Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The terms nonossifying fibroma (nonosteogenic fibroma), fibrous cortical defect, and metaphyseal fibrous defect have been applied to the same metaphyseal fibrohistiocytic process. The location of the lesion in the growing portions of long tubular bones of skeletally immature patients and its frequent spontaneous resolution indicate that it represents a self-limiting process most likely related to incomplete ossification.
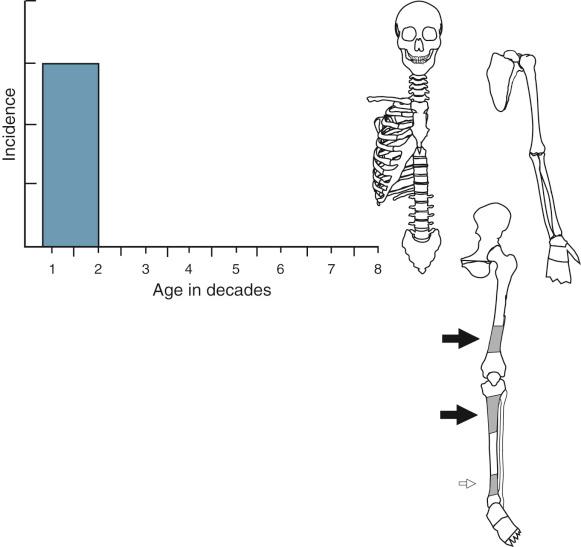
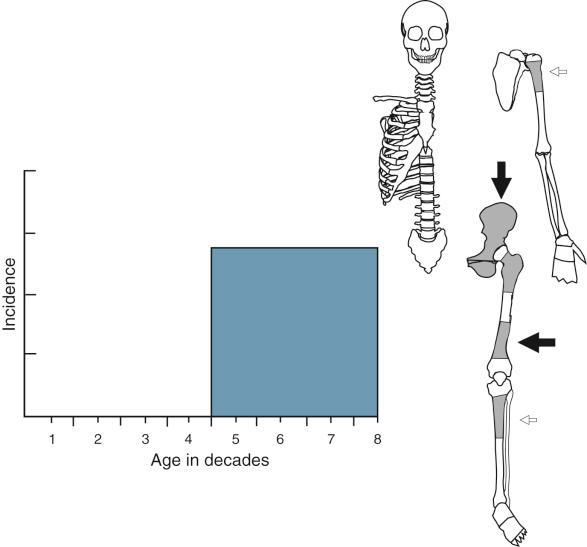
The peak age incidence and the most frequent sites of skeletal involvement are shown in Figure 9-1 . Radiographic evidence of fibrous cortical defect of long tubular bones can be found in approximately 30% of young individuals with unfused growth plates. The majority of lesions are asymptomatic and are incidental radiographic findings. A small percentage of these lesions can produce larger lytic defects that can become symptomatic. The larger ones are prone to pathologic fracture and may require prophylactic curettage. The peak incidence is in the second decade of life. The distal femoral metaphysis is the most common site. The second most frequent site is the proximal tibial metaphysis followed by the distal tibial metaphysis. Nonossifying fibroma occurs less commonly in the upper extremity, where it may be found in the proximal humerus and distal radius. Occasionally the lesions may be multifocal, simultaneously involving several symmetric metaphyseal sites, most frequently in the long bones of the lower extremity. The lesion is metaphyseal but can also involve the diaphysis. Its long axis is parallel to the long axis of the affected bone. The radiographic findings are specific, and the diagnosis can be made with certainty if the lesion is present in a typical skeletal site and appropriate age group. On this basis, such a lesion can be observed untreated. The rarity of both fibrous cortical defects and nonossifying fibroma on radiographs of adults is indirect evidence of a natural history characterized by spontaneous regression of most of these lesions over time.
Most nonossifying fibromas are asymptomatic. The lesion is usually incidentally identified on radiographs obtained for other reasons such as trauma. Larger lesions can cause pain or expansion of the affected area. In some instances, pathologic fracture can be a presenting symptom. Superimposed aneurysmal bone cyst with the development of an expansive, rapidly enlarging mass can also call attention to the lesion.
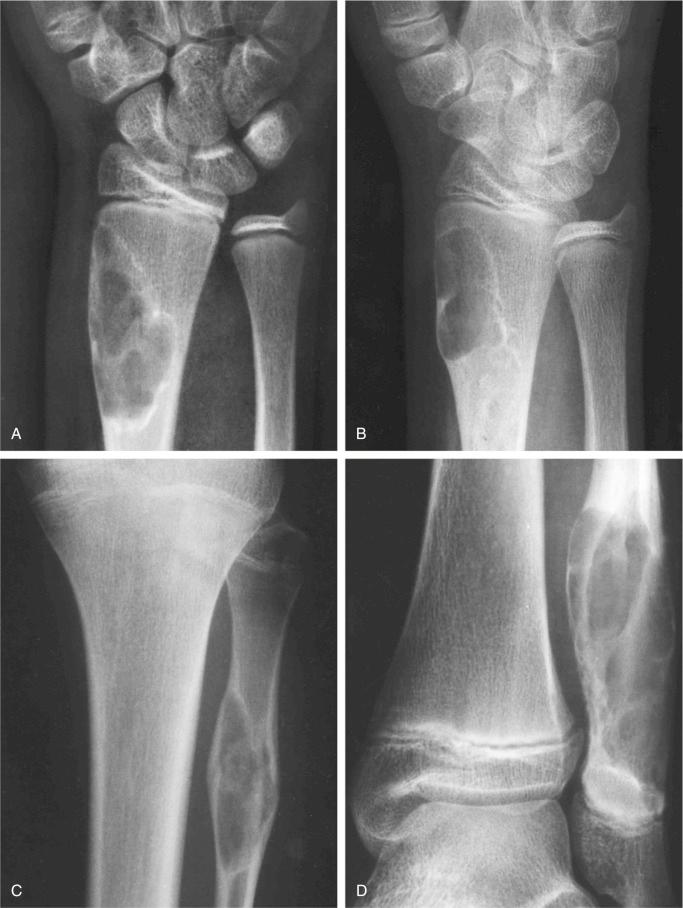
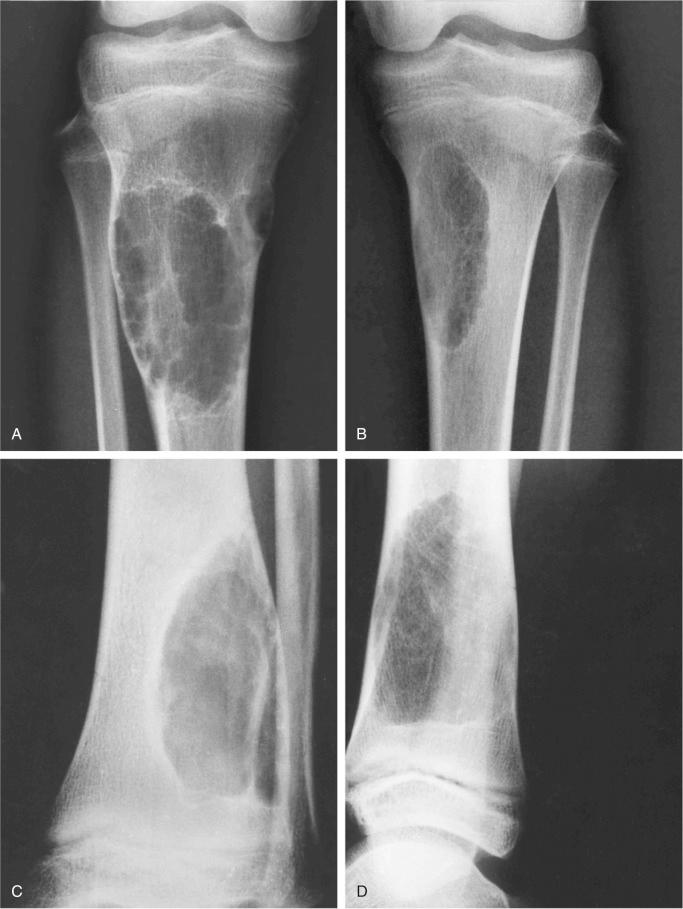
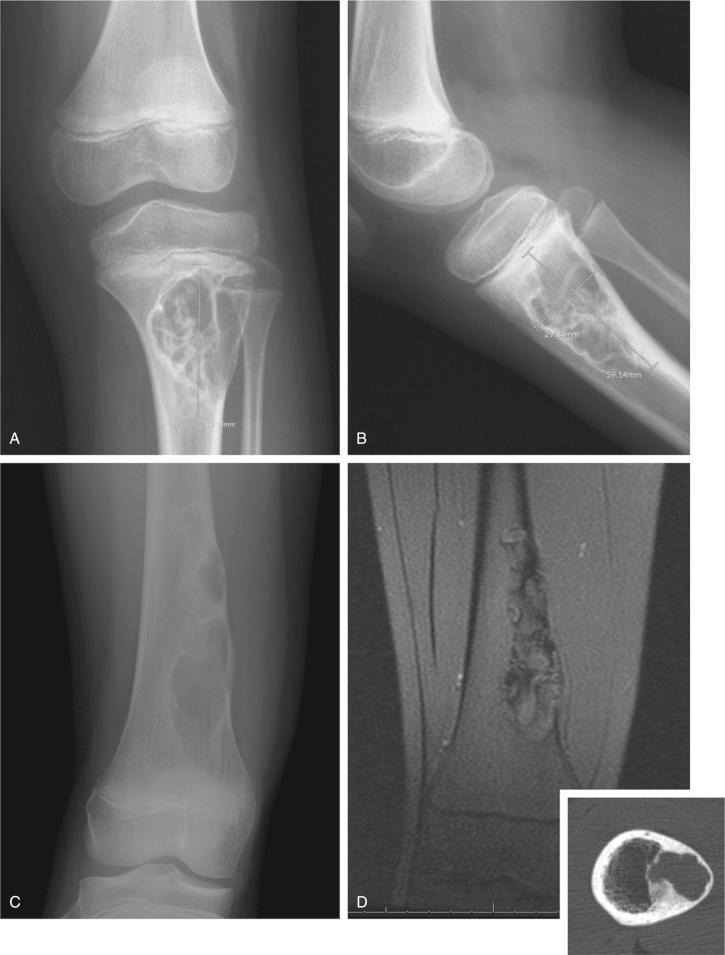
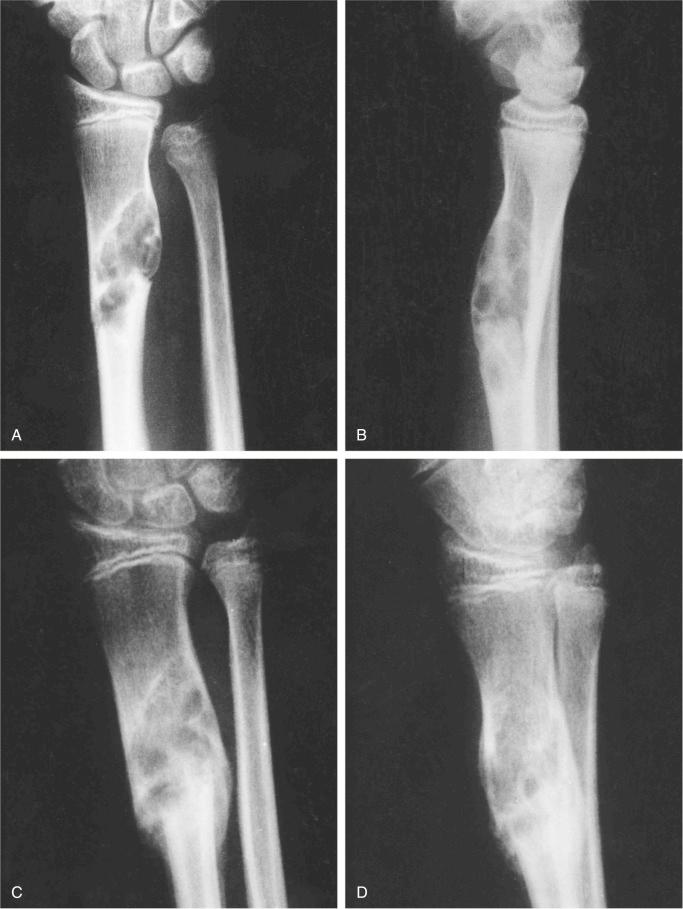
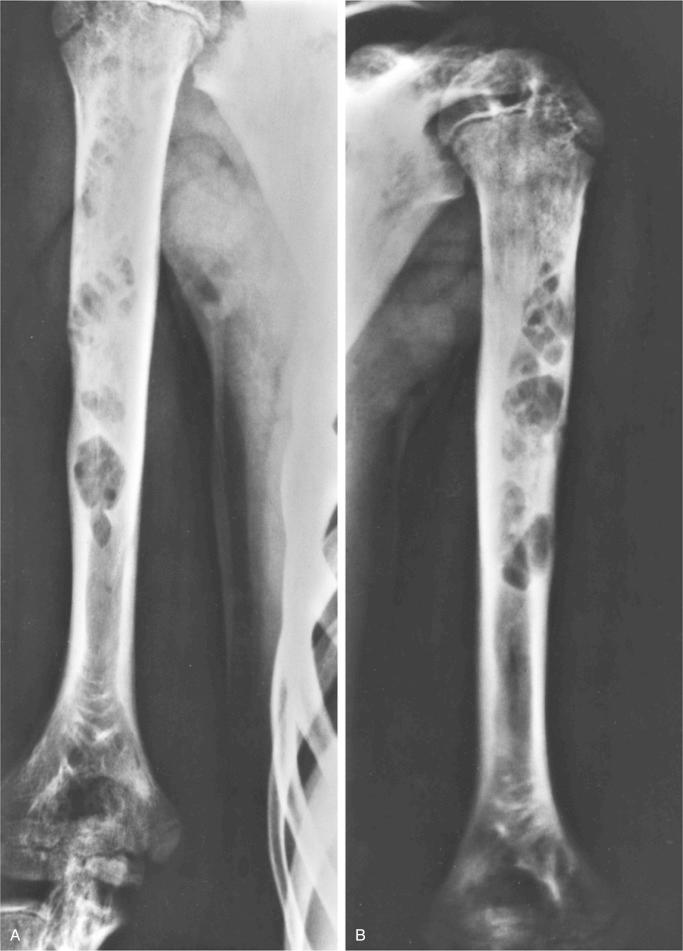
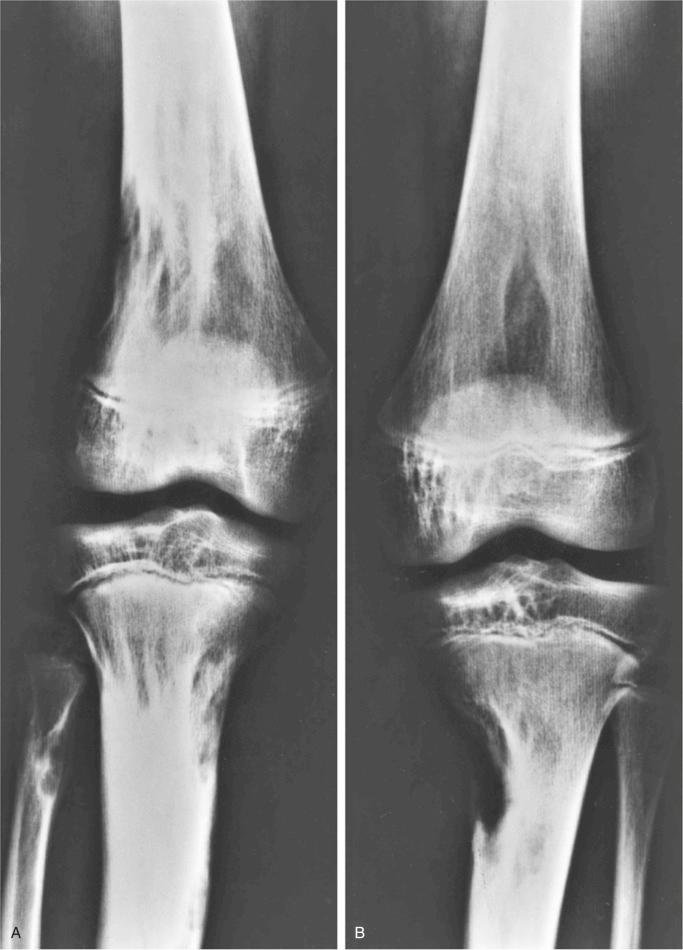
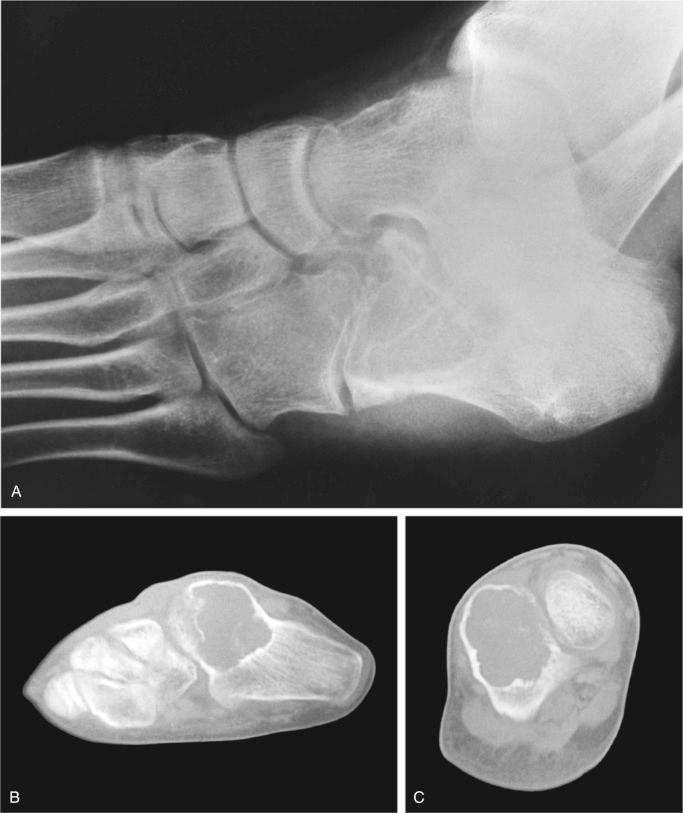
Metaphyseal fibrous defect produces an eccentric cortical-based lytic lesion with distinct and frequently scalloped, sclerotic margins ( Figs. 9-2 to 9-6 ). The cortex overlying the lesion is usually minimally to moderately expanded and markedly thinned but intact ( Figs. 9-2 to 9-5 ). There is no matrix mineralization, but in older patients evidence of bone sclerosis can accompany healing of the lesion. The distance from the growth plate usually increases with age and in older adolescents. The lesion appears to migrate in a shaftward direction as a result of skeletal growth. The lesions typically are lytic. Incomplete trabeculation is frequently seen and is accentuated in the peripheral parts of the defect ( Figs. 9-3 and 9-4 ). Older lesions may show prominent marginal or intralesional sclerosis ( Figs. 9-4 and 9-5 ). In smaller bones, such as the fibula, an eccentric location is less common, and the lesion may appear as a central, expansile trabeculated lytic defect ( Fig. 9-2 ). Pathologic fracture is a frequent presenting sign ( Fig. 9-7 ). Computed tomography (CT), magnetic resonance imaging (MRI), or both modalities can precisely document the relationship between the lesion, the medullary cavity, the cortex, and the periosteum ( Figs. 9-4 to 9-6 ). These imaging techniques show that the lesion represents an eccentric defect delineated from the medullary cavity by a rim of sclerotic bone and covered by a thinned, intact cortical bone and periosteum (see Figs. 9-4 to 9-6 ).
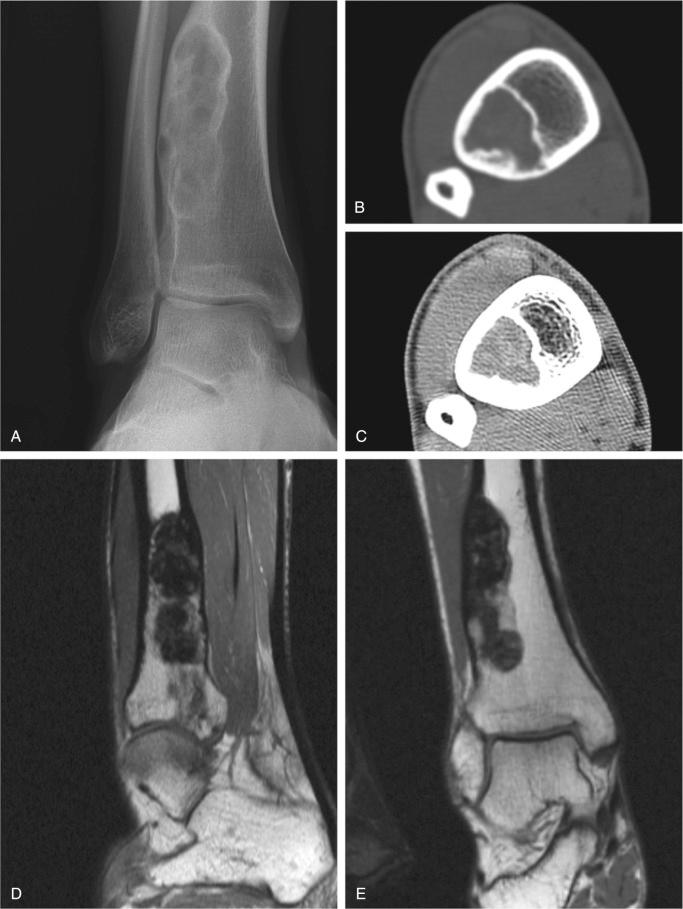
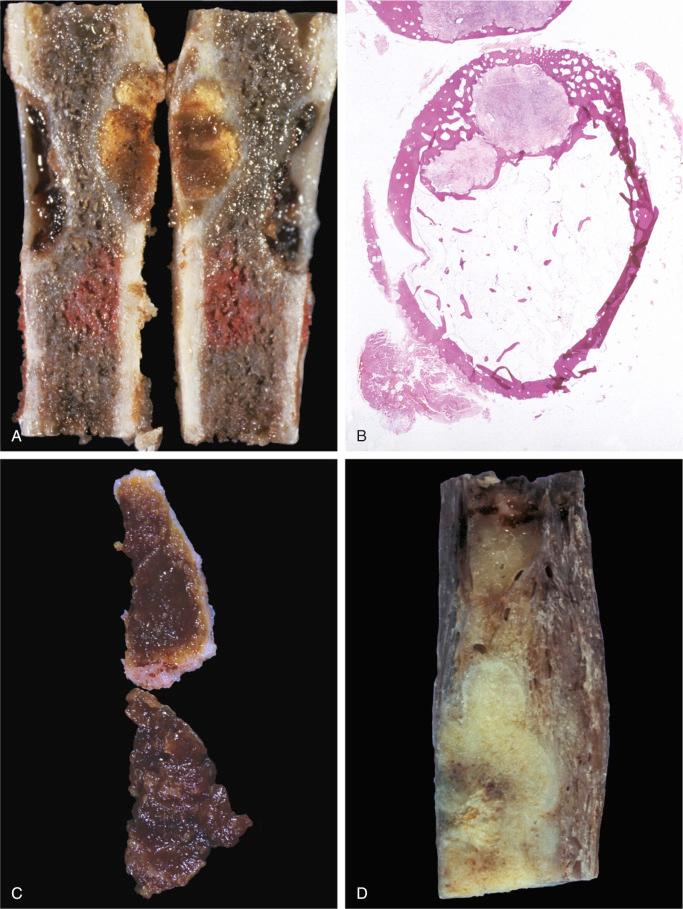
When evaluated intact in its setting, nonossifying fibroma is an eccentric and cortically based lesion with an inner boundary that is scalloped and distinctly demarcated. The cortex can be markedly thinned and expanded but intact. The tissue is fibrous, fleshy, and yellow or tan-brown ( Fig. 9-8 ). There may be evidence of cystic change, hemorrhage, or extensive necrosis, particularly in association with pathologic fracture of larger lesions.
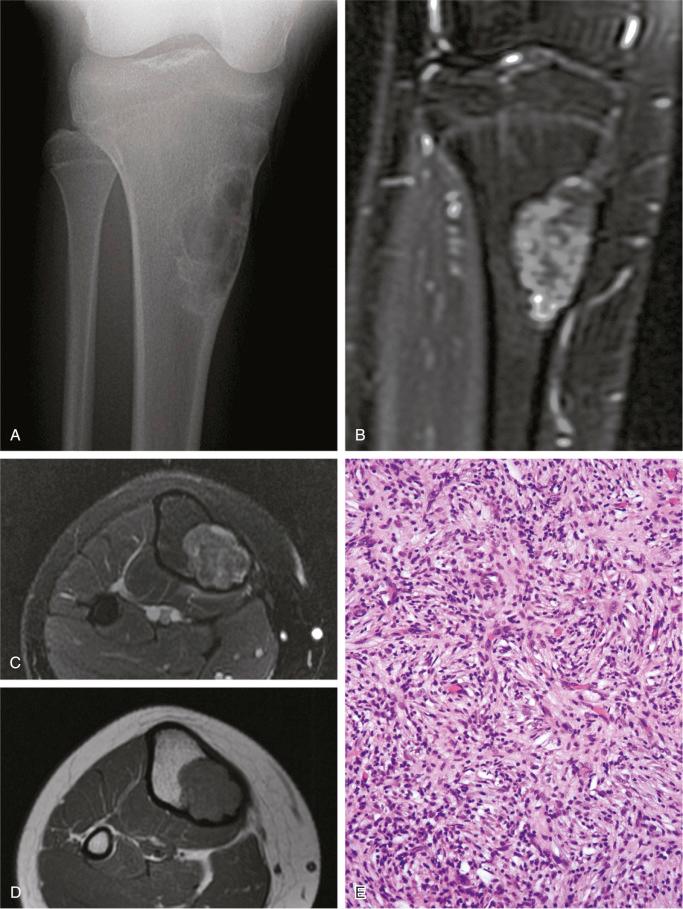
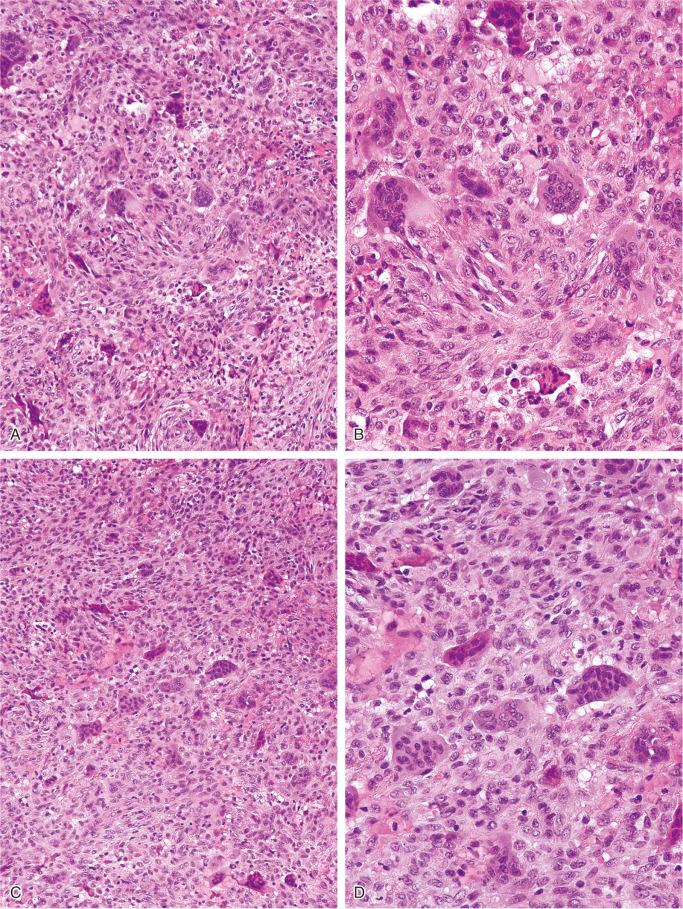
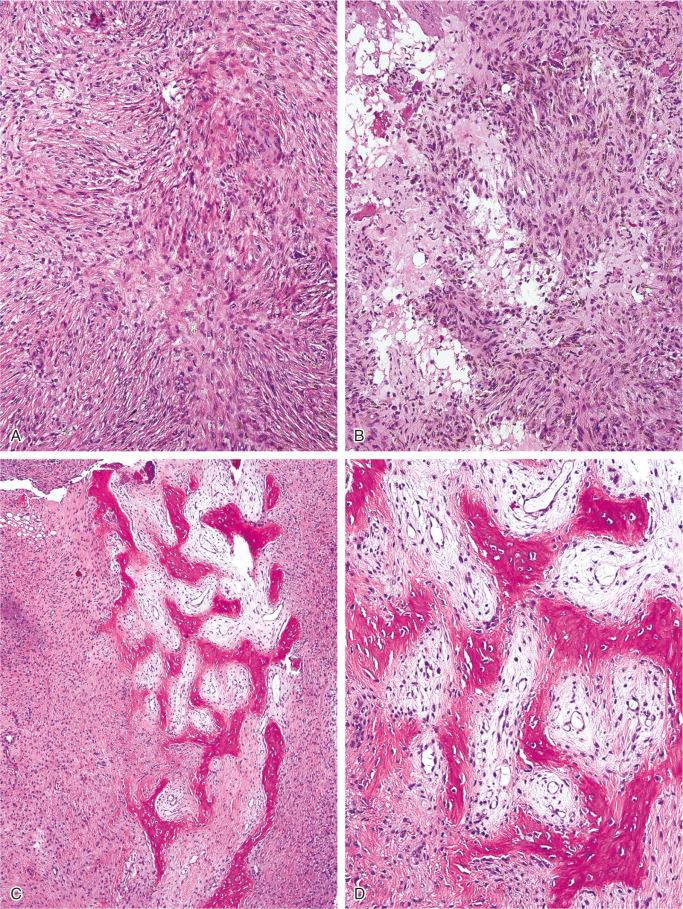
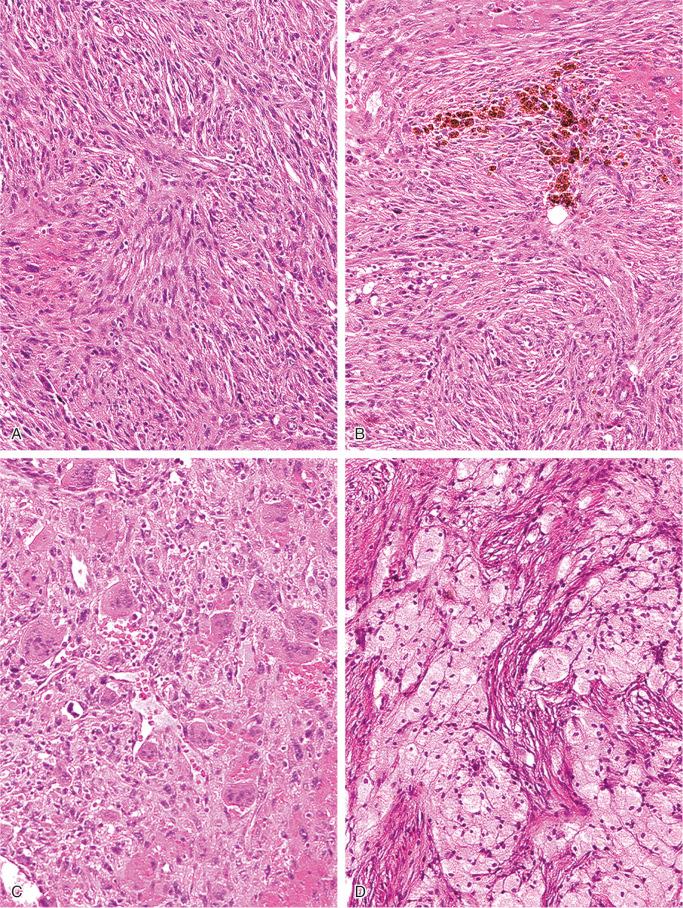
The nonossifying fibroma is composed of fibroblastic, usually highly cellular, stromal tissue with a prominent storiform pattern ( Fig. 9-9 ). In this stroma, there are a few multinucleated giant cells and foci of xanthomatous reaction with foamy histiocytes present ( Figs. 9-9 to 9-11 and 9-16 ). Usually, there is a substantial amount of hemosiderin pigment clearly visible in the stromal cells ( Fig. 9-16 ). Minimal numbers of mononuclear inflammatory cells may be present. Fresh hemorrhage and cystic change similar to that seen in aneurysmal bone cyst may be present. Secondary aneurysmal bone cyst formation can lead to rapid enlargement and a “blowout” expansion into the soft tissue. Occasionally multinucleated giant cells dominate, but they are usually focally distributed ( Fig. 9-14 ). With low power magnification, it is possible to observe the localization of prominent giant cell reaction to areas of hemorrhage. Ghosts of giant cells and patterns demonstrating the storiform architecture of the lesion may be perceived in areas of necrosis. Necrosis, as well as the presence of occasional normal mitotic figures, are not worrisome features in otherwise typical nonossifying fibroma and should not be considered as microscopic features of malignancy. Atypical mitoses are absent. Rarely nonossifying fibroma can be markedly hypercellular and is composed of plump fibrohistiocytic cells with enlarged nuclei ( Figs. 9-12 to 9-14 ). Such lesions may raise suspicion of malignancy but the true cytologic atypia and atypical mitosis are absent. In addition, correlation with radiographic features showing typical radiographic presentation of nonossifying fibroma with the absence of imaging features indicating an aggressive growth pattern, confirm the benign nature of such lesions. Focal myxoid change as well as areas of reactive bone formations can be present ( Fig. 9-15 ). In summary, the lesion has microscopic features that overlap with those of the fibrohistiocytic reactive lesions commonly seen in extraskeletal sites and with the reactive changes superimposed on various preexisting skeletal conditions. The best example of this is giant cell tumor of bone, which may contain a substantial component of reactive fibrohistiocytic changes resembling nonossifying fibroma or benign fibrous histiocytoma. As a consequence, nonossifying fibroma can be confused with many other conditions if its microscopic appearance is evaluated without clinicoradiologic correlation.
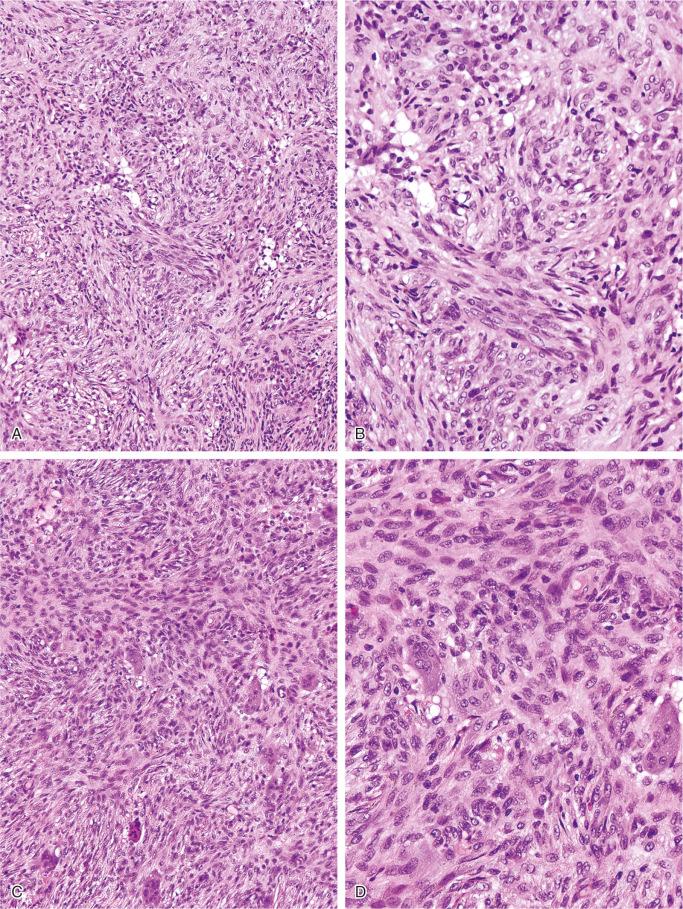
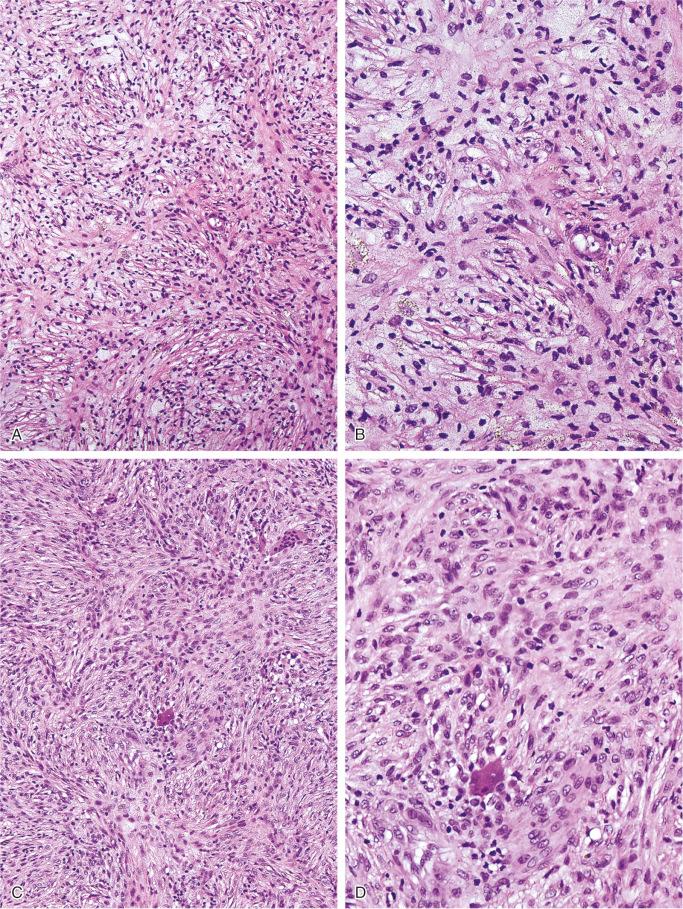
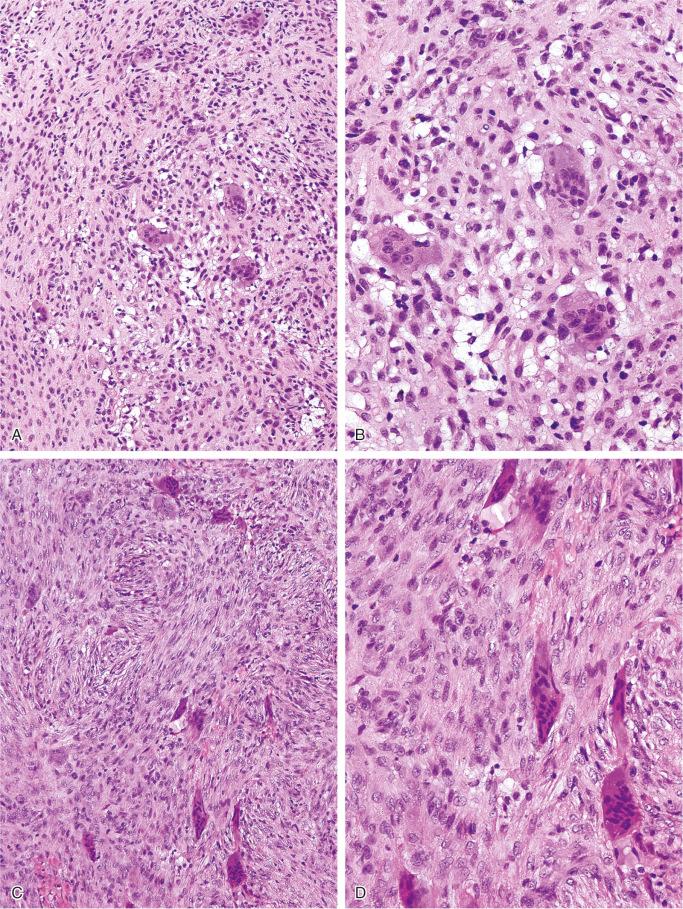
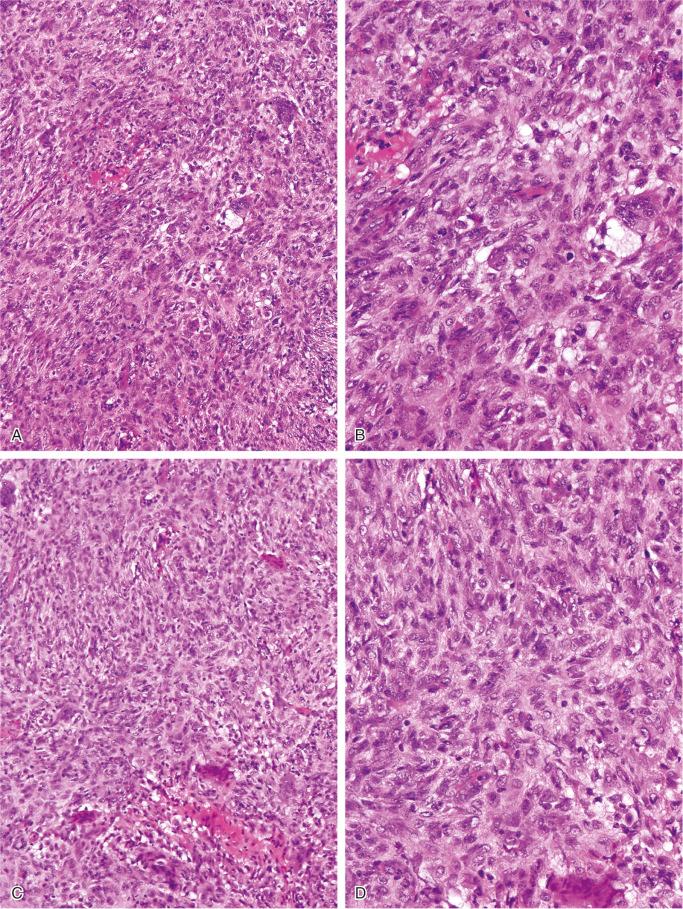
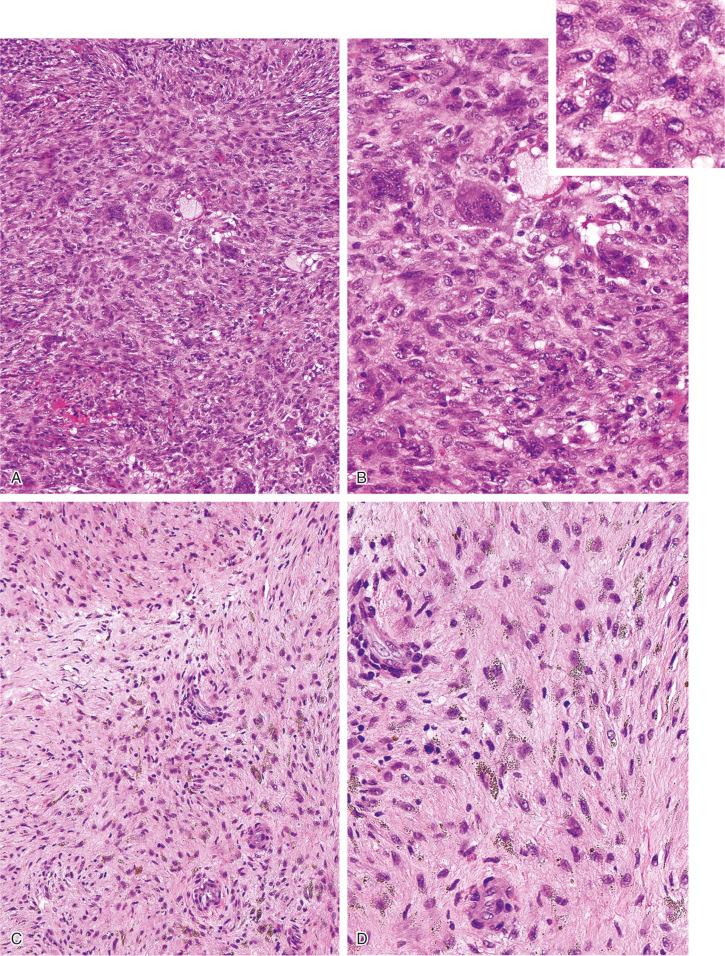
Ultrastructural studies suggest the histiocytic rather than fibroblastic origin of nonossifying fibroma. This observation is consistent with the strong positivity of nonossifying fibroma for CD68. Cytogenetically, nonossifying fibromas have a near-diploid chromosomal complement. Several clonal chromosomal aberrations, including translocation t(1;4)(p31;q34), suggest the true neoplastic nature of at least a subset of nonossifying fibromas.
The presence of multinucleated giant cells in a spindle-cell background without the formation of bone or cartilage matrix may raise the question of giant cell tumor in the microscopic differential diagnosis of nonossifying fibroma. The distinction is easily made on clinical and radiologic grounds because giant cell tumor occurs almost exclusively in adult patients at the ends of long bones, not in the metaphyses.
Fibrous dysplasia may be considered when small cortical fractures occur in nonossifying fibroma and lead to reactive bone formation. Biopsy samples from such areas show parallel arrays of immature bone trabeculae with bordering osteoblasts. Although a storiform pattern is sometimes present in fibrous dysplasia, the multinucleated giant cells seen in nonossifying fibroma are not a common feature of fibrous dysplasia. Furthermore, the radiologic eccentricity of nonossifying fibroma is not characteristic of fibrous dysplasia.
Desmoplastic fibroma may be mimicked by the more heavily collagenized areas of nonossifying fibromas undergoing involutional changes. Eccentric radiolucent lesions of the medial supracondylar side of the femur in the cortical irregularity syndrome can have overlapping features with fibrous cortical defects, but this avulsive lesion usually does not show the multinucleated giant cell elements or storiform pattern of nonossifying fibroma.
Benign fibrous histiocytoma is a term applied to lesions that are histologically identical with nonossifying fibroma but that occur in unusual locations such as the flat bones, ribs, or vertebrae.
As stated, nonossifying fibromas are frequently asymptomatic, self-healing lesions that do not require treatment. The larger lesions that expand the bone, develop secondary aneurysmal bone cysts, or pose a danger of fracture are treated by curettage and bone grafting. Most lesions stabilize at puberty and show signs of healing by peripheral sclerosis or solid opacification. In rare instances nonossifying fibroma may continue to grow and show considerable increase in size over time.
In one study of 23 cases of pathologic fractures through nonossifying fibromas, it was found that the distal tibia was most frequently involved. Lesions that occupied more than 50% of the transverse diameter of the involved bone were likely to fracture. Pathologic fractures through these lesions can be treated by cast bracing until the fracture heals; then curettage with or without bone grafting can be done.
Cases in which a malignant tumor was present independently in a bone that also contained a nonossifying fibroma have been reported. The malignancies have all been osteosarcomas.
Several rare cases of apparent malignant transformation of nonossifying fibroma have been described. In all of these cases, the original radiographic presentation of the lesion was not characteristic of nonossifying fibroma. Bone production and periosteal reaction were present in the first instance and loss of anterior cortex in the second. In the third example, the lesion involved the mandible, which is a very unlikely location for nonossifying fibroma. This raises the possibility that the original lesion in all of those instances was most likely malignant from the time of presentation. Furthermore, benign-appearing, whorled fibrous tissue and foam cells, which microscopically characterize nonossifying fibroma, can be seen focally in several bone malignancies.
Surprisingly, nonossifying fibromas are still occasionally diagnosed histologically as giant cell tumors, even though review of the clinical and radiologic features would suffice to exclude this diagnosis when considering a metaphyseal lesion in a preadolescent patient. Radiologic correlation would also serve to explain the presence of reactive bone in curetted material when a recent or old pathologic fracture is present in the thin cortex of such a lesion. Nonossifying fibromas produce eccentrically oriented lucencies, whereas a focus of fibrous dysplasia is indicative of a central intramedullary lesion. Knowledge of the latter helps prevent the less frequent error of diagnosing a nonossifying fibroma with reactive bone as fibrous dysplasia.
Multifocal nonossifying fibromas can occur in several extremely rare clinical settings. The first is familial multifocal nonossifying fibromas. Evans and Park reported three patients in the same family who had multiple nonossifying fibromas in long bones with many associated abnormalities. Patients with neurofibromatosis (von Recklinghausen's disease) have a greater likelihood of developing multifocal nonossifying fibromas in the typical long bone sites. Moser et al. found that 5% of 900 patients with multifocal nonossifying fibromas had underlying neurofibromatosis. In both of these clinical syndromes, the nonossifying fibromas are found in the usual sites (i.e., metaphyses of long bones), and they may be bilateral and symmetric.
Jaffe-Campanacci syndrome is an uncommon, congenital disorder in which multifocal nonossifying fibromas of bone are associated with café au lait pigmentation, various nonskeletal anomalies, and mental retardation. Originally, it was thought that neurofibromatosis is not a component of this syndrome. More recent observations indicate, however, that Jaffe-Campanacci syndrome may represent a unique presentation of neurofibromatosis type 1 because some of these patients may have other stigmata of neurofibromatosis, such as cutaneous neurofibromas. The nonossifying fibromas in Jaffe-Campanacci syndrome tend to be more pronounced in the metaphyseal areas of the long tubular bones, but they also form separate diaphyseal lesions. Widespread, more or less symmetric involvement of the long tubular bones of the extremities is typical in this syndrome ( Figs. 9-17 and 9-18 ). In Jaffe-Campanacci syndrome, the trunk, axial skeleton, and craniofacial bones are also typically involved. Each individual lesion has the radiographic appearance of a nonossifying fibroma, but occasionally each focus coalesces, especially in the metaphyseal parts, and forms a larger trabeculated lesion. Pathologic fracture frequently occurs in this syndrome, especially in younger patients. Similar to ordinary nonossifying fibroma, skeletal lesions in Jaffe-Campanacci syndrome have a tendency to stabilize and heal after puberty. The nonskeletal anomalies in this syndrome are hypogonadism, cryptorchidism, ocular anomalies, and cardiovascular malformations, including mitral insufficiency and aortic stenosis. In some cases, precocious puberty and kyphoscoliosis have been reported. These patients appear to have a generalized defect of mesenchymal tissue. Rarely, familial distribution has been reported. Nonossifying fibromas in Jaffe-Campanacci syndrome have microscopic features identical to those seen in ordinary nonossifying fibromas.
Benign fibrous histiocytoma of bone is a spindle-cell lesion composed of fibroblast-like cells with a storiform pattern. Foamy histiocytes and multinucleated giant cells are usually present. When xanthomatous changes dominate the histologic picture, the diagnosis of xanthoma, xanthofibroma, or fibrous xanthoma of bone may be suggested. Microscopically, it is similar to the more common benign fibrous histiocytoma of soft tissue. In bone, it is microscopically indistinguishable from nonossifying fibroma or from the prominent fibrohistiocytic reaction found in some giant cell tumors. There is controversy about whether this is a distinct benign bone neoplasm or merely a reactive lesion superimposed on a preexisting condition that cannot be easily identified.
Benign fibrous histiocytoma is a very rare entity with strict radiologic and microscopic criteria. Lesions with the clinical and radiographic features of nonossifying fibroma or giant cell tumor should be excluded from the category of benign fibrous histiocytoma. Patient age has a wide range from 5 to 75 years, and there is no clear sex predilection. The ilium and ribs are the most frequently involved sites. Individual cases have been reported in flat bones, including the craniofacial bones, vertebrae, and sacrum. Occasionally, benign fibrous histiocytoma may present as a central intramedullary diaphyseal lesion of the long tubular bones. Examples of benign fibrous histiocytoma involving the acral skeleton have also been described. The age distribution and typical sites of skeletal involvement are shown in Figure 9-19 .
The most frequent symptom is pain, ranging in duration from months to several years. Pathologic fracture is rarely a presenting symptom.
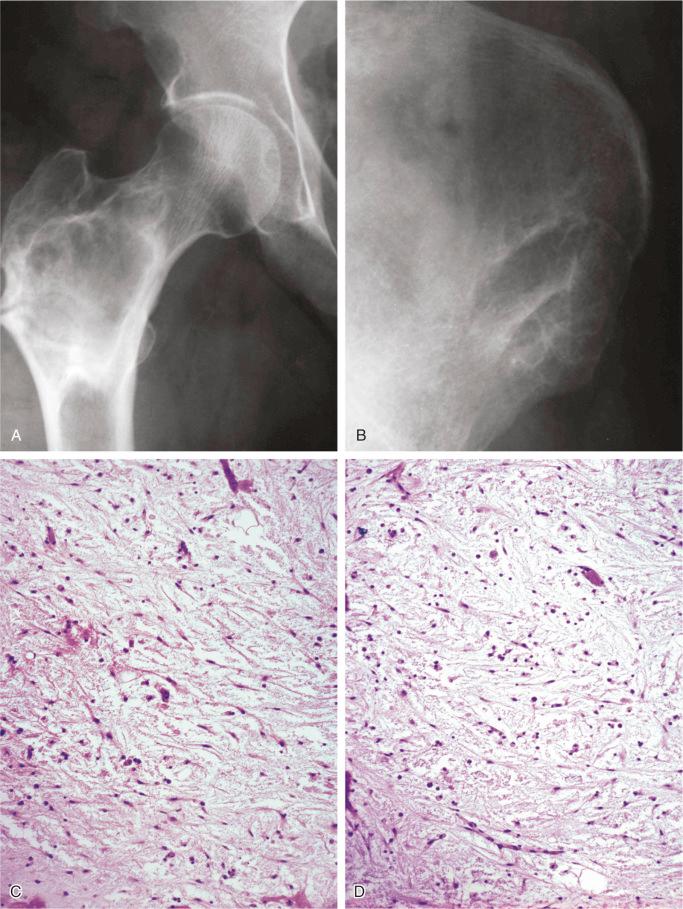
On radiographs, benign fibrous histiocytoma appears as a purely lytic, sharply demarcated lesion. A sclerotic rim and fine trabeculations may be present ( Figs. 9-20 and 9-21 ). The tumor can disrupt the cortex and expand into soft tissue, but usually it is delineated by a thin rim of newly formed bone. In the long bones, the diaphysis and metaphysis are usually involved. The reported cases located in the ends of long tubular bones are controversial because it is difficult, or perhaps even impossible, to exclude a giant cell tumor with a prominent fibrohistiocytic component.
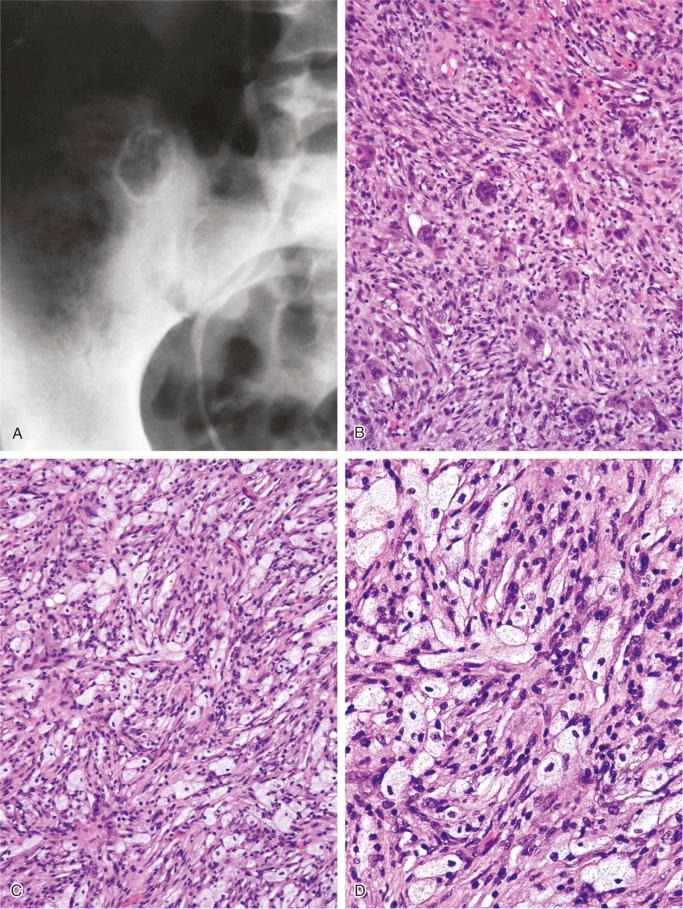
The lesion evaluated in situ represents an intramedullary, well-demarcated lesion that is tan-gray with yellow and brown areas related to the presence of lipids and hemorrhage.
The spindle-shaped, fibroblast-like cells have elongated plump nuclei and small prominent nucleoli. A storiform pattern is typically present, at least focally. Foamy, lipid-filled macrophages are interspersed among spindle cells and occasionally form larger solid areas ( Fig. 9-20 ). Multinucleated giant cells are almost always present and are sparsely distributed among the other elements. Features of fresh and old hemorrhage with hemosiderin deposits can be found. Cystic changes can frequently be seen microscopically. Occasional mitotic figures can be present, but atypical mitoses are absent. Overall, the cellularity can be high with occasional hyperchromatic nuclei, but true nuclear atypia is absent. Similar to nonossifying fibroma, ultrastructural studies confirmed the histiocytic nature of cells in benign fibrous histiocytoma, which are CD68 positive.
Benign fibrous histiocytoma is histologically indistinguishable from nonossifying fibroma, differing only in its clinical and radiographic features. In the ribs, pelvis, skull, scapula, and vertebral column, such lesions may contain large amounts of lipids and may be referred to as xanthofibromas or xanthogranulomas of bone. Indeed, such lipid-filled lesions in ribs have been mistaken for localized lesions of Erdheim-Chester disease.
Giant cell tumor of bone may be characterized by fibrohistiocytic elements with a prominent storiform pattern, abundant lipids within histiocytes, and reactive bone formation. These changes can be very extensive or only focal. Such areas sampled in small biopsy fragments may be interpreted as benign fibrous histiocytoma. The radiographic features of such cases are usually diagnostic for giant cell tumor of bone. Indeed, adequate sampling of a lytic tumor in the end of a long bone of an adult patient that shows “benign fibrous histiocytoma” features usually reveals small areas of classic giant cell tumor.
The diagnosis of benign fibrous histiocytoma of bone is made by exclusion of other entities (eosinophilic granuloma, fibrous dysplasia, giant cell tumor, and giant cell reparative granuloma) that may exhibit prominent fibrohistiocytic reactions.
Benign fibrous histiocytoma should be treated by curettage and bone grafting. Lesions that can be resected (i.e., rib, fibula) because of their anatomic site should be treated by en bloc excision. Recurrences can occur in the bone after curettage, but local aggressive behavior and particularly distant metastasis indicate that the diagnosis of benign fibrous histiocytoma was inappropriate. These cases, in our opinion, were in all probability misdiagnosed examples of giant cell tumor or low-grade malignant fibrous histiocytoma of bone.
This controversial entity is undoubtedly overdiagnosed when the designation is applied to giant cell tumors containing extensive areas of fibrohistiocytic reaction. A thorough histologic search for foci of conventional giant cell tumor usually reveals the true nature of the lesion, but sometimes all the available material is composed of fibrohistiocytic reaction. In such cases, the characteristic radiographic features of a lytic lesion at the end of a long bone in a skeletally mature individual allow the inference to be drawn that the lesion is a giant cell tumor with secondary changes. This is analogous to making a diagnosis of giant cell tumor or any other bone lesion in which secondary aneurysmal bone cyst formation is so profuse that it obscures the precursor lesion.
It is best to reserve the term benign fibrous histiocytoma for lesions that are indistinguishable from nonossifying fibroma but are located in unusual skeletal sites or present in older patients.
Fibromyxoma is a rare lesion that bears certain similarities to chondromyxoid fibroma, but the few cases described in the literature occurred in older individuals. In addition, such lesions lack the lobular architecture and the chondroid matrix that are characteristic of chondromyxoid fibroma. Radiographically, fibromyxoma appears as a lytic defect with scalloped margins ( Fig. 9-22 ). Microscopically, fibromyxoma is uniformly myxoid with low to intermediate cellularity ( Fig. 9-22 ). Some investigators believe that these lesions represent variants of chondromyxoid fibroma.
In 1979, Enzinger described a distinct tumor predominantly occurring in the subcutaneous soft tissue of children and young adults that is composed of solid nests of histiocytoid cells with multiple blood-filled cystic spaces. He proposed the term angiomatoid malignant fibrous histiocytoma. This lesion has been renamed angiomatoid fibrous histiocytoma to reflect the relative rarity of metastasis and the overall favorable prognosis. The originally suggested fibrohistiocytic origin of the lesion is questionable. We have seen five examples of this lesion arising in bone. Additional single case report of angiomatoid fibrous histiocytoma involving bone have been discussed in the literature.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here