Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Growth during fetal life is characterized by an early period of cell division and organogenesis, followed by a more prolonged period of growth and refinement of organ development. Growth factors are peptides or proteins that serve as key regulators of cell proliferation and differentiation that exert their effects by binding to specific receptors, which mediate signal transduction across cell membranes. This binding triggers a cascade of responses, including those involving second messengers, which ultimately result in cell mitogenesis or differentiation. Various growth factors are expressed in diverse tissues during embryonic life, which may have endocrine as well as autocrine-paracrine effects. Our understanding of growth factor regulation of fetal growth has developed through in vitro, animal, and clinical studies, which will be reviewed in this chapter.
The primary regulator of postnatal somatic growth is growth hormone (GH). In the liver and other target cells, GH induces the production of insulin-like growth factors (IGFs), IGF-1 and IGF-2. The IGFs, particularly IGF-1, have direct endocrine effects on somatic growth and on the proliferation of many tissues and cell types. Both IGF-1 and IGF-2 are expressed during fetal development in a GH-independent fashion. In addition to their anabolic effects, IGFs also are thought to be significant autocrine-paracrine factors involved in cellular and tissue function. , Locally produced IGFs have been demonstrated in nearly all tissue types, where they are considered to be responsible for cell growth and differentiation. GH is thought to play only a minor role in fetal growth, limited to the third trimester, and infants born with congenital GH deficiency are normally grown at birth. This chapter presents evidence that the IGFs and their regulatory proteins play a central role in the regulation of fetal growth.
IGF-1 and IGF-2 are two closely related peptide hormones approximately 7 kDa in size that share a high degree of structural similarity with proinsulin. Like proinsulin, they are composed of A, B, and C domains; however, they also include a D domain. Together, these components form the mature IGF peptide. The IGFs are synthesized with an additional extension peptide known as the E peptide, which is removed during post-translational processing. Both IGF-1 and IGF-2 are encoded by a single gene each, which by alternative splicing results in several messenger RNA (mRNA) species. The IGF1 gene spans 95 kilobases (kb) on the long arm of chromosome 12 and contains 6 exons; the IGF2 gene has a total genomic size of 35 kb and is made up of 9 exons. IGF2 is located on the short arm of chromosome 11 (near the insulin gene), in an area that is paternally imprinted in all mammalian species. This paternal imprinting phenomenon is related to the fact that IGF-2 is a major fetal growth factor.
The IGFs interact with specific receptors, designated type 1 and type 2 IGF receptors, as well as with the insulin receptor, which can bind IGF-1 and IGF-2, but with much lower affinity than for insulin. The type 1 IGF receptor binds IGF-1 with high affinity, IGF-2 with slightly lower affinity, and insulin with low affinity. This receptor mediates the known mitogenic effects of the IGFs. The type 1 IGF receptor and the closely related insulin receptor are heterotetramers composed of two pairs of α and β subunits, the result of post-translational processing of a single gene product of a single gene that encodes for the entire receptor. The two α subunits, which are involved in ligand binding, are linked by disulfide bonds and are primarily extracellular ( Fig. 140.1 ). The β subunits are connected to the α subunits by disulfide bonds and function as intracellular tyrosine kinases. On ligand binding, the subunits undergo a conformational change that enables them to bind ATP and become autophosphorylated. Subsequently, these kinases phosphorylate cytoplasmic molecules known as the insulin receptor substrates (IR , S-1 and -2), which are involved in mediating many of the effects of insulin and IGFs. IRS proteins couple insulin and IGF receptors to the phosphoinositide 3-kinase (PI3-kinase) and extracellular signal-regulated kinase (ERK) cascades. Products of PI3-kinase, including phosphatidylinositol-3,4-biphosphate and phosphatidylinositol 3,4,5-triphosphate, attract serine kinases to the plasma membrane, including the phosphoinositide-dependent kinase (PDK1 and PDK2) and at least three protein kinases B (PKB)/AKT isoforms. During co-localization at the plasma membrane, PDK1 or PDK2 phosphorylates and activates PKB-1, -2, or -3. The activated PKB/AKT phosphorylates many substrates to control various biologic signaling cascades, including glucose transport, protein synthesis, glycogen synthesis, and cellular proliferation and survival in various cells and tissues.
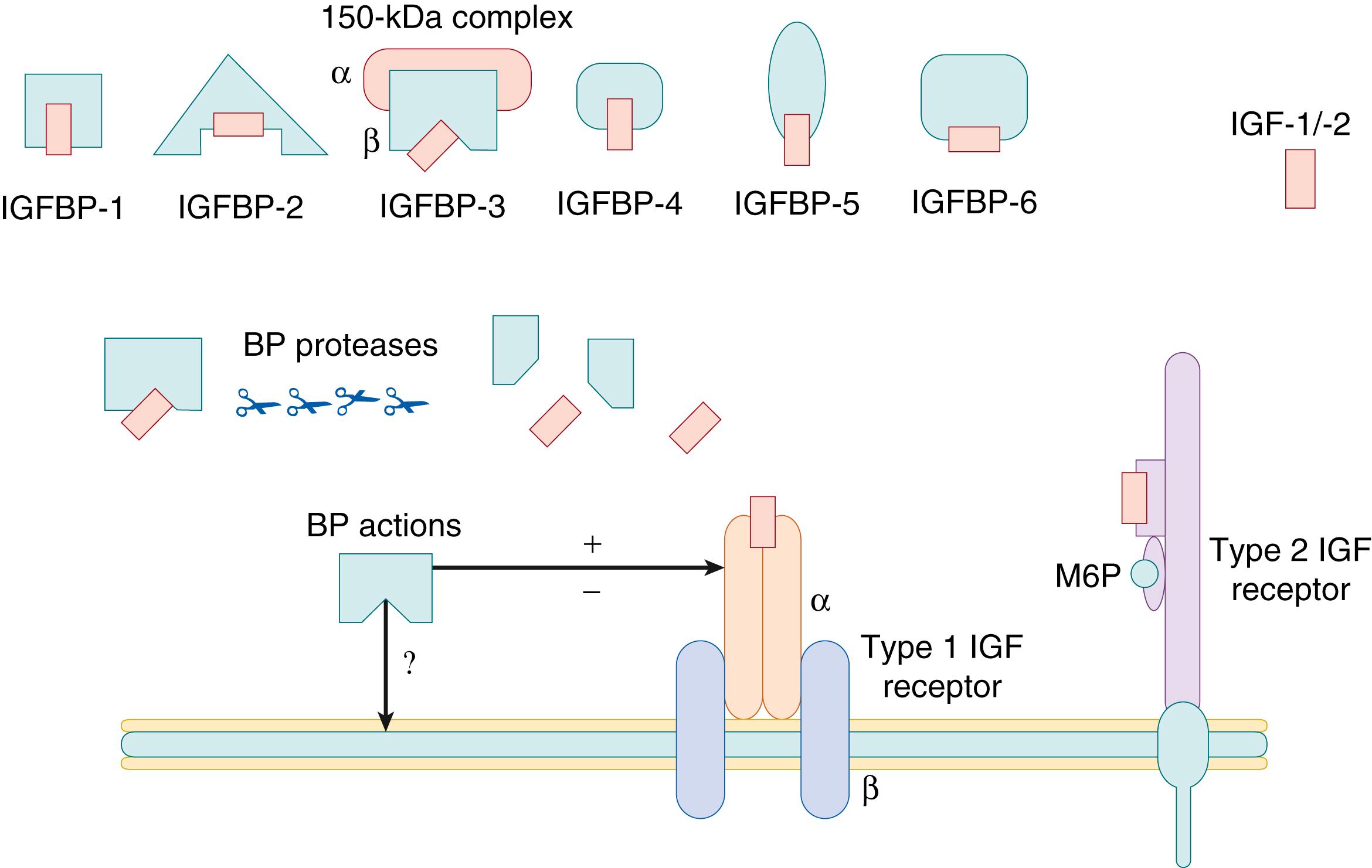
The insulin receptor itself exists in two isoforms in humans, produced by tissue-specific alternative splicing of exon 11 of the insulin receptor gene, IR. Relative expression of the different insulin receptor isoforms, the type A insulin receptor (IR-A) and type B insulin receptor (IR-B), is not only tissue-specific but also developmentally regulated, with IR-A preferentially expressed in fetal tissues. In addition, IR-A binds IGF-2 with an affinity equal to that for insulin, whereas IR-B has less affinity for IGF-2. Insulin binding to IR-A stimulates the metabolic effects of insulin receptor activation, while IGF-2 binding to IR-A more effectively stimulates mitogenic processes. This differential effect may result from less insulin receptor endocytosis following IGF-2 binding relative to insulin binding. ,
The type 2 IGF receptor is structurally distinct from the type 1 receptor and the insulin receptor and primarily binds IGF-2. Unlike the insulin receptor and the type 1 IGF receptor, the type 2 IGF receptor is not a tyrosine kinase. Instead, it is a monomeric receptor with a large extracellular domain made up of 15 repeat sequences and a small region homologous to the collagen-binding domain of fibronectin. The type 2 IGF receptor is 270 kDa in size and functions also as the cation-independent mannose-6-phosphate receptor. The receptor does not have a signaling domain and is thought to be recycled between the plasma membrane and intracellular compartments. It also has been suggested that the type 2 IGF receptor targets excess IGF-2 for lysosomal destruction during fetal life. The type 2 receptor is maternally imprinted in mice but not in humans.
Additional members of the class of receptors for the IGF-insulin family are less well characterized. One such receptor is composed of an insulin receptor α-β heterodimer binding to a type 1 IGF receptor α-β heterodimer and thus has been labeled the hybrid receptor. Analysis of tissues in which this receptor appears to be common (such as placenta) and transfection experiments in cell lines have demonstrated two isoforms of this hybrid receptor—a hybrid receptor containing insulin receptor isoform A (Hybrid-Rs A ) and one containing insulin receptor isoform B (Hybrid-Rs B ). Hybrid-Rs A has biologic properties intermediate between those of the insulin and the type 1 IGF receptor: It binds both insulin and IGFs with high affinity and stimulates cell proliferation and migration. (By contrast, Hybrid-Rs B binds IGF-1 with high affinity but IGF-2 with only low affinity, whereas insulin has little to no affinity at all.) The complementary DNA (cDNA) for an additional receptor has been cloned, and because of its high homology with the insulin receptor, it has been designated the insulin receptor–related receptor (IRR). IRR has been regarded as an orphan receptor of the insulin receptor family that does not bind insulin or insulin-like peptide and can be expressed as variably spliced isoforms. IRR is expressed specifically in developing renal and neural tissues. IRR also is expressed in pancreatic islets, but the lack of this receptor in mice does not affect β cell development and function. It is activated in mildly alkaline media and may play a role in acid-base balance. An additional atypical IGF receptor with altered ligand binding also has been described. This may represent a post-translationally modified type 1 IGF receptor of unclear function.
The IGF-binding proteins (IGFBPs), a family of proteins with high affinity for the IGFs, have been shown to be involved in the modulation of the mitogenic effects of IGFs on cells. In vivo, the IGFs are bound to this family of six structurally and evolutionarily related IGFBPs (IGFBP-1 to IGFBP-6). In serum, a majority (70% to 80%) of IGFs exist in a 150-kDa complex composed of one IGF molecule, IGFBP-3, and the acid-labile subunit (ALS). A smaller proportion (approximately 20%) of the IGFs are associated with other serum IGFBPs within a 50-kDa complex, and less than 5% of the IGFs are found in the free form of 7.5 kDa. ALS is a protein that binds to the IGF/IGFBP-3 binary complex, primarily in serum, prolonging the t 1/2 of serum IGFs and facilitating their endocrine actions. These IGFBPs appear to regulate the availability of free IGFs for interaction with the IGF receptors, as well as to interact directly with cell function. They are part of a superfamily that comprises six high-affinity IGFBPs and at least four other low-affinity IGF binders, termed insulin-like growth factor–binding protein-related proteins (IGFBP-rPs) . In addition, the cell surface proteoglycan glypican-3 (GPC3) binds both IGF-2 and the type 1 IGF receptor, facilitating IGF-2 mediated signaling. Mutations of this causes the overgrowth syndrome known as Simpson-Golabi-Behmel syndrome type 1 and GPC3 may also have a role in oncogenesis.
IGFBP-1 is a 25-kDa protein that is found in high concentrations in amniotic fluid; it also is secreted by hepatocytes under negative regulation by insulin. IGFBP-2 is a 31-kDa protein found in serum, cerebrospinal fluid, and seminal plasma. It is expressed in many fetal and adult tissues. IGFBP-3 is the major binding protein in postnatal serum and is synthesized by hepatocytes and other cells. In plasma, IGFBP-3 is found as part of a 150-kDa complex, which also includes an ALS and an IGF molecule, all of which are GH-dependent. IGFBP-4 is a 24-kDa protein that has been identified in serum, seminal plasma, and numerous cell types. IGFBP-5 appears to be an IGF-enhancing protein that is found in cerebrospinal fluid and serum. It also is observed in rapidly growing fetal tissues. IGFBP-6 has relative specificity for IGF-2 over IGF-1. It is found in cerebrospinal fluid and serum and is produced by many cells. The six cloned cDNAs for the IGFBPs show a high degree of structural similarity and sequence conservation across species. IGFBPs are tightly regulated by various endocrine factors and are uniquely expressed during ontogeny. Three possible models for the interaction of the IGFBPs with the IGFs and their receptors have been proposed. The first is that these molecules limit the availability of free IGFs for interaction with the IGF receptors. For example, the addition of IGFBPs to many cell cultures in vitro results in the inhibition of IGF actions within these experimental systems. However, in other systems, IGFBPs have been demonstrated to enhance IGF action under circumstances that may involve cellular processing of the IGFBPs. Finally, as has been shown in several in vitro systems, IGFBPs may inhibit cell growth in an IGF-independent fashion, acting through their own receptors. ,
IGFBP-3 is a mediator of apoptosis under the control of a number of cell cycle regulators. In studies using specific antisense oligonucleotides and neutralizing anti-IGFBP-3 antibodies to block IGFBP-3 expression and action, IGFBP-3 was identified as the mediator of apoptosis induced by retinoic acid and transforming growth factor-β (TGF-β).
A group of enzymes that are capable of cleaving IGFBPs have been recognized as potential modulators of IGF action. First identified in pregnancy serum, IGFBP-3 proteolytic activity is responsible for the disappearance of intact IGFBP-3 from the serum of pregnant women, with no change in IGFBP-3 immunoreactivity and an increase in lower-molecular-weight fragments. , IGFBP proteases also have been reported in the serum of severely ill patients in a state of cachexia, in critically ill neonates, in patients with GH receptor deficiency, and in patients with in prostate cancer and other malignancies. Prostate-specific antigen was the first kallikrein protein demonstrated to be an IGFBP protease. Subsequently, it has been determined that other members of the kallikrein family, such as nerve growth factor (NGF), also serve as specific IGFBP proteases. Cathepsins are another class of IGFBP proteases that have been shown to be active in acid pH. Finally, matrix metalloproteinases, which are known to cleave collagen and other extracellular matrix components, are a group of IGFBP proteases. It has been speculated that the IGFBP proteases are important modulators of IGF bioavailability and bioactivity through their modification of the IGF carrier proteins. The proteolytic activity may play a role in regulating IGF availability at the tissue level by decreasing the affinity of the binding proteins for the growth factors, thereby releasing free IGFs and allowing increased receptor binding. , , Mutations in pregnancy-associated plasma protein A2 (PAPP-A2), a protease that cleaves IGFBP-3 and -5, have recently been shown to cause short stature with elevated circulating IGF-1 concentrations and reduced free IGF-1 levels. , Although this confirms the role of proteases in modulating height in childhood and circulating bioavailable IGF-1, the role of these proteases in fetal growth is not known. Birth length was variable in the five reported children who had pathogenic PAPPA-A2 mutations, with Z-scores ranging from −0.07 to −2.75. Low concentrations of IGFBP proteases have been described during the first trimester in pregnancies at higher risk of subsequent preeclampsia and growth restriction, suggesting that reduction in proteases in pregnancies may reduce IGF bioavailability for placental development. ,
Fibroblast growth factors (FGFs) are a family of peptide cytokines that are important in the regulation of many tissues. To date, 23 different FGFs have been identified, including the best-characterized acidic FGF (aFGF), or FGF1; basic FGF (bFGF), or FGF2; and keratinocyte growth factor (KGF), or FGF7. The FGFs exert their actions by binding to one of a family of four membrane-bound FGF receptors, FGFR1 to FGFR4, and a heparin sulfate proteoglycan co-receptor to form a three-part signaling complex. FGFs appear to have primarily autocrine-paracrine functions in organ growth and differentiation, bone formation, and cell differentiation and migration, as well as in carcinogenesis. The role of FGF in osteogenesis and growth initially was demonstrated by the discovery of a gain-of-function mutation in FGFR3 as the cause of achondroplasia in humans. , Other forms of chondrodysplasia, as well as other skeletal dysplasias, were subsequently found to be caused by mutations in the FGFRs, further emphasizing the importance of FGF signaling in bone formation.
The complex array of cells that make up the nervous system is under the regulatory influence of specific growth factors, such as NGF, neurotensin, and the neural-derived growth factors (NDGFs). NGF is synthesized in peripheral cells and transported in retrograde fashion up the neuronal axis to the cell body—a process that suggests that NGF is a neurotropic agent directing neurons to sites of innervation. NGF exerts its effects on cells derived from the neural crest, including central nervous system and peripheral nervous system neurons, and some non-neurons, such as immune cells and endocrine cells. NGF stimulates mitosis in some cell types and is capable of inducing differentiation events in neural tissue. NGF synthesis is controlled in part by hormones, such as testosterone and thyroxine. It also has been demonstrated that the γ subunit of NGF serves as an IGFBP protease.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here