Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Over half of all critically ill patients develop some degree of acute kidney injury (AKI), and nearly 5% require renal replacement therapy (RRT). For those patients with severe AKI requiring RRT, mortality can be as high as 70%, and up to 30% of surviving patients remain dialysis dependent. AKI may be a consequence of prerenal causes resulting in hypoperfusion of the kidneys, intrinsic kidney disease, and postrenal or obstructive causes. In critically ill patients, most cases of AKI are related to ischemic or toxic acute tubular injury, which are treated with supportive measures and are often reversible. Acute glomerulonephritis (GN) accounts for about 4% of all cases of AKI. In addition to supportive care, initiation of correct treatment is paramount for patient and renal survival. The focus in this chapter is on GN, a cause of AKI for which there is often disease-specific therapy.
In GN, patients present with nephritic syndrome characterized by hematuria, proteinuria, AKI, edema, and hypertension. Hematuria may be microscopic or macroscopic. The urine sediment demonstrates dysmorphic red blood cells (RBCs) and RBC casts. Urinary protein excretion typically exceeds 1 gram per day, and the degree of proteinuria can be rapidly assessed using simultaneous measurement of the urine protein-to-creatinine ratio (urine PCR) and the urine albumin-to-creatinine ratio (urine ACR). In general, the urine albumin-to-protein ratio (urine ACR/urine PCR) exceeds 0.5 in patients with glomerular disease. In contrast, an albumin-to-protein ratio of <0.4 is most consistent with tubular proteinuria when the urine PCR is <3. In some instances, patients may have nephrotic-range proteinuria (>3 g/d) with associated clinical manifestations, including edema, hypoalbuminemia, and hyperlipidemia. Leukocyturia with or without white blood cell casts may be observed with GN of inflammatory origin.
In renal biopsy series of patients with unexplained AKI, the most common diagnoses included various forms of GN (pauci-immune GN, immunoglobulin [Ig]A nephropathy, postinfectious GN, lupus nephritis, anti–glomerular basement membrane [anti-GBM] disease) and acute interstitial nephritis (AIN). , Indeed, the third most common cause of end-stage renal disease (ESRD) in the United States and Europe is GN. Distinguishing the type of GN with kidney biopsy is critical for diagnosis and for assessing the degree of acute versus chronic disease, which helps guide treatment and informs prognosis.
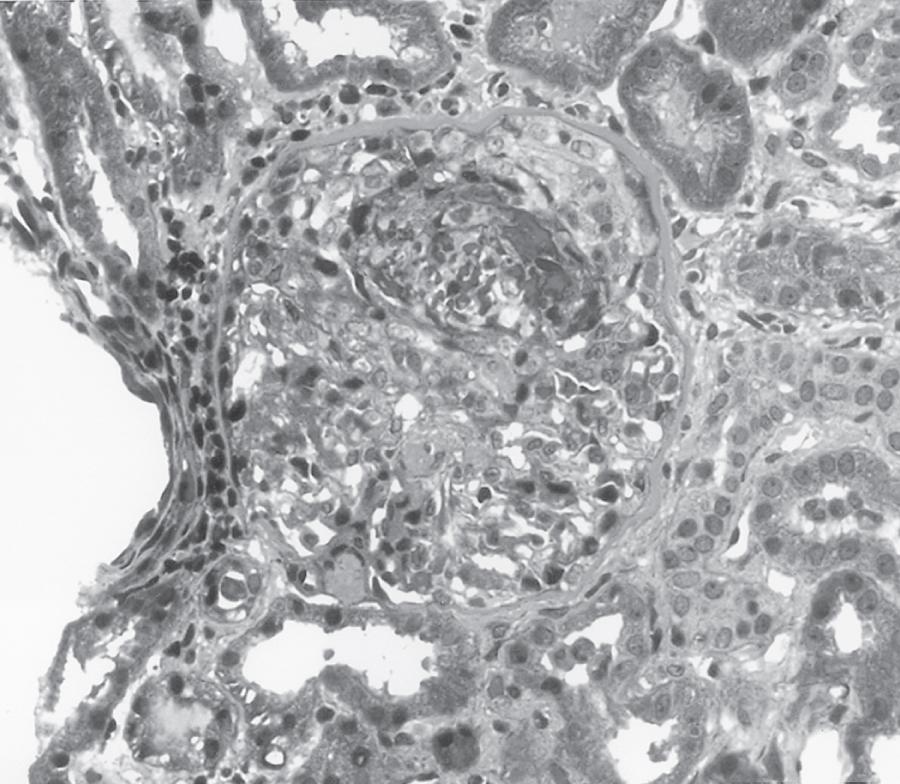
The most aggressive form of GN is described clinically as rapidly progressive glomerulonephritis (RPGN). Rather than a single disease entity, RPGN is the most severe clinical presentation of many glomerular diseases that are divided into renal-limited etiologies and systemic diseases that involve the kidneys ( Table 103.1 ). RPGN is defined as rapidly declining renal function, progressive oliguria, hematuria, proteinuria, and hypertension. Although many critically ill patients may have hematuria associated with infection or trauma, hematuria and AKI should always prompt consideration of acute GN. Kidney ultrasound characteristically shows normal to slightly enlarged kidneys, although reduced kidney size may be present when the diagnosis is delayed. Kidney biopsy reveals a high degree of glomerular injury with extensive crescent formation ( Fig. 103.1 ). Importantly, the transition from acute cellular crescentic disease to chronic, irreversible injury may occur rapidly over days. The rapid progression of such cases constitutes a need for prompt diagnosis with early intervention and therapy to interrupt a natural progression to chronic kidney disease. In adults, the most common cause of RPGN is pauci-immune GN associated with antineutrophil cytoplasmic antibodies (ANCAs), followed by immune-complex diseases such as lupus nephritis or mixed cryoglobulinemia. Anti-GBM disease (formerly called Goodpasture disease ) is less common. , Immunofluorescence microscopy shows pauci-immune staining in ANCA-associated GN; linear IgG staining of the GBM in anti-GBM disease; and immune complex deposition in lupus nephritis, IgA nephropathy, and infection-related GN.
| Renal Limited | |
| IgA nephropathy | |
| Infection-related glomerulonephritis | Low complement, streptococcal serologies, bacterial cultures |
| ANCA-associated glomerulonephritis (pauci-immune glomerulonephritis) | ANCA titers |
| Anti-GBM disease (Goodpasture syndrome) | Anti-GBM antibodies |
| Systemic Disorders | |
| Lupus nephritis | Low complement, ANA, dsDNA antibodies |
| ANCA-associated small vessel vasculitis | ANCA titers |
| Anti-GBM disease | Anti-GBM antibodies |
| Henoch-Schönlein purpura | None |
| Cryoglobulinemic vasculitis | Low complement, cryoglobulins, hepatitis C serologies, positive rheumatoid factor |
Pulmonary-renal syndrome, characterized by RPGN and diffuse alveolar hemorrhage (DAH), is a medical emergency requiring early aggressive treatment. Untreated, it is associated with high mortality and rapid progression to ESRD. Admission to the intensive care unit (ICU) and mortality are related to both the disease itself and infectious complications. The clinical presentation is typically characterized by dyspnea, cough, hemoptysis, and in some cases fever, with chest radiography documenting pulmonary infiltrates. It may be difficult to distinguish from pneumonia, especially in patients without hemoptysis. Roughly 30% of patients with DAH do not have hemoptysis. The presence of renal dysfunction and hematuria in patients presenting with pulmonary symptoms should raise suspicion for a pulmonary-renal syndrome. Although Goodpasture syndrome was first used in 1958 to describe patients presenting with pulmonary hemorrhage and GN, the most common cause of pulmonary-renal syndrome is ANCA-associated vasculitis. Anti-GBM disease (formerly Goodpasture disease) now refers to the triad of DAH, RPGN, and the presence of anti-GBM antibodies. It is the second most common cause of pulmonary-renal syndrome. Much less common causes of pulmonary-renal syndromes are systemic lupus erythematosus (SLE), thrombotic microangiopathies, and other forms of systemic vasculitis.
A thorough history and physical examination may provide evidence for a systemic vasculitis (e.g., scleritis, purpuric rash, oral or sinus lesions). Bronchoscopy is critical to confirm DAH and evaluate for infection. The gold standard for diagnosis is renal or pulmonary biopsy, but critically ill patients are often at high risk for these procedures. The majority of patients have either ANCA-associated vasculitis or anti-GBM disease. Because both diseases are treated similarly in the acute setting of RPGN and DAH, treatment with plasma exchange, corticosteroids, and cyclophosphamide may be initiated rapidly before the availability of serologic results.
Pauci-immune necrotizing GN or ANCA-associated GN may present as a systemic vasculitis, pulmonary-renal syndrome, or renal-limited disease. The spectrum of disease includes microscopic polyangiitis (MPA), granulomatosis with polyangiitis (GPA), and eosinophilic granulomatosis with polyangiitis (EGPA or Churg-Strauss disease). , Kidney biopsy shows focal and segmental crescentic GN, fibrinoid necrosis, and an absence of Ig or complement within the glomeruli by immunofluorescence microscopy. Either anti-myeloperoxidase (MPO) or anti–proteinase 3 (PR3) antibodies are detectable in most patients. However, a minority of patients with characteristic clinical manifestations of these diseases and pauci-immune GN do not have detectable antibodies.
The mortality of untreated disease is roughly 90% at 2 years after disease onset. However, advances in treatment have led to improved patient outcomes, with 80%–85% of patients achieving remission with effective immunosuppressive strategies. Treatment consists of pulse intravenous (IV) methylprednisolone followed by oral corticosteroids and IV cyclophosphamide, rituximab, or both. , Even patients who are dialysis dependent on presentation often recover kidney function with appropriate treatment. Poor prognostic indicators for patient and renal survival are the presence of DAH, severe kidney dysfunction at the time of diagnosis, a high degree of glomerular injury and tubulointerstitial fibrosis on biopsy, and older age. Patients with DAH have a high mortality rate, and plasma exchange may improve patient survival. Coagulation factors should be replaced in patients with active hemorrhage. , For severe pulmonary disease, a few patients have been successfully treated with extracorporeal membrane oxygenation (ECMO). , Patients with severe renal disease have a greater likelihood of renal recovery when treated with plasma exchange. , , , ,
With appropriate treatment, roughly 80%–90% of patients achieve remission. , , , , Treatment resistance is more common in females, African Americans, and patients with severe renal disease. Relapse is more common in patients with anti-PR3 antibodies and in those with pulmonary and upper respiratory tract involvement. MPO ANCA is more commonly associated with MPA or renal-limited vasculitis. The ANCA-associated vasculitides (AAVs) follow a remitting and relapsing course, with the exception of drug-induced AAV (e.g., levamisole-adulterated cocaine, hydralazine), making long-term monitoring a key component to patient and kidney survival.
Anti-GBM disease (formerly Goodpasture disease) presents as DAH and RPGN with evidence of anti-GBM antibodies on serologic testing that target the noncollagenous domain of the α3 chain of type IV collagen. Kidney biopsy shows linear deposition of antibodies, most commonly IgG and C3, along the GBM with glomerular crescent formation. It is the most aggressive form of RPGN, and roughly 30%–40% of patients present with renal-limited disease without pulmonary involvement. It commonly affects Caucasians in a bimodal age distribution with peaks during the third and seventh decades. ,
Untreated disease is highly fatal, and death is usually caused by pulmonary hemorrhage or renal failure. Treatment with therapeutic plasma exchange, cytotoxic agents, and corticosteroids was introduced in the 1970s, resulting in improved patient and renal survival. In patients with pulmonary and renal involvement, plasma exchange is crucial for rapid clearance of anti-GBM antibodies and should be continued daily until antibodies are undetectable. Long-term outcomes are related to the degree of pulmonary compromise and renal dysfunction at presentation. With appropriate treatment, survival rates may exceed 90% for acute disease, but patients requiring RRT on initial presentation have lower survival rates. , , In fact, in patients with renal-limited anti-GBM disease requiring RRT at presentation, less than 10% recover renal function at 1 year despite treatment with plasma exchange, corticosteroids, and cyclophosphamide. ,
In contrast, patients with serum creatinine (SCr) levels below 5.7 mg/dL on presentation in one study had 100% 1-year patient and 95% renal survival. In addition to dialysis dependence and elevated SCr, predictors of poor renal outcome include oligoanuria, high anti-GBM antibody titers, and a high percentage of glomeruli with crescent formation and extensive tubulointerstitial disease on kidney biopsy. , , , Although patient and renal survival is generally worse with anti-GBM disease than with ANCA-associated disease, late recurrence of anti-GBM disease almost never occurs, whereas recurrence of ANCA-associated disease is common. ,
Both ANCA-associated vasculitis and anti-GBM disease are rare. Interestingly, a subset of patients have both types of antibodies detected on serologic studies. Approximately 15%–30% of patients with ANCA-associated disease also have anti-GBM antibodies, whereas only 5%–10% of patients with anti-GBM antibodies also have detectable ANCA titers. , , , Although outcome data are limited in this small group of patients, the outcomes of these patients may be better than patients with only anti-GBM antibodies.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here