Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Fine needle aspiration (FNA) of the kidney is a useful technique for diagnosing a specific subset of renal lesions. Most renal lesions in adults are either benign cysts (based on their radiographic appearance) that can be left alone or are sufficiently worrisome that resection is indicated regardless of the results of FNA. It is estimated that only 10% to 30% of all renal masses are candidates for FNA.
Until recently, renal FNA in the pediatric population was contradicted because aspiration of a suspected Wilms tumor, the most common renal tumor in children, resulted in clinical “upstaging.” This has been changed and has resulted in an increase in the use of FNA in pediatric patients in some centers. Interpretation of these specimens is challenging, and it is discussed more fully in the Fine Needle Aspiration in Pediatric Patients “section at the end of” this chapter.
Both FNA and core needle biopsy can be performed. Immunohistochemistry and genetic studies can be done on both types of specimens. Although each technique has its own advantages and disadvantages, the combination of FNA and core needle biopsy has the highest yield. However, in some cases an aspirator may be willing to do an FNA but unwilling to do a core needle biopsy because of the risk for hemorrhage. Thus the ability to interpret FNA material remains an important skill. Because the interpretation of core biopsies is similar to that of routine histologic assessment, which is discussed in more detail in other chapters of this textbook, this chapter will focus on interpretation of FNA specimens. Regardless of the biopsy method, accurate identification of some unusual subtypes of renal cell carcinoma (RCC) may depend on both adequate sampling and ancillary studies, and these may not always be available. As a result, accurate subtyping of some unusual types of RCC may not always be possible with either FNA or core needle biopsy.
FNA has several indications. First, FNA can provide a diagnosis in a patient who is not a candidate for resection. This includes patients whose primary tumor is high stage, patients with presumptive metastatic disease, or patients who are not medically able to undergo resection. In addition, many urologists will consider following rather than resecting benign lesions such as oncocytoma, and this is a relatively common indication for FNA. Second, FNA has traditionally been used to evaluate patients with radiographically indeterminate lesions, usually cysts. However, the utility of the technique in this setting is quite limited; see the section on renal cysts. Finally, although the ultimate decision to perform a partial rather than total nephrectomy is a surgical one, the results of FNA can be useful in triaging these patients. Candidates for partial nephrectomy include patients with small lesions, young patients, and patients who are at risk for multiple and bilateral lesions, such as patients with von Hippel–Lindau syndrome. More and more patients are potential candidates for partial nephrectomy, in part because higher-resolution imaging techniques can identify smaller lesions than in the past. FNA can provide a specific or differential diagnosis in most of these patients. Finally, in the patient with focal bacterial pyelonephritis or a renal abscess, needle placement permits both diagnosis and therapeutic drainage.
Virtually all renal aspirations are performed percutaneously by radiologists using ultrasound, computed tomography, or magnetic resonance imaging. Rarely a surgeon might obtain an FNA intraoperatively. Most of the literature is on alcohol-fixed specimens prepared with either Papanicolaou (Pap) or hematoxylin and eosin stains, or air-dried Giemsa-stained material. Cell blocks can be particularly helpful for identifying papillae and other architectural features, and many lesions are best diagnosed with immunohistochemical support (e.g., angiomyolipoma). Genetic studies can be useful, and sometimes necessary, in subtyping RCCs. Fluorescence in situ hybridization can be performed on unstained cytospins, monolayers, smears, and cell block sections.
The complications of renal FNA include hemorrhage, pneumothorax, infection, arteriovenous fistula, and urinoma. All are uncommon. Morbidity, mortality, and needle track cancer seeding are rare in the modern era with the use of smaller (less than 18 gauge) needles.
Based on historical studies, renal FNA accurately distinguishes benign from malignant lesions in 73% to 94% of cases; correct subclassification of RCC is achieved in 74% to 80% of cases, and up to 90% for the most common subtypes of RCC using routine cytology. More recent studies show that accuracy can be increased up to 99% on core biopsy material for the most common subtypes of RCC by using immunohistochemistry for carbonic anhydrase IX, CD117, α-methylacyl-coenzyme A racemase, cytokeratin (CK) 7, and CD10. Other panels have also been shown to be of value. To date, subtyping RCC is generally not necessary for the treatment of most patients, although it may provide prognostic information. As therapies other than surgery continue to improve, this may not remain true, especially if some of these therapies prove subtype specific. As noted earlier, recognition of the common subtypes is highly reliable. The recognition of unusual RCC subtypes on limited material without adequate ancillary studies may be impossible, and this may drive the need for the development of more thorough sampling protocols. Unfortunately, no specific immunohistochemical stain exists for oncocytoma, making this a diagnosis of exclusion, and perhaps less reliable than a diagnosis of RCC.
Renal FNA is performed infrequently, and many cytopathologists may not have much experience evaluating these specimens. False-positive results have occurred when xanthogranulomatous pyelonephritis, angiomyolipoma, benign hepatocytes, benign tubular cells, glomeruli, and benign adrenal cortical cells have been misinterpreted as RCC. In the past, interpretation of renal FNA has been one of the poorest performers on survey material, with a false-positive rate of 30.4% and a false-negative rate of 13.9%. Many of these errors relate to misinterpretation of normal elements as tumor. Recent surveys have shown improved performance.
Cellularity is important to consider when interpreting a kidney FNA; rare atypical cells are common in many benign lesions. Hypocellular kidney aspirates therefore should never be diagnosed as positive.
Up to 30% of renal aspirates are nondiagnostic (inadequate); repeat aspiration is helpful in approximately half of cases. Most inadequate specimens are related to technical failure to obtain adequate diagnostic material. Although specific adequacy criteria are not well defined, awareness of the radiographic appearance of the lesion is important. For example, an aspirate composed exclusively of macrophages from a known Bosniak category 1 lesion (see later discussion of cysts for definition) being drained for symptomatic relief is both adequate and diagnostic (benign); in contrast, the same aspirate from a solid Bosniak category 2 or 3 lesion is neither adequate nor diagnostic, and is best reported as “nondiagnostic” rather than negative.
Normal elements are commonly present in renal FNA.
Glomeruli ( Fig. 4.1 ) are highly cellular globular structures that may mimic the papillae that are seen in papillary RCC, especially at low power. In contrast with papillary RCC, however, endothelial cells lining the capillary loops can be seen at the extreme edge of many glomeruli. In RCC the neoplastic cells may be at the edge of a large cluster, but the cytoplasm of the cell almost always extends even more peripherally than the nucleus, and the nucleus is rounder than that of an endothelial cell.
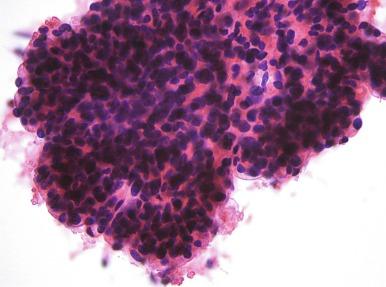
Proximal tubular cells ( Fig. 4.2 ) consist of single cells with round bland nuclei, small but easily seen nucleoli, and abundant, granular cytoplasm. These normal cells are often ripped from their tubules, and as a result, the cell membrane is usually torn and the granules often appear to be spilling out of the cells. Less commonly, entire tubules may be aspirated, resulting in small round to elongate nests of cells. These cells are similar, if not identical, to those seen in aspirates from oncocytoma and some cases of papillary and chromophobe RCC. However, FNAs of tumors are typically quite cellular. Papillary tumors have papillae that are best seen on cell block preparations. Chromophobe RCC tumor cells are often binucleate; there is variation in cell and nuclear size and shape. The cell borders of the tumor cells are usually well defined because these cells are only loosely cohesive and relatively easily removed from the tumor, and chromophobe RCC rarely produces round nests of cells.
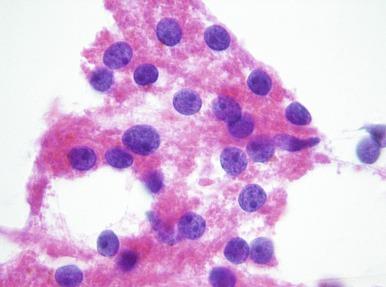
Distal tubular cells ( Fig. 4.3 ) are small, single or cohesive cells in sheets of up to about 20 cells, with scant clear to slightly granular cytoplasm and small, round nuclei with or without very small nucleoli. Cell membranes are usually intact, but may be difficult to see because the cytoplasm is scant. What little cytoplasm that is present is clear to minimally granulated, but should not be vacuolated. These cells are identical to those seen in either low-grade clear cell or papillary RCC. However, aspirates of tumors should be more cellular, both tumors may have intracytoplasmic hemosiderin, and papillary RCC may form papillae and spherules.
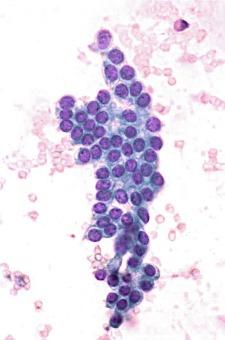
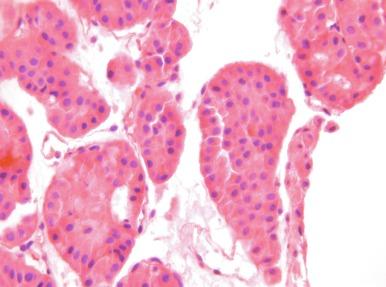
Oncocytoma comprises 3% to 5% of all renal tumors and is benign, although rare metastases have been reported (see Chapter 2 for additional discussion of the metastatic potential of oncocytoma). The tumor cells have abundant granular cytoplasm and uniform round nuclei, with variably prominent nucleoli, and in many cases salt-and-pepper type chromatin. Occasional large bizarre nuclei may be present (similar to “endocrine atypia”), but mitotic figures are absent or rare. The arrangement of the neoplastic cells in rounded nests is distinctive and stands in contrast with the trabeculae (ribbons) of chromophobe RCC. In addition, although the classic gross description of this tumor as a mahogany brown lesion with a central scar continues to be perpetuated in most textbooks, a more accurate description is a well-circumscribed lesion that is exactly the same color as the adjacent cortex, which is mahogany brown if the kidney is removed without draining the blood first or more pale gray or tan if the kidney has been drained before resection.
Cytologically, the cells of oncocytoma are easily dissociated and aspirates are typically highly cellular. The specimens are composed of numerous single cells and loosely cohesive small clusters of up to about 10 cells, with abundant, eosinophilic, granular cytoplasm, well-defined cell membranes, frequent binucleation, and round nuclei with small but distinct nucleoli, and in many but not all cases, salt-and-pepper chromatin ( Fig. 4.4 ). Although atypia can be present in oncocytoma, it is of a specific type. In general this is “endocrine” atypia and consists of nuclei with marked size variation and relatively dark chromatin without prominent nucleoli. This is in marked contrast with the prominent nucleoli and fine chromatin of most RCCs. Although hyaline globules have been reported in oncocytomas, they are not specific.
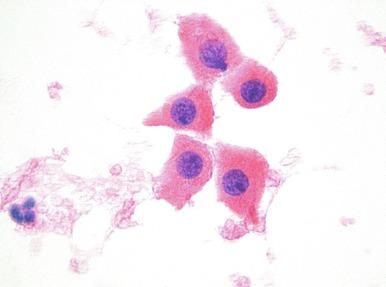
The differential diagnosis includes normal proximal tubules, clear cell RCC, chromophobe RCC, papillary RCC, and hepatocytes. The cells of proximal tubules are identical, but aspirates are scant and the cell membranes are rarely intact. Even though oncocytoma may have cohesive cells, it rarely forms the rounded nests that one sees in normal proximal tubules. Instead the clusters of cells are more loosely cohesive and irregular. Although clear cell RCC is always listed in the differential diagnosis of oncocytoma, this is usually easy to resolve. The aspirates of clear cell RCC are more cohesive, have fewer single cells, more nuclear atypia, and less uniformly granular cytoplasm. Although some clear cell tumors may have abundant granular cytoplasm, most are higher-grade tumors. In addition, clear cell RCC is CD117 − , whereas oncocytoma is CD117 + . In contrast, chromophobe RCC may be extremely difficult to distinguish from oncocytoma, but in general has less uniformly granular cytoplasm and often more nuclear outline irregularity; nucleoli are rare and the chromatin is finer (although it may be either light or dark). Chromophobe tumors show diffuse cytoplasmic positivity with Hale colloidal iron staining, a finding that supports the diagnosis. However, oncocytoma usually shows focal to moderate membranous staining with Hale colloidal iron stain, which can be difficult to interpret. Both tumors are CD117 + . Chromophobe RCC is reported to be more diffusely positive for CK7, CK20, and PAX8 than oncocytoma, but reactivity is often patchy in both tumors.
Rare low-grade papillary tumors can have cells that are identical to those seen in oncocytoma, but the papillary architecture is diagnostic, and these tumors should be diffusely CK7 + . As in clear cell tumors, most papillary tumors with granular cytoplasm are higher-grade tumors that are not typically in the differential with oncocytoma. Inadvertent sampling of the liver can superficially resemble oncocytoma because hepatocytes have abundant granular cytoplasm. However, hepatocytes are usually aspirated in groups as well as singly, may contain bile, and have more variation in the size of the cells and their nuclei.
It is currently controversial whether oncocytoma should be diagnosed on FNA alone. There is no specific immunohistochemical marker for oncocytoma, and currently the diagnosis of oncocytoma on aspirate material alone is a diagnosis of exclusion. Individual cells in both clear cell and papillary tumors can be identical to those seen in oncocytoma, and chromophobe RCC can strongly resemble oncocytoma even on resection. Hybrid tumors that have features of both chromophobe RCC and oncocytoma have been described, both sporadically and in association with Birt-Hogg-Dube syndrome. In general the best diagnosis for these lesions is oncocytic neoplasm with a differential diagnosis of oncocytoma versus RCC, including chromophobe RCC. If a definitive diagnosis is mandatory, a core needle biopsy ( Fig. 4.5 ) exhibiting the nested architecture may assist in distinguishing the lesion from chromophobe RCC. However, it is often possible to satisfy the clinical needs of the patient without making a definitive diagnosis. For example, in an elderly patient with multiple comorbidities and a relatively limited life expectancy, a diagnosis of favor oncocytoma is often enough for the urologist to forgo excision and likely represents the best clinical care.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here