Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Central nervous system (CNS) abnormalities are among the most common congenital anomalies. A systematic evaluation of the head and spine should be performed to include, at a minimum, the following structures listed in the practice guideline for the standard obstetrical ultrasound examination: the lateral cerebral ventricles, the choroid plexus, the midline falx, the cavum septi pellucidi, the cerebellum, the cisterna magna, the upper lip, and the cervical, thoracic, lumbar, and sacral spine.
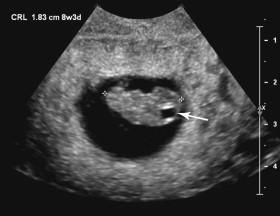
The embryonic head can be distinguished from the body by 7 to 8 weeks. Beginning at 7 to 8 weeks until approximately 10 weeks, ultrasound depicts a prominent cystic structure in the posteroinferior aspect of the head, corresponding to a cystic space in the rhombencephalon (hindbrain; Fig. 15-1 ). This cystic structure should not be mistaken for an abnormality such as anencephaly, a choroid plexus cyst, or a Dandy-Walker malformation.
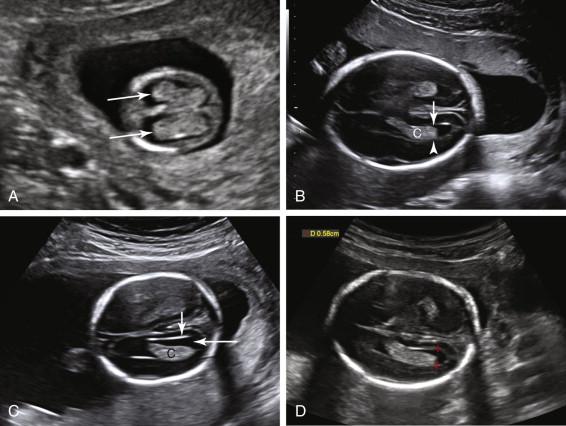
During the second and third trimesters of pregnancy, assessment of intracranial anatomy includes scans in multiple planes. Axial imaging at the level of the thalami depicts the cavum septi pellucidi as a rectangular or triangular anechoic structure between the frontal horns of the lateral ventricles, immediately anterior to the thalami ( Fig. 15-2A and B ). The cavum septi pellucidi is anechoic, without an internal midline echogenic line. Identification of a midline echogenic line indicates that the columns of the fornix, and not the cavum septi pellucidi, have been imaged (see Fig. 15-2C ). The midline echo corresponds to the interface between the columns of the fornix. Slight adjustment of the scan plane superior and anterior to the level of the columns of the fornix should demonstrate the cavum septi pellucidi when it is present (see Fig. 15-2D ). The third ventricle is depicted as a slitlike structure in the midline, between the thalami ( Fig. 15-3A ). Although images of the corpus callosum are not a required component of the standard obstetrical ultrasound examination, the corpus callosum can be visualized in a midline sagittal view of the head (see Fig. 15-3B ). It is also occasionally visualized in the coronal plane. The falx cerebri is identified in the midline as an echogenic linear structure extending between the cerebral hemispheres ( Fig. 15-4 ).
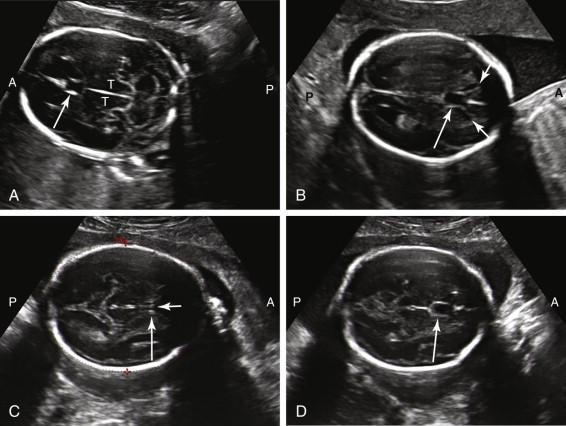
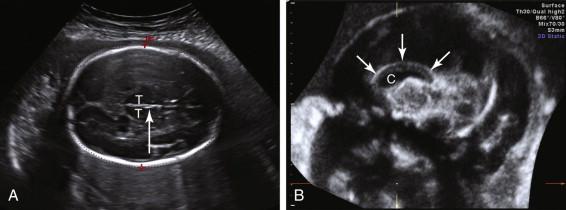
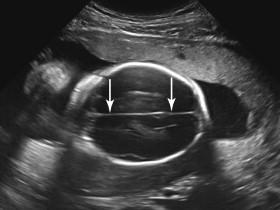
The lateral ventricles can be visualized beginning late in the first trimester by identifying the echogenic choroid plexus, which fills the majority of the ventricle at this stage of development ( Fig. 15-5A ). The ventricles remain relatively constant in size throughout gestation, with head growth accounted for by enlargement of the brain parenchyma surrounding the ventricles. In normal fetuses the choroid plexus should fill more than 60% of the width of the atria of the lateral ventricles (see Fig. 15-5B and C ). The lateral ventricles should be measured on an axial image of the atrium, near the posterior aspect of the choroid plexus (see Fig. 15-5D ). When possible, the measurement should be obtained on an image that demonstrates a substantial length of choroid plexus superior to the level of the thalami. To obtain this view it may be necessary to angle the posterior portion of the scan plane inferiorly, particularly during the third trimester. The generally accepted upper limit of normal for the width of the atrium of the lateral ventricle is 10 mm.
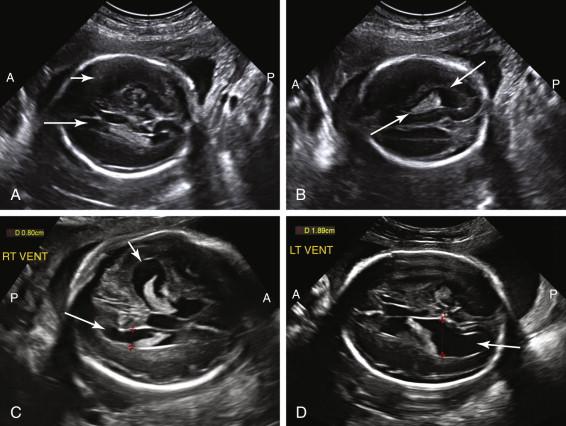
Because of near-field reverberation artifact from the calvarium, frequently only the lateral ventricle in the far field is identified in the true axial conventional scan plane for measurement ( Fig. 15-6A ). The difficulty of imaging the near-field ventricle can be mitigated by adjusting the transducer to image the near-field ventricle in an oblique scan plane (see Fig. 15-6B ). Although both lateral ventricles are similar in size in most fetuses, occasionally ventricular enlargement is asymmetric or unilateral. Consequently, visualization of one normal-size lateral ventricle does not ensure the contralateral ventricle has normal dimensions. If the far-field ventricle is enlarged or if an oblique view of the near-field ventricle suggests that it is dilated, measurements of the near-field ventricle should, when possible, be obtained after the fetus has turned and the ventricle initially in the near field shifts to the far field (see Fig. 15-6C and D ).
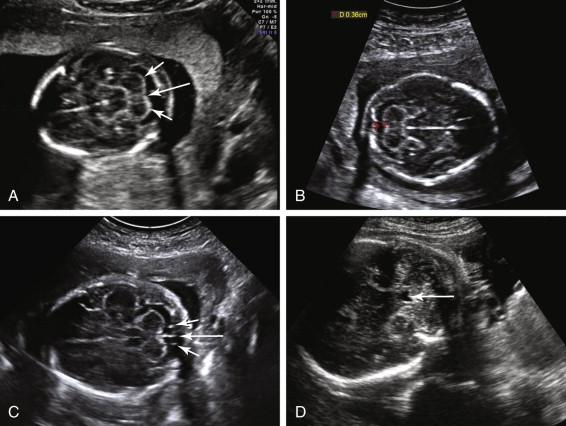
The posterior fossa is routinely imaged in an axial view, at the level of the thalami or midbrain, with the transducer tilted inferiorly toward the occiput. The cerebellum is bilobed, identified by two rounded hypoechoic hemispheres laterally and the vermis in the midline ( Fig. 15-7A ). The cisterna magna is a fluid-containing space between the cerebellum and the occiput. The cisterna magna is measured from the posterior margin of the cerebellar vermis to the inside of the occipital bone. The normal anteroposterior (AP) diameter of the cisterna magna is between 2 and 10 mm (see Fig. 15-7B ). The cisterna magna contains anechoic cerebrospinal fluid and linear, hyperechoic septa extending from the midline (termed falx cerebelli ) and from the junction between the cerebellar vermis and the cerebellar hemisphere on each side of the occipital bone (see Fig. 15-7C ). The fourth ventricle can be seen in the anterior portion of the posterior fossa in the majority of normal fetuses during the second and third trimesters if specifically sought (see Fig. 15-7D ). The normal fourth ventricle increases in size with gestational age, in both AP and width dimensions, measuring up to 8 mm in width by term. Its shape can be slitlike, triangular, or ovoid or resemble a boomerang.
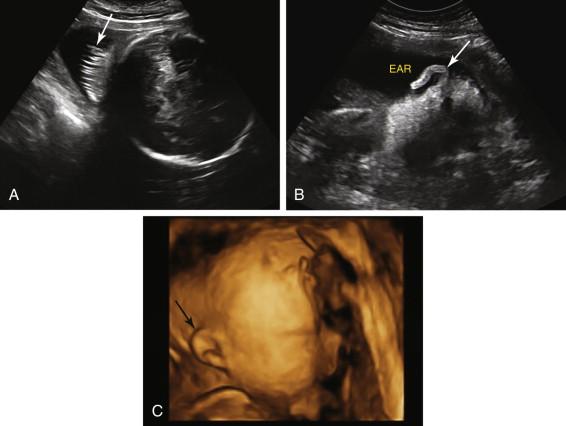
Normal structures adjacent to the calvarium include subcutaneous tissue, skin, hair, and ears, and should not be confused with pathologic conditions ( Fig. 15-8 ).
Ventriculomegaly refers to enlargement of the lateral ventricles. Dilatation of the third and fourth ventricles in the setting of dilated lateral ventricles is variable and depends on the etiology and severity of the ventriculomegaly. Hydrocephalus refers to ventriculomegaly secondary to increased intraventricular pressure. Ventriculomegaly also occurs in the absence of increased pressure due to overproduction of cerebrospinal fluid, developmental abnormalities, and destructive processes such as vascular and infectious insults.
When the lateral ventricle is dilated, the posterior portion of the choroid plexus droops toward the gravity-dependent wall of the ventricle, resulting in an appearance termed the dangling choroid plexus ( Fig. 15-9A ). This occurs because the anterior portion of the choroid plexus is tethered as it courses toward the third ventricle at the foramen of Monro, but the posterior portion is free floating and gravity dependent. When the lateral ventricle is enlarged, the choroid plexus is often diminutive in size (see Fig. 15-9B ).
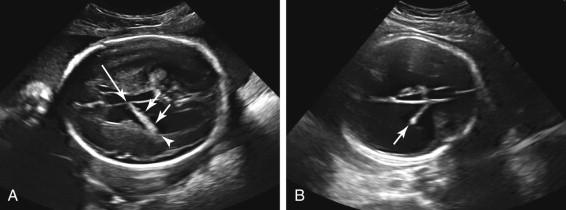
The upper limit of normal for the width of the lateral ventricle is 10 mm. Measurements between 10 and 12 mm are typically considered as mild ventriculomegaly, between 12 and 15 mm as moderate ventriculomegaly, and greater than 15 mm as severe ventriculomegaly ( Fig. 15-10 ). Enlargement of the fetal head occurs in some fetuses with severe ventriculomegaly, due to increased intraventricular pressure, and is a sign of hydrocephalus. Hydrocephalus can also be diagnosed if a mass causing ventricular obstruction is directly visualized. Ultrasound depicts a dilated third ventricle as a rounded or triangular fluid collection exceeding 3 mm in diameter between the thalami ( Fig. 15-11 ).
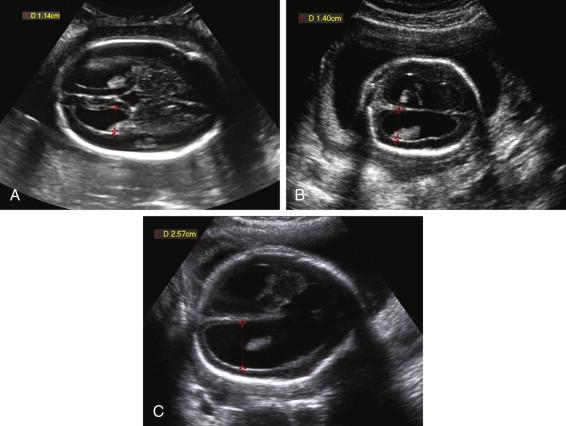
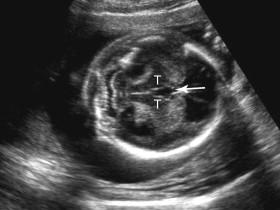
The range of fetal outcomes in the setting of ventriculomegaly is wide, with outcome depending on both the severity and underlying etiology of the dilatation. Even in the setting of mild ventriculomegaly, a detailed anatomic scan and follow-up sonograms should be performed to assess for additional fetal abnormalities and progressive ventricular enlargement. Fetuses with mild ventriculomegaly are at increased risk of aneuploidy, and fetal karyotyping is typically offered in this setting ( Fig. 15-12 ). Isolated mild ventriculomegaly is often associated with a normal outcome but can also be a harbinger of less favorable outcomes such as progressive dilatation on follow-up examinations or postnatal developmental delay.
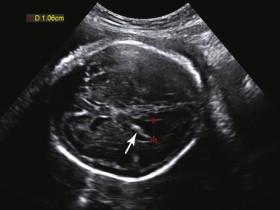
Ventriculomegaly is a nonspecific finding seen in the setting of both CNS and extra-CNS disorders. Common CNS etiologies of ventriculomegaly include aqueductal stenosis, Chiari II malformation, Dandy-Walker abnormality, agenesis of the corpus callosum (AGCC), cephalocele, infection, intracranial hemorrhage, and vein of Galen malformation ( Box 15-1 ).
Agenesis of the corpus callosum
Aqueductal stenosis
Cephalocele
Chiari II malformation
Dandy-Walker abnormality
Infection
Intracranial hemorrhage
Vein of Galen malformation
CNS, Central nervous system.
Aqueductal stenosis occurs when there is obstruction of the aqueduct of Sylvius, the narrow channel connecting the third and fourth ventricles. It is a common cause of hydrocephalus, and is characterized by dilatation of the lateral and third ventricles without a dilated fourth ventricle ( Fig. 15-13 ). Ventriculomegaly may be marked and progressive, and is sometimes a late finding, first seen in the third trimester after visualization of normal-size ventricles on earlier sonograms. Aqueductal stenosis is a diagnosis of exclusion that only can be made after the other aforementioned etiologies of ventriculomegaly have been ruled out. A minority of cases are X-linked recessive and this form, only seen in male fetuses, can be associated with adducted flexed thumbs.
Severe ventriculomegaly can cause such marked dilatation that the ventricles lose their normal configuration and occupy most of the volume of the brain. As a result, it can be difficult to distinguish markedly dilated ventricles from other entities that cause large intracranial fluid collections such as hydranencephaly and alobar holoprosencephaly, both of which are discussed in the following sections.
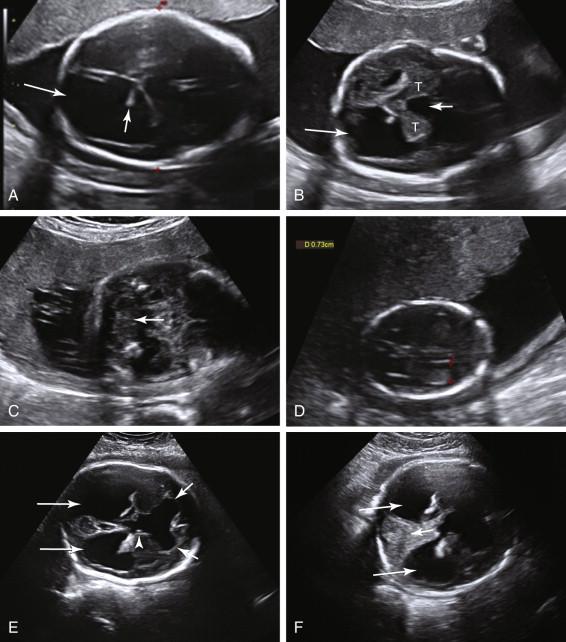
Hydranencephaly is a rare condition in which the cerebral hemispheres are replaced by a large fluid-filled cavity, secondary to occlusion of the middle cerebral or internal carotid arteries. Hydranencephaly is a lethal condition. The vast majority of affected babies die in utero or within the first year of life. Underlying disorders include vascular accidents and other ischemic events, infections, toxins, and death of one fetus in a monochorionic multiple gestation pregnancy. The distribution of brain destruction often follows the distribution of the affected artery, resulting in variable sparing of parts of the frontal, occipital, and temporal lobes as well as the posterior fossa, thalami, and brain-stem structures because the posterior circulation is not affected. Sonography reveals a large fluid-filled intracranial cavity superiorly, with preservation of the thalami and brain stem at the base of the cavity ( Fig. 15-14A ). A portion of the falx cerebri is often seen. Head size is variable and ranges from small to enlarged. The main differential diagnosis includes severe hydrocephalus, alobar holoprosencephaly, and severe schizencephaly. The presence of a thin rim of preserved cortical tissue peripherally distinguishes severe hydrocephalus from hydranencephaly (see Fig. 15-14B ). Identification of the falx cerebri or thalami that are not fused distinguishes hydranencephaly from alobar holoprosencephaly. Schizencephaly causes clefts in the brain. In some cases it may not be possible to distinguish between these entities on an ultrasound, and in such cases magnetic resonance imaging (MRI) may be helpful.
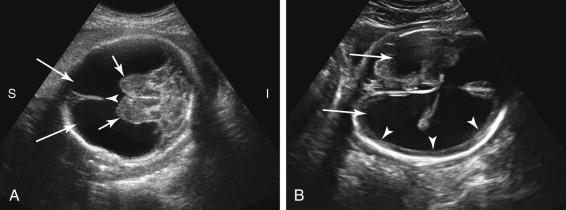
Hydranencephaly is often considered the most severe form of porencephaly. Porencephaly refers to the presence of one or more cystic cavities in the brain ( Fig. 15-15 ). Similar to hydranencephaly, porencephaly is likely vascular in etiology. The cystic area frequently communicates with the adjacent lateral ventricle. The porencephalic cyst may enlarge with advancing gestation, cause mass effect on brain structures, or protrude through the suture lines to extend outside the calvarium. The differential diagnosis for a porencephalic cyst includes schizencephaly and an arachnoid cyst.
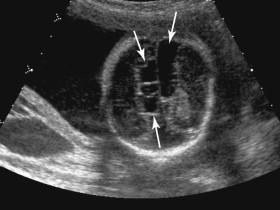
Schizencephaly is a rare disorder in which a brain defect lined by gray matter extends between the lateral ventricle and the cerebral cortex to communicate with the subarachnoid space. Open-lip schizencephaly is characterized by separation of the walls of the cleft by cerebrospinal fluid, facilitating identification at antenatal ultrasound. Ultrasound depicts a fluid-filled cleft in the brain parenchyma extending laterally from the lateral ventricle to the brain surface ( Fig. 15-16A ). Closed-lip schizencephaly occurs when the walls of the cleft are in contact with each other and consequently is difficult to diagnose on ultrasound. Schizencephaly can be unilateral or bilateral (see Fig. 15-16B ). Severe cases of schizencephaly can be difficult to distinguish from severe hydrocephalus and hydranencephaly.
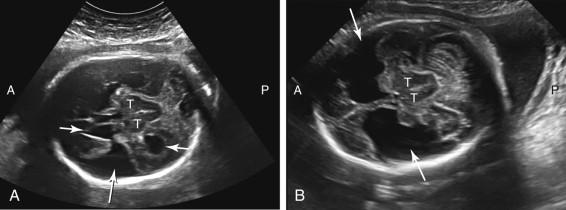
An arachnoid cyst is a fluid collection between the cerebral meninges that does not communicate with the lateral ventricle. It can be found in numerous locations along the brain such as in the cerebral fissures, posterior fossa, and suprasellar region and over the cerebral convexity ( Fig. 15-17 ). A very large arachnoid cyst can compress brain parenchyma and cause hydrocephalus. It may not be possible to distinguish an arachnoid cyst definitively from other cystic processes in a similar location.
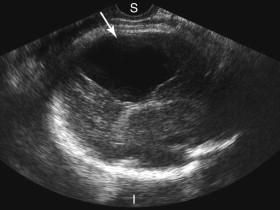
A choroid plexus cyst is the most common cystic brain lesion identified at antenatal sonography ( Fig. 15-18 ). It is associated with an increased risk of trisomy 18. If, however, the fetus has a normal karyotype, the choroid plexus cyst is not associated with adverse sequelae. Choroid plexus cysts are discussed in greater detail in Chapter 22 .
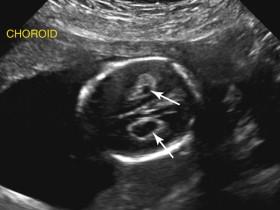
Holoprosencephaly includes a spectrum of abnormalities of the midline brain and facial structures. It occurs due to incomplete cleavage of brain parenchyma during embryologic development of the forebrain. Holoprosencephaly is frequently associated with chromosomal abnormalities. The most commonly associated aneuploidy is trisomy 13; the next most frequent is triploidy. Holoprosencephaly is classified into three main types: alobar, semilobar, and lobar holoprosencephaly (from the most to the least severe).
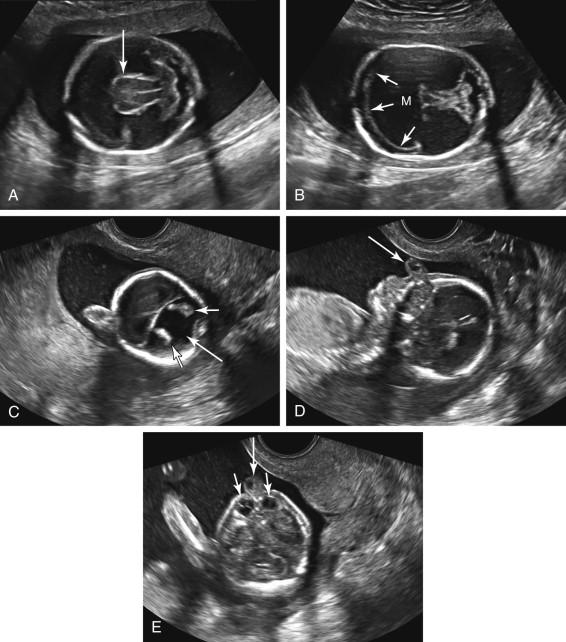
Alobar holoprosencephaly is characterized by fused thalami, a single monoventricular cavity, absence of midline structures such as the falx cerebri and cavum septi pellucidi, and facial abnormalities ( Fig. 15-19 ). Brain tissue surrounds the monoventricular cavity, forming an anterior wedge of tissue resembling a boomerang. The monoventricular cavity may communicate with a dorsal cyst, replacing the usual configuration of the ventricular system. A wide range of midline facial abnormalities such as cyclopia, proboscis, hypotelorism, and cleft lip/palate are seen ( ![]() and
and ![]() ; e-Figs. 15-1 and 15-2
; e-Figs. 15-1 and 15-2 ![]() ). Facial anomalies tend to be more severe when the brain abnormality is severe. The overall head size can be normal, macrocephalic, or microcephalic.
). Facial anomalies tend to be more severe when the brain abnormality is severe. The overall head size can be normal, macrocephalic, or microcephalic.
![e -F igure 15-1, Profile ( e-Fig. 15-1 ) and axial views ( e-Fig. 15-2 ) of fetal face show a proboscis (long arrows) and severe hypotelorism with no significant space between the orbits ( short arrows [ e-Fig. 15-2 ]). See also Videos 15-1 and 15-2 . e -F igure 15-1, Profile ( e-Fig. 15-1 ) and axial views ( e-Fig. 15-2 ) of fetal face show a proboscis (long arrows) and severe hypotelorism with no significant space between the orbits ( short arrows [ e-Fig. 15-2 ]). See also Videos 15-1 and 15-2 .](https://storage.googleapis.com/dl.dentistrykey.com/clinical/FetalCentralNervousSystemFaceandNeck/19_3s20B9780323086189000158.jpg)
![e -F igure 15-2, Profile ( e-Fig. 15-1 ) and axial views ( e-Fig. 15-2 ) of fetal face show a proboscis (long arrows) and severe hypotelorism with no significant space between the orbits ( short arrows [ e-Fig. 15-2 ]). See also Videos 15-1 and 15-2 . e -F igure 15-2, Profile ( e-Fig. 15-1 ) and axial views ( e-Fig. 15-2 ) of fetal face show a proboscis (long arrows) and severe hypotelorism with no significant space between the orbits ( short arrows [ e-Fig. 15-2 ]). See also Videos 15-1 and 15-2 .](https://storage.googleapis.com/dl.dentistrykey.com/clinical/FetalCentralNervousSystemFaceandNeck/20_3s20B9780323086189000158.jpg)
Semilobar holoprosencephaly is of intermediate severity and consists of at least partial separation of the lateral ventricles posteriorly with a monoventricular cavity anteriorly. The falx cerebri is absent. As in the alobar form, the thalami are fused and facial anomalies are common. The least severe form, lobar holoprosencephaly, is characterized by two discrete cerebral hemispheres, absence of the cavum septi pellucidi, and fused frontal horns, often with a squared configuration. The head is typically normal in size. The lobar form is the most difficult to diagnose at antenatal sonography because findings such as nonvisualization of the cavum septi pellucidi are relatively subtle and resemble other brain disorders including septo-optic dysplasia and agenesis of the corpus callosum (AGCC).
AGCC can be partial or complete. Ventriculomegaly is common in complete AGCC. The ventricular dilatation may dominate the sonographic picture and be the only finding recognized antenatally in some fetuses, with the specific diagnosis of AGCC not appreciated antenatally. It is important to distinguish AGCC, which is a nonobstructive form of ventriculomegaly, from obstructive hydrocephalus, because the nonobstructive ventriculomegaly occurring in AGCC does not improve with ventricular shunting.
Ultrasound features that suggest the diagnosis of AGCC include dilated ventricles and failure to identify the cavum septi pellucidi ( Fig. 15-20 ). Because documentation of the cavum septi pellucidi is a required component of the basic fetal anatomy scan, nonvisualization is often an early clue to the diagnosis. Colpocephaly (disproportionate enlargement of the posterior portion of the lateral ventricles) is a common finding and can result in a teardrop configuration to the lateral ventricles. Additional ultrasound features include enlargement of the third ventricle and extension of the third ventricle above the level of the thalami due to absence of the callosal tract superior to it. The third ventricle may herniate between the cerebral hemispheres, resulting in a midline interhemispheric cyst. The lateral ventricles tend to be located farther laterally and oriented more parallel to each other than usual. The frontal horns may exhibit a comma-shaped concavity medially instead of the typical lateral concavity, and be spread further apart due to prominent longitudinal Probst bundles. Because the cavum septi pellucidi is absent, a radiating pattern of gyri can sometimes be seen extending peripherally from the interhemispheric fissure in the sagittal scan plane.
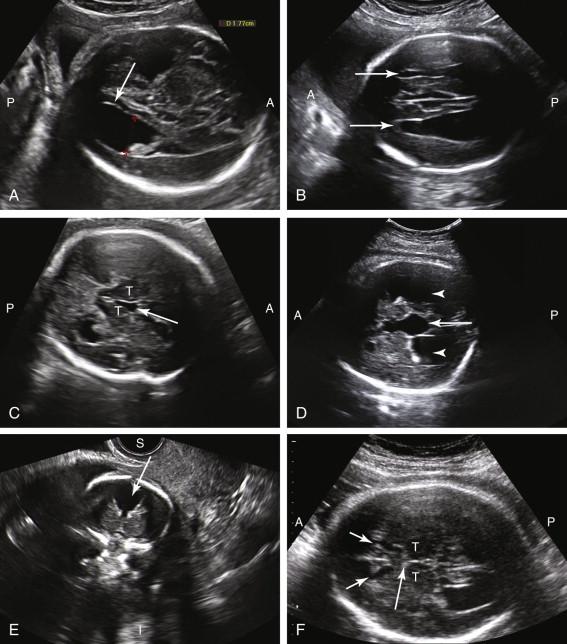
The partial form of AGCC is characterized by absence of the posterior portion of the corpus callosum. The anterior portion is intact because embryologic development of the corpus callosum starts anteriorly and progresses posteriorly. Because the cavum septi pellucidi and the anterior component of the corpus callosum are present, partial AGCC can be difficult to diagnose at antenatal sonography.
AGCC can be an isolated anomaly or can be seen in association with chromosomal abnormalities, additional CNS defects (e.g., Dandy-Walker malformation, holoprosencephaly, and Arnold-Chiari II malformation), and a wide variety of congenital syndromes. Transvaginal ultrasound evaluation (if the head is in vertex presentation), three-dimensional (3D) ultrasound reconstruction, and/or an MRI may prove of value in directly imaging the region of the corpus callosum and assessing for additional brain abnormalities.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here