Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The disparity between supply and demand of organs for transplantation continues to grow, and the organ shortage has made it necessary to liberalize acceptance criteria to include donors beyond traditional criteria. These donors collectively are known as extended criteria donors . (ECDs) In the past, an “ideal” donor was defined as age less than 40 years, trauma as the cause of death, donation after brain death (DBD), hemodynamic stability, and absence of steatosis, chronic liver disease, or transmissible disease. Use of an allograft from an ECD versus from an ideal donor puts the recipient at higher risk for postoperative complications such as impaired allograft function (delayed graft function or primary graft nonfunction) or transmission of a donor-derived disease (infection, malignancy, or other disease). Although organs from ECDs may not be ideal, they are viable alternatives while awaiting liver transplantation. Centers across the world are using grafts from ECDs with outcomes comparable to grafts from normal donors. Although there is no precise definition of an ECD in liver transplantation, referenced donor factors that have been associated with poorer outcomes or disease transmission include advanced age, hepatic steatosis, positive viral serological study results, donation after cardiac death (DCD), and history of infection, malignancy, or metabolic disease. This chapter examines how each of these factors defines an ECD and affects the use of liver allografts.
As the population ages and the availability of suitable organs decreases, the acceptable age of potential donors has increased. Organs from elderly donors now represent the largest expanding component of the donor organ pool, and their use has had a significant impact on the rate of liver transplantation. Several studies in the literature have demonstrated that outcomes after liver transplantation from elderly donors are comparable to outcomes from younger donors. These studies demonstrate good outcomes with livers from donors older than 60, 70, and even 80 years. Although there is no absolute age limit, several points about older liver allografts deserve emphasis.
Elderly donors have decreased physiological reserve and frequent comorbidities that may affect organ procurement. The recovery surgeon should work in concert with the organ procurement, anesthesia, and hospital teams to optimize donor physiological condition and operating room conditions. The presence of atherosclerotic disease, aneurysmal disease, and prior cardiothoracic or abdominal surgery may require modification of the methods normally used to procure the liver safely. Characteristic features of older liver allografts include smaller size, steatosis, capsular fibrosis, and arterial atherosclerosis. Careful inspection of the liver before and after cold perfusion is necessary to evaluate the degree of these features and how they might affect the intended recipient. In addition, the higher incidence of undiagnosed malignancy in elderly donors requires meticulous examination of the entire operative field to safeguard against the transmission of a previously unknown cancer.
Allografts from elderly donors are more susceptible to cold ischemia time and can have increased delayed graft function and prolonged cholestasis consistent with significant ischemia-reperfusion injury. Thus minimizing cold ischemia time is of paramount importance. In addition, selected recipients of older allografts should be able to endure a degree of delayed graft function. Older liver allografts also have an increased risk for hepatitis C virus recurrence and should be used with caution in patients with hepatitis C–related liver disease. Data in the literature consistently demonstrate earlier hepatitis C recurrence, increased graft failure, and decreased survival in hepatitis C–positive recipients of allografts from donors older than 60 years.
In summary, liver transplantation outcomes with allografts from elderly donors can be excellent provided there are no additional risk factors. Age alone therefore should not exclude potential liver donors, especially for recipients who might otherwise have a prolonged wait on the list. Use of allografts from elderly donors should be limited to those with minimal steatosis. Attention to other donor risk factors, recipient factors, and especially cold ischemia time is necessary to ensure optimal outcomes after transplantation of older liver allografts.
Hepatic steatosis is one of the most important factors affecting allograft function. Early functional recovery and regenerative capacity of liver allografts are significantly impaired when steatosis is present, likely because of more severe ischemia-reperfusion injury. With the growing obesity epidemic, transplant surgeons are encountering more hepatic steatosis in donors. The prevalence of steatosis within developed countries ranges between 10% and 30% of the population. Steatosis is most common in donors with a history of obesity, diabetes, and alcohol consumption, and the presence of these factors should prompt careful evaluation for steatosis at the time of procurement. Inspection at procurement helps detect steatosis ( Fig. 41-1 ), but biopsy is the gold standard to grade the degree of steatosis.
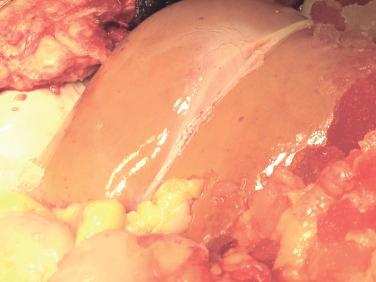
Steatosis can be subdivided into microvesicular or macrovesicular steatosis. In microvesicular steatosis, multiple small fat vacuoles occupy the cytoplasm of the hepatocyte. In macrovesicular steatosis, one large fat vacuole replaces most of the cytoplasm and displaces the nucleus of the hepatocyte. Whereas microvesicular steatosis has minimal effect on graft function, macrovesicular steatosis has been associated with poor graft function. Macrovesicular steatosis can be subcategorized as mild (<30%), moderate (30% to 60%), and severe (>60%). Substantial data have correlated the extent of macrovesicular steatosis with the incidence of graft dysfunction. Mild macrosteatosis has minimal impact on graft function, provided that the cold ischemia time is short. Allografts with mild macrosteatosis can be used safely in select recipients. Moderate macrosteatosis has variable effect on outcomes, and use of these grafts remains challenging. The incidence of delayed function approaches 35%, and primary nonfunction is reported as high as 15% in grafts with moderate macrosteatosis. Careful donor evaluation to exclude additional risk factors, careful selection of recipients who can tolerate potential delayed graft function, and minimization of cold ischemia time are paramount to the successful use of moderately steatotic allografts. Allografts with severe macrosteatosis have a high rate of primary nonfunction and should not be used. Any degree of fibrosis associated with steatosis also should preclude the use of the graft.
Liver allografts from donors with positive viral serologic study results should be used only if the recipient is already infected with the same agent or has a critical need for liver transplantation. Use of serologically positive allografts requires a frank discussion with the recipient and informed consent before transplantation. Several studies in the literature report favorable outcomes with the use of liver allografts from patients with positive serological study results for hepatitis C virus (HCV), hepatitis B virus (HBV), and human T-cell lymphotropic virus 1/2 (HTLV-1/2).
Approximately 5% of all potential organ donors in the United States and Europe are positive for antibody to HCV, and half of these donors are positive for HCV RNA by polymerase chain reaction. HCV-positive allografts without severe inflammation or fibrosis are best used for recipients with HCV-related liver disease. Data in the literature clearly indicate that there is no difference in HCV recurrence, graft survival, or patient survival with the use of HCV-positive allografts in HCV-positive recipients compared to recipients receiving an HCV-negative allograft. Studies also have examined the dynamic interaction of donor and recipient HCV strains after liver transplantation and found no consistent pattern of viral repopulation. Liver recipients in whom the donor HCV strain becomes predominant can have significantly longer disease-free survival than recipients who retain their own HCV strain. Because genotype is predictive only of response to interferon-based therapy and not disease severity, genotype is not an important consideration in the decision to transplant a liver from an HCV-positive donor. In summary, the literature shows that transplanting an HCV-positive allograft into an HCV-positive recipient is safe with long-term outcomes comparable to HCV-negative allografts. The procurement team should consider donor liver biopsy to evaluate for severe inflammation or fibrosis, the presence of which should preclude use of the allograft. The use of HCV-positive liver allografts for HCV-negative recipients or for HCV-positive recipients with an undetectable viral load should be reserved for cases of extreme necessity.
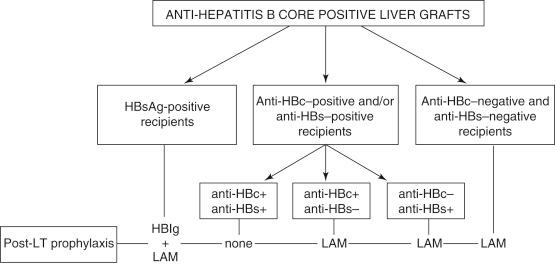
The risk for transmission of HBV via liver transplantation is high. However, allografts from hepatitis B surface antigen (HBsAg)-negative/hepatitis B core antibody (HBcAb)-positive donors are being used to expand the donor pool with comparable graft and patient survivals. HBcAb-positive donors represent 3% to 6% of the donor pool in the United States, 8% to 15% of the donor pool in Europe, and 50% to 55% of the donor pool in Asia, and the need to use HBcAb-positive allografts varies with the regional incidence of HBV. Persistent intrahepatic HBV DNA in HBsAg-negative/HBcAb-positive patients is a reservoir for potential viral reactivation, particularly in the setting of posttransplant immunosuppression. Thus, in the absence of postoperative antiviral prophylaxis, the rate of de novo HBV infection is reported as high as 75% to 80% in HBsAg-negative/HBcAb-negative recipients, 15% to 20% in HBsAg-positive or HBcAb-positive recipients, and 5% to 10% in hepatitis B surface antibody (HBsAb)-positive/HBcAb-positive recipients. The presence of antibodies to hepatitis B surface antigen and hepatitis B core antigen in the recipient reduces the rate of HBV infection after transplantation with an HBcAb-positive liver allograft. Liver allografts from HBcAb-positive donors thus should be used preferentially in recipients who are undergoing transplantation for HBV-related liver disease. Because of the effectiveness of anti-HBV medications, HBcAb-positive liver allografts also can be used safely in HBsAg-negative/HBcAb-negative recipients but should be reserved for cases of urgent need and in areas with high HBV prevalence when possible. Lifelong compliance with anti-HBV prophylaxis and permanent surveillance post transplantation are imperative ( Fig. 41-2 ). Should de novo infection occur in the recipient, various treatment strategies with anti-HBV medications and hepatitis B immune globulin are ultimately successful in HBV DNA negative seroconversion.
Transplantation of liver allografts from donors with HTLV-1/2 infection risks transmission to the recipient and subsequent development of either adult T-cell leukemia or myelopathy (tropical spastic paraparesis). The majority of HTLV-1 infections do not cause disease in immunocompetent hosts, and HTLV-2 has not been linked to human disease. However, the immunocompromised status of liver transplant recipients may increase their risk for developing HTLV-associated disease. In the United States the prevalence of HTLV-1/2 infection is estimated to be 0.05% to 0.1%, and therefore most donors are not routinely tested for HTLV-1/2. The prevalence can be as high as 30% in endemic areas such as the Caribbean, South America, Africa, and Asia. Studies from across the world thus demonstrate varying rates of transmission of infection and development of disease. Despite the small but real risk for developing HTLV-associated disease after liver transplantation, HTLV-positive allografts can be used for select recipients, including those who have urgent need, older recipients, and patients who are HTLV-positive before transplantation. Recipients of HTLV-positive liver allografts should undergo heightened screening after transplantation.
Unexpected transmission of human immunodeficiency virus (HIV), HCV, and HBV through organ transplantation remains a patient safety and public health concern. The Centers for Disease Control and Prevention (CDC) has identified certain criteria that confer increased risk for recent HIV, HCV, and HBV infection in the donor ( Table 41-1 ). Donors who meet these criteria are designated as CDC high-risk . Despite advances in screening for these pathogens, the risk for transmission through liver transplantation persists. Infection in the donor may go undetected because multiple blood transfusions confound testing by serum dilution or because the testing happened during the window period for the infection. When considering an allograft from a CDC high-risk donor, the transplant team must weigh the risk for transmission of a potentially fatal disease against the risk for recipient death on the waiting list. These risks can be difficult to quantify and must be individualized to the specific donor and recipient. Frank disclosure about the risks and voluntary consent from the recipient are mandatory before transplantation. After transplantation, recipients of allografts from CDC high-risk donors should undergo appropriately timed screening for HIV, HCV, and HBV.
|
|
|
|
|
|
|
|
|
|
|
Donors who meet the following criterion should be identified as being at increased risk for recent HCV infection only:
|
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here