Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Dr. Porter would like to acknowledge the assistance in manuscript preparation from Anoka Padubidri, MD, orthopedic resident, Indiana University; and Madison Walrod and Nicole Heffern, students, Butler University.
Tarsal tunnel syndrome (TTS) is classified as a focal compressive neuropathy of the posterior tibial nerve (PTN) or one of its associated branches individually or collectively within the tarsal tunnel. The tarsal tunnel is a fibro-osseous space that is defined by the medial malleolus (superiorly), tibia (anterior border), posterior process of the talus (posterior border), calcaneus (lateral border), abductor hallucis (inferior border), and flexor retinaculum (laciniate ligament), which lies over the tibial nerve to create an enclosed space. Within the tarsal tunnel lies the long flexor tendons and posterior tibial artery and vein along with the tibial nerve and its branches. Because of the inelasticity of the tunnel, any enlargement of the structures inside the tunnel can increase pressure leading to nerve compression. In general, nerve injury in this area is secondary to another pathology. The intrinsic and extrinsic etiologies of TTS primarily stem from posttraumatic, biomechanical, inflammatory, and morphological conditions; however, it can also be classified as an idiopathic and iatrogenic syndrome. Characteristic clinical manifestations of TTS include paresthesia, dysesthesia, and hyperesthesia radiating from the retro-malleolar region to either the sole, heel, digits of the forefoot, or any combination of these areas. In some cases, pain has been shown to radiate proximally up the calf. The patient may report foot weakness as well. In addition to the symptoms described above, swelling over the tibial nerve may be palpable. Symptoms are typically made worse with increased activity such as standing, walking, or running. The varying location and degree of symptoms depends on the location and the degree of compression of the tibial nerve or nerve branches.
While rare in the general population, TTS has become increasingly more common in athletes. Neurologic conditions account for 10% to 15% of all exercise-induced leg pain among running athletes. Although the exact incidence of TTS in athletes is unknown, a study conducted by Kinoshita et al. identified surgically treated TTS patients, and found that between 1986 and 2002, 2.7 patients were treated annually with TTS. While this number is small, the percentage of sports-related cases among them is relatively high (39.1%). As mentioned above, TTS typically due to a secondary pathology. The increased frequency in athletes could be related to direct contusion, ill-fitting shoe wear, space occupying pathology (e.g., venous stasis, varicosities, accessory muscles, and ganglion cysts), or lower limb malalignment. Specific examples include tenosynovitis, accessory muscle (accessory soleus, accessory flexor tendons), fracture, or, more rarely, ligament fibrosis from chronic medial/deltoid ankle sprains. Repetitive trauma in the setting of predisposing malalignments is commonly seen in athletes. Researchers believe that injury to the PTN, because of its location and vulnerability at the tarsal tunnel, is due to the repetitive nature of running accompanied with abnormal or excessive pronation. Valgus deformity or flat feet can also exacerbate the problem. Additionally, sports that require repetitive hyper dorsiflexion may predispose athletes to TTS because dorsiflexion increases the overall pressure within the tarsal tunnel.
Athletes with TTS will experience symptoms similar to nonathletes. Individuals with TTS may report medial ankle pain with cramping, burning, and tingling that radiates into the plantar arch of the foot and is exacerbated by activity, such as running, jumping, and prolonged standing. The pain can be sharp, shooting, or dull. Swelling, as mentioned previously, may also be present. Individuals may get relief from symptoms with rest, elevation, and removal of tight shoe wear.
Medical causes of leg/ankle-pain have many similar presenting features, which makes TTS frequently misdiagnosed or underdiagnosed. Underdiagnoses is one possible reason why the incidence of tarsal tunnel in the general population and athletes seems low. Clinical presentations may appear similar, but in fact have a different pathophysiology relating to separate vascular abnormalities, compartment syndrome or neurological dysfunction. For example, Sanger et al. reviewed the case of a 19-year-old female collegiate soccer player complaining of unilateral foot pain that later transformed into bilateral foot pain with numbness, tingling, and cramping. This led to the diagnosis of TTS; however, the initial diagnosis was plantar fasciitis because of the location of pain and the description of symptoms. The misdiagnosis subsequently delayed her return to sport. Early diagnosis is key because a delay in diagnosis can lead to worsening of symptoms, inferior athletic performance, missed competition, and delayed return to sport. Clinicians should consider several general principles to facilitate the diagnosis and management of neurologic symptom: (1) maintain high level of index of suspicion for neurologic syndromes; (2) recognize common presentations of neuropathic pain; (3) perform a meticulous physical examination with detailed palpation of individual anatomic structures; (4) consider a broad differential diagnosis (neurologic and non-neurologic); (5) use diagnostic testing appropriately—electromyography/nerve conduction velocity test (EMG/NCV) and magnetic resonance imaging (MRI); and (6) make rational clinical decisions, including referral for second opinion when indicated.
Tarsal tunnel syndrome diagnosis greatly relies on an in-depth history and physical examination because of the many possible causes of leg/ankle-pain. The aim of the history in a patient with possible neurologic leg/ankle pain is to gather occupational and sporting history, to determine the location and timing of the pain, its mechanism of onset, degree of irritability, aggravating and relieving factors, presence of other joint pain or swelling, previous local trauma, and presence of neurological signs or symptoms. The physical exam combines the use of many investigations to properly diagnosis TTS. The Hoffmann-Tinel sign is the most commonly used clinical test of TTS. A positive Tinel sign or nerve percussion/compression test will cause paresthesia either locally or radiating along the course of the nerve when the suspected site of compression is percussed. It is suggested that greater than 50% of patients with compression neuropathy of the tarsal tunnel will demonstrate a positive Tinel’s sign of the PTN. Patients may also exhibit loss of two-point discrimination or occasionally, an abnormal Semmes-Weinstein test for pressure. Dorsiflexion-eversion test has been shown to increase strain on the tibial nerve and illicit symptoms also. Kinoshita et al. report that the dorsiflexion-eversion test reproduces or aggravates symptoms in 36 out of 44 feet in their study, with no replication evident in the control group. However, it is this author’s opinion that local pain with palpation specific to the PTN nerve and the medial plantar nerve (MPN) is still the hallmark of TTS and its diagnosis.
Radiological imaging modalities such as isotope bone scanning, ultrasound, and magnetic resonance imaging (MRI) relate to the detection of bone and soft tissue lesions rather than neurological disease for the etiology of TTS. MRI, the gold standard in identifying suspected compression of the tarsal tunnel caused by obstructive masses, can be used to confirm space-occupying pathology. MRI has been shown to be effective in 88% of patients with a firm clinical diagnosis of TTS. Not only does it confirm a suspected lesion, but it defines the depth, extent, and margins of the lesion for accurate characterization. Ultrasounds can also effectively visualize structures within the tarsal tunnel and accurately identify compressive lesions and/or focal changes in the tarsal tunnel cross-section area. Diagnostic evaluation should also include weight-bearing foot and ankle radiographs to evaluate for deformity, talocalcaneal coalition, fracture malunions, and osteophytes.
We have noted an increasing occurrence of an accessory muscle that acts to compress the PTN in the tarsal tunnel in the athlete. This can present as either an accessory soleus, which is outside the tarsal tunnel but still causes pressure on the PTN, or an accessory flexor tendon within the tarsal tunnel. The accessory flexor tendon is unnamed but is thought to be an accessory flexor hallucis tendon ( Fig. 11.1 ).
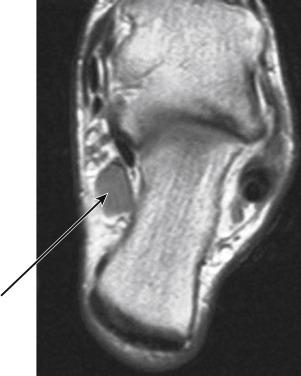
In some circumstances, the accessory flexor tendon in the tarsal tunnel will only affect the lateral plantar nerve. In this isolated lateral plantar nerve compression, the athlete complains only of pain in the lateral plantar foot (see case study). This presentation can be more confounding, and we have seen athletes misdiagnosed as fifth MT stress fractures, peroneal tendon injury, and cuboid stress fracture. Failure to improve with nonweight bearing (NWB) or immobilization is often a clue to nerve pain in the athlete.
In addition, electrodiagnostic investigations, which include nerve conduction studies and EMG, can assist in the diagnosis of TTS. While it has been concluded that sensory nerve conduction is more sensitive, yet less specific in identifying nerve injury than motor nerve conduction, it is recommended that both motor and sensory nerve conductions are evaluated. Unfortunately, these studies often yield an unacceptable level of false-negative results with inexperienced electromyographers, and should be utilized as an adjunctive assessment to confirm physical examination findings. Because of the poor sensitivity and specificity for TTS, these investigations may be most useful in ruling out proximal nerve pathology. We do believe, however, that with experience, detection of TTS with EMG/NCV can be more sensitive and specific. We therefore recommend developing a relationship with an interested lower extremity electromyographer to enhance their capabilities diagnosing the condition with the technique.
Researchers have recently recommended that a firm diagnosis of TTS be made only when the following triad exists: (1) foot pain and paresthesia, (2) positive nerve percussion sign/Tinel sign, and (3) positive electrodiagnostic studies. If only two exist, then the term probable TTS is recommended, and if only one exists, the diagnosis should be reconsidered.
Nonoperative, conservative treatments are the preferred initial treatments even though they are often unsuccessful. Treatment includes activity modification, immobilization, oral or topical nonsteroid antiinflammatory drugs (NSAIDs), neuromodulator medications (e.g., Gabapentin, Pregabalin), physical therapy, biomechanical management, and steroid injections. Physical therapy includes strengthening the foot intrinsic and medial arch supporting muscles, Achilles stretching in subtalar neutral, lower limb kinetic chain rehabilitation, and proprioception-enriched rehabilitation in cases of ankle or subtalar joint instability. Special attention should be paid to Achilles tendon flexibility and foot intrinsic and ankle support muscle strength and coordination. Biomechanical management varies with clinical presentation and can be assisted with the use of motion control shoe, medial heel wedge, medial sole wedge/medial buttress, ankle stirrup brace, heel lift, or fixed ankle walking brace. In severe cases, medial wedges and arch supports may exacerbate symptoms, and more rigid immobilization via molded hindfoot orthosis, ankle-foot orthosis, or walking boot/cast may be needed. Tarsal tunnel injections may assist with treatment by reducing swelling in the tarsal tunnel providing relief of symptomatic nerve compression. However, for those infrequently injecting the tarsal tunnel or if there is a poor pulse to palpate, ultrasound guidance should be considered when doing steroid injections to minimize risk of neurovascular injury.
In the result of failed conservative treatments, surgery should be considered. Surgical procedures involve decompressing the tibial nerve or involved branches, while also repairing the pathology responsible for increased tunnel pressure. This may involve dissection of the flexor retinaculum, neurolysis of the affected nerve, tenosynovectomy, excision of bony fragments, stretching of the neurovascular bundle around foot deformities, or removal of space-occupying masses. While pain relief can be immediate with surgery, neuropathic symptoms generally improve 6 weeks postoperatively, but maximal recovery time may take 6 months or more. Kinoshita and colleagues reported a complete return to sports in 12 of 18 athletes postsurgery. Many studies have revealed that surgical release may improve or resolve the overall symptoms of TTS in 85%–90% of cases. Alternatively, several studies have also reported little to no improvement of symptoms from surgical intervention. Researchers believe that this could be due to continual clinical dichotomy between objective and subjective surgical outcome parameters, making definitive results highly variable. While failure is described to be less than 5%, initial releases often fail because of failure to control hemostasis, which leads to scarring and neuritis or nerve damage. Therefore, we advise meticulous hemostasis after tourniquet release before definitive skin closure. Failures of surgical intervention often result from incorrect initial diagnosis, incomplete release of the tarsal tunnel (releasing the PTN, without releasing the MPN in the abductor canal), adhesive neuritis following initial decompression measures, intraneural damage associated with direct neural trauma or systemic disease, presence of space-occupying lesion, or double crush syndrome.
The athlete was a 21-year-old white female senior basketball player who plays the 4-5 position on a Division I program majoring in Sports Management, who was originally from Serbia. She was evaluated for right lateral foot pain. She presented with a chronic 3-year history of right foot pain that was intermittent, achy, throbbing, sharp, and burning in nature. She states that she does not remember a specific injury but has a history of “recurrent stress fractures/stress reactions in the right foot” that never improved with immobilization, rest, even NWB status. Because of her lateral foot pain we were concerned for a peroneal tendon or abductor digitii quniti minimi (ADQM) injury. An MRI was normal and particularly showed no stress fractures and normal peroneal tendons. We tried an injection in the ADQM with no relief. We then ordered an EMG/NCV test, and it showed changes in the lateral plantar mixed nerve only with decreased amplitudes (essentially absent) but near-normal medial plantar mixed nerve amplitudes. There were severe changes on the needle exam to the ADQM with severe motor unit drop-out and severe loss of recruitment consistent with lateral plantar nerve involvement. The needle exam to the MPN was essentially normal.
The exam was consistent with pain over the PTN in the tarsal tunnel. There was some sharp tenderness locally and radiation into the lateral plantar foot that reproduced her symptoms. The plain x-rays were normal.
We performed an injection into the right tarsal tunnel placing some local anesthetic with steroid around the PTN and the MPN. We were able to confirm we were near the nerve by some mild radiation into the distal nerve distribution with the injection. It took 10 days to 2 weeks to get relief (a little longer than typical), but the athlete reported near-complete relief with her lateral foot pain. Since we were still in the off-season, she elected to proceed with a complete tarsal tunnel release . We followed a typical postoperative protocol that included cold compression therapy intermittently; boot immobilization for 2–4 weeks; then gradual return to biking, Stairmaster, and elliptical; and then running with a lace-up and velcro ankle brace. A functional progression program followed after she could run comfortably for 30–40 minutes on a treadmill. She returned to practice at 3 months and led the team in scoring and rebounding her senior year. She returned overseas after the season and pursued professional basketball. She obtained complete relief of her preoperative pain.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here