Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Acute, severe, exfoliative and necrotizing diseases of skin and underlying structures may cause significant morbidity and mortality in the afflicted patient. The problems associated with these diseases, such as wound infection, sepsis, inadequate nutrition, and pain, are similar to those seen in patients with major burns. Thus burn centers, with their multidisciplinary teams, have been advocated to provide the treatment and management for this unique, critically ill patient population. This chapter describes the pathophysiological processes of severe exfoliative skin disorders, their diagnosis, and the specialized treatment offered by burn centers.
This classification of exfoliative diseases of the skin and mucous membranes historically consisted of three specific maladies, distinguished by the amount of skin involvement: erythema multiforme major (EM), Stevens–Johnson syndrome (SJS), and toxic epidermal necrolysis (TEN). For the terms of this chapter, we are maintaining that approach. However, some recent classifications list EM as its own distinct disease entity separate from SJS and TEN and most commonly associated with the herpes simplex. This new designation does not affect the classifications of SJS and TEN: Each are still determined by the extent of total body surface (TBSA) affected by the disease process. SJS is considered to affect <10% total body surface area (TBSA), whereas TEN covers >30% TSBA, leaving a zone of overlap between 10% and 30% TBSA, which is referred to as SJS/TEN. The most common characteristics of these disease entities are defined in Table 42.1 .
| Erythema Multiforme | Stevens–Johnson Syndrome | Toxic Epidermal Necrolysis | |
|---|---|---|---|
| Prodrome | Absent | High fever, malaise | High fever, malaise |
| Acute phase | 4–8 days | 4–8 days Sensation of skin burning or tenderness |
Sudden onset, 1–2 days Sensation of skin burning or tenderness |
| Skin lesions | Symmetrical, primarily located on the extremities, some target lesions without blisters | Variable distribution, individual vesicles on an erythematous base <10% TBSA Nikolsky's positive |
Diffuse generalized epidermal detachment, absence of target lesions, large confluent plaques >30% TBSA Nikolsky's positive |
| Mucosal involvement | Limited to one surface, usually oral | Severe, two or more surfaces involved | Severe, two or more surfaces involved |
| Histopathology | Dermoepidermal separation, mononuclear perivascular cell infiltrate, small areas of epidermal detachment associated with target lesions | Dermoepidermal separation, more intense dermal infiltrate, areas of epidermal detachment | Epidermal necrosis, dermoepidermal separation, minimal dermal inflammatory infiltrate, large areas of epidermal detachment |
| Recovery | 1–4 weeks | 1–6 weeks | 1–6 weeks |
| Mortality | 0% | 0–38% | 25–80% |
TEN represents the severe extreme of the spectrum of necrolytic disorders. A rare disease, the incidence of TEN in this country is estimated at 0.4–1.9 cases per million persons per year and that of SJS has been reported to be only slightly higher at 1–7 cases per million persons per year. This incidence is similar to 1.89 cases of TEN per million persons per year reported for Europe in 1996, but higher than reported for Asia. The overall rarity of this disease process has been, and continues to be, a barrier to research. However, over the past several decades, the concentration of physician experience with TEN into a limited set of regional burn centers has facilitated an increasing quantity and quality of active research.
These exfoliative disorders occur in all age groups; however the incidence is increased in the elderly and females. In addition, TEN has been linked to certain infectious disease processes. The annual incidence of TEN in the human immunovirus (HIV)-infected population is 1000-fold higher than in the general public. Whether this increase is due to their immunocompromised state or to the increased prescription of high-risk drugs, particularly sulfonamides, is debated. However cases with no drug history or preceding illness have been reported. An additional infectious disease process associated with SJS/TEN is Mycoplasma pneumonia. Finally idiopathic cases not related to drugs account for 3–4% of TEN.
On the surface, these exfoliative disorders may mimic the symptoms of a partial-thickness thermal injury, specifically in relation to the involvement of the skin. However these diseases also affect all epithelial surfaces, thus increasing the risk and incidences of infections, other complications, and death. Patients suffering from particularly complicated cases of SJS may benefit from the expertise of a burn center, although these patients are generally referred on an as-needed basis. In contrast, those diagnosed with TEN carry a specific set of severe and complicated medical needs that are clearly best addressed in the context of a specialized burn center.
Mortality of TEN ranges from 25% to 80%, with morbidity being reported as high as 65%. However reports are variable and usually based only on small patient populations. Death may occur early in the course of the disease, with sepsis being the most frequent cause. Pseudomonas aeruginosa and Staphylococcus aureus are the predominant organisms involved. Pulmonary embolism and gastrointestinal hemorrhage are other causes of death.
Mortality is increased significantly in those patients at the extremes of age and in relation to the percentage of denuded skin and serum urea nitrogen levels. Bastuji-Garin et al. in 2000 created the Score of Toxic Epidermal Necrolysis (SCORTEN), a scoring system to predict mortality in the adult population presenting with TEN. The scoring system uses seven independent risk factors to predict mortality by allotting 1 point for each variable:
Age older than 40
Heart rate greater than 120 beats per minute
Comorbid malignancy
Epidermal detachment of greater than 10% of body surface area (BSA) on day 1
Blood urea nitrogen of greater than 28 mg/dL
Glucose of greater than 252 mg/dL
Bicarbonate less than 20 mEq/L.
The scoring system should be performed on day 1 and day 3 postadmission to maximize its predictive value. Mortality from TEN increases from 3.2% for a score of 0–1 to 90% for a score greater than 5 ( Table 42.2 ).
| SCORTEN (Sum of Scores) | Predicted Mortality |
|---|---|
| 0–1 | 3.2% |
| 2 | 12.1% |
| 3 | 35.8% |
| 4 | 58.3% |
| >5 | 90% |
While originally developed for adults, the scoring system has been validated in the pediatric population, but overall has been challenged for its lack of incorporating no more than one morbidity (malignancy) in its calculation. While other investigators have developed formulas based on logistic regression analysis, the SCORTEN system is the most widely utilized.
SJS is associated with a mortality rate of 0–38%. EM rarely causes death.
Keratinocyte apoptosis and secondary epidermal necrosis represent the pathopneumonic cellular process of TEN. The origins of the cellular process remain an active area of research, with an intricate meshwork of overlapping risk factors and processes starting to emerge as the picture sharpens. What has become clear is that the TEN disease process results from a complicated interaction of multiple processes and risk factors: genetic predisposition, environmental triggers, immunologic reaction, inflammatory mediators, and cellular apoptotic machinery.
TEN appears to be driven by immunological reactions to foreign antigens, often referred to as “triggers.” Medications are by far the most commonly identified trigger, implicated in 77–94% cases of TEN. Antimicrobials, anticonvulsants analgesics, and nonsteroidal antiinflammatory agents of the oxicam type have been implicated. Naturally the particular profile of TEN instigators will vary from population to population depending on the epidemiology of medication exposures in any given population.
Attempts to identify drugs suspected of having caused exfoliative necrolysis by skin test and laboratory tests seldom have been successful. While some assays are trialed in research settings, there are currently no fully vetted, reliable methods available for identifying the inciting trigger in a patient with TEN. Historically the medication-specific trigger for any given case of TEN was ultimately a matter of speculation. However Sassolas et al. in 2010 developed an algorithm of drug causality for epidermal necrolysis termed ALDEN. The algorithm assigns a score of 1 to 10 for each drug based on six parameters. The score is then categorized as very probable (>6), probable (4–5), possible (2–3), unlikely (0–1), and very unlikely (<0) ( Table 42.3 ). The results of this algorithm were compared to a case-controlled analysis of the European Study of Severe Cutaneous Adverse Reactions (EuroSCAR) and found to have good accordance. The use of ALDEN in the clinical setting may greatly improve the ability for clinicians and researchers to link medication-specific triggers and TEN.
| Criterion | Values | Rules to Apply | |
|---|---|---|---|
| Delay from initial drug component intake to onset of reaction (index day) | Suggestive +3 | From 5 to 28 days | −3 to 3 |
| Compatible +2 | From 29 to 56 days | ||
| Likely +1 | From 1 to 4 days | ||
| Unlikely −1 | >56 Days | ||
| Excluded −3 | Drug started on or after the index day | ||
| In case of previous reaction to the same drug, only changes for: | |||
| Suggestive: +3: from 1 to 4 days | |||
| Likely: +1: from 5 to 56 days | |||
| Drug present in the body on index day | Define 0 | Drug continued up to index day or stopped at a time point less than five times the elimination half-life a before the index day | −3 to 0 |
| Doubtful −1 | Drug stopped at a time point prior to the index day by more than five times the elimination half-life a but liver or kidney function alterations or suspected drug interactions b are present | ||
| Excluded −3 | Drug stopped at a time point prior to the index day by more than five times the elimination half-life a , without liver or kidney function alterations or suspected drug interactions b | ||
| Prechallenge/rechallenge | Positive specific for disease and drug: 4 | SJS/TEN after use of same drug | −2 to 4 |
| Positive specific for disease and drug: 2 | SJS/TEN after use of similar c drug or other reaction with same drug | ||
| Positive unspecific: 1 | Other reaction after use of similar c drug | ||
| Not done/unknown: 0 | No known previous exposure to this drug | ||
| Negative −2 | Exposure to this drug without any reaction (before or after reaction) | ||
| Dechallenge | Neutral 0 | Drug stopped (or unknown) | −2 or 0 |
| Negative −2 | Drug continued without harm | ||
| Type of drug (notoriety) | Strongly associated 3 | Drug of the “high-risk” list according to previous case–control studies | −1 to 3 |
| Associated 2 | Drug with definite but lower risk according to previous case–control studiesd | ||
| Suspected 1 | Several previous reports, ambiguous epidemiology results (drug “under surveillance”) | ||
| Unknown 0 | All other drugs including newly released ones | ||
| Not suspected −1 | No evidence of association from previous epidemiology stud with sufficient number of exposed controls c | ||
| Intermediate score = total of all previous criteria | −11 to 10 | ||
| Other cause | Possible −1 | Rank all drugs from highest to lowest intermediate score | −1 |
| If at least one has an intermediate score >3, subtract 1 point from the score of each of the other drugs taken by the patient (another cause is more likely) | |||
| Final score | −12 to 10 |
a Drug (or active metabolite) elimination half-life from serum and/or tissues (according to pharmacology textbooks, tentative list available in complementary table), taking into account kidney function for drugs predominantly cleared by kidney and liver function for those with high hepatic clearance.
b Suspected interaction was considered when more than five drugs were present in a patient's body at the same time.
c Similar drug = same ATC code up to the fourth level (chemical subgroups), see Methods.
While advances have been made in correlating medications as the causative agent of TEN, the role of nonmedication triggers in the etiology of TEN remains murky. Upper respiratory tract infections, pharyngitis, otitis media, or viral illness are frequently reported as preceding or coinciding with the development of TEN in some patients. Mycoplasma pneumoniae and herpes viruses (cytomegalovirus, Epstein–Barr virus, herpes simplex, and varicella zoster) have been implicated in the cause of EM and SJS, but not TEN. Quantifying the association between TEN and antecedent viral syndromes is complicated by the rarity of TEN and the similarities between prodromal symptoms and other more common disorders. This leads to patients presenting with relatively prominent prodromal symptoms who are typically misdiagnosed as having a simple viral syndrome. Because of the delay between early prodromal symptoms and the more specific TEN rash, it is often impossible to determine in retrospect whether the pertinent symptoms were actually prodromal or if the patient had an antecedent viral infection. Furthermore, these prodromal symptoms are often treated with medications before the TEN diagnosis is considered. As such, medications used to treat prodromal symptoms are frequently misinterpreted as TEN triggers.
Genetics clearly plays an important role in the pathophysiology of TEN, with multiple alleles identified as potent risk factors. Interestingly, the genetic risk factors identified thus far all appear to be trigger-specific. That is, a particular allele may place a person at risk for developing TEN in response to one drug, but not increase that same person's risk for developing TEN in response to a different medication. The majority of those alleles identified thus far appear to increase risk through two separate mechanisms: drug–antigen metabolism and antigen presentation. As early as 1986, Shear et al. identified a pattern of “slow” sulfonamide metabolism in patients with a history of severe cutaneous sulfonamide reactions. Insights and technologies developed through the maturation of the field have allowed pharmacogeneticists to trace such associations with polymorphisms in genes encoding for components of the cytochrome P450 machinery. Investigators studying TEN patients' genotypes have assembled a sizable catalog of cytochrome P450C variants that convey an increased risk for developing TEN, with each polymorphism associated with a specific trigger agent. The largest family of genes with a documented association with TEN include polymorphisms at a number of HLA loci that have been associated with drug-specific risks for TEN. These polymorphisms presumably alter the morphology of the HLA molecules on antigen-presenting cells, making them more or less inclined to “recognize” a particular antigen. The “antigen” may be the medication molecule, a drug metabolite, a byproduct of drug metabolism, or a hapten created by the binding of the drug, metabolite, or byproduct to another peptide.
The clinical application of these genetic markers is complicated by the fact that their predictive value varies widely when applied to populations of different ethnic makeups. An example of this strong association between HLA, drug sensitivity, and ethnic background was discovered by Chung et al. They demonstrated a strong association between HLA-B*1502, SJS, and carbamazepine in Han Chinese. This unique relationship was later observed in a Thai population, but was absent in the Japanese and European populations. In fact, RegiSCAR, a large European study, suggested that HLA-B*1502 is not a marker for carbamazepine, sulfamethoxazole, lamotrigine, or NSAID oxicam-type-induced SJS or TEN.
Although the HLA-B*1502 and carbamazepine association demonstrated significant ethnic variance, this is not the observation for all HLA–medication interactions. A second strong relationship among HLA, drug sensitivity, and SJS/TEN was observed for allopurinol and HLA-B*5801. While the strength of the relationship continued to vary between ethnicities, the correlation persisted in Han Chinese, Japanese, Thai, and Europeans, unlike for HLA-B*1502 and carbamazepine.
In summary, for many of these markers, prevalence patterns make the marker too rare to function as a screening tool outside the context of specific ethnicities. Beyond issues of prevalence, some markers are associated with a TEN reaction in one population, but not in another: a phenomenon that likely traces to interactions between the “maker” allele and other genetic variables.
Multiple lines of evidence point to T cells as critical effectors of the TEN process. T cells are the predominant cell type found in the blister fluid and exudate of patients with acute TEN.
Suppression or cytotoxic T-cell infiltrates are observed in the epidermis in TEN. The observation of blebbing of the keratinocyte plasma membrane in TEN is considered a reliable morphological finding of cytotoxic T-lymphocyte cytolysis. Serum and exudate levels of markers of T-cell activation have been shown to correlate with disease activity and resolution.
TEN is perhaps best understood as an immunologically mediated burn. For the most part, TEN resembles a classic type IV (delayed) hypersensitivity disorder. The delay between exposure and reaction, the critical role of T cells, and the accelerated reaction reported in cases of re-exposure to an initial trigger all favor this characterization. While some have pointed to immunofluorescence microscopy findings of IgM and C3 deposited along the dermoepidermal junction and dermal vessels in cases of postherpetic SJS and EM as evidence of an associated type II hypersensitivity reaction, others attribute this to simple nonspecific exudation. The exact antigen that activates the T-cell receptor varies depending on the specific trigger agent: the recognized moiety could be a molecule within the medication, a byproduct of agent metabolism, or a complex formed between an endogenous peptide and either of the preceding. Keratinocyte apoptosis is central to the pathogenesis of TEN. This event is thought to be mediated by ligand/receptor interaction of the tumor necrosis factor (TNF) superfamily (as TNF-α/TNF receptor or FasL/Fas interaction). Through a series of experiments, Viard et al. observed, in vitro, that TEN patients expressed lytically active Fas ligand and that the action of this ligand could be blocked by both a monoclonal antibody and human immunoglobins. In SJS, keratinocyte DNA fragmentation has been found in about 90% of cases associated with dermal perforin-positive lymphocytes.
A prodromal phase of TEN is frequently identifiable in retrospect and usually characterized by some combination of low-grade fever, malaise, cough, conjunctivitis, and dysuria. These symptoms typically precede any cutaneous manifestation by 1–21 days, but usually last for 2–3 days. This prodrome precedes the development of a frank rash, although patches of tender erythema and inflamed mucosal membranes are sometimes present. Skin involvement usually begins with subtle patches of tender erythema and localized morbilliform eruptions or discrete erythematous or purpuric macules. Later vesicles and large bullae emerge from areas of erythema, either en masse or via coalescence of initial morbilliform eruptions. On light digital pressure, the epidermis desquamates in sheets: Nikolsky's sign is positive ( Fig. 42.1 ). The TEN rash is extremely painful, even when bullae are still intact. Generally, a lag period of 1–3 weeks is observed from initiation of drug until skin eruption, but this may be shorter, particularly in cases of re-exposure in a previously sensitized individual.
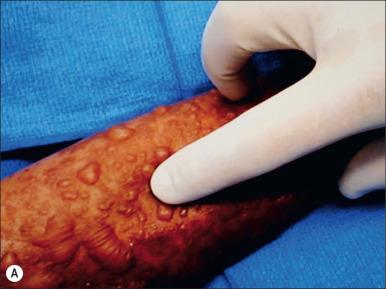
Mucosal involvement is extremely common in TEN, with two or more areas typically involved. These mucosal lesions are often most problematic in that they can cause significant immediate and long-term complications and typically persist (symptomatically) longer than cutaneous lesions. Site involvement usually follows this order of frequency: oropharynx (93%), ocular (78%), genitalia (63%), and occasionally the anal mucosa.
Mucosal involvement precedes skin lesions by 1–3 days in one-third of cases. As such, any case presenting with skin involvement with no mucosal involvement should raise the suspicion for an alternative diagnosis. However it should be noted that detection of mucosal involvement often requires a significant index of suspicion: the fulminant skin findings can frequently distract from more subtle mucosal findings, such as enteral mucosal involvement and bronchial involvement, which are often more easily inferred from symptoms rather than direct visualization.
An early skin biopsy is essential for diagnosis. Skin manifestations vary from patient to patient and with the age of the lesion ( Fig. 42.2 ). Advancing edges of the target lesions show scattered necrotic keratinocytes in the epidermis and only mild dermal inflammation. In older lesions and central zones of target lesions, the dusky appearance corresponds to areas of extensive keratinocyte necrosis, often with the formation of subepidermal bullae and dermoepidermal separation. The surrounding erythematous zone shows papillary dermal edema, vascular dilation with endothelial cell swelling, and perivascular mononuclear cell infiltrate. Extravasated erythrocytes may be seen in the surrounding papillary dermis. The reticular dermis is normal.
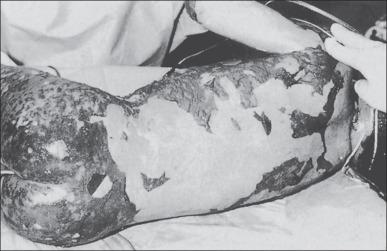
Epidermal and dermoepidermal suppressor or cytotoxic T lymphocytes, in addition to dermal infiltrates of helper T lymphocytes, have been demonstrated. Hertl has confirmed that these epidermal cells are cytotoxic T cells. Langerhans cells appear to be reduced in the epidermis, although numerous dermal macrophages are observed. A more intense dermal cell infiltrate is present in SJS, especially in postherpetic cases. Dendritic lymphoid cells are observed, opposed to damaged dermal macrophages and necrotic keratinocytes. Furthermore, at the point where the cytoplasmic processes contact the keratinocyte, the plasma membrane of the keratinocyte is absent. Aberrant expression of HLA-DR on keratinocytes has been observed, a phenomenon that has been present in many other inflammatory skin disorders.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here