Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
| Symptomatic and disease-modifying treatment |
|
| Disease-modifying treatments: amyloid plaques |
|
| Disease-modifying treatments: neurofibrillary tangles |
|
| Beyond amyloid and tau |
|
Because of the rapid aging of the population and the accompanying increase in the prevalence of Alzheimer’s disease, there is enormous interest from both the federal government and private industry in developing new and better treatments. In general, these treatments are aimed at two targets: treating the symptoms of Alzheimer’s disease and attempting to slow the progression of the disease.
Patients for whom we prescribe a cholinesterase inhibitor, memantine, or a combination of the two often ask how these medications work and what else can be done. After explaining the mechanism of these standard of care, U.S Food and Drug Administration (FDA)-approved medications, we often have a discussion of some of the types of treatment that are currently being developed. There are two basic reasons why we discuss future treatments. One is to give patients (and their children who are often worried about developing Alzheimer’s disease when they are older) hope for the future. The second is that some patients may be interested in participating in one of the numerous clinical trials of novel medications that are being developed for Alzheimer’s disease, available in many large medical centers and Alzheimer’s disease specialty clinics/clinical research centers.
One strategy to further help patients with Alzheimer’s disease is to facilitate neuronal transmission, that is, communication between brain cells. The cholinesterase inhibitors and memantine improve the function of neurons that use acetylcholine, glutamate, and dopamine as their neurotransmitters. There are, however, other neurotransmitter systems in addition to these that can be augmented. It is now well documented that Alzheimer’s disease affects many neurotransmitter systems. Table 23.1 summarizes these transmitter systems. At some point we might envision a drug cocktail that will boost the level of multiple neurotransmitter systems, thereby facilitating cognition. We are now at the beginning of this type of strategy when we combine memantine with a cholinesterase inhibitor. We may eventually become sufficiently sophisticated to tailor the cocktail based on the specific symptoms that the patient is experiencing or even the patient’s genotype.
| Transmitter System | Degree of Involvement in Alzheimer’s Disease | Approved Medications | Clinical Trials Ongoing? |
|---|---|---|---|
| Acetylcholine | ✓✓✓ | donepezil (Aricept), rivastigmine (Excelon), galantamine (Razadyne) | ✓✓ |
| Glutamate | ✓✓✓ | memantine (Namenda) | ✓ |
| Serotonin | ✓✓ | — | ✓✓ |
| Norepinephrine | ✓✓ | — | ✓ |
| Dopamine | ✓ | memantine (Namenda) | ✓ |
| GABA | ✓ | — | ✓ |
| Peptides | ✓ | — | ✓ |
There continues to be research ongoing aimed at developing additional symptomatic treatments. Recent trials include drugs to affect acetylcholine, N -methyl-D-aspartic and serotonin activity as well as trials to examine the effect of anti-inflammatory, metabolic, and hormonal treatments. Many of these trials are evaluating the effects of adding drugs to the current standard of care (cholinesterase inhibitors) ( ). Additionally, in a recent clinical trial, suvorexant showed a modest improvement in Alzheimer’s disease patients with sleep disorder and based on this was approved by the FDA for treatment of insomnia in Alzheimer’s disease dementia and can now be prescribed ( ).
Development of symptomatic treatments notwithstanding, the major focus of new drug development for Alzheimer’s disease is on disease-modifying drugs. That is, drugs that address the underlying cause of the disease and in doing so prevent cell death. If this strategy is successful, then the progression of the disease could theoretically be slowed and perhaps even be halted ( Fig. 23.1 ). If this strategy could be combined with both early diagnosis—or even preclinical diagnosis—and the use of symptomatic drugs, we could be on the pathway of successfully managing Alzheimer’s disease.
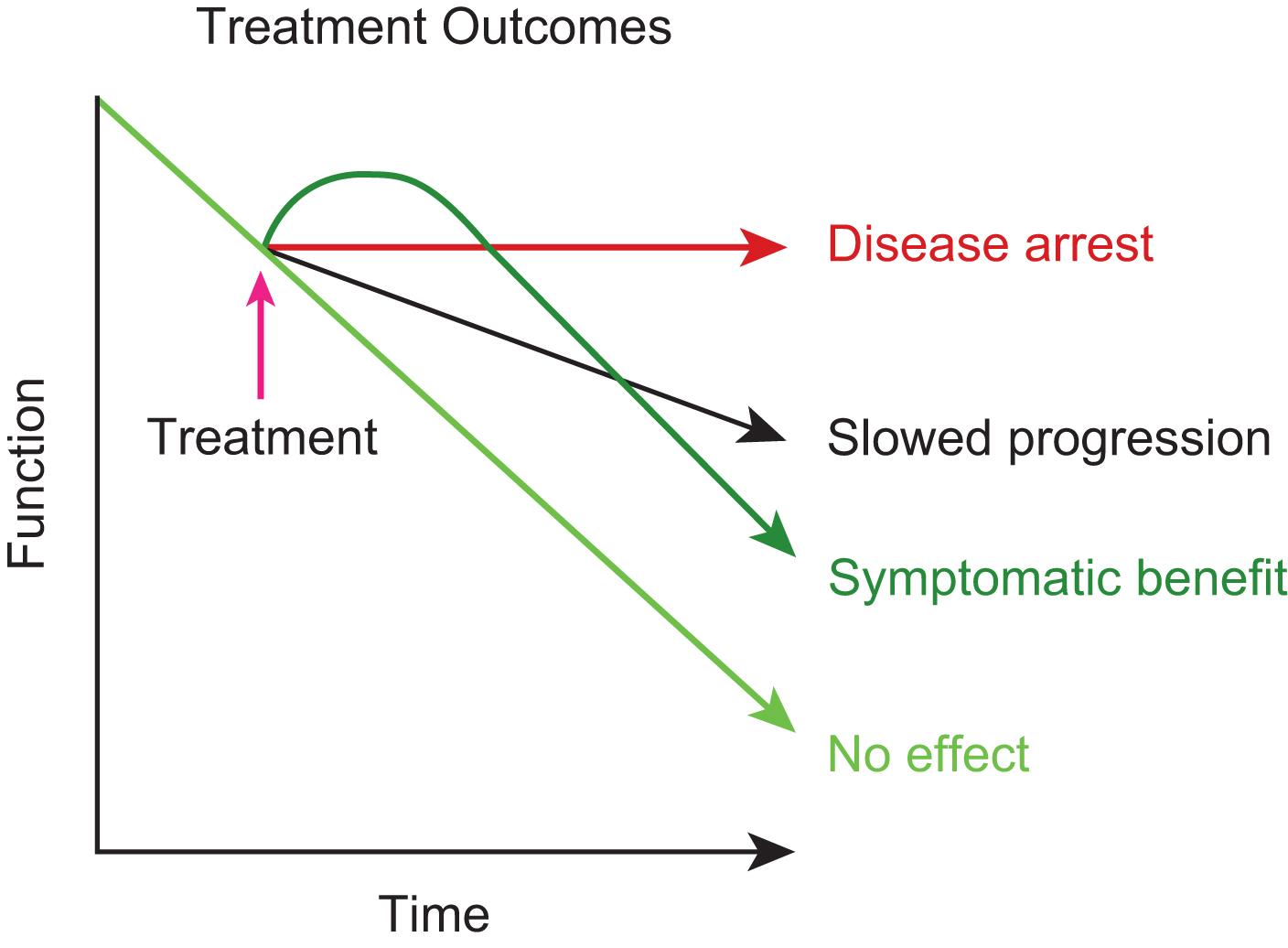
Although the cause of Alzheimer’s disease is unknown, there are a number of promising hypotheses regarding the pathogenesis of the disease, and these hypotheses have led to the development of potential disease-modifying treatments.
When Alois Alzheimer had the opportunity to examine the brain of his first patient, Auguste D, in 1906, he characterized the two forms of pathology that are the neuropathological hallmarks of the disease that now bears his name: senile plaques and neurofibrillary tangles.
Senile plaques appear to have a fluffy central core surrounded by thick irregular processes ( Fig. 23.2 ). The central core is made up of a sticky protein called amyloid, and the surrounding material is a combination of dystrophic processes (axons and dendrites) and astrocytes. These plaques are primarily found in the association areas of the frontal, parietal, and temporal cortices and the piriform cortex, hippocampus, and amygdala ( Fig. 2.1 ). Neurofibrillary tangles are intraneuronal cytoplasmic structures that are composed of paired filaments. Under the microscope they look like skeins of yarn (see Fig. 23.2 ). These tangles appear to consist primarily of a hyperphosphorylated form of the microtubule-associated protein tau . Microtubules are one of three major constituents of the neuronal cytoskeleton; neurofilaments and microfilaments are the other two. All of these can be thought of as infrastructural elements of neurons that participate in functions such as axonal transport and maintenance of the structural integrity of the cell. In Alzheimer’s disease, neurofibrillary tangles are present in the cortex, hippocampus, amygdala, nucleus basalis of Meynert, dorsal raphe, other brainstem nuclei, and ultimately in many other brain regions (see Chapter 4 for additional information on senile plaques and neurofibrillary tangles).
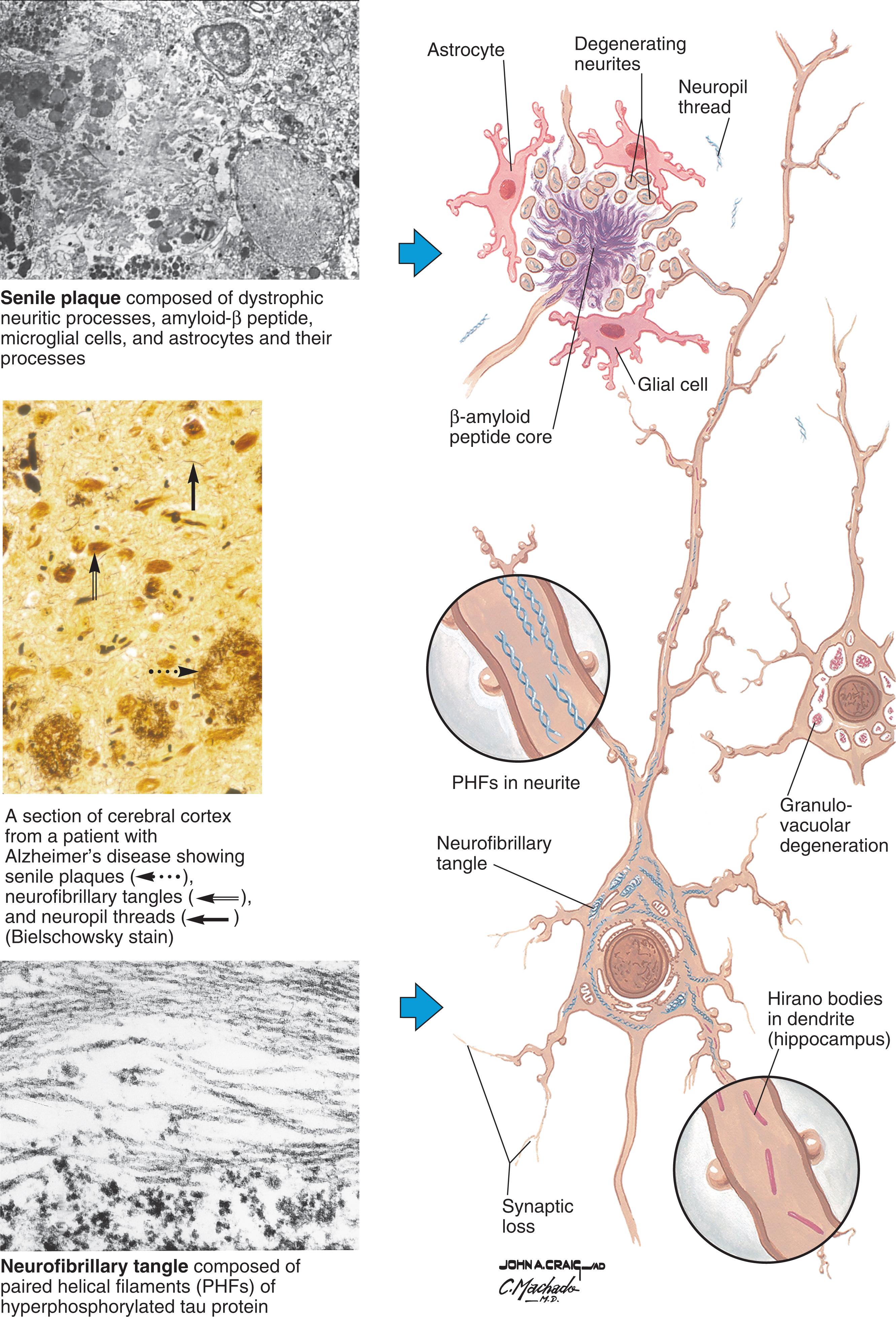
For many years there was an ongoing debate regarding which form of pathology, senile plaques or neurofibrillary tangles, is the primary culprit in Alzheimer’s disease. The debate became known as the “baptists” versus the “tauists.” The baptists favored the view that β-amyloid protein and β-amyloid plaques are the culprits that start an inexorable cascade that ultimately destroys neurons. They are referred to as baptists because β-amyloid protein (or amyloid plaque) can be shortened to βAP but is now more commonly known as β-amyloid or βA. The tauists put forth the view that neurofibrillary tangles are the primary cause of cell death. They were called tauists because of the role of hyperphosphorylated tau in forming the tangles.
As with many scientific debates, there is often a synthesis of the two competing views and this is now the case in the baptist-tauist debate. The current view is that the earliest pathological event is the formation of amyloid. There is now evidence that the accumulation of brain amyloid precedes the emergence of cognitive symptoms by at least and perhaps up to 20 years ( ). There is also evidence that β-amyloid is toxic to neurons, but the primary culprit in cell death is now believed to hyperphosphorylated tau. Hyperphosphorylated tau breaks down the infrastructure of the neuron and in doing so leads to neuronal death. The current view further hypothesizes that through mechanisms that are not fully understood, β-amyloid promotes the emergence of hyperphosphorylated tau and that once tau invades a neuron it can spread throughout the brain from neuron to neuron ( ). It is important to realize that although amyloid plaques are unique to Alzheimer’s disease, neurofibrillary tangles are present in other neurodegenerative diseases and are generally considered markers of cell death.
The amyloid cascade hypothesis has been the dominant heuristic driving effort to produce disease modifying treatments. Although most of the effort to date has been focused on βA, more recently in addition to the continuing work related to amyloid, there are also now increasing efforts to address the role of tau and other downstream effects of amyloid in the progression of Alzheimer’s disease ( ).
The hypothesis starts with the premise that the earliest sign of the pathogenesis of Alzheimer’s disease is the accumulation of toxic forms of amyloid. It then follows that the focus of treatment of disease-modifying drugs is to slow and eventually stop the accumulation of amyloid.
Fig. 4.10 summarizes the amyloid cascade hypothesis ( ). As Fig. 4.10 shows, the initial assumption is that different gene defects can lead, either directly or indirectly, to an increase in toxic forms of β-amyloid. This increase may be because of either overproduction of or failure to clear these toxic forms of amyloid. Gradual accumulation of aggregated β-amyloid protein leads to a multistep cascade that includes inflammation, neuritic and synaptic changes, neurofibrillary tangles, neurotransmitter loss, and gliosis, and ultimately results in cell death and dementia.
β-Amyloid is a small piece of a much larger protein called the amyloid precursor protein (APP). The APP is a transmembrane protein that, when activated, is cut into smaller segments which operate either inside or outside the neuron. There are several ways that the APP can be cut, one of which leads to the production of β-amyloid (see Figs. 23.3 and 4.11 ).
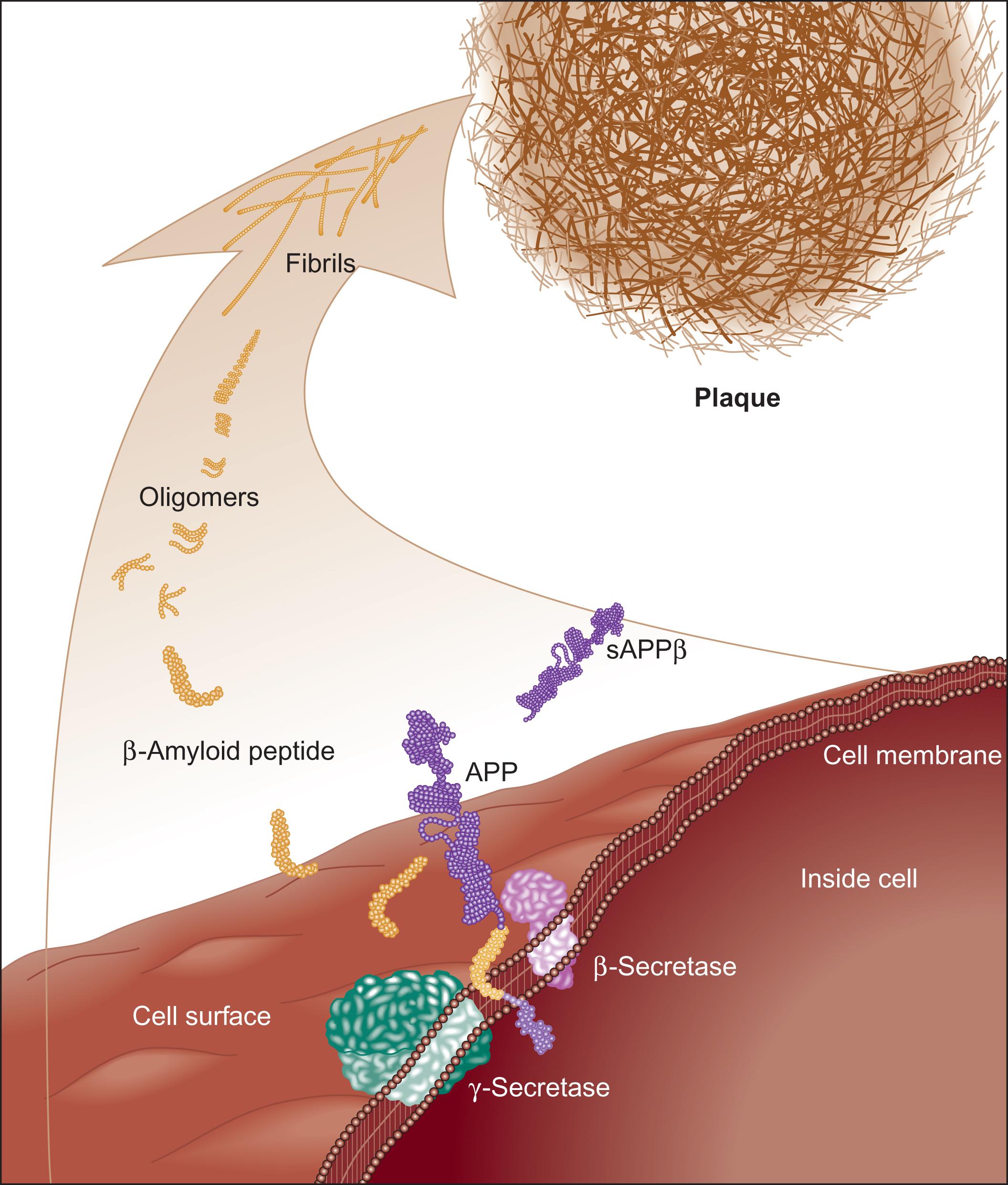
There is considerable evidence to support the amyloid cascade hypothesis, for example:
In a few hundred extended families worldwide, researchers have discovered mutations in genes that virtually guarantee an individual will develop Alzheimer’s disease at a young age. Each of these abnormal genes increases β-amyloid production (those in the APP, presenilin 1, or presenilin 2).
All Alzheimer’s disease patients have amyloid plaque counts that far exceed those found in normal aging.
Down’s syndrome patients, who invariably develop Alzheimer’s disease pathology by age 50 years, produce too much β-amyloid protein from birth (presumably because they have a third copy of the gene coding for the APP, found on chromosome 21).
β-Amyloid fibrils damage neurons in culture and activate brain inflammatory cells (microglia).
It is hypothesized, with increasing supportive evidence, that β-amyloid can also induce hyperphosphorylated tau which leads to the formation of neurofibrillary tangles and that these tangles can spread from neuron to neuron throughout the brain.
Despite this evidence, the amyloid cascade hypothesis is still a hypothesis, and as with all hypotheses there are unanswered questions. Nevertheless, the amyloid cascade hypothesis continues to be the most widely accepted view of the pathogenesis of Alzheimer’s disease and as such amyloid has been and continues to be a major therapeutic target.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here