Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Records of obstetrics and gynecology date back to the time of Hippocrates in 400 BCE. He was probably the first physician to describe midwifery, menstruation, sterility, symptoms of pregnancy, and puerperal (the period after labor) infections. Most of the early gynecologic history stems from Soranus in the 2nd century CE. His works included chapters on anatomy, menstruation, fertility, signs of pregnancy, labor, care of the infant, dysmenorrhea (painful menstruation), uterine hemorrhage, and even the use of vaginal specula.
William Harvey, who devised the theory of blood circulation, was also responsible for a monumental treatise on obstetrics. This work, published in 1651, included a detailed assessment of uterine changes throughout life.
The 18th century was a period of a further understanding of pregnancy, labor, and fertility. However, it was not until the 19th century that diseases of the female genitalia were better understood. As recently as 1872, Emil Noeggerath published his investigations on gonorrhea, which ultimately changed the opinion of the medical world about the significance of this disorder. He was the first to suggest that “latent gonorrhea” was associated with sterility in women. Although the first cesarean section was described in 1596 by Scipione Mercurio, the development of the current technique of Max Sänger was described in 1882.
In 2018, cancer of the uterine corpus, also known as endometrial cancer, the most common cancer of the female reproductive organs, accounted for 7% of all cancers and 4% of all cancer deaths in women in the United States. It is the fourth most common cancer found in women, after breast cancer, lung cancer, and colorectal cancer. Obesity increases the risk of endometrial cancer because estrogen is produced in adipose tissue. Forty percent of uterine cancer cases are linked to obesity. Increased estrogen exposure is a strong risk factor for endometrial cancer. Other factors that increase estrogen exposure include menopausal estrogen therapy (without use of progestin), early menarche, late menopause, low parity, and a history of polycystic ovary syndrome (PCOS) due to chronic anovulation. In 2018, there were 63,230 new cases and 11,350 deaths from cancer of the uterus. The lifetime risk for development of cancer of the uterus is 1 per 35. For all cases of cancer of the uterus, the 5-year relative survival rate is 81%. The 5-year survival rate is 95%, 68%, or 16%, if the cancer is diagnosed at a local, regional, or distant stage, respectively. Although the mortality rate has declined slightly since the 1980s among white women, it has remained stable among other racial and ethnic groups. Although the incidence rate of uterine cancer is lower for black women than for white women, the mortality rate among black women is higher, at 82% and 60% respectively. Black women are more likely to be diagnosed with a later-stage disease, and black and Hispanic women have been shown to present with more aggressive disease.
Between the mid-1950s and 1992, deaths from invasive cancer of the cervix in the United States dropped by 74%. The decline in mortality from cervical cancer is largely attributed to early detection by physical examination. It has been estimated that noninvasive cervical cancer (carcinoma in situ) is approximately four times more common than invasive cervical cancer. In the United States, the widespread use of the Papanicolaou (Pap) 1
1 Named for George N. Papanicolaou, the physician who developed this screening technique. When properly performed, the Pap test can accurately diagnose cervical carcinoma in 98% of cases.
test has decreased the incidence and mortality rate by 40% since the mid-1970s. Most invasive cervical carcinomas are found in women who have not had regular Pap tests. In 2018, there were 13,240 new cases of invasive cervical cancer diagnosed, and 4170 women died from this disease. The death rate continues to decline by approximately 2% per year. An American woman has a 0.78% lifetime risk (1 per 128) for development of cervical cancer and a 0.27% risk of dying from the disease. The 5-year relative survival rate for the earliest stage of invasive cervical cancer is 92%, and the overall (all cases considered together) 5-year survival rate is 65%.
Of the many risk factors that have been evaluated, young age at first sexual intercourse, multiple sexual partners, infection with the human papillomavirus (HPV), infection with herpes simplex virus, infection with human immunodeficiency virus, immunosuppression, a history of cervical dysplasia, and smoking are most often associated with an increased risk of cervical cancer.
The most important risk factor for cervical cancer is infection with a high-risk strain of HPV. HPVs are a group of more than 100 types of viruses, some of which can cause warts, or papillomas; these are noncancerous (benign) tumors. Certain other types of HPV can cause cancer of the cervix. These are called high-risk or oncogenic types of HPV, and approximately 70% of all cervical cancers are caused by HPV types 16 and 18. It has been shown that younger women, in their teens and early 20s, have the highest incidence of HPV infection, but are able to mount an immune response to clear the infection. The current guidelines indicate that a woman should be screened with cervical cytology, or Pap test, at the age of 21, regardless of when she initiated sexual activity. In women older than 30 years, an HPV test should be conducted at the same time as a Pap test. 2
2 Final Recommendation Statement: Cervical Cancer: Screening . U.S. Preventive Services Task Force. December 2016. https://www.uspreventiveservicestaskforce.org/Page/Document/RecommendationStatementFinal/cervical-cancer-screening . Assessed December 31, 2018.
Vaccines have been developed that protect against infection with certain strains of HPV, which may reduce cervical cancer rates in the future. Gardasil9 is now the only approved vaccine in the United States, replacing the previously approved quadrivalent HPV recombinant vaccine (Gardasil) and HPV bivalent vaccine (Cervarix). Gardasil9 protects against types 6, 11, 16, 18, 31, 33, 45, 52, and 58. The Gardasil9 vaccine entails a series of three injections over a 6-month period. In October 2018, the FDA expanded the approved use of Gardasil9 for use in females and males 9 to 45 years of age (previously approved for use from age 9 to 26) to prevent all HPV-related lesions and cancers. 3
3 https://www.fda.gov/biologicsbloodvaccines/vaccines/approvedproducts/ucm426445.htm . Updated October 10, 2018. Assessed December 31, 2018.
These vaccines cannot protect against established infections, nor do they protect against all HPV types.
Although ovarian carcinoma accounts for only 3% of all cancers in women, it is the cause of 5% of all cancer deaths in women. It is the fifth leading cause of cancer death and the leading gynecologic malignancy in women in the United States. Cancer of the ovary accounts for nearly 50% of all deaths from gynecologic malignancies. This is due to the advanced stage at diagnosis in most patients. The most important risk factor is a strong family history of breast or ovarian cancer. In 2015, there were 21,429 new cases of ovarian cancer and 13,920 deaths from it. The lifetime risk for development of ovarian cancer is 1 per 59; the incidence is 1.4 per 100,000 women younger than 40 years, but it increases to 45 per 100,000 women older than 60 years. The incidence has been declining by 1% per year since 1992. The overall 5-year relative survival rate is 47%, with a 92% survival rate for the earliest stage of ovarian cancer and a 29% 5-year survival rate in late-stage disease. A carefully performed bimanual pelvic examination has been shown to be the cornerstone of diagnosis of ovarian cancer.
The external female genitalia, known collectively as the vulva, are shown in Fig. 19.1 . The vulva consists of the mons veneris, the labia majora, the labia minora, the clitoris, the vestibule and its glands, the urethral meatus, the fourchette, and the perineum. The mons veneris is a rounded prominence of fat tissue overlying the pubic symphysis. The labia majora are two large skinfolds that form the lateral boundaries of the vulva. They meet anteriorly at the mons veneris to form the anterior commissure. The labia majora and the mons veneris have hair follicles and sebaceous glands. The labia majora correspond to the scrotum in the man. The labia minora are two narrow, pigmented skinfolds that lie between the labia majora and enclose the vestibule, which is the area lying between the labia minora.
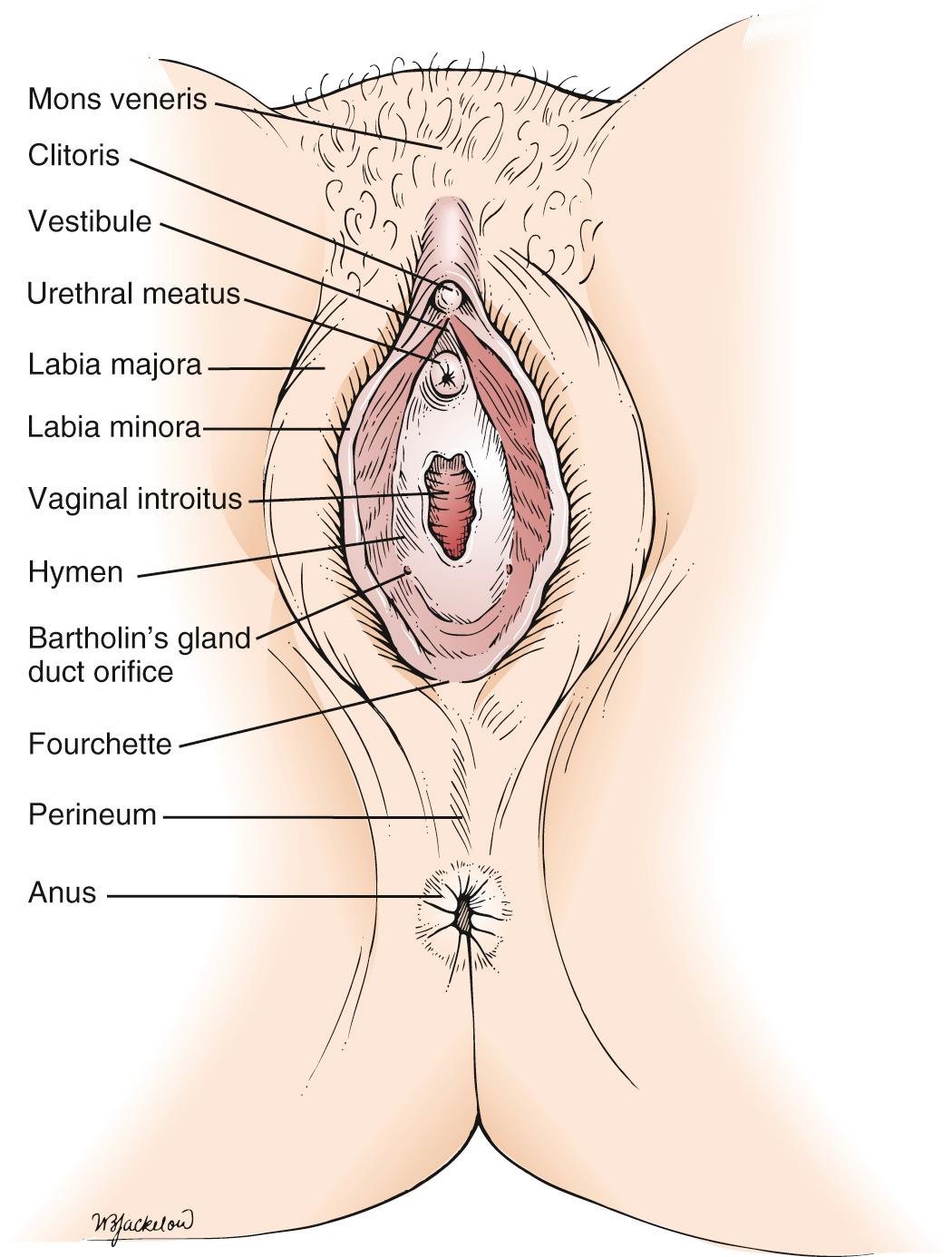
Anteriorly, the two labia minora join to form the prepuce of the clitoris. The clitoris, analogous to the penis, consists of erectile tissue and a rich supply of nerve endings. It has a glans, two corpora cavernosa (the body), and the crura. The crura are attached to the undersurface of the pubic ramus and are covered by the ischiocavernosus muscles. The external urethral meatus is located in the anterior portion of the vestibule below the clitoris. Paraurethral glands, or Skene glands, are small glands that open lateral to the urethra. Secretion of sebaceous glands in this area protects the vulnerable tissues against urine. The major vestibular glands are known as Bartholin glands, or vulvovaginal glands. These pea-sized glands correspond to the male Cowper glands. Each Bartholin gland lies posterolaterally to the vaginal orifice. During sexual intercourse, a watery fluid is secreted that serves as a vaginal lubricant. Posteriorly, the labia minora unite at the posterior commissure to form the fourchette. The perineum is the area between the fourchette and the anus.
If the labia minora are reflected laterally, the vaginal introitus and hymenal ring are exposed. The hymen is a circular fold of tissue that partially occludes the vaginal introitus. There are marked variations in its size, as well as in the number of openings in it. The vaginal introitus is the border between the external and internal genitalia and is located in the lower portion of the vestibule.
The majority of the vulva is innervated by the pudendal nerve, with the exception of the area anterior to the urethral meatus, which is innervated by the ilioinguinal and genitofemoral nerves. The blood supply to the external genitalia and perineum is predominantly from the internal pudendal arteries. The lymphatic drainage is into the superficial and deep inguinal nodes.
The internal genitalia are shown in Fig. 19.2 . The vagina is a muscularly walled, hollow canal that passes upward and slightly backward, at a right angle to the uterus. The vagina lies between the urinary bladder anteriorly and the rectum posteriorly. The vaginal walls are lined by transverse rugae, or folds. The lower portion of the cervix projects into the upper portion of the vagina and divides it into four fornices. The anterior fornix is shallow and is just posterior to the bladder. The posterior fornix is deep and is just anterior to the rectovaginal pouch, known as the cul-de-sac (pouch) of Douglas , and the pelvic viscera lie immediately above this pouch. The lateral fornices contain the broad ligaments. The fallopian tubes and ovaries may be palpated in the lateral fornices. The superficial cells of the vagina contain glycogen, which is acted on by the normal vaginal flora to produce lactic acid. This is in part responsible for the resistance of the vagina to infection.
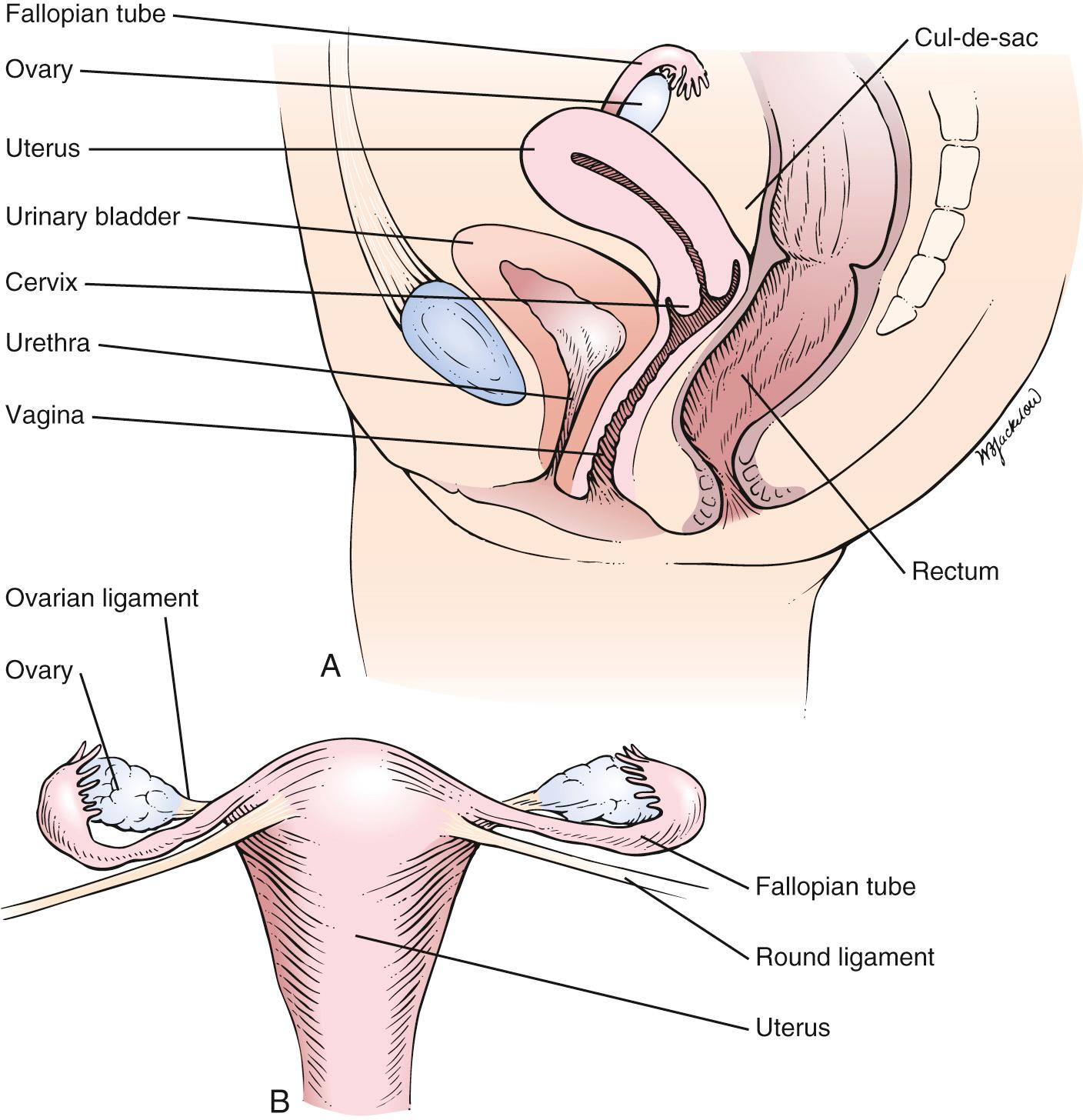
The arterial supply to the vagina is derived from the internal iliac, uterine, and middle hemorrhoidal arteries. The lymphatic channels of the lower third of the vagina drain into the inguinal nodes. The lymphatic channels of the upper two-thirds enter the hypogastric and sacral nodes.
The uterus is a hollow muscular organ with a small central cavity. The lower end is the cervix, and the upper portion is the fundus. The size of the uterus is different during various stages of life. At birth, the uterus is only 3 to 4 cm long. The adult uterus is 7 to 8 cm long and 3.5 cm wide, with an average wall thickness of 2 to 3 cm. The growth of the uterus and the relationship of the size of the fundus to the size of the cervix are shown in Fig. 19.3 .
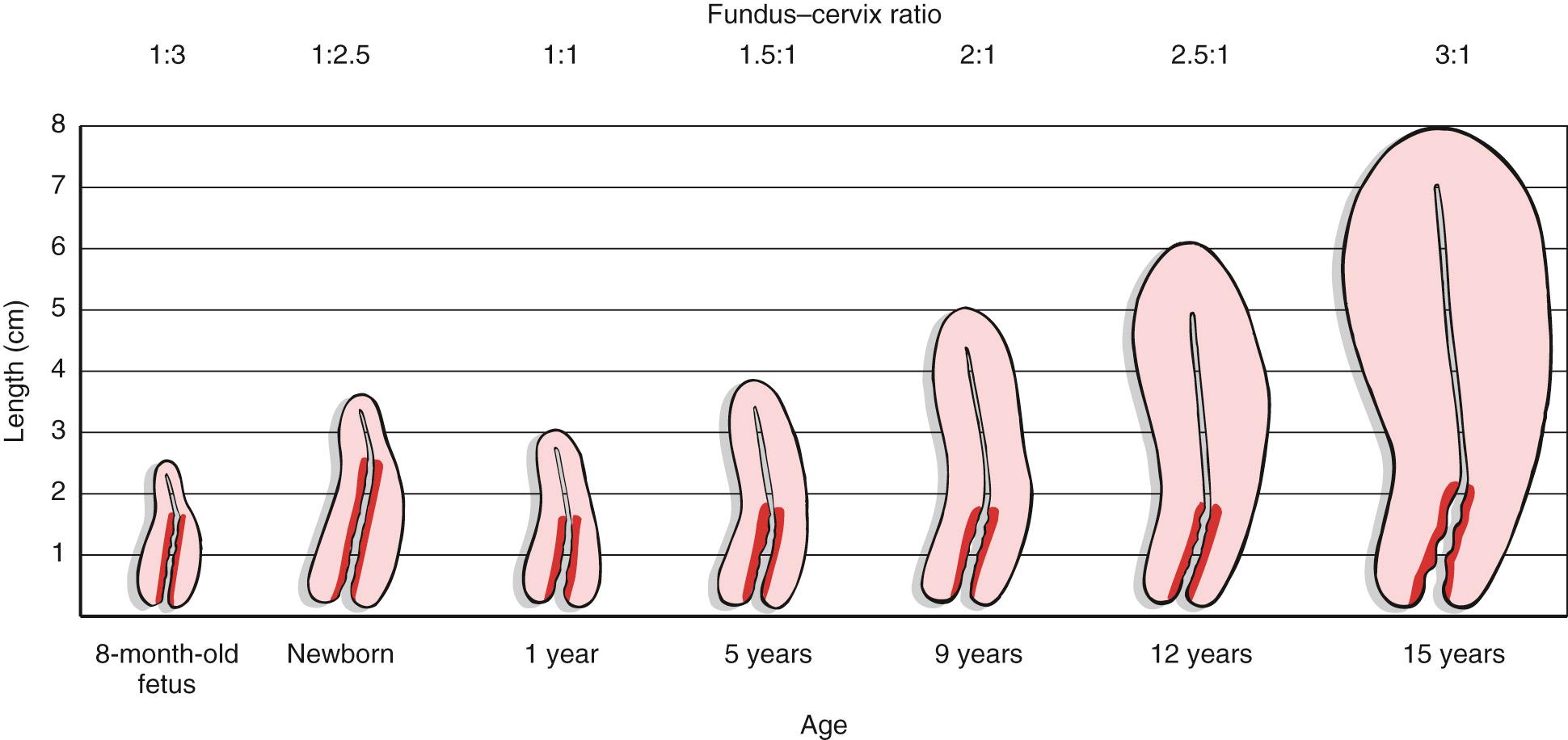
The triangular uterine cavity is 6 to 7 cm in length and is bounded by the internal cervical os inferiorly and the entrances of the fallopian tubes superiorly. The uterus can be anteverted, axial, or retroverted in position related to the axis extending from cervix to the uterine fundus. The uterine fundus may also be folded forward (anteflexed) or backward (retroflexed).
The uterus is freely mobile and is located centrally in the pelvic cavity. It is supported by the broad and uterosacral ligaments, as well as by the pelvic floor. The peritoneum covers the fundus anteriorly down to the level of the internal cervical os. Posteriorly, the peritoneum covers the uterus down to the pouch of Douglas. Fig. 19.4 is a detailed anatomic representation of the uterus.
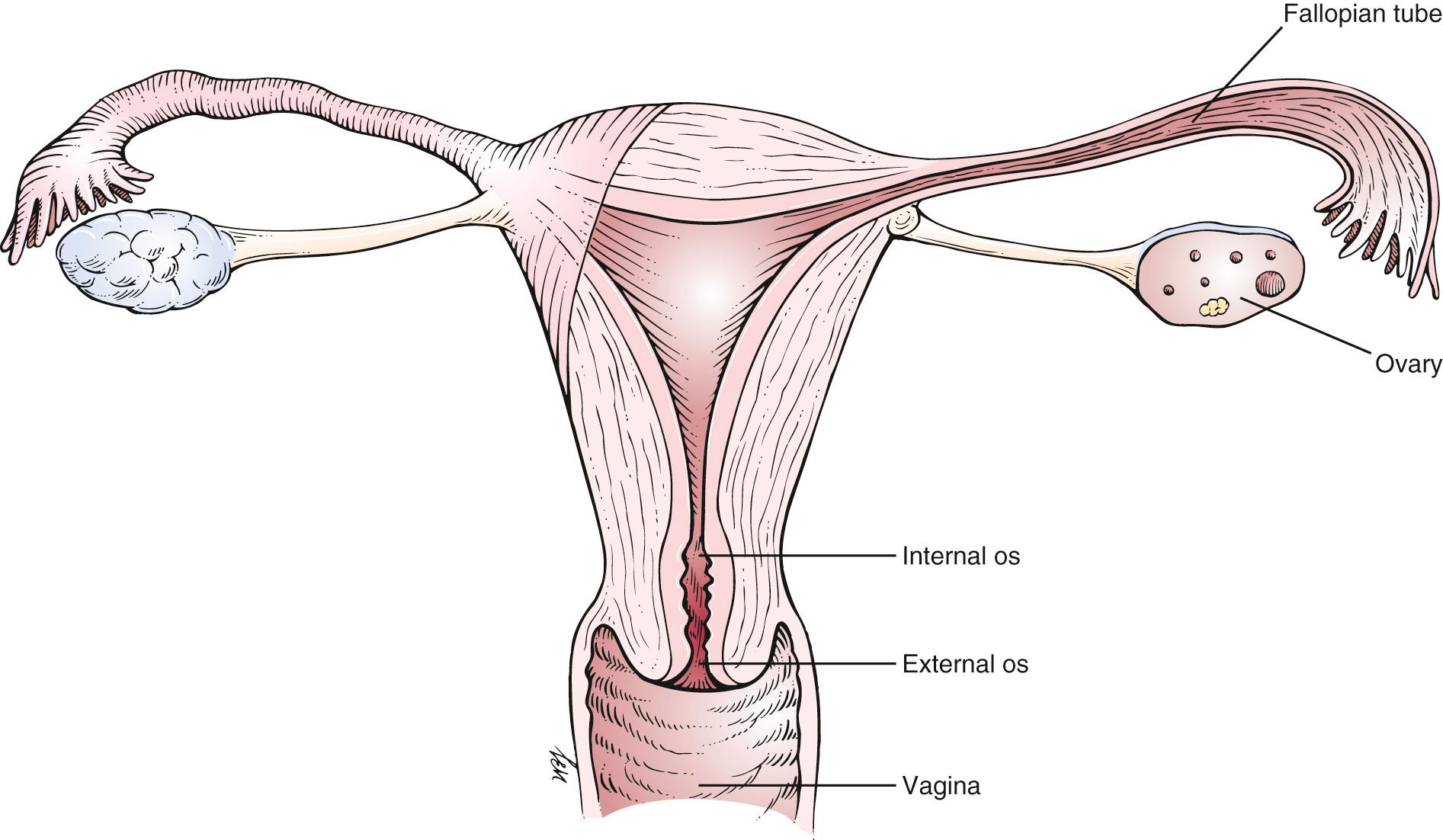
The cervix is the vaginal portion of the uterus. The greater portion of the cervix has no peritoneal covering. The cervical canal extends from the external cervical os to the internal cervical os, where it continues into the endometrial cavity up to the fundus of the uterus. The external cervical os in women who have not given birth vaginally is small and circular. In women who have had vaginal deliveries, the external cervical os is linear or oval.
With increasing levels of estrogens, the external cervical os begins to dilate, and cervical mucous secretion becomes clear and watery. With high levels of estrogens, cervical mucus, when placed between two glass slides that are then pulled apart, can be stretched 15 to 20 cm before breaking. This property of cervical mucus—the ability to be drawn into a fine thread—is termed spinnbarkeit. When cervical mucus is allowed to dry on a glass slide and is examined under low power by light microscopy, a fern pattern made up of salt crystals may be seen. Spinnbarkeit and ferning reach a maximum at the midpoint of the menstrual cycle. Sperm can more easily penetrate mucus with these characteristics.
The blood supply to the uterus comes from the uterine and ovarian arteries. The lymphatic vessels of the fundus enter into the lumbar nodes.
The fallopian tubes, or oviducts, enter the fundus at its superior aspect. They are small muscular tubes that extend outward into the broad ligament toward the pelvic wall. The other end of the oviduct opens into the peritoneal cavity near the ovary. These endings are surrounded by fringe-shaped projections called fimbriae. The primary function of the fallopian tube is to provide a conduit for the egg to convey it from the corresponding ovary to the uterus, a trip that takes several days. Sperm traverse the fallopian tube in the opposite direction, and it is usually in the fallopian tube that fertilization takes place.
The ovaries are almond-shaped structures about 3 to 4 cm long and are attached to the broad ligament. The blood supply to the ovaries arise from the ovarian arteries from the abdominal aorta; while the left ovarian vein drains into the left renal vein and the right ovarian vein drains directly into the inferior vena cava. The primary functions of the ovary are oogenesis and hormone production.
The ovaries, fallopian tubes, and supporting ligaments are termed the adnexa.
The female reproductive system is under the influence of the hypothalamus, whose releasing factors control the secretion of the anterior pituitary gonadotropic hormones: follicle-stimulating hormone and luteinizing hormone. In response to these hormones, the ovarian Graafian follicle secretes estrogens and discharges its ovum. After ovulation, the ovarian follicle is termed the corpus luteum, which secretes estrogens and progesterone. With the secretion of progesterone, the basal body temperature rises. This is a reliable sign of ovulation. Under the influence of the ovarian hormones, the uterus and breasts undergo the characteristic changes of the menstrual cycle.
If pregnancy does not occur, the corpus luteum regresses, and the level of ovarian hormones begins to fall. At this time, before menstruation, many women have symptoms of weakness, depression, and irritability. Breast tenderness is also common. These symptoms are termed premenstrual syndrome. Approximately 5 days after the fall in the level of hormones, the menstrual period begins. Menstrual fluid throughout the 5-day period measures 50 to 150 mL, only half of which is blood; the remainder is mucus. Because menstrual blood does not contain fibrin, it does not clot. When the menstrual flow is heavy, as it is on days 1 to 2, “clots” may be described. These clots are not fibrin clots but are combinations of red blood cells, glycoproteins, and mucoid substances that are believed to form in the vagina rather than in the uterine cavity.
Some of the hormone-dependent changes related to the menstrual cycle are shown in Fig. 19.5 .
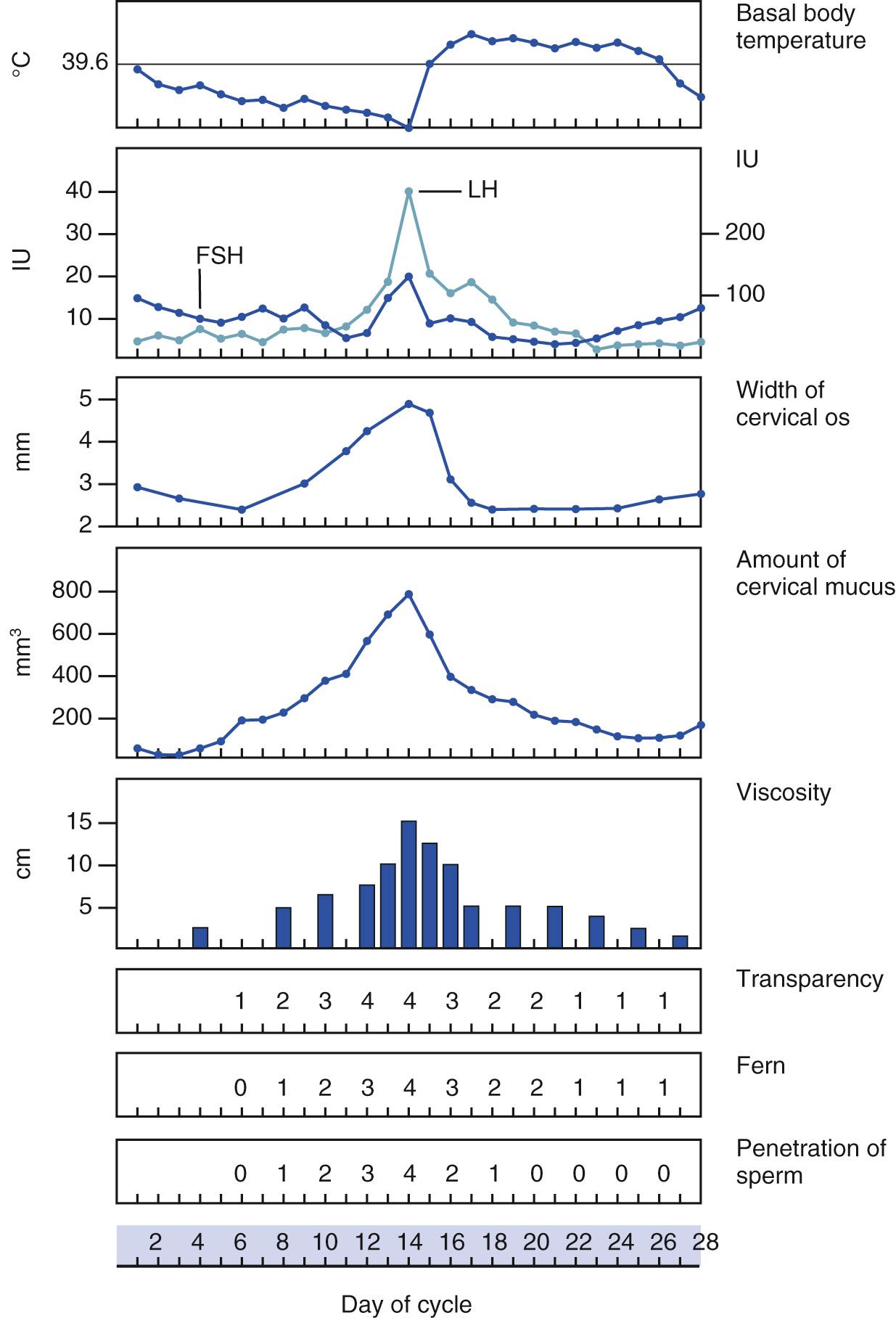
Approximately 1.5 years before puberty, gonadotropins are measurable in the urine. The ovaries enter a period of rapid growth at, on average, 8 to 9 years of age, which marks the onset of puberty. Secretion of estrogens begins to increase rapidly at approximately 11 years of age. Concomitant with estrogen production, the sexual organs begin to mature. During puberty, the secondary sex characteristics begin to develop. The breasts enlarge, hair develops on the pubis, the vulva enlarges, the labia minora become pigmented, and the body contour changes. Puberty lasts for 4 to 5 years. The first menstrual cycle, called menarche, occurs at the end of puberty at approximately 12.5 years of age. There is, however, a wide variation in the age at menarche. The cycles continue approximately every 28 days, with a flow lasting 3 to 5 days. The first day of the period is designated the first day of the cycle. It is rare for a woman to be absolutely regular, and cycles of 21 to 35 days are considered normal.
At the time of menarche, the menstrual cycle is usually anovulatory 4
4 Not accompanied by the release of an ovum from the ovaries.
and irregular. After 1 to 2 years, ovulation begins. After stabilization of the menses, ovulation occurs approximately midcycle in a woman with a regular cycle.
Menopause marks the ends of menstruation. Menopause is defined as the last uterine bleeding induced by ovarian function. It usually occurs between 45 and 55 years of age. Ovulation and corpus luteum formation no longer occur, and the ovaries decrease in size. The period after menopause is termed postmenopausal.
Even in the absence of specific symptoms, all women, regardless of age, should be asked several important questions to provide a complete gynecologic, obstetric, and reproductive history. A comprehensive gynecologic history includes the following basic information: menstrual history, obstetric history, sexual history, types of contraception (past and present), pap smear history, history of sexually transmitted infections (STIs), history of gynecology-related problems (such as PCOS, fibroids, and ovarian cysts), a gynecologic specific review of symptoms, and screening for intimate partner violence. It is always important to remember to use nonmedical terminology when taking a history.
A menstrual history should begin by summarizing the age at menarche, the cycle length, and the duration of bleeding. If a woman reached menarche at age 12 years and has had regular periods every 29 days lasting for 5 days, her menstrual history is written as “12 × 29 × 5.” Examples of questions to use to elicit a menstrual history include:
“When was the first day of your last menstrual period (LMP)?”
“At what age did you start getting your period?”
“How often do your periods occur?”
“Are they regular?”
“How many days does the bleeding last?”
“How many pads or tampons do you use on the heaviest day of your period?”
“During your period, do you experience any menstrual cramps? Mood swings? Breast tenderness or breast pain? Bloating? Swelling? Headache?”
“Do you have any bleeding between periods?”
“Do you have any bleeding after intercourse?” (postcoital bleeding)
“Have you had any episodes of bleeding since stopping your periods?” (postmenopausal patients)
The obstetric history includes the number of total pregnancies regardless of outcome, known as gravidity, and the number of pregnancies reaching 20 weeks or beyond, known as parity. Parity is further broken down to:
t —term pregnancies (37 to 42 weeks)
p —preterm pregnancies (20 to <37 weeks)
a —spontaneous or induced abortions and ectopic pregnancies
l —live children.
Gravidity and parity are typically written as such: G # P t-p-a-l . For example: a woman who has two children (one born at term, one born at 34 weeks), a history of an ectopic pregnancy and two miscarriages in the first trimester, and is currently pregnant would have a gravidity/parity representation of G6 P1132 . A second example: a patient with a history of two prior full-term children and a 22-week miscarriage would have a gravidity/parity representation of G3 P2102 .
When asking about abortions or miscarriages, it is advisable to keep in mind that in the United States, and in some other countries, the word “abortion” itself may be charged with many religious, political, and cultural feelings in different women. Maintain sensitivity while asking the question, reminding the patient that the medical term “abortion” means loss of any pregnancy, including voluntary terminations, ectopic pregnancy, or spontaneous miscarriage.
Examples of questions for obtaining an obstetric history include:
“Have you ever been pregnant?”
“Do you have a history of any miscarriages, abortions, or ectopic pregnancies?” or “What was the outcome of your pregnancies?”
“How were these pregnancies treated? Did you take medication or did you need surgery?”
“For each delivery, what was the date of delivery? How many weeks pregnant were you when you delivered?”
“Did you have any complications during your delivery?”
“Did you have any complications during your pregnancy, such has high blood pressure or diabetes?”
“Did any of your children have complications after delivery?”
Discussing sexual health with a patient can be very challenging for both the patient and the physician. A great majority of women with sexual concerns report that they are not asked about sexual health during medical visits, and if sexual health is discussed during their encounter, the conversation is initiated by the patient. Health care providers should become comfortable asking open-ended questions about a patient's sexual habits at an initial visit, an annual visit, and when there is concern for an STI or sexual dysfunction disorder. Examples of questions for obtaining a sexual history include:
“Are you currently sexually active?”
If yes, “In the past 6? 12? months, how many sexual partners have you had?”
“When you are sexually active, are your partners male, female, or both?”
“What kind of sexual contact do you have? Genital? Anal? Oral?” (Ask for both male and female partners)
“Do you use any type of contraception?”
“Do you use any barrier protection to protect against STIs? (Male/female condoms)”
“Every time, most times, sometimes?”
If no to contraception and condoms, “Are you trying to become pregnant?”
If no contraception and not trying to conceive , “Do you want to discuss contraception today?”
“Do you (or any partners?) have a history of any STIs, such as herpes, HIV, gonorrhea, and chlamydia?” (Listing infections specifically can trigger patient's memory)
“When were you last tested for STIs?”
“Do you ever experience pain with intercourse? Pain on insertion, deep pelvic pain, etc.”
“Do you have concerns about sexual function, like ability to achieve an orgasm? Changes in sexual desire or arousal? Or satisfaction during sex?”
“Do you feel safe in your sexual relationships?”
The most common symptoms of female genitourinary disease are as follows:
Abnormal (uterine/vaginal) bleeding
Dysmenorrhea (painful menses)
Masses or lesions
Vaginal discharge
Vaginal itching
Abdominal or pelvic pain
Dyspareunia (pain with intercourse)
Changes in hair distribution
Changes in urinary pattern
Infertility
Ask these questions of any woman with abnormal vaginal bleeding:
“How long have you noticed the vaginal bleeding?”
“Do you use contraceptives?” If yes , “What types of contraceptives do you use?”
”Are your periods regular or irregular?”
“How often are your periods?”
“How many days do your periods last?”
“How many tampons or pads do you use on the heaviest days of your period?”
“Are there any clots of blood?”
“What was the first day of your last period?”
“Have you noticed bleeding between your periods?”
“Do you have abdominal/pelvic pain during your periods?”
“Do you have hot flashes? Cold sweats?”
“Do you think you might be pregnant?”
“Have you ever been pregnant before?” If yes, “Tell me about all of your pregnancies.”
“Are you under any unusual emotional stress?”
“Have you noticed that you were warm in a room and others were not? Have you noticed that you were cold in a room and others were not?”
“Have you noticed a change in your vision?”
“Have you had any headaches? Nausea? Change in hair pattern? Milk discharge from your nipples?”
“What is your diet like?”
Abnormal uterine bleeding (AUB) is classified by the bleeding pattern and its underlying cause. In 2011, a new classification system was introduced to characterize AUB. 5
5 Munro MG, Critchley HOD, Fraser IS. The FIGO systems for nomenclature and classification of causes of abnormal uterine bleeding in the reproductive years: who needs them? Am J Obstet & Gyn. 2012;207(4): 259–265.
AUB can be due to structural or nonstructural causes. Examples of structural causes of AUB include endometrial polyps, adenomyosis, leiomyomas, and endometrial hyperplasia and carcinoma. Nonstructural causes of AUB include coagulopathies, ovulatory dysfunction, or idiopathic causes.
The term menorrhagia has now been replaced with heavy menstrual bleeding (AUB/HMB) and is excessive bleeding at the time of the menstrual period. The flow may be increased, the duration may be increased, or both may occur. The number of pads or tampons a patient uses each day of the cycle helps quantify the flow. AUB/HMB in some cases may be associated with blood disorders such as leukemia, inherited clotting abnormalities, and decreased platelet states. Uterine fibroids are a leading cause of AUB/HMB. Metrorrhagia is now described as intermenstrual bleeding (AUB/IMB) and is uterine bleeding of normal amount at irregular, noncyclic intervals, particularly between normal menstrual cycles. Foreign bodies such as intrauterine devices, as well as uterine or cervical polyps, and ovarian and uterine tumors, can cause AUB/IMB. Often there is increased bleeding between cycles as well as heavier periods; this is described as heavy and IMB, previously known as menometrorrhagia.
Amenorrhea is the cessation or nonappearance of menstruation. Before puberty, amenorrhea is physiologic, as it is during pregnancy and after menopause. In primary amenorrhea, menstruation has never occurred; in secondary amenorrhea, menstruation has occurred but has ceased, as in pregnancy. Long-distance joggers, patients with anorexia, or any woman with abnormally low body fat may have secondary amenorrhea. Diseases of the hypothalamus, pituitary gland, ovary, uterus, and thyroid gland are associated with amenorrhea. Galactorrhea , or milk discharge from the nipples, occurs in many individuals with pituitary tumors. Chronic disease is also frequently associated with secondary amenorrhea.
Menopause is typically defined as having no menstrual cycle for 12 months.
Bleeding that occurs after this 12-month time period is termed postmenopausal bleeding. Because many cancers of reproductive organs may present with bleeding, any postmenopausal bleeding must be investigated. Uterine fibroids or tumors of the cervix, uterus, or ovary may be responsible. Lower genital tract disease, such as vaginal atrophy, or even urinary tract problems may present with postmenopausal bleeding.
Dysmenorrhea, or painful menstruation, is a common symptom. It is often difficult to define as abnormal because many healthy women have some degree of menstrual discomfort. In most women, these cramps subside soon after the commencement of the menstrual flow. There are two types of dysmenorrhea: primary and secondary. Primary dysmenorrhea is far more common. It begins shortly after menarche, is associated with colicky uterine contractions, and occurs with every period. Childbirth frequently alleviates this state permanently. Secondary dysmenorrhea is caused by acquired disorders within the uterine cavity (e.g., intrauterine devices, polyps, or fibroids), obstruction to flow (e.g., cervical stenosis), or disorders of the pelvic peritoneum (e.g., endometriosis 6
6 Endometriosis is the presence of endometrial tissue outside the uterus and is a common cause of chronic pelvic pain.
or pelvic inflammatory disease [PID]). It usually occurs after several years of painless periods. Regardless of its cause, dysmenorrhea is described as intermittent, crampy pain accompanying the menstrual flow. The pain is felt in the lower abdomen and back, sometimes radiating down the legs. In severe cases, fainting, nausea, or vomiting may occur.
Masses or lesions of the external genitalia are common. They may be related to STIs, cysts, skin changes, pre-malignancies, or cancerous lesions. Ask these questions of any woman with a lesion on the genitalia:
“When did you first notice the mass (lesion)?”
“Has it changed since you first noticed it?”
“Have you ever had it before?”
“Is it painful?” “Is it itchy?”
“Are you sexually active?” If yes, “When was the last time you engaged in sexual activity?”
“Have you been exposed to anyone with a known STI?”
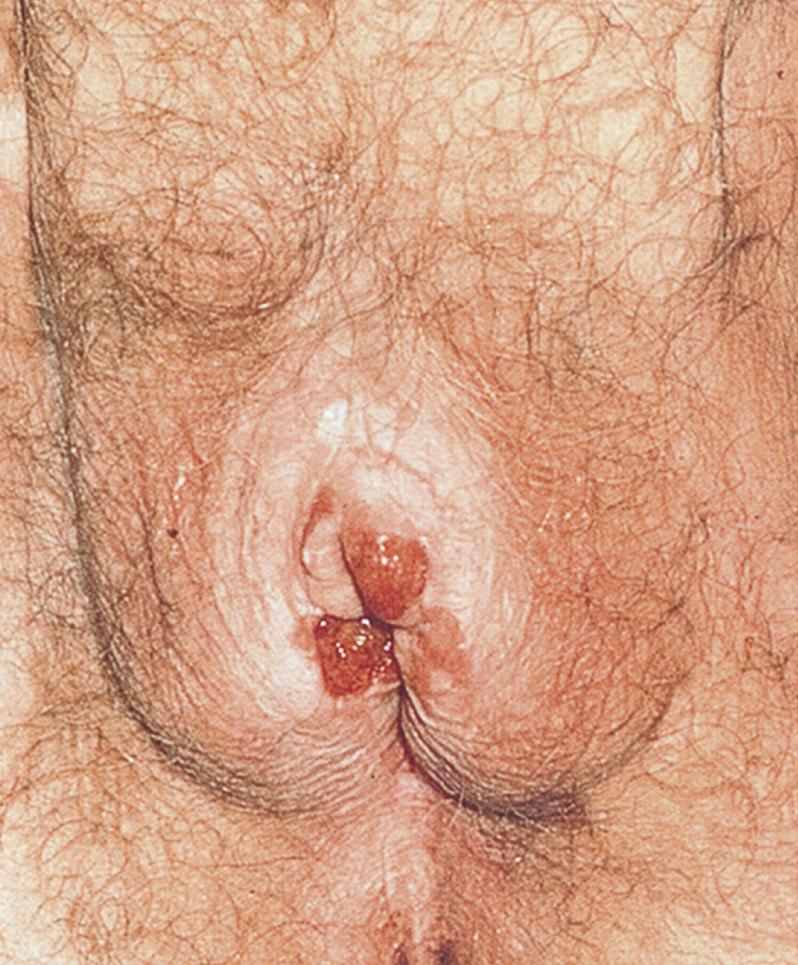
Syphilis may result in a chancre on the labia (see Fig. 19.33 ). Often unnoticed, it is a small, painless ulcer with a sharply demarcated border. Small, acutely painful ulcers may be chancroid (see Fig. 19.34 ) or genital herpes (see Fig. 19.32 ). A patient with an abscess of Bartholin gland (see Fig. 19.17 ) may present with an extremely tender mass in the vulva. Benign lesions, such as genital warts (condylomata acuminate, see Figs. 19.14 and 19.15 ), and malignant conditions manifest as a mass on the external genitalia.
Some affected patients complain of a sensation of fullness or mass in the pelvis, incontinence, and sexual dysfunction as a result of pelvic organ prolapse. Pelvic organ prolapse refers to the descent or protrusion of the vaginal walls, cervix, or uterus into the vagina or through the vaginal introitus. This is caused by a weakening of the pelvic support ligaments. The anterior vaginal wall can descend, producing a cystocele that triggers urinary symptoms such as frequency and stress incontinence. The posterior vaginal wall can descend, producing a rectocele, which triggers bowel symptoms such as constipation, tenesmus, or incontinence. The uterus can also descend, which results in uterine prolapse. In the most severe state, the uterus may lie outside the vulva with complete vaginal inversion, a condition known as procidentia. The consequences of pelvic organ prolapse are discussed further in the section titled “ Clinicopathologic Correlations ” later in this chapter.
Vaginal discharges, also known as leukorrhea, are common. Is there an associated foul odor? Although a whitish discharge is often normally present, a malodorous discharge often indicates a pathologic problem. The most common pathologic odor, associated with bacterial vaginosis, is a foul, fishy odor related to the volatilization of amines that are produced by anaerobic metabolism of a variety of bacteria. Is itching also present? Women with candidiasis complain of intense pruritus and a white, dry discharge that looks like cottage cheese. Has the woman recently taken any medications, such as antibiotics? Antibiotics change the normal vaginal flora, and an overgrowth of Candida may result. Table 19.1 summarizes the important characteristics of vaginal discharge.
| Feature | Physiologic Discharge | Nonspecific Vaginitis | Trichomonas | Candida | Gonococcal |
|---|---|---|---|---|---|
| Color | White | Gray | Grayish-yellow | White | Greenish-yellow |
| Fishy odor | Absent | Present | Present | Absent | Absent |
| Consistency | Nonhomogeneous | Homogeneous | Purulent, often with bubbles | Cottage cheese–like | Mucopurulent |
| Location | Dependent | Adherent to walls | Often pooled in fornix | Adherent to walls | Adherent to walls |
| Discharge at introitus | Rare | Common | Common | Common | Common |
| Vulva | Normal | Normal | Usually normal | Erythematous | Erythematous |
| Vaginal mucosa | Normal | Normal | Usually normal | Erythematous | Normal |
| Cervix | Normal | Normal | May have red spots | Has patches of discharge | Has pus in os |
Vaginal and vulvar itching is often associated with infections, glycosuria, 7
7 High levels of glucose in the urine, as in diabetes.
vulvar dermatitis, vulvar dystrophies, or vulvar neoplasia. Pruritus may also be a symptom of a systemic disease, such as Crohn disease.
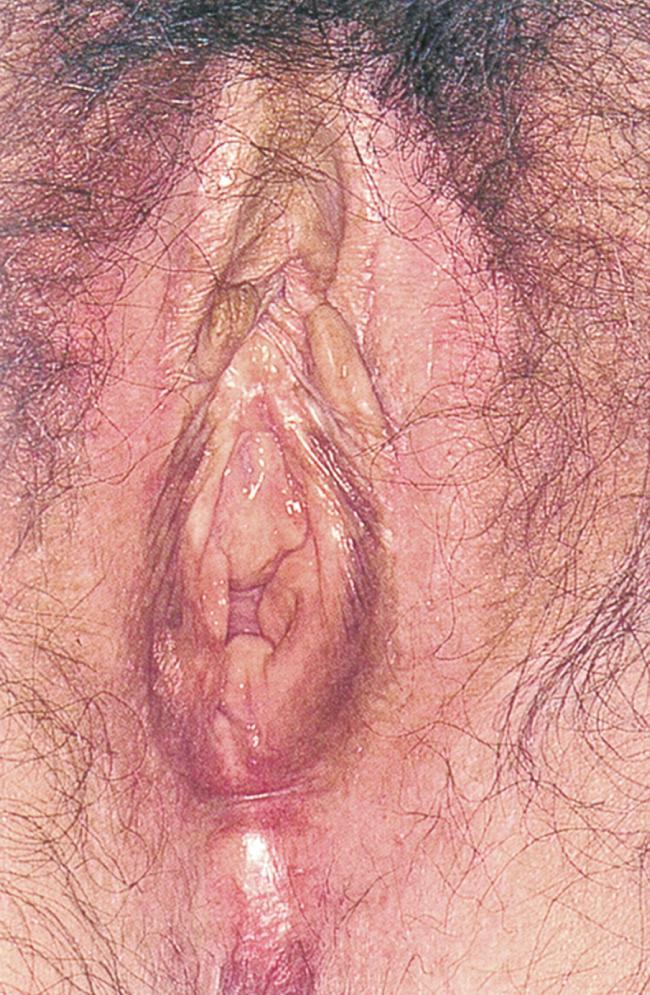
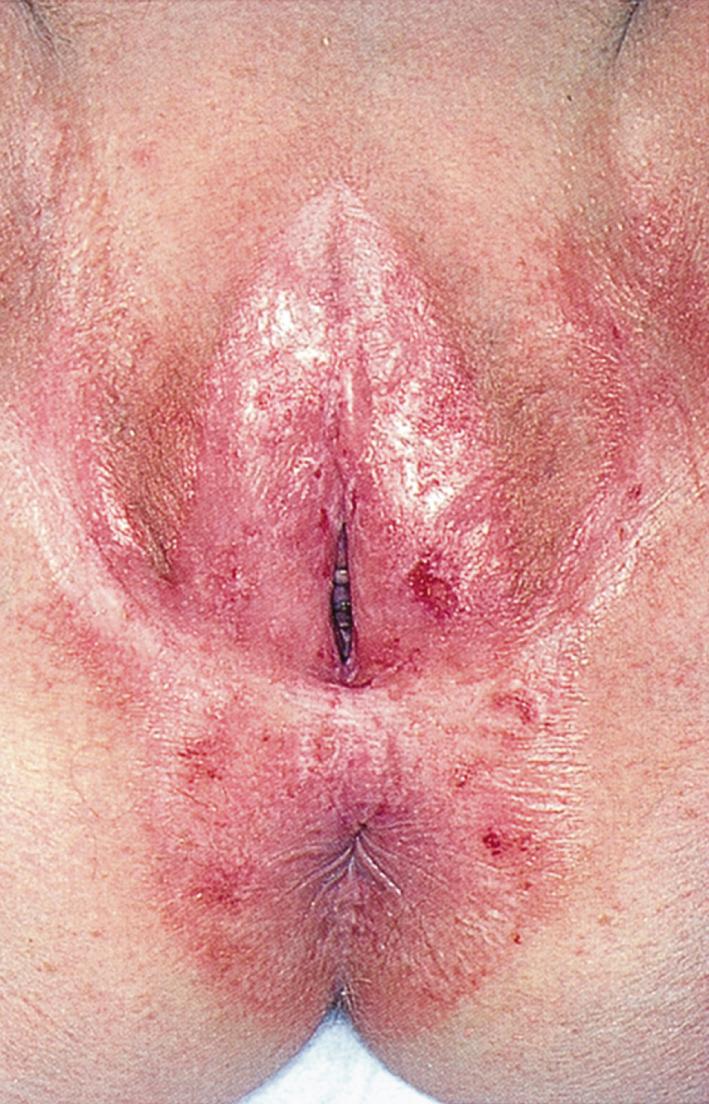
An example of a vulvar dystrophy is lichen sclerosus, a relatively common condition in which the genital skin shows a uniform reddened, smooth, shiny, almost transparent appearance. It is a destructive inflammatory condition with a predilection for genital skin. It is much more common in women, although it can be seen in men with involvement of the glans penis and foreskin. Whitish atrophic patches of thin skin are typical, as is fine crinkling of the skin. Pruritus is a common symptom, and the fragile skin is susceptible to secondary infection. Although most common in white and Latino postmenopausal women, lichen sclerosus may be seen in patients of all ages. It is rare in African-American women. Lichen sclerosus should be thought of as a premalignant lesion because one complication is the development of squamous cell carcinoma. Fig. 19.6 shows an early stage of lichen sclerosus in a female patient. Notice the resorption of the labia minora; the clitoris is preserved. Fig. 19.7 shows a later stage in another patient. Notice the classic whitish crinkling of the skin and the resorption of the labia and clitoris. Fig. 19.8 shows vulvar squamous cell carcinoma with the background of lichen sclerosus in another patient.
Ask the following questions, in addition to those listed in Chapter 17 , The Abdomen, of any woman with pelvic pain:
“How long have you been experiencing the pain?”
“What was the first day of your last menstrual period?”
“Are you using any contraceptives?” If no , “Could you be pregnant?”
“Do you notice the pain at any particular time of your menstrual cycle?”
“Are you sexually active?” If yes, “When were you last sexually active?”
“Is the pain related to intercourse?”
“Have you been sexually active with a partner with any known STIs?”
“Do you have abnormal vaginal discharge?”
“Do you experience a burning sensation when you urinate?”
Pelvic pain may be acute or chronic. Is the patient pregnant? Acute abdominal pain may be a complication of pregnancy. Spontaneous abortion, uterine perforation, and ectopic tubal pregnancy all are life-threatening situations. Acute inflammation, or PID, is caused by vaginal bacteria, such as gonorrhea, chlamydia, or bacterial vaginosis, moving from the vagina to the uterus, fallopian tubes, and ovaries. Acute lower pelvic pain localized to one side that occurs at the time of ovulation is termed mittelschmerz . This pain is related to a small amount of intraperitoneal bleeding at the time of ovum release. Urinary tract infection may also cause acute suprapubic pain. Patients with urinary tract infections usually have associated urinary symptoms of burning sensation or frequency.
Chronic pelvic pain may result from ectopic endometrial tissue known as endometriosis, chronic PID, and fibroids, as well as from nongynecologic causes such as interstitial cystitis, inflammatory bowel disease, musculoskeletal conditions, and psychological factors.
Dyspareunia is pain during or after sexual intercourse. To aid in the evaluation and diagnosis of dyspareunia, dyspareunia should be categorized as pain with insertion, vaginal pain, or deep pelvic pain during intercourse. Causes of pain with insertion may include vulvovaginal infections or dystrophies and vaginismus. Vaginismus is a condition of severe pelvic pain and involuntary spasm when there is any attempt at penetration. Patients with this condition may have a history of depression/anxiety, or trauma or abuse, and may require psychological therapy or couples therapy with desensitization to overcome this problem. Mid-vaginal pain is usually attributed to lack of lubrication during intercourse. This can be alleviated with use of lubricants during intercourse, or vaginal estrogen cream if there is vaginal atrophy present. Women with sexual dysfunction, particularly arousal disorders, may also experience lack of genital lubrication during intercourse. This is often treated symptomatically with lubricants and vaginal estrogen in the case of atrophy. Deep pelvic pain during intercourse is often due to endometriosis, PID, interstitial cystitis, irritable bowel syndrome, or a history of sexual abuse.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here