Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Normal embryonic development in vertebrates requires the highly coordinated action of thyroid hormones. In mammals, these iodothyronine hormones (T3 and T4) are derived from both maternal and fetal sources and regulate the proliferation, differentiation, and apoptosis of developing tissues in a temporally and anatomically precise manner. During most of gestation, extracellular concentrations of active thyroid hormones in the fetus are low (as compared with maternal serum) and the local action of thyroid hormones is regulated by the dynamic expression of cellular thyroid hormone transporters, metabolizing enzymes, and nuclear receptors. During the second trimester, concentrations of thyroid hormone in fetal serum rise, and after delivery the infant begins transitioning to an adult pattern of thyroid hormone metabolism over the ensuing 2 to 4 weeks. This transition may be altered by the gestational age at the time of delivery, illness, exogenous medications administered to the mother or infant, abnormal development of the hypothalamic-pituitary-thyroid (HPT) axis, or disorders of thyroid hormone production and/or metabolism.
Progress in human genetics has helped define many of the key mechanisms required for normal thyroid development, migration, and function and several of the transcription factors critical for thyroid gland development also play an important role in non-thyroid organogenesis ( Table 145.1 ). , These patients have expanded our understanding of thyroid hormone physiology and underscore the role that local thyroid hormone signaling plays in fetal and neonatal development.
| Gene (OMIM) | Name | Thyroid Phenotype | Additional Features |
|---|---|---|---|
| NKX2-1 ( 600635 ) | NK2 homeobox 1 or thyroid transcription factor 1 | Thyroid hypoplasia, hemiagenesis, or athyreosis | Brain-lung-thyroid syndrome; benign hereditary chorea, infant respiratory distress syndrome (surfactant deficiency) |
| PAX8 ( 167415 ) | Paired box 8 | Thyroid hypoplasia, ectopy, or athyreosis | Urogenital tract abnormalities |
| FOXE1 ( 602617 ) | Forkhead box protein E1 or thyroid transcription factor 2 | Thyroid hypoplasia or athyreosis | Bamforth-Lazarus syndrome; cleft palate, choanal atresia, bifid epiglottis, and spiky hair |
| TSHR ( 603372 ) | Thyroid-stimulating hormone receptor | Thyroid hypoplasia | None |
| HHEX ( 604420 ) | Hematopoietically expressed homeobox | Thyroid hypoplasia and ectopy | None |
The primordial thyroid begins as an involution of the foregut on the midline of the embryonic mouth referred to as the median anlage. In humans, this anlage appears at embryonic day 20 to 22 with caudal migration through the thyroglossal duct to its mature location at the base of the neck starting at embryonic day 30 to 40 ( Fig. 145.1 ). By embryonic day 50, migration is complete and the thyroglossal duct has involuted. Notably, thyroid follicular cells (the cell type that biosynthesizes thyroid hormones) arise from this median anlage. The second secretory cell type found within the mature thyroid gland, the calcitonin-secreting para follicular or C cells, are believed to arise from the lateral anlages of the ultimobranchial bodies, a pair of embryonic structures that migrate from the fourth pharyngeal pouches and fuse into the definitive thyroid gland around embryonic day 60. Of note, more recent studies have raised the possibility that the C cells may arise in the endoderm germ layer. Over this time, the embryonic thyroid gland begins to express thyroid-specific genes required for T4 and T3 biosynthesis. The mature anatomy of the thyroid gland is a bilobed structure with two lateral lobes connected by the isthmus. The pyramidal lobe of the thyroid gland is a normal anatomic variant found in up to 65% of patients that extends superiorly from the isthmus or lateral lobe. ,
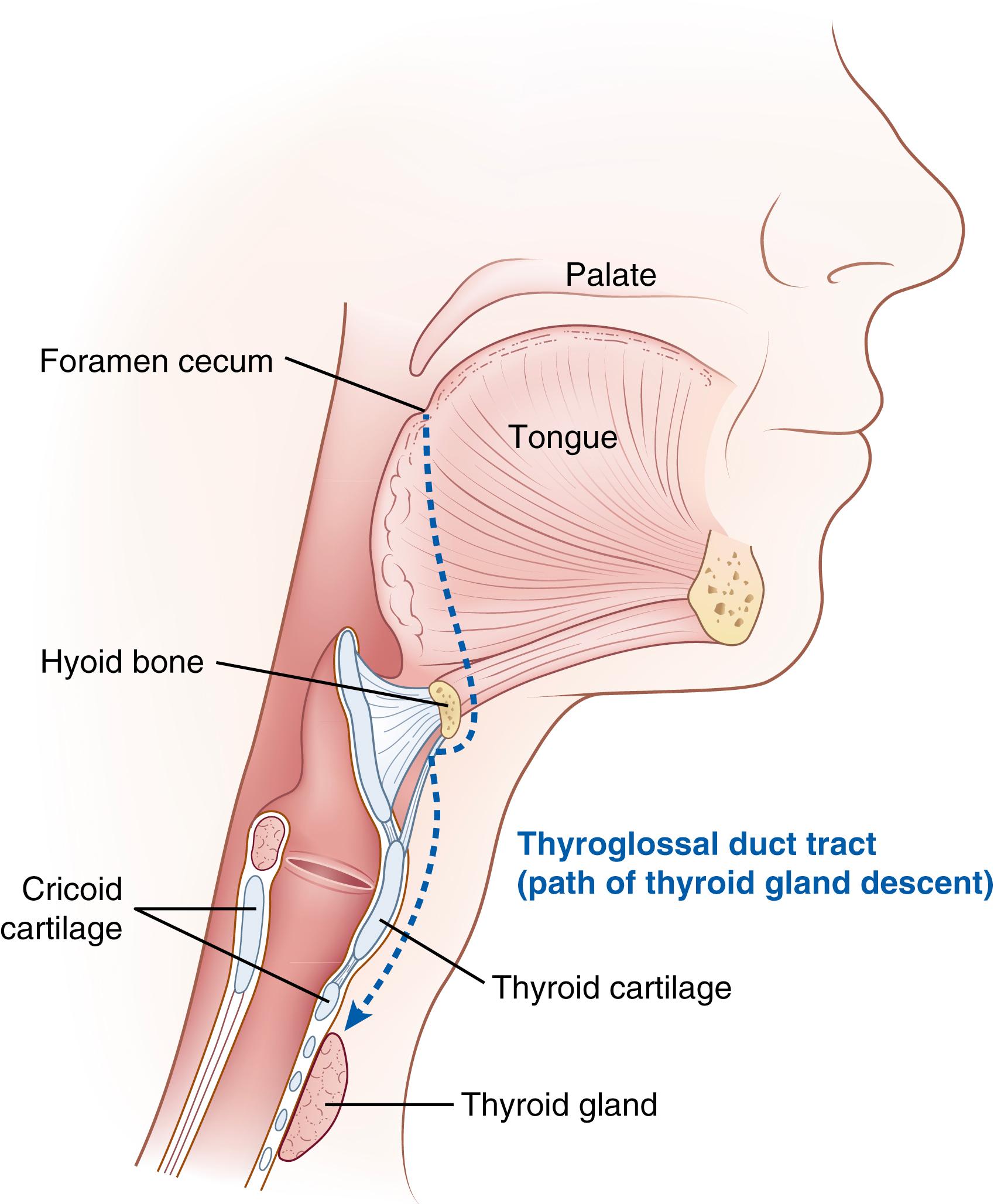
Thyroid dysgenesis (TD; abnormal development of the embryonic thyroid), including the most severe form, agenesis (the complete absence of the thyroid), is the most common cause of permanent hypothyroidism in humans, representing approximately 65% of the 1 in 3000 children who are born with congenital hypothyroidism (CH). The remaining 35% of infants with CH have a eutopic thyroid (located in the correct anatomic position), with approximately 50% of these infants having an identifiable defect in thyroid hormone biosynthesis (thyroid dyshormonogenesis). TD is typically considered a sporadic disease with ongoing investigation if familial cases follow an autosomal dominant, Mendelian mode of inheritance. , In contrast, the majority of germline mutations causing thyroid dyshormonogenesis are inherited in an autosomal recessive manner. ,
The most common form of TD is thyroid ectopy (75% of cases) where the glandular tissue is located in the tongue (lingual thyroid) or anywhere else along the normal migratory path (see Fig. 145.1 ). , The expression of the transcription factors HHEX , NKX2-1 , FOXE1 , and PAX8 is critical for specification of the ventral endoderm cells that ultimately comprise the median and lateral anlagen, with NKX2-1 , FOXE1 , and PAX8 critical for thyroid folliculogenesis ( NKX2-1 and PAX8 ) and migration ( FOXE1 and CDCA8 /Borealin). , Several of these transcription factors are also involved in non-thyroid related organogenesis with mutations resulting in syndromic-forms of CH. As an example, expression of NKX2-1 is critical in the development and function of interneurons in the central nervous system (CNS), surfactant producing cells in the lungs, and expression of thyroid peroxidase (TPO) and thyroglobulin. Mutations in NKX2-1 are associated with brain-lung-thyroid syndrome characterized by benign hereditary chorea, infant respiratory distress syndrome, and CH secondary to varying severity of TD. Similarly, mutations in FOXE1 are associated with craniofacial alterations, including bilateral choanal atresia, cleft palate, bifid epiglottis, spiky hair, and athyreosis (Bamforth-Lazarus syndrome; see Table 145.1 ). ,
The only biologically essential function of the thyroid gland is to produce adequate amounts of the iodothyronine hormones, 3′,3,5-triiodothyronine (T3) and tetra-iodothyronine (T4). Thyroid hormone producing cells are arranged as spheres or “follicles” in which the thyroid follicular cells surround a lumen of colloid. This structure facilitates a unique feature of the thyroid among endocrine glands, which is its ability to store large amounts of preformed hormone. This adaptation allows the thyroid to secrete adequate amounts of thyroid hormone during limited interruptions in dietary iodine intake. The thyroidal C-cells secrete calcitonin, a hormone involved in calcium balance through inhibition of osteoclast activity and decreased resorption of calcium in the kidneys. These activities oppose the action of parathyroid hormone; however, there is no significant bone morbidity or negative alternation in calcium homeostasis associated with the loss of endogenous calcitonin production in patients with thyroid agenesis or in patients who have undergone thyroidectomy.
Each thyroid follicular cell contains the machinery required for thyroid hormone synthesis ( Fig. 145.2 ). Only T3 and T4 are capable of binding to the nuclear thyroid hormone receptors (TRs). T3 binds to the TRs with approximately 12-fold higher affinity compared to T4 and because of this T3 is accepted to be the active form of thyroid hormone and T4 its prohormone. Thyroid hormone synthesis requires the nutritional precursor of iodine, which is absorbed from the gut and circulates in plasma as iodide. Plasma iodide is actively transported or “trapped” into thyroid follicular cells through the sodium-iodine symporter (NIS). Using H 2 O 2 generated by calcium-dependent dual oxidase (DUOX) enzymes, intracellular iodide in thyroid follicular cells is oxidized and then incorporated onto tyrosine residues within the thyroglobulin glycoprotein. The latter process is termed iodine organification and is catalyzed by the heme-containing TPO protein. The addition of a single iodine atom to tyrosine results in monoiodotyrosine (MIT), and the addition of two iodine atoms produces diiodotyrosine (DIT). Within the thyroglobulin protein, these iodotyrosine molecules fuse or “couple” to form either T3 (from DIT + MIT coupling) or T4 (from DIT + DIT coupling). This coupling reaction is also catalyzed by TPO. The dietary requirements of iodine are based on age, with increased daily requirements from 150 µg/day for women during pre-pregnancy planning to 250 µg/day during pregnancy and lactation, secondary to increased maternal thyroid hormone production and fetal iodine requirements. ,
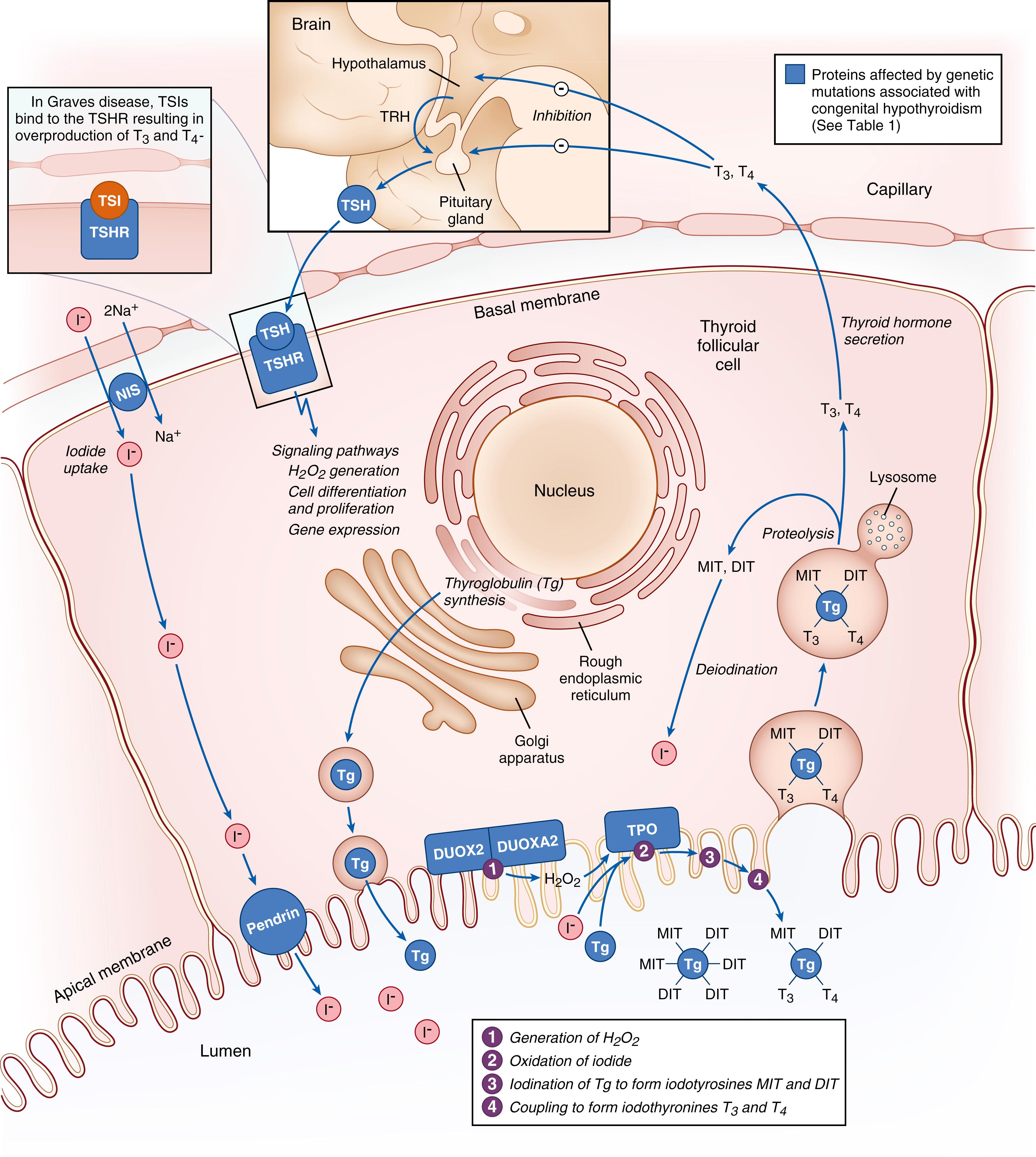
The binding of thyroid stimulating hormone (TSH) to the TSH-receptor (TSHR) increases synthesis of iodothyronines as well as secretion of T3 and T4. At the apical membrane of the thyrocyte, colloid is endocytosed into the follicular cell and thyroglobulin undergoes lysosomal proteolysis to release T4 and T3 into the circulation. Uncoupled iodotyrosines are also released by proteolysis and then deiodinated by intracellular iodotyrosine dehalogenases, allowing their iodine to be recycled for new organification reactions. In a healthy, iodine-replete individual, the ratio of T4/T3 (T4:T3) secretion is approximately 14:1 with only 20% of circulating T3 originating from the thyroid; the remainder (80%) is converted from the prohormone T4 in peripheral tissues.
Genetic alterations in the genes involved in thyroid hormone biosynthesis are collectively called thyroid dyshormonogenesis ( Table 145.2 ). Loss of function mutations in the genes that encode NIS (SLC5A5) , pendrin (SLC26A4) , DUOX1/DUOX2 and DUOXA2, TPO (TPO) , thyroglobulin (Tg) , and iodotyrosine deiodinase or dehalogenase (IYD) are well recognized causes of dyshormonogenesis associated CH. The majority of these syndromes are inherited in an autosomal recessive pattern. Interestingly, mutations in TSHR may follow an autosomal dominant pattern, associated with TSH resistance (varying degrees of elevated TSH with normal to low T4), or an autosomal recessive pattern, associated with thyroid hypoplasia.
| Gene (OMIM) | Name | Mode of Inheritance | Phenotype |
|---|---|---|---|
| TSHR ( 603372 ) | Thyroid-stimulating hormone receptor | AD | TSH resistance with elevated TSH and T4 in the normal range |
| SLC5A5 ( 601843 ) | NIS: sodium-iodide symporter | AR | Absent or low iodide uptake on scintigraphy with elevated serum thyroglobulin (Tg). Variable hypothyroidism and goiter |
| SLC26A4 ( 605646 ) | Pendrin: Anion transporter | AR | High level of uptake on scintigraphy with positive perchlorate discharge test and elevated serum Tg. Sensorineural hearing loss with enlarged vestibular aqueduct hypothyroidism and goiter |
| DUOX1/DUOX2 ( 606758 / 606759 ) | Dual oxydase 1 and 2 | AR or AD | High level of uptake on scintigraphy with positive perchlorate discharge test and elevated serum Tg. Transient or permanent hypothyroidism and goiter |
| DUOXA2 ( 612772 ) | Dual oxidase associated protein | AR | High level of uptake on scintigraphy with positive perchlorate discharge test and elevated serum Tg. Transient or permanent hypothyroidism and goiter |
| TPO ( 606765 ) | Thyroid peroxidase | AR | High level of uptake on scintigraphy with positive perchlorate discharge test and elevated serum Tg. Severe hypothyroidism and goiter |
| Tg ( 188450 ) | Thyroglobulin | AR | Positive uptake on thyroid scintigraphy and low to undetectable serum Tg. Variable hypothyroidism and goiter |
| IYD/DEHAL1 ( 612025 ) | Iodotyrosine dehalogenase | AR | Positive uptake on thyroid scintigraphy and elevated serum Tg. Variable hypothyroidism and goiter |
| GNAS ( 139320 ) | Alpha subunit of the stimulatory guanine nucleotide-binding G-protein | Imprinted gene | Hypothyroidism with partial TSH resistance |
The pattern and timing of hypothyroidism and goiter onset may vary based on the genetic etiology. Mutations in Tg , TPO , and DUOXA2 have been reported as etiologies of fetal goiter. Prenatal US in the second trimester is an accurate means of assessing for fetal thyroid volume, and treatment should be considered based on the severity and progression of polyhydramnios as well as concerns of premature labor and airway compromise after delivery. Mutations in DUOX2 can cause either transient (monoallelic mutation) or permanent (biallelic mutation) CH, and mutations in SLC26A4 (pendrin) and IYD may present in early infancy or have delay in presentation into late childhood.
Under normal physiologic circumstances, thyroid hormone secretion is tightly regulated by the HPT axis (see Fig. 145.2 ). The hypothalamus secretes thyrotropin-releasing hormone (TRH), which stimulates the pituitary to secrete thyrotropin (also called TSH ). Thyrotropin in turn stimulates the thyroid gland to secrete T4 and T3, both by stimulating hormone biosynthesis and by promoting the growth of thyroid tissue. Acting through the plasma membrane TSHR, TSH signals through phospholipase C, cyclic adenosine monophosphate, and inositol-phosphate diacylglycerol pathways to increase H 2 O 2 production, iodine organification, iodide uptake, and the expression of NIS, TPO, and Tg (see Fig. 145.2 ). Postnatally, TRH and TSH secretion are negatively regulated by feedback from circulating T4 and T3.
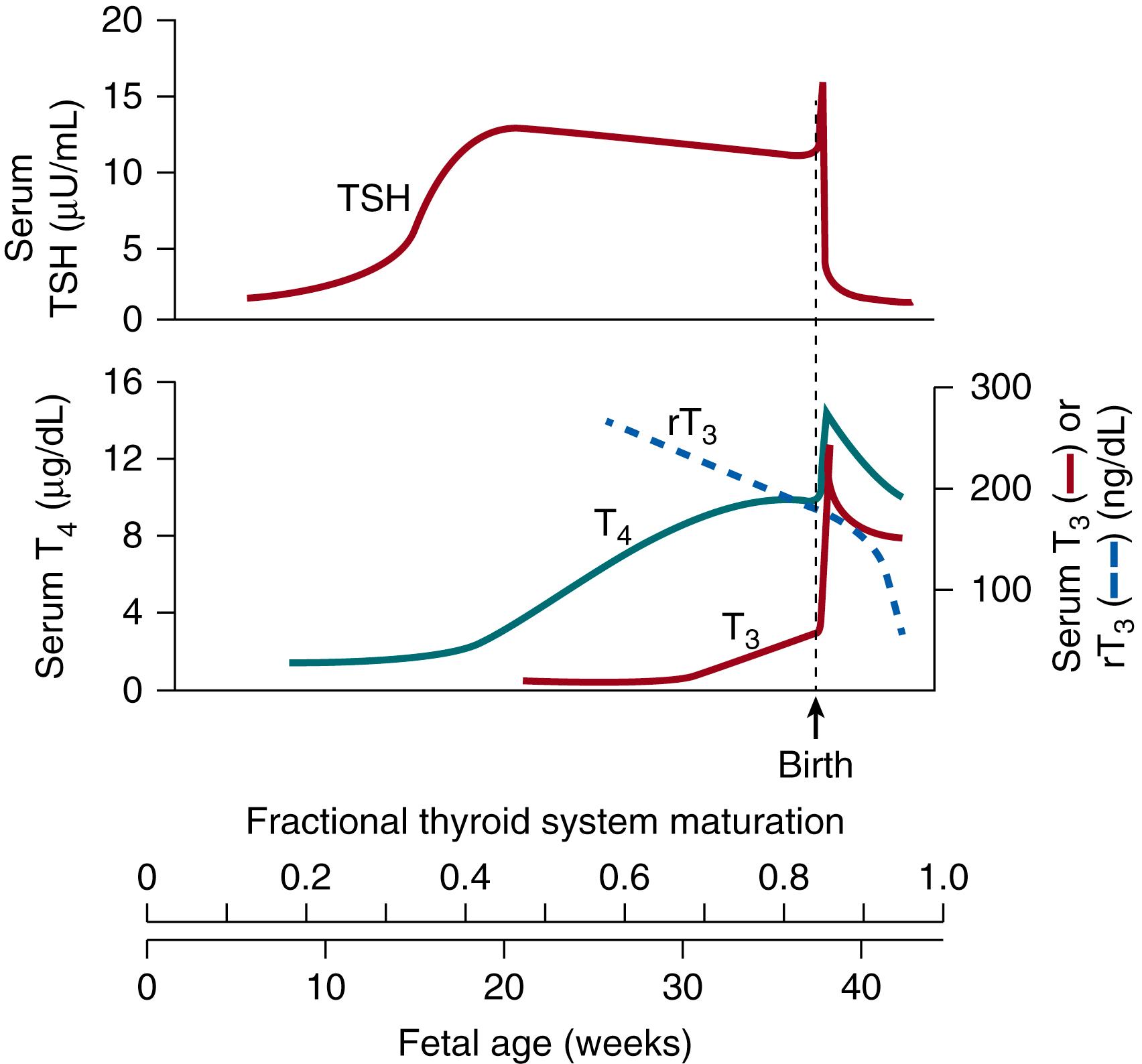
Studies of fetal serum show that the secretion of fetal TSH is minimal until mid-gestation (18 to 20 weeks), when the fetal thyroid gland begins to concentrate iodine, with subsequent increases in T3 and T4 secretion between 20 weeks and term gestation ( Fig. 145.3 ). During the first trimester, maternal T4 production increases by approximately 20% under stimulation of placental human chorionic gonadotropin (hCG). Maternal T4 then crosses the placenta to drive fetal organogenesis and development after conversion to T3 by type 2 deiodination within fetal tissues. In the early third trimester (25 weeks) the fetal pituitary response to TRH develops as well as maturation of the negative feedback control of pituitary TSH from T3 and T4. The ability of the fetal pituitary to both stimulate thyroid secretion and to sense negative feedback is intact prior to delivery, as has been demonstrated by (1) development of fetal goiter in the offspring of mothers with Graves disease overtreated with antithyroid medication (due to transplacental transfer of the antithyroid medication, blockade of fetal thyroid hormone production, and subsequent increase in fetal TSH), and (2) neonatal thyrotoxicosis with elevated T3 and T4 and suppressed TSH in infants born to mothers with Graves disease (due to transplacental transfer of maternal TSHR-stimulating antibodies).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here