Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
As this is the first chapter considering ovarian tumors, some remarks concerning their evaluation are appropriate. Most of these comments are basic and familiar to experienced pathologists but may be occasionally forgotten with potential failure to frame a correct differential diagnosis. Many, such as a thorough history, pertain to surgical pathology in general and other areas of gynecologic tumor evaluation. Table 13.1 and a further table specific to the ovary highlighting associations that may be helpful ( Table 13.2 ) are derived from tables prepared primarily by the late Dr. Robert E. Scully for an essay considering the evaluation of ovarian tumors based on their patterns and cell types (Young and Scully). We have expanded upon those tables based on our own experience. The various issues are also noted in the text at the appropriate points.
|
|
Although some tumors occur at any age (such as dermoid cysts), others are much more common in specific age groups. Surface epithelial carcinomas are relatively rare before the age of 30 with a progressive increase in frequency thereafter. Conversely, primitive germ cell tumors are usually seen before the age of 30 and only rarely thereafter. These age differences, for example, are helpful in the diagnosis of tumors that occasionally having overlapping morphology, such as yolk sac tumor versus clear cell carcinoma. Another vital clinical feature is the history of a prior neoplasm. Even in the absence of such a history a pathologist may be confronted with an incomplete history requiring a search for more details from the medical record, the clinician, or even the patient. One example is a malignant tumor with oxyphilic cells containing probable melanin pigment making metastatic malignant melanoma a realistic consideration. In this scenario and some others (for example, breast carcinoma and endometrial stromal sarcoma), the history might be so remote as to be unknown to the clinician and not even mentioned by the patient who might think it irrelevant to her current complaint. Similarly, with regard to operative findings, it is obvious that knowledge of any synchronous extra-ovarian tumor might be crucial in reaching the correct diagnosis. For example, a mucinous carcinoma involving the ovary should always raise concern for metastasis if extra-ovarian disease is present. Subtle tumor on the tubal serosa, potentially overlooked especially if there is a much larger ovarian tumor, can also be diagnostically important. Gross features can drive the differential diagnosis or in some cases can be diagnostic (such as a dermoid cyst). Bilateral tumors (particularly with morphology unusual for a primary neoplasm) or mucinous or endometrioid morphology should cause concern for the possibility of metastasis to the ovary. Awareness of the great diversity of patterns and cell types of ovarian tumors and what is potentially encountered within individual categories of neoplasia is obviously crucial in the differential diagnosis (Young and Scully). For example, it is crucial to be aware that sex-cord-like patterns in endometrioid carcinomas are much more common than in other surface epithelial neoplasms, occasionally almost perfectly mimicking sex cord neoplasia. Another example, but pertaining to cell type, is that clear cells can be seen in diverse tumors other than clear cell carcinomas, and without the typical patterns of clear cell carcinoma, that diagnosis should be made with caution.
If after thorough microscopic examination, the diagnosis is still uncertain, additional sampling of the specimen can often facilitate the diagnosis. For example, in the differential of endometrioid carcinoma versus a sex cord tumor, even a minute focus of squamous differentiation on additional sampling would immediately exclude a sex cord tumor. Conventional special stains such as for glycogen, mucin, reticulin, and fat are now infrequently performed but can still have a role. The reticulin stain is particularly helpful in determining whether a granulosa cell component is present in a sex cord–stromal tumor given the overlap in the morphology of granulosa cell tumors and thecomas. Mucin stains, glycogen stains, and fat stains can still potentially aid in evaluating clear cell tumors. Immunohistochemistry can of course have a crucial diagnostic role as indicated in the following chapters but the immunohistochemical staining results should always be placed in context of standard clinical, gross, and conventional microscopic findings. Electron microscopy has largely been replaced by immunohistochemistry but occasionally it may have a role, particularly if a sample is limited, such as lymphoma versus carcinoma or to confirm the presence of neuroendocrine differentiation.
Table 13.2 lists a variety of important gross and microscopic findings associated with ovarian tumors that may be helpful in differential diagnosis. For example, in one case we know of, several experienced pathologists overlooked subtle gross evidence of a dermoid cyst that would have facilitated the diagnosis of oxyphilic struma and prompted confirmatory immunohistochemical stains. One reported case of dermoid cyst with ‘granulosa cell tumor’ was suspect given the propensity for some strumas to mimic a granulosa cell tumor; re-evaluation of that case showed that the ‘granulosa cell tumor’ was thyroglobulin-positive. An ovarian sarcoma, particularly in a young person, may be explained histogenetically if minor foci of associated teratomatous or Sertoli–Leydig elements are identified, sometimes only after rigorous sampling. As noted above, the differential between an endometrioid carcinoma with sex cord-like patterns and a sex cord tumor can be facilitated if additional sampling discloses an endometrioid adenofibromatous component indicating an endometrioid carcinoma with sex cord-like patterns. Perhaps one of the most helpful associated findings in ovarian tumor evaluation is the presence of endometriosis (such as an endometriotic cyst), a finding that would suggest tumors that are commonly endometriosis-related. Some high-grade or undifferentiated ovarian carcinomas arise from low-grade endometrioid neoplasms or a low-grade mucinous cystic tumor, findings that may explain the histogenesis of the undifferentiated cancer. As both Brenner and Sertoli–Leydig cell tumors may occasionally have a prominent mucinous component, the latter's origin is explained if minor elements of the former tumors are identified. Finally, the differential diagnosis of an ovarian mass versus a para-ovarian mass generally differs, although some neoplasms can occur at both sites. Nonetheless the likelihood of a specific tumor is heightened if it is established that the tumor is ovarian or para-ovarian. As there is a tendency for clinicians and pathologists to assume a sizeable adnexal mass is ovarian, if there is any doubt the gross specimen should be re-evaluated to confirm the primary site.
Epithelial ovarian tumors, which are composed of epithelial elements and often a stromal component that is usually fibrous and derived from ovarian stroma, are traditionally considered to originate from the ovarian surface epithelium (or its derivatives, epithelial inclusion glands, and cysts) and the ovarian stroma. However some of these tumors have a different histogenesis .
Recent observations suggest that a subset of ‘ovarian’ serous carcinomas are of tubal origin based on observations from the use of the SEE-FIM protocol ( Chapter 11 ), although what proportion of serous carcinomas are of tubal origin is currently not certain.
Most clear cell and many endometrioid tumors arise from the epithelium of endometriosis or an adenofibroma.
Some mucinous tumors arise from the mucinous component of a teratoma.
Squamous carcinomas usually arise from the squamous component of a teratoma or metaplastic squamous epithelium within an endometriotic cyst.
The tumors are subclassified by (a) their epithelial cell type(s); (b) the relative amounts of their epithelial and stromal components; (c) the location of their epithelial elements: surface (exophytic), cystic (endophytic), or both; and (d) their growth patterns and nuclear features.
Benign tumors have absent or minimal cellular stratification or atypia and no invasion.
Borderline tumors (tumors of low malignant potential) have epithelial stratification and atypia less than that of carcinoma but may have the appearance of intraepithelial carcinoma. The latter term is used most often in mucinous tumors and should be used cautiously in other categories, particularly serous (where we avoid it). Borderline tumors lack overt invasion but can have microinvasive foci, usually defined as individual foci <3 mm or <10 mm 2 .
Carcinomas usually exhibit obvious and often extensive invasion although some intracystic carcinomas lack invasion of the cyst wall. Such tumors should still be diagnosed as carcinoma as stromal invasion within the intracystic component can result in a malignant course. Similarly, surface foci of serous carcinoma (with or without ovarian stromal invasion), because of their location, are more likely to be clinically malignant than intracystic neoplasms.
Aside from Brenner tumors that typically contain a prominent, often predominant, stromal component, when the stroma of a tumor occupies an area greater than that of the glands, the suffix ‘fibroma’ is added (e.g., serous adenofibroma). When a grossly significant cystic component is present the designation cystadenofibroma is appropriate, although of no clinical relevance.
Admixtures of neoplastic elements are common in surface epithelial tumors, including those with two or more types of neoplastic epithelium, or variable combinations of benign, borderline, and carcinomatous components. Careful gross examination and thorough histologic sampling of these tumors are necessary for accurate classification. The extent of required sampling varies between categories; mucinous tumors particularly are often more heterogeneous than others.
Serous tumors
Mucinous tumors, endocervical-like and intestinal type
Endometrioid tumors
Clear cell tumors
Transitional cell tumors
Squamous cell tumors
Undifferentiated carcinoma |
Epithelial tumors account for 50% of all ovarian tumors and for 90% of ovarian cancers in the western world.
The clinical presentation is usually pelvic or abdominal pain or abdominal swelling. Some tumors may cause vague abdominal symptoms, malaise, or gastrointestinal complaints related to metastatic spread. Rare tumors cause paraneoplastic, paraendocrine, or endocrine symptoms (Clement et al.).
Examination usually reveals an adnexal mass or masses; ascites or evidence of peritoneal spread is often present in patients with borderline tumors and even more so with carcinomas. The serum level of CA125 is frequently elevated, as is CA19.9 in mucinous tumors.
The stromal component can secrete estrogenic or androgenic hormones with endocrine manifestations (‘tumors with functioning stroma’, Chapter 18 ).
The steroidogenic stromal cells are often luteinized to at least some degree and reactive for inhibin and calretinin. Even phenotypically normal stromal cells may produce hormones and stain for inhibin and calretinin, so-called ‘enzymatically active stromal cells’ (Scully and Cohen).
Estrogenic manifestations may include atypical endometrial hyperplasia in about a quarter of ovarian epithelial cancers (all types) and in as many as half of endometrioid carcinomas (Mingels, Masadah et al.).
Stage is the most important prognostic factor.
The 2014 FIGO staging system for ovarian, tubal, and peritoneal cancer ( Table 13.4 ) requires assignment of the primary site (ovary, tube, peritoneum) whenever possible.
| Stage I | Tumor is confined to ovaries or fallopian tube(s) |
| IA | Tumor limited to one ovary (capsule intact) or fallopian tube, no malignant cells in ascites or peritoneal washings |
| IB | Tumor limited to both ovaries or fallopian tubes, no malignant cells in ascites or peritoneal washings |
| IC | Tumor limited to one or both ovaries or fallopian tubes with any of the following |
| IC1 | Surgical spill |
| IC2 | Capsule ruptured before surgery or tumor on ovarian or fallopian tube surface |
| IC3 | Malignant cells in the ascites or peritoneal washings |
| Stage II | Tumor involves one or both ovaries or fallopian tubes with pelvic extension or primary peritoneal cancer |
| IIA | Extension and/or implants on uterus and/or fallopian tubes and/or ovaries |
| IIB | Extension to and/or implants on other pelvis tissues |
| Stage III | Tumor involves one or both ovaries or fallopian tubes, or primary peritoneal cancer, with microscopically confirmed peritoneal metastasis outside the pelvis and/or metastasis to the retroperitoneal (pelvic and/or para-aortic) lymph nodes |
| IIIA1 | Positive retroperitoneal lymph nodes only (histologically confirmed) |
| IIIA1(i) | Metastasis ≤10 mm in greatest dimension |
| IIIA1(ii) | Metastasis >10 mm in greatest dimension |
| IIIA2 | Microscopic extrapelvic (above the pelvic brim) peritoneal involvement with or without positive retroperitoneal lymph nodes |
| IIIB | Macroscopic peritoneal metastasis beyond pelvis 2 cm or less in greatest dimension with or without metastasis to the retroperitoneal lymph nodes |
| IIIC | Macroscopic peritoneal metastasis beyond the pelvis more than 2 cm in greatest dimension with or without metastasis to the retroperitoneal lymph nodes (includes extension of tumor to capsule of liver and spleen without parenchymal involvement of either organ) |
| Stage IV | Distant metastasis excluding peritoneal metastases |
| IVA | Pleural effusion with positive cytology |
| IVB | Liver or splenic parenchymal metastases; metastases to extra-abdominal organs (including inguinal lymph nodes and lymph nodes outside the abdominal cavity); transmural involvement of intestine |
Singh et al. (2014, 2015, 2106) and McCluggage et al. have proposed guidelines for site assignment of high-grade serous carcinomas (HGSCs) (see Table 11.2 ) based on gross and microscopic findings using the SEE-FIM protocol for examination of the fallopian tube ( Chapter 11 ).
Using these guidelines, Kulkarni et al. classified nonuterine HGSCs as tubal (72%), ovarian (20%), and peritoneal (8%) in marked contrast to their site assignment of the same cases using conventional criteria: 40% (tubal), 59% (ovarian), and 1% (peritoneal).
However, we and others believe further studies are needed before firm conclusions can be made with regard to the tubal or ovarian origin of most tumors that present clinically as an ovarian mass.
Grade is also of prognostic importance.
Grading is based on architectural features, sometimes combined with nuclear features. A two-tier grading system is now generally used for serous carcinoma. Our approach to grading for each major subtype is indicated under their respective headings.
While grading has generally been to found to have prognostic significance in early stage ovarian carcinomas, Kommoss et al. found that no grading system has prognostic value in high-stage carcinomas.
Other adverse prognostic factors have included older age, macroscopic residual tumor, ascites, positive peritoneal cytology, and aneuploidy.
Sassen et al. found that the only favorable prognostic features after neoadjuvant chemotherapy in advanced-stage ovarian cancers were absence of residual tumor or tumor foci ≤5 mm in size. More recently Said et al. have detailed a chemotherapy response score (CRS) for high-grade serous carcinomas (see corresponding heading).
Family history is the strongest risk factor for ovarian epithelial carcinomas, at least 10% of them being related to hereditary susceptibility. These cancers occur at a younger average age than their sporadic counterparts.
Three syndromes have been recognized: site-specific ovarian cancer, the breast–ovarian cancer syndrome (both of which are linked to BRCA abnormalities), and the hereditary nonpolyposis colorectal cancer (HNPCC, Lynch) syndrome, which is related to mutations in DNA mismatch repair genes, especially MLH1, MSH2, and MSH6 .
In addition Kang et al. reported germline mutations in a wide variety of other genes in women with pelvic HGSCs suggesting the need for multigene testing to assess hereditary cancer risk.
Most site-specific ovarian cancers and those of the breast and ovarian cancer syndrome are high-grade serous carcinomas (HGSCs); BRCA abnormalities account for most hereditary HGSCs.
found BRCA abnormalities in 50% of HGSCs: BRCA1 or BRCA2 germline or somatic mutations in 30% (20% BRCA1, 10% BRCA2 ) and methylation of BRCA1 in another 20%. BRCA1 mutation carriers are associated with a lifetime risk of 40–50% for HGSC; the corresponding figure for BRCA2 carriers is 20–30%.
The same study found that although BRCA mutation, TP53 abnormalities, and host immune infiltrates in HGSCs were associated with improved survival in univariate analysis, these features were not independently prognostic.
Alsop et al., however, in a study of 1001 patients, found that germ-line BRCA mutations were associated with a ‘major’ improvement in PFS and OS, even in those with early relapse after primary treatment.
Many BRCA1 and BRCA2 carriers undergo risk-reducing salpingo-oophorectomy (RRSO), optimally by the age of 40, a procedure that dramatically reduces the risk of subsequent HGSC. In toto histologic examination of these specimens should be performed ( Chapter 11 , SEE-FIM protocol).
Finch et al. found HGSCs in RRSO specimens in 6.4% of BRCA1 carriers and 1.5% of BRCA2 carriers. Powell et al. had a detection rate of 17% in both BRCA1 and BRCA2 carriers whereas Carcangiu et al. found HGSCs in 16% of BRCA1 carriers but in none of the BRCA2 carriers.
Most HGSCs in RRSO specimens are within the tubal fimbria, and although often microscopic, they may be associated with metastases. Ayres et al. argue that bilateral salpingectomy alone may not prevent the development of a fimbrial HGSC as they found microscopic foci of fimbrial tissue adherent to the ovaries in 15% of RRSO specimens.
BRCA-related tubal carcinomas and their precursors are considered further in Chapter 11 . The histologic features of BRCA-related HGSCs involving the ovary are considered under a separate heading in this chapter.
Women with HNPPC (Lynch syndrome) have a 40–60% lifetime risk of developing endometrial carcinoma ( Chapter 8 ) and an up to 20% lifetime risk of ovarian cancer (Ryan et al.). Occasionally the ovarian and endometrial carcinomas are synchronous.
There appears to be no increased risk of tubal carcinomas, although synchronous endometrial and tubal cancers were found in an MSH2 mutation carrier (Palma et al.).
Endometrioid (ECs) and clear cell carcinomas (CCCs) are over-represented in HNPCC-related ovarian cancers, accounting for 35% and 17% of such tumors, respectively in one series (Ketabi et al.) and 26% and 13% in a literature review (Downes et al.).
Chui et al. found that 90% of germline mutation-confirmed HNPCC ovarian cancers were EC (pure or mixed) and 10% were CCCs.
Chui et al. found MMR deficiency in 21% of nonserous ovarian carcinomas in their institution over a 1-year period, all of which were EC or CCC, supporting MMR and/or MSI testing for all newly diagnosed such tumors.
In their review of HNPCC-related ovarian cancers with MMR testing, Downes et al. found that 48% had MSH2 mutation, 38% MLH1 , and 14% MSH6.
The predominance of early stage disease accounts for the good prognosis of HNPPC-related ovarian cancers (80% 5-year survival); high-stage tumors have a poor survival (40% 5-year survival) (Ryan et al.).
These tumors account for 60% of serous tumors. They may occur at any age, but are most common in the reproductive era.
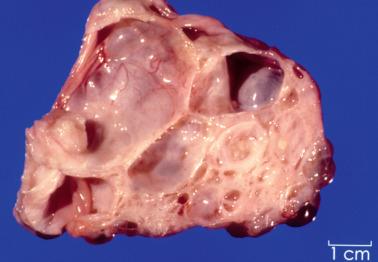
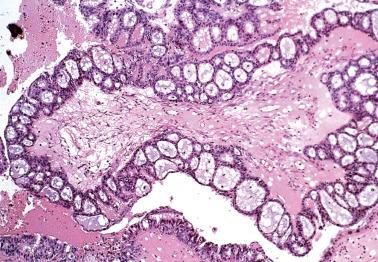
Serous cystadenomas are composed of one or more thin-walled cysts with watery fluid and a lining that is smooth or with soft to firm, polypoid excrescences composed almost entirely of stroma. The tumors are bilateral in up to 20% of cases.
Serous surface papillomas appear as polypoid excrescences on the outer surface of one or both ovaries.
Serous adenofibromas and cystadenofibromas are typically hard, white to yellow-white, predominantly solid, fibromatous tumors that contain glands or fluid-filled cysts that may bear polypoid excrescences that vary from firm to soft.
The excrescences and the hard consistency of the stromal component in some tumors may prompt an erroneous gross impression of carcinoma.
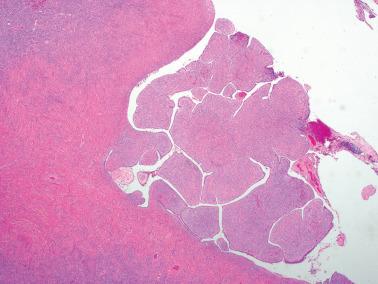
The cysts and polypoid stromal excrescences (that may be densely collagenous or markedly edematous) are typically lined by an epithelium similar to that of the fallopian tube, including ciliated cells and less numerous nonciliated secretory cells.
There is usually a single layer of cells but particularly when the wall is somewhat fibrous, benign glands may involve the cyst wall and even exhibit a cribriform pattern.
Tumors lined entirely by nonspecific, cuboidal or columnar nonciliated epithelium are also generally classified as serous, but some may actually be of endometrioid type, the distinction being subjective in some cases. A search for often subtle foci of endometriosis usually indicates an endometrioid nature.
Focal limited proliferation less than that of a borderline tumor may be seen and the suffix ‘with focal atypia’ or ‘with focal proliferation’ may be added.
Psammoma bodies may be present, but are generally inconspicuous.
The stroma of some tumors, presumably due to denudation of the cyst lining, may contain histiocytes.
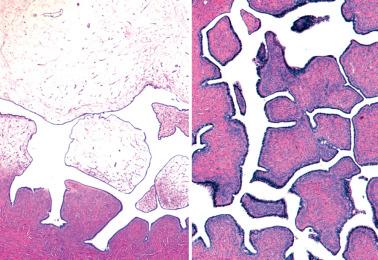
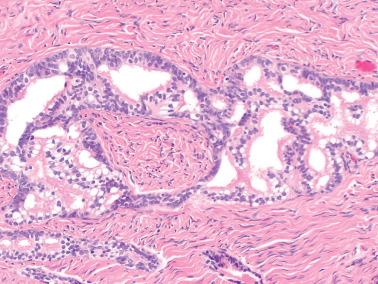
Epithelial inclusion cyst with serous features. This distinction is based arbitrarily on size, the lesion being considered neoplastic if >1 cm.
Surface stromal proliferations ( Chapter 12 ). These, in contrast to surface serous papillomas, are usually multifocal and microscopic.
Endometriotic cyst. This diagnosis may be suggested by collections of foamy and/or pigmented histiocytes, but endometriotic epithelium and stroma are absent.
Endometrioid adenofibroma. These tumors have stratified, predominantly nonciliated epithelial cells; squamous differentiation may be present.
Rete cystadenoma ( Chapter 17 ). Differential features include a hilar location, mural hilus cells or smooth muscle, and a lining with small crevices and cells with rare or no cilia.
Cystic struma ovarii ( Chapter 15 ). Follicles (often rare and attenuated within the cyst wall) with colloid, an absence of cilia, and reactivity for TTF1 or thyroglobulin establish the diagnosis.
These tumors (SBTs) account for ~10% of serous tumors. They are most common in the reproductive era; in the largest study, the age range was 12–89 years (mean, 42; median, 50).
SBTs are FIGO stage I in about 70% of cases, with most of the rest being stage II (spread within the pelvis) or stage III (spread to the abdomen or lymph nodes). Rare stage IV tumors (<1% of cases) have included those presenting with cervical node involvement.
Gru et al. reported two high-stage ovarian SBTs (one with progression to a low-grade serous carcinoma) that were associated with pulmonary thrombotic microangiopathy due to tumor emboli to the pulmonary vessels.
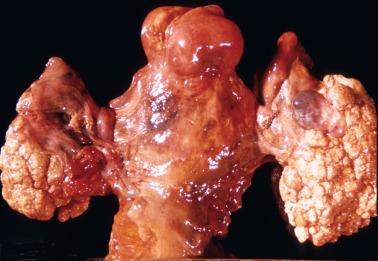
SBTs are bilateral in 25–40% of cases and typically unilocular or multilocular cystic masses with a mean diameter of 10 cm. The intracystic fluid is usually watery, but may be mucoid, suggesting a mucinous tumor. Solid areas, present in a third of tumors, usually represent a fibromatous component (serous adenofibroma or cystadenofibroma of borderline malignancy).
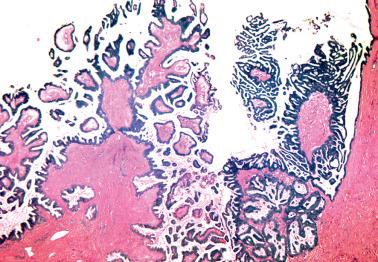
Polypoid to papillary excrescences (imparting a velvety appearance) usually form part or most of the cyst lining, the ovarian surface (serous surface papilloma of borderline malignancy), or both. Surface involvement is present in 30% (stage I) to 50% (all stages) of tumors.
Occasional tumors are a serous surface papillary tumor of borderline malignancy in which velvety surface tumor often covers an otherwise normal ovary.
Proposed sampling recommendations are:
Primary tumors: at least 1 block/cm of tumor diameter for tumors <10 cm and 2 blocks/cm for larger tumors. Sections should also be taken from areas that differ in appearance (fleshy, necrotic, hemorrhagic) to exclude serous carcinoma. Blocks of exophytic (surface) tumor and ruptured locules should be separately identified.
Omentum (if grossly negative): at least 1 block/2 cm of the largest dimension.
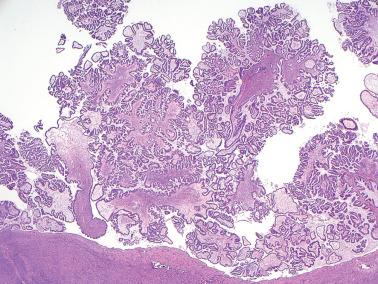
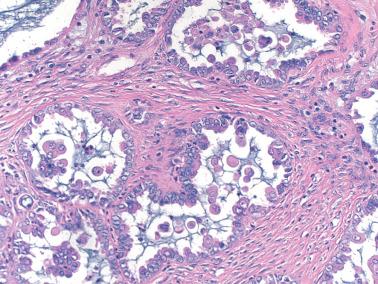
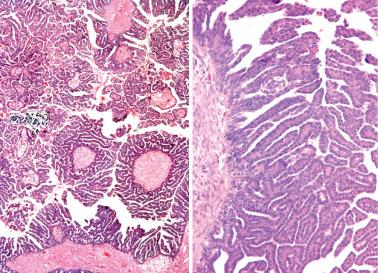
Low-power examination typically reveals intracystic or serosal papillae with fibrous or edematous cores often accompanied by an orderly invagination of papillae and glands into a fibromatous stroma.
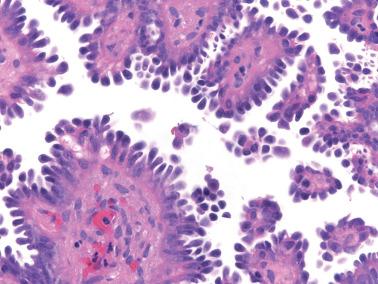
The papillae are typically complex with a hierarchical branching pattern in which progressively smaller fibrous-cored papillae are in turn covered by stratified tumor cells that form cellular tufts or buds. Some tumor cells are exfoliated or appear detached because their point of attachment is not in the plane of section.
The tumor cells are mostly columnar with a modest amount of cytoplasm. Some cells, typically apical, contain copious eosinophilic cytoplasm; ciliated cells are also usually present and hobnail cells may also be seen. Some glands may have an endometrioid-like appearance. Luminal mucin may be conspicuous and cytoplasmic mucin, if present, is confined to the tips of the cells.
The nuclear atypia varies from mild to moderate, but rare cells with greater atypia may be present. Mitotic figures are usually infrequent and nucleoli are only occasionally conspicuous.
Luminal or stromal psammoma bodies are often present and may be numerous. Stromal infiltrates of foamy histiocytes, sometimes in response to denudation of the epithelial lining cells, should not be misinterpreted as indicating an origin from endometriosis with which SBTs are only rarely associated.
Infarction of the fibrous-cored papillae may be seen that may elicit a desmoplastic response with entrapment of tumor cells, potentially mimicking an autoimplant (see next heading) or invasion.
Small foci of borderline tumor may be a focal or multifocal finding in an otherwise typical serous cystadenoma.
When <10% of the tumor is borderline tumor, some have used the terms ‘focally proliferative’ or ‘focally atypical’ serous cystadenoma because of an uneventful follow-up in some studies. Allison et al. found that when the borderline foci accounted for <20% of the tumor and were exclusively intracystic, there were no associated implants.
Ramalingam et al., however, found that rare tumors with these features have been associated with extraovarian implants. Additionally, Longacre et al. have found that some tumors with <10% borderline component are followed by an ipsilateral or contralateral typical SBT.
Descriptive diagnoses such as ‘serous cystadenoma with focal proliferation’ may be used with an accompanying notation specifying the proportion of the tumor that is proliferative (borderline appearing). Some may prefer to designate such a tumor as ‘benign serous tumor with a minor borderline component’.
Whatever terminology is used for these neoplasms, the proportion of the tumor that is composed of borderline neoplasia and whether or not the latter involves the ovarian surface should be specified. Irrespective of the specific term elected, communication with the treating physician is most important.
The stroma of SBTs usually is paucicellular and fibrous but may have a loose edematous appearance and/or contain inflammatory cells. A hypercellular stroma (that is most common in endometrioid tumors) is only occasionally seen and rarely may suggest a granulosa cell tumor although in one reported case of this type that component was FOXL2 negative.
Florid mesothelial hyperplasia in the wall and on the surface of the tumor is occasionally present ( Chapter 20 ).
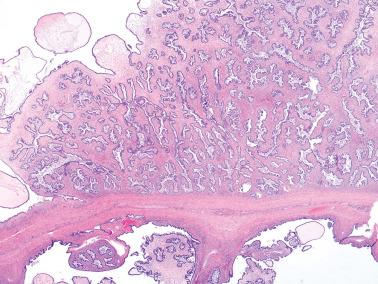
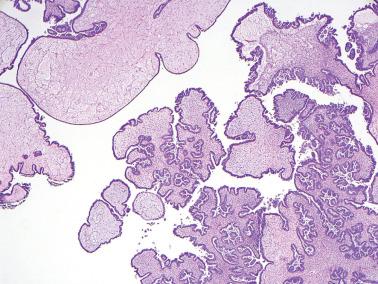
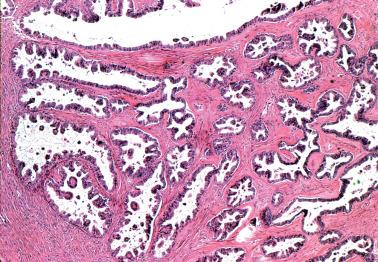
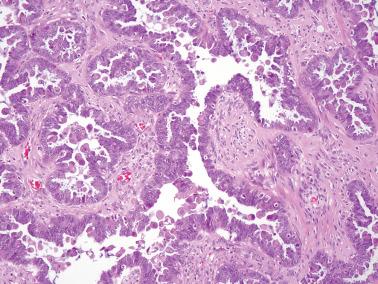
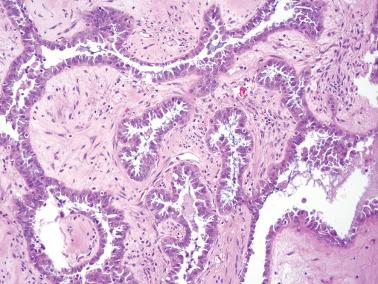
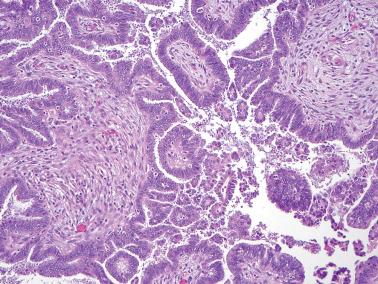
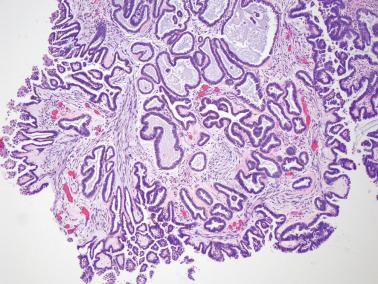
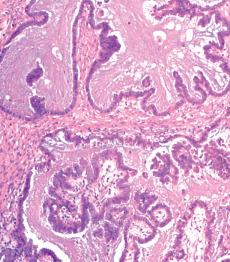
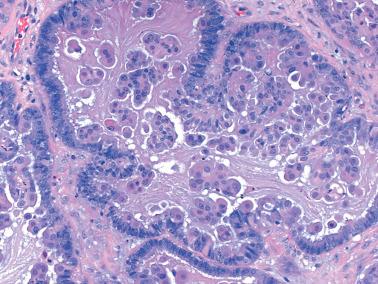
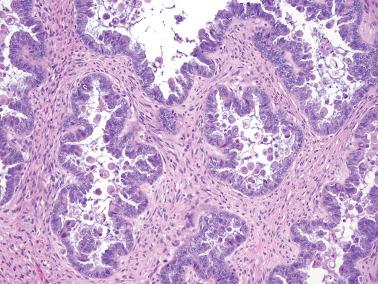
Micropapillary (including cribriform) pattern.
This pattern, which occurs in 5–10% of SBTs, is synonymously referred to in the 2014 WHO classification as ‘noninvasive low-grade serous carcinoma’. We and others prefer and continue to use (and do so here) ‘micropapillary SBT’ (MSBT) to denote this finding as long as cytologic findings do not warrant a diagnosis of carcinoma.
A size requirement of ≥5 mm for the micropapillary focus was initially proposed for a diagnosis of MSBT, although Vang et al. (2017) in a later study with long follow-up found that even a 1 mm micropapillary focus was prognostically significant.
Compared to typical SBTs, MSBTs more often exhibit or are associated with bilaterality, a surface component, microinvasion, stage II or III disease due to extraovarian implants (even in entirely intracystic tumors), invasive implants, and lymph node involvement that may represent metastases or independent primaries.
Long (arbitrarily defined as 5× longer than wide), filiform, nonbranching papillae with minimal or absent stromal cores emanate directly from large, smoothly contoured, stromal-cored papillae or from the cyst lining, resulting in a nonhierarchical, often filigree, pattern.
A cribriform pattern, which may be pure or coexist with the micropapillary pattern, is included in the MSBT category by most investigators.
The neoplastic cells with micropapillary and cribriform foci are more monomorphic than those of typical SBTs, being round, nonciliated, and with scanty cytoplasm, mild to moderate nuclear atypia, and often a small, occasionally prominent, nucleolus. High-grade atypia, other than in rare cells, is absent. If the latter is more than minimal, caution must be taken to exclude noninvasive serous carcinoma.
Rare MSBTs are associated with an invasive serous carcinoma that is usually low-grade (LGSC, see corresponding heading) and thus should be thoroughly sampled. May et al. have found that MSBTs and LGSCs have similar gene expression profiles.
Solid patterns. Rare SBTs have florid cellular stratification to the extent that the tumor cells fill and even obliterate the cyst lumens, resulting in solid sheets of cells.
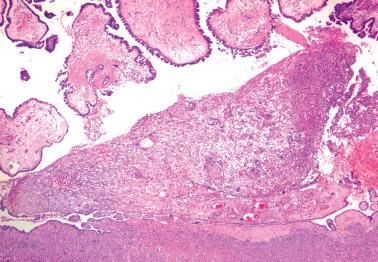
Autoimplants. These mimic the noninvasive desmoplastic implants that occur on the extraovarian peritoneum (see below), appearing as sharply delimited desmoplastic foci that typically occur on the serosal surface but occasionally on the inner (cystic) surface of the tumor. In some cases, an appearance similar to that of an autoimplant may be a result of infarction.
In 90% of cases, the autoimplants are associated with extraovarian implants that are usually noninvasive.
The desmoplasia within an autoimplant can suggest serous carcinoma. Features favoring an autoimplant include a location between tumor papillae or on the ovarian surface, circumscription, a high stromal:epithelial ratio, and epithelial cells like those of typical SBT.
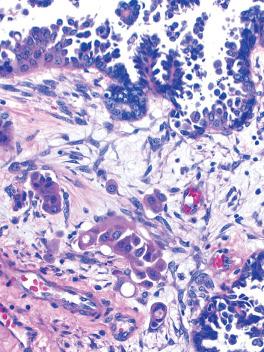
Microinvasion (in 5–10% of SBTs).
The foci of microinvasion, which are often multifocal, are characterized by tumor cells that often have abundant eosinophilic cytoplasm and are disposed singly or as small clusters, cribriform arrangements, and small papillae.
There is usually little or no stromal response. The invasive cells may occupy spaces that may result from fluid secretion by the tumor cells or may represent lymphatics (see last point below).
Upper size limits for each focus of 3 mm or 5 mm in maximal dimension or 10 mm 2 in area have been suggested, although found that microinvasion exceeding these size limits was not a prognostically adverse finding (see Prognostic Factors).
Maniar et al. found that the eosinophilic cells in microinvasive foci (and lymph nodes) showed loss of ER, PR, WT1, and Ki-67 and staining for M30 (an apoptotic marker) suggesting that these cells are senescent. The opposite findings were found in the noneosinophilic cuboidal/columnar cells of SBTs as well as in LGSCs.
Lymphatic invasion has been associated with microinvasion in several studies.
Focal ‘noninvasive’ carcinoma. Most SBTs are homogeneous throughout but occasional tumors have focal high-grade cytologic atypia indicating transformation to carcinoma. We have seen cases of this type with extraovarian invasive tumor confirming that the primary focus was frank carcinoma.
Invasive serous carcinoma.
Occasional SBTs are admixed with an invasive serous carcinoma that is usually low-grade, either micropapillary or as discrete small, rounded nests lacking the eosinophilic cells of microinvasion and with more cytologic atypia. Rare SBTs are associated with an invasive HGSC. The proportion of the tumor that is carcinomatous and whether it involves the surface should be noted.
Some SBTs are followed by a serous carcinoma that in at least some cases likely represents direct progression from the SBT (Xing, Zeppernick et al.)
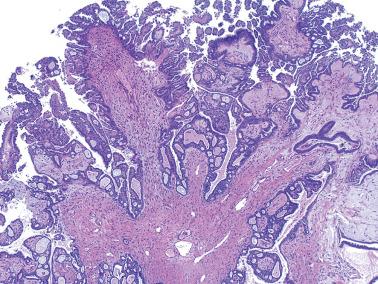
Extraovarian serous peritoneal lesions associated with SBTs include: endosalpingiosis ( Chapter 19 ), a finding that does not affect the stage; noninvasive implants; invasive implants; and low- or high-grade serous carcinomas. The last, in particular, are likely independent primary tumors. As noted earlier, implants are present in about 30% of SBTs; in some studies, the frequency is higher.
That extraovarian implants are metastatic in most cases is suggested by their much higher frequency in SBTs with a surface component than in those that are entirely intracystic: 62% vs 4% (Segal and Hart) and 69% vs 16% (Longacre et al.). However, MSBTs without a surface component are associated with implants in up to 35% of cases.
Implants may be grossly visible as small nodules or plaques, but are often microscopic.
Noninvasive implants, which account for ~90% of implants, are composed predominantly of neoplastic epithelial cells (epithelial implants) or of desmoplastic stroma (desmoplastic implants). Psammoma bodies may be absent, occasional, or conspicuous. The implants involve the peritoneal surface including invaginations between omental fat lobules that may be misinterpreted as invasion.
Noninvasive epithelial implants resemble the primary ovarian tumor with absent or limited stromal response that may include adhesions. Occasionally, likely due to maturation, glands indistinguishable from endosalpingiosis may be seen.
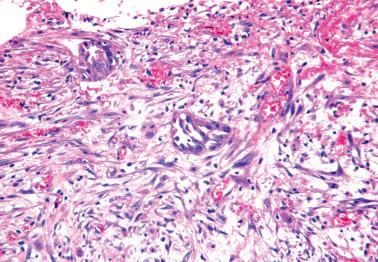
Noninvasive desmoplastic implants form generally well-delineated peritoneal plaques or nodules and are composed of small nests, glands, papillae, or singly disposed tumor cells within a desmoplastic or granulation tissue-type stroma that almost always exceeds the volume of the epithelial component, sometimes accounting for most of the implant. Fibrin and acute inflammatory cells may be conspicuous, particularly superficially.
If the biopsy specimen does not include underlying tissue, the lesion is considered ‘consistent with a noninvasive implant’ on the assumption that it has been stripped off with ease.
Noninvasive implants may have a micropapillary pattern or small nests of cells surrounded by a clear zone or cleft. The micropapillary pattern may be exophytic (as in primary tumors) or endophytic, with a complex network of irregular spaces; Bell KA et al. consider these patterns ‘invasive’ even without invasion of underlying tissues. found them to be more frequent in implants that were invasive by usual criteria but were not independently prognostic.
Invasive implants account for no more than 5% of implants; the frequency is even lower in some studies.
The 2014 WHO classification has replaced the term ‘invasive implants’ with ‘low-grade serous carcinoma’ but some prefer the former term. As Prat states, ‘the term “invasive peritoneal implant” fits better with the synchronous presence of a noninvasive ovarian tumor and describes more properly a small and superficial tumor that may or may not progress to peritoneal carcinomatosis (LGSC) after several years’.
Low-power examination reveals destructive invasion and replacement of underlying tissue by an irregular and haphazard infiltration of small nests (sometimes surrounded by clefts), cribriform glands, and micropapillae, usually within a desmoplastic stroma. The epithelial:stromal ratio is usually higher than in noninvasive desmoplastic implants.
The tumor cells may have more basophilic cytoplasm and more uniformly atypical nuclei than in noninvasive implants, in some cases resembling those of a low-grade serous carcinoma.
In some cases, an implant cannot be confidently classified as invasive or noninvasive; such implants were considered ‘indeterminate’ in the Longacre study. Given the rarity of invasive implants, the bias should be towards diagnosing noninvasive implants unless there is convincing evidence to the contrary.
Differential diagnosis and/or associated features of implants:
Endosalpingiosis ( Chapter 19 ). This is a common finding involving pelvic and omental peritoneum in the reproductive era, and is especially common in women with SBTs. Endosalpingiotic glands are lined by bland tubal-type epithelium without the cellular stratification, papillarity, tufting, and nuclear atypia of SBT implants.
Primary extraovarian SBT ( Chapter 19 ).
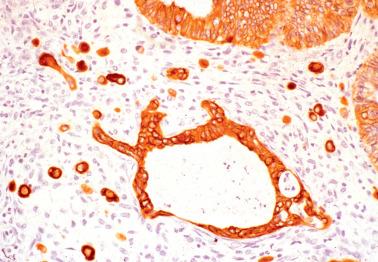
Mesothelial hyperplasia ( Chapter 20 ). This common finding associated with SBTs may complicate determining if cells in a peritoneal biopsy specimen are tumor cells or mesothelial cells, or both. The former usually immunoreact with epithelial markers (we prefer claudin-4; only membranous staining is considered positive) but not for calretinin, whereas the opposite findings indicate mesothelial cells.
Histiocytic infiltrates ( Chapter 20 ). An implant will sometimes elicit a striking peritoneal histiocytic infiltrate (sometimes with admixed hyperplastic mesothelial cells) that may obscure the neoplastic cells of the implant. Immunostains for histiocytic markers (such as CD68) and epithelial markers can be helpful.
Implants of low-grade serous carcinoma. These can occasionally appear noninvasive but usually have a higher epithelial:stromal ratio than noninvasive desmoplastic implants of a SBT, and typically have the morphologic features of low-grade serous carcinoma (see below) including uniformly atypical nuclei.
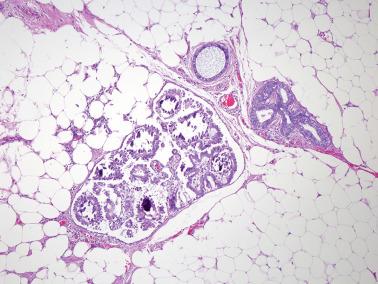
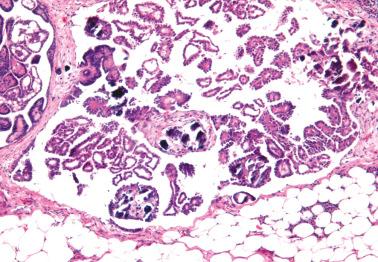
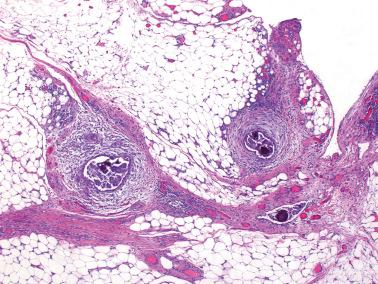
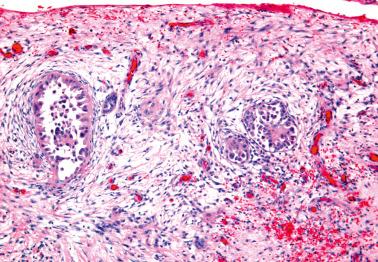
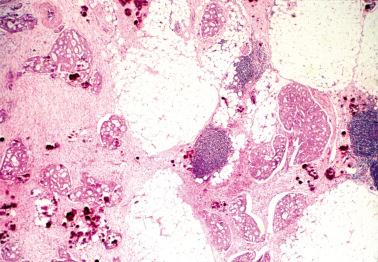
Involvement of the fallopian tube lumen or mucosa by psammoma bodies is common in SBTs. found psammoma bodies in 24% of stage I SBTs and in 51% of stage II and III cases; Wolsky et al. had similar findings. The latter authors also found intraluminal detached papillae, buds, clusters, or nests of tumor cells in 42% of stage IC–III SBTs but in none of their stage IA or IB tumors suggesting spread of detached tumor cells into the tubal lumina.
Endosalpingiosis. Wolksy et al. found tubal serosal endosalpingiosis in 18% of stage IC–III SBTs but in none of the stage IA–B tumors.
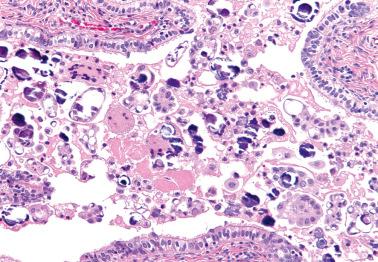
The tumor cells are positive for most epithelial markers, and ER, PR, WT1, and p53 (wild-type pattern), but these are rarely needed for diagnosis.
found that only 8% of SBTs exhibit functional p53 mutations compared to 51% of HGSCs.
Ho et al. found that almost 90% of SBTs harbor BRAF or KRAS mutations that were also present in adjacent cystadenomatous epithelium, suggesting that the mutations precede the development of SBT.
Most peritoneal implants are clonal (with the same KRAS or BRAF mutations as in the primary SBT) supporting metastatic spread (Ardigheiri et al., Horn et al., Sieben et al.) (also see molecular findings below). Zuo et al. found KRAS mutations in 61% of invasive implants vs 22% of noninvasive implants.
Nodal involvement (LNI) has been found in 20–40% of patients with SBTs undergoing lymphadenectomy. Patients with LNI have been reported to be younger than those without it.
Maniar et al. found that LNI had a strong association with ovarian surface involvement and peritoneal implants, suggesting that LNI in some cases may be due to spread of exfoliated tumor cells rather than via lymphatics from the primary tumor.
found that the involved nodes were, in order of frequency, pelvic, mesenteric/omental, para-aortic, and supradiaphragmatic. The number of involved nodes varied from 1 to 20 (mean, 3).
found four patterns of intranodal SBT:
Individual cells, nests, and simple papillae within sinuses or the parenchyma (90% of cases).
Intraglandular pattern within the parenchyma: epithelium-lined spaces containing complex hierarchical papillae (68% of cases), sometimes with contiguous foci of microinvasion similar to that in primary SBTs.
Tumor cells with copious eosinophilic cytoplasm (52% of cases) in the sinuses and/or parenchyma similar to those seen in primary SBTs and their implants.
Micropapillary pattern (16% of cases) in the parenchyma consisting of delicate papillae without fibrovascular cores and/or a complex maze of slit-like spaces.
LNI has a strong association with intranodal endosalpingiosis, from which the intranodal SBT may arise, usually with the second pattern noted above. Features favoring metastatic intranodal SBT include an absence of endosalpingiosis; sinus involvement; intranodal SBT as single cells, nests, or papillae; and microinvasion, sometimes with LVI, in the primary tumor. Occasionally, LNI may be a result of both synchronous primary neoplasia and metastasis.
Most studies have found that LNI is not an adverse prognostic factor independent of stage. , however, found that nodules (>1 mm in size) of confluent tumor without intervening lymphoid tissue, often with a micropapillary pattern and a desmoplastic stroma, may be an adverse prognostic factor, although we would consider this pattern to represent LGSC.
The differential of LNI in SBT includes, in addition to endosalpingiosis and primary intranodal SBT, intranodal hyperplastic mesothelial cells (see Chapter 20 ).
Lymph nodes may be the site of a recurrence (see below). In such cases, the involved nodes may be supradiaphragmatic and the intranodal tumor may resemble the primary SBT, LGSC, or even, rarely, HGSC.
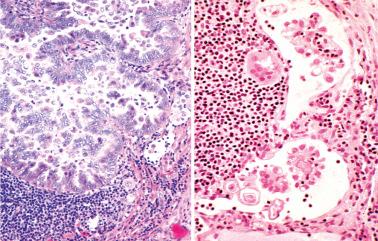
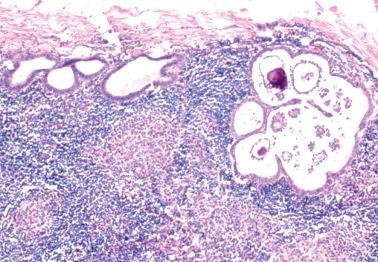
reported 5 cases of LNI by LGSC in women with ovarian SBTs. The LNI was synchronous (one case) or metachronous (4 cases) being found from 7 months before to 5 months after the ovarian SBT.
The LGSCs were supraclavicular (2 cases) and cervical, intramammary, and para-aortic nodes (1 case each) and replaced the lymph node or diffusely involved the sinusoids or cortices. Two patients also had extensive intranodal SBT and endosalpingiosis in pelvic nodes.
The ovarian SBTs exhibited surface excrescences (4 cases) and microinvasion (3 cases) but lacked micropapillary/cribriform patterns. All had noninvasive implants and one also had invasive implants.
Two patients had local recurrences and died of disease. Another patient, who initially had supraclavicular LNI by LGSC, developed abdominopelvic lymphadenopathy and was alive with disease at last follow-up.
The authors concluded that, as with intranodal SBTs, intranodal LGSCs may arise from intranodal endosalpingiosis and progress independently of the ovarian SBT.
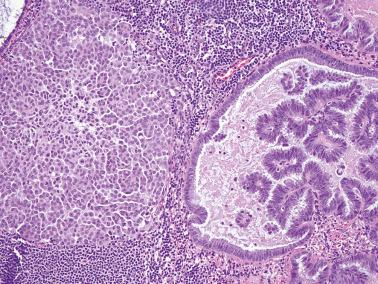
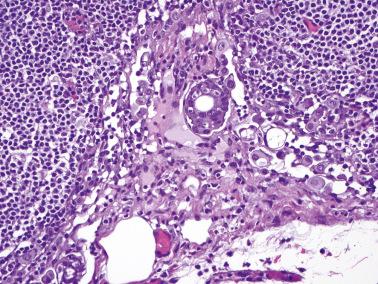
Müllerian borderline tumors of mixed cell type (see Chapter 14 ). These tumors often contain foci resembling SBT but also have cells with intracellular mucin and in some cases, endometrioid and squamous elements.
LGSCs. These differ from SBTs by the presence of stromal invasion greater than microinvasion. Additionally, there are small nests, more uniform papillae than in most SBTs, and greater cytologic atypia, an appearance differing from that of microinvasion of an SBT.
HGSC lacking stromal invasion. These can be misdiagnosed as a SBT if severe cytologic atypia is overlooked. Most such tumors, however, also have more florid epithelial proliferations off the surfaces of papillae with the formation of slit-like spaces and other typical formations of serous carcinoma unacceptable for an SBT.
Endometrioid papillary tumor of borderline malignancy and endometrioid and clear cell carcinomas ( Chapter 14 ).
Retiform Sertoli–Leydig cell tumors ( Chapter 16 ).
FIGO stage .
Longacre et al. found OS rates of 95% (98% stage I; 91% stage II–IV) and DFS rates of 78% (87% stage I; 65% stage II–IV). Overall, 5% of patients were dead of disease and another 6% had persistent disease at last follow-up.
Mortality rates from a literature review ( ) were 6% for stage II tumors and 19% for stage III tumors.
With long follow-up (mean, 16 years), found recurrence and fatality rates of 6.8% and 5% in stage I tumors. Surprisingly, the only finding that predicted recurrence was a higher frequency of endosalpingiosis (72.7% vs 12.5%). Some of the ‘recurrences’ in this study were likely new tumors given the long post-oophorectomy interval and that most of the recurrent tumors were serous carcinomas.
Macroscopic residual disease . This is an important predictor of recurrence and survival in most studies.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here