Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The exact pathogenesis of bile duct carcinoma has not been described, but predisposing factors are similar to those causing intrahepatic bile duct neoplasms. It is believed that long-standing inflammation causes metaplasia and, finally, carcinogenesis.
Tumors of the biliary tract constitute 2% of all cancers found at autopsy. The vast majority of bile duct tumors are extrahepatic (87% to 92%). Patients are typically older than those with intrahepatic bile duct tumors (60 to 70 years of age vs. 50 to 60 years), and there is a slight male preponderance. Survival rates are high (50% at 1 year, 39% at 5 years), reflecting the slow course of the disease and better prognosis than intrahepatic tumors as a result of later metastases. Established risk factors for the development of bile duct cancer are similar to those for intrahepatic biliary tumors and include causes of biliary inflammation such as choledochal cysts, parasitic infestation, biliary stones, inflammatory bowel disease; inherited disorders such as alpha-1 antitrypsin deficiency; toxic exposures such a thorium dioxide; and certain medications (e.g., isoniazid).
In general, cholangiocarcinomas are slow growing and locally infiltrative and they metastasize late. Extrahepatic and perihilar tumors have a more indolent clinical course than intrahepatic tumors, with a better prognosis after complete resection.
Symptoms of cholangiocarcinoma result from biliary obstruction with subsequent cholestasis and hyperbilirubinemia. Thus, extrahepatic and perihilar tumors manifest early in the course of disease. Patients develop jaundice and pruritus and may also report acholic (clay-colored) stools and bilirubinuria (dark urine).
Other symptoms include increased bleeding and bruising, right upper quadrant or epigastric pain, and diarrhea. Prolonged biliary obstruction is associated with complications such as cholangitis, cirrhosis, renal dysfunction, and progressive malnutrition. On physical examination, hepatomegaly may be present. With tumors distal to the cystic duct takeoff, a painless enlarged gallbladder may be identified (Courvoisier's sign).
The liver consists of bile-secreting hepatocytes draining into bile canaliculi, which merge into hepatic ductules to form the biliary tree. The main right and left hepatic ducts drain their respective lobes and converge just outside the liver to form the common hepatic duct. The convergence of the left and right hepatic ducts is extrahepatic. The common hepatic duct is then joined by the cystic duct to form the common bile duct (CBD). The CBD continues inferiorly, joining the pancreatic duct at the ampulla of Vater to drain into the second part of the duodenum ( Figure 55-1 ).
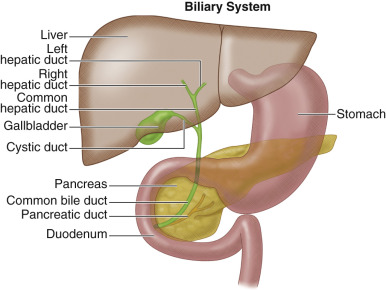
Bile duct tumors are classified as extrahepatic (87% to 92%) or intrahepatic (8% to 13%). Extrahepatic tumors occur in the major bile ducts, whereas intrahepatic tumors develop in smaller ductal branches within the liver. Tumors located at the hepatic duct bifurcation are the least common and are termed perihilar or Klatskin's tumors. Morphologically, extrahepatic biliary tumors may exhibit exophytic, polypoid, infiltrating, or combined growth patterns.
The extrahepatic biliary tree is divided into proximal, middle, and distal regions, with anatomic divisions at the level of the cystic duct and upper duodenum. Of bile duct tumors, 50% to 75% are localized in the upper third, 10% to 25% in the middle third, and 10% to 20% in the lower third. Among proximal extrahepatic tumors, 10% to 26% involve the confluence of the ducts (Klatskin's tumor). Among distal tumors, the common hepatic duct is involved in 14% to 37%, the cystic duct in 6%, and the ampulla of Vater in 0.2% of cases.
Cholangiocarcinomas most commonly disseminate via the lymphatic system. Extrahepatic tumors spread to the cystic and CBD nodes in 32% of cases, the celiac nodes in 16% of cases, and the peripancreatic and superior mesenteric nodes on rare occasions. Hematogenous spread to the liver, peritoneum, and lungs is extremely rare. Direct extension of tumor results in infiltration of adjacent liver in 23% of cases and in peritoneal seeding in 9% of cases.
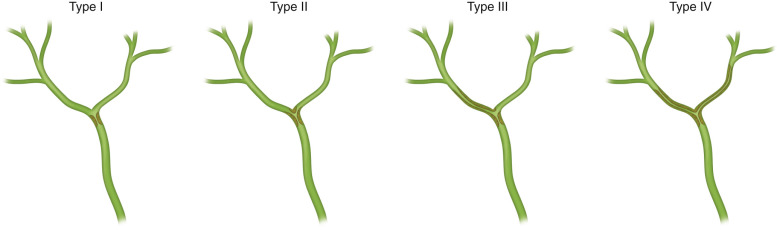
Staging of extrahepatic cholangiocarcinoma is performed using the American Joint Committee on Cancer Staging Tumor, Node, Metastasis (TNM) system of classification ( Table 55-1 ). Klatskin's tumors are categorized using the Bismuth classification ( Table 55-2 and Figure 55-2 ).
| A: STAGING FOR PERIHILAR BILE DUCT TUMORS | |
|---|---|
| TNM Definition | Tumor Location |
| TX | Primary tumor cannot be assessed |
| T0 | No evidence of primary tumor |
| Tis | Carcinoma in situ |
| T1 | Tumor confined to the bile duct, with extension up to the muscle layer or fibrous tissue |
| T2a | Tumor invades beyond wall of the bile duct to surrounding adipose tissue |
| T2b | Tumor invades adjacent hepatic parenchyma |
| T3 | Tumor invades unilateral branches of the portal vein or hepatic artery |
| T4 | Tumor invades main portal vein or its branches bilaterally; or the common hepatic artery; or the second-order biliary radicals bilaterally; or unilateral second-order biliary radicals with contralateral portal vein or hepatic artery involvement |
| NX | Regional lymph nodes cannot be assessed |
| N0 | No regional lymph node metastasis |
| N1 | Regional lymph node metastasis (including nodes along the cystic duct, common bile duct, hepatic artery, and portal vein) |
| N2 | Metastasis to periaortic, pericaval, superior mesentery artery, and/or celiac artery lymph nodes |
| M0 | No distant metastasis |
| M1 | Distant metastasis |
| Type | Involvement |
|---|---|
| I | Common hepatic duct |
| II | Bifurcation without secondary intrahepatic ducts |
| IIIA | Right secondary intrahepatic duct |
| IIIB | Left secondary intrahepatic duct |
| IV | Bilateral secondary intrahepatic ducts |
| V | Junction of common bile duct and cystic duct |
Rarely, extrahepatic bile duct tumors may represent adenomas, hamartomas, cystadenomas, and papillomas, but the vast majority are malignant cholangiocarcinomas. Lesions are thought to develop through an adenoma-carcinoma sequence, stimulated by recurrent biliary obstruction and inflammation with resulting cholestasis.
Histologically, the majority (95%) of malignant cancers are mucin-secreting adenocarcinomas with varying levels of differentiation like their intrahepatic counterpart. Nonsecretory apudomas have occasionally been reported in the hilar region.
Pathologic subtypes include scirrhous, nodular, and papillary cancers. Extrahepatic scirrhous tumors can be seen in the hilum. The nodular variety forms irregular strictures in the ductal walls and is most common in the distal ducts and ampulla. Papillary tumors are typically intraluminal, highly vascular and friable, and most commonly seen in the intrahepatic ducts.
Microscopic findings of extrahepatic cholangiocarcinomas mirror intrahepatic cholangiocarcinoma and anisonucleosis, and distended intracytoplasmic lumens support the diagnosis of bile duct cancer. Histologically dedifferentiated tumors have a worse prognosis. Immunohistochemical staining for carcinoembryonic antigen (CEA) and CA19-9 may facilitate diagnosis. Experimental studies have also identified mutations in the TP53 tumor suppressor gene, c-erb oncogene, chromosomes 5 and 17, epidermal growth factors, and nuclear antigens.
Tumors located distal to the cystic and pancreatic ducts also may disrupt the functions of the gallbladder and pancreas, respectively.
Bile duct tumors are highly variable in location and morphology and may require multiple imaging studies for complete characterization. In general, extrahepatic tumors are smaller than their intrahepatic counterparts and more difficult to identify with imaging. Imaging is also used to evaluate vascular and ductal anatomy, secondary signs of obstruction, and metastatic disease.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here