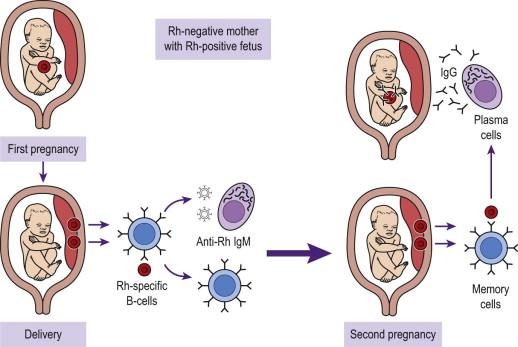
Around 17% of RhD-negative women are immunized by a single RhD-positive pregnancy, if anti-D immunoglobulin is not administered.
50% of sensitized women will have affected neonates with mild or no anaemia;
25–30% will have moderate anaemia requiring neonatal treatment;
20–25% will develop hydrops fetalis due to severe anaemia, and heart failure, leading to intrauterine or neonatal death unless fetal treatment is given ( Figures 19-2 and Figure 19-3 A and B ).
Routine first-trimester antenatal care involves testing for ABO blood group and Rh status, as well as an antibody screen. A further antibody test is offered at 28 weeks' gestation to detect any red cell antibodies that may have developed in the pregnancy.
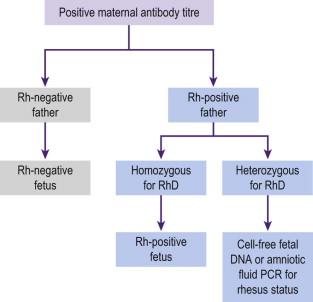
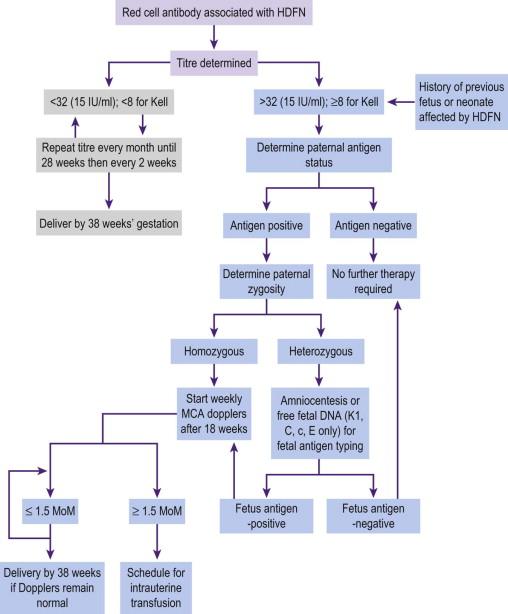
If maternal serum red cell antibodies are detected, the partner's blood is analyzed for blood group (genotyping may be required) ( Figure 19-4 ). If the partner is homozygous for D antigen the fetus will be D antigen-positive and at risk of developing fetal anaemia. If the partner is heterozygous there is a 50% chance that the fetus will be RhD-negative and unaffected by the antibodies.
Prenatal testing of the fetal genotype was initially done using polymerase chain reaction method on fetal DNA from amniotic fluid or chorionic villus cells. This method is invasive, carries a risk of miscarriage and can theoretically worsen fetal anaemia by increasing maternal antibody levels (particularly following chorionic villus sampling). A major advance in the management of rhesus alloimmunization was the identification of free fetal DNA (found in maternal blood from as early as 7 weeks' gestation) to detect the presence of the RhD gene by real-time PCR techniques; this has been available in the UK since 2001. This approach has the advantage of being non-invasive and in addition to RhD this technology can determine a growing number of fetal blood group antigens including Kell, c and E.
In a pregnancy at risk of alloimmunization, maternal antibody levels are measured serially as the outcome of pregnancy is related to their concentration. Antibody titres are measured using the indirect Coombs' test and are expressed as the reciprocal of the highest dilution of serum that causes agglutination. Most laboratories use a critical titre, often 1 : 32, above which the risk of fetal anaemia is considered high. However, levels do not always correlate with severity of fetal disease, reflecting the importance of antibody quality, including IgG subclass. In Europe the levels of the antibodies are often expressed in international units/mL. Follow-up of the fetus is recommended when the antibody level is more than 15 IU/mL, (or the titre is 1 in 128). Often the trend in antibody level/titres is more significant than the absolute value.
In pregnancies at risk, ultrasound assessment is necessary to detect early signs of fetal anaemia before hydrops develops. Doppler ultrasound evaluation of the middle cerebral artery is the most widely used tool for assessment of fetal anaemia. Fetal anaemia is treated with intrauterine transfusion (IUT), performed using ultrasound-guided cordocentesis. Survival rates of more than 90% are now common, and the vast majority of survivors appear to have a normal long-term development.
Although the RhD antigen is traditionally the most common cause of red cell alloimmunization and HDFN, over 50 other red cell antibodies have been associated with HDFN. Table 19-2 shows the incidence of haemolytic disease of the newborn (HDN) with atypical maternal red blood cell antibodies. The most significant non-RhD antibodies include K (Kell system) and c of the rhesus system. Although these antibodies are less common, they remain a cause of fetal disease as no prophylactic immunoglobulin is available to prevent antibody formation. These non-RhD antibodies develop in 1–2% of people after blood transfusion and account for 2% of cases of HDFN. The pathophysiology is the same as for RhD isoimmunization. Anti-Kell alloantibodies not only cause fetal red cell destruction but because erythroid precursor cells express the Kell antigen, also reduce red cell production. This may explain the more rapid and earlier development of fetal anaemia in Kell alloimmunization. Anti-Kell antibody levels do not correlate with disease severity and management of affected pregnancies therefore requires serial ultrasound and Doppler assessment to screen for anaemia.
| Frequency of HDN | Antibody |
|---|---|
| Common | c |
| Kell | |
| E | |
| Uncommon | e |
| Cw | |
| C | |
| Ce | |
| Kp a , Kp b | |
| cE | |
| K | |
| S | |
| Wr a | |
| Fy a | |
| Very rare | S |
| U | |
| M | |
| Fy b | |
| N | |
| Do a | |
| Co a | |
| Di a , Di b | |
| Lu a | |
| Yt a | |
| Jk a , Jk b | |
| No occurrence | Le a , Le b |
| p |
Maternal screening for these antibodies is now routine in the UK with an algorithm for the clinical management of the non-RhD alloimmunized pregnancy ( Figure 19-5 ).
Parvovirus B19 is a single-stranded DNA, non-enveloped virus. Known as ‘fifth disease’ or ‘slapped cheek syndrome’, it is such a common childhood viral infection that approximately 65% of pregnant women are immune. It is spread by respiratory droplets, by blood or blood product transfusion or by transplacental passage. The incidence of acute parvovirus B19 infection in pregnancy is 1–2%. Cases are often asymptomatic although prodromal symptoms may be present after an incubation period of 4–14 days. In some cases, the more definable symptoms of rash (erythema infectiosum) and arthralgia are present 7 days after the prodromal illness.
Transplacental transmission of the virus occurs in 33% of pregnant women following infection. Parvovirus B19 causes fetal anaemia by inhibiting erythropoiesis leading to an aplastic crisis. In healthy adults this crisis is well-tolerated with minimal anaemia. However, the fetal demand for red blood cells is greater with a larger red cell mass and rapid cell turnover. This renders the fetus particularly vulnerable to any insult to erythropoiesis and a profound anaemia may result from parvovirus B19 infection. In the fetus the virus mainly affects the bone marrow but can also affect extra-medullary haematopoiesis in the liver or spleen. The fetal anaemia, as well as associated hepatitis, hypoalbuminaemia and myocarditis, can lead to cardiac failure and subsequent hydrops fetalis.
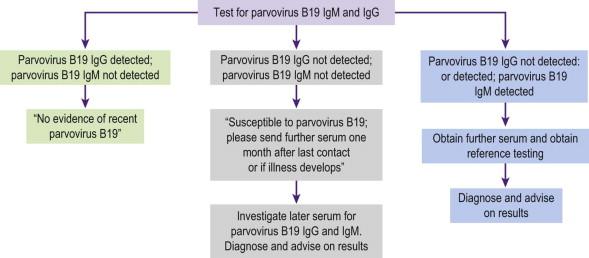
Pregnant women presenting with a rash suggestive of parvovirus B19 or following contact with an infected individual can be tested for specific IgM and IgG ( Figure 19-6 ). Some fetuses that develop hydrops may have false-negative maternal serum IgM titres, as a result of which IUT may be delayed or even withheld. In cases of established hydrops thought to be secondary to fetal anaemia, testing the amniotic fluid for parvovirus B19 may be beneficial, even if the serology is negative.
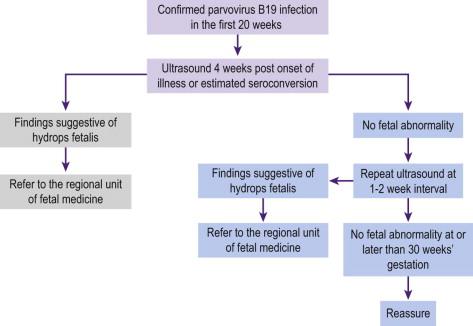
Once serological diagnosis of acute maternal infection is made, fetal ultrasonography is performed to exclude fetal anaemia and hydrops fetalis. Fetal anaemia generally develops 6 weeks (range 3–12) after maternal infection and is most commonly diagnosed at a gestation of 22 weeks. It is very unusual to see severe fetal anaemia secondary to maternal seroconversion after 21 weeks. The management of fetal anaemia secondary to a primary parvovirus B19 infection requires serial scanning for anaemia every 1–2 weeks starting from 4 weeks up until 12 weeks after maternal infection. In some cases the anaemia resolves spontaneously (resolving fetal infection) and in others IUT is required ( Figure 19-7 ). In most cases, one transfusion is sufficient to treat the anaemia and reverse the hydrops. Ascites may persist for several weeks and should not be regarded as treatment failure. Other congenital infections that can cause fetal anaemia include cytomegalovirus, syphilis and toxoplasmosis, however the anaemia is generally not severe enough to cause fetal hydrops.
Other causes of anaemia feto-maternal haemorrhage can occur spontaneously or following maternal abdominal trauma. This can be assessed and quantified by maternal Kleihauer-Betke test.
Rare causes of fetal anaemia include Blackfan-Diamond anaemia, a disorder of ribsosome function caused by mutations affecting genes encoding ribosomal proteins. DBA is characterised by physical abnormalities, cranio-facial and thumb abnormalities, short stature, red cell aplasia and an increased risk of malignancy. There is a wide range of severity and different patterns of inheritance have been reported, most commonly autosomal dominant in familial DBA.
Congenital dyserythropoietic anaemias are a heterogeneous group of rare inherited anaemias characterised by ineffective erythropoiesis, morphological abnormalities of mature red blood cells and morphological and functional abnormalities of erythroblasts. The anaemia is usually mild-moderate, severe transfusion-dependent cases are uncommon. They have been classified into three types (CDA I, II and III) in relation to their pattern of inheritance and bone marrow morphology. CDA II is the most common type and has an autosomal recessive mode of inheritance.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here