Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Valve disease is globally common, affecting approximately 2.5% of the population.
In industrially underdeveloped countries, rheumatic disease (RhD) is the most common cause. Endomyocardial fibrosis is an underresearched disease common in equatorial Africa.
In industrially developed regions, diseases of old age predominate, particularly calcific aortic stenosis and secondary mitral regurgitation.
In the United States, valve disease is most common among the elderly, with a prevalence of 13% among those older than 75 years.
Drug-induced valve disease is increasing as a result of 5-HT2B receptor agonists.
Infective endocarditis is increasingly related to medical devices and intravenous drug use.
Failure of biological replacement valves is a major burden in all regions of the world.
Substantial variation in access to health care exists in all countries, including those that are industrially developed.
The main global challenge is to prevent chronic RhD, which will require collaborations among social, political, and medical programs.
Heart valve disease is the term for any deviation from normal valve anatomy or function. This includes a wide spectrum of severity, from mild mitral prolapse to a flail mitral leaflet or aortic sclerosis to critical aortic stenosis. The term does not describe a unitary diagnosis but instead a set of conditions affecting one or more of the four heart valves caused by genetic, environmental, or acquired pathologic processes that vary with geography and demography.
Our knowledge of the epidemiology of valve disease is incomplete. The global disease survey , used 56,356 unique sources of data from 195 countries to estimate incidence, prevalence, mortality, and disability rates arising from 328 diseases and injuries. Rheumatic disease (RhD) and infective endocarditis (IE) were the only valve diseases recorded.
National or large-scale screening programs for RhD exist in some countries (e.g., Australia, Cambodia, Fiji, India, Laos, Mali, Mozambique, New Caledonia, New Zealand, Nicaragua, Pakistan, Samoa, South Africa, Tonga, Yemen), but most surveys of RhD are of children and young adults and are restricted geographically and by time. Countrywide estimates are usually extrapolated from these small studies to people of all ages and socioeconomic classes and therefore remain uncertain. In the industrially developed world, there have been population surveys of all types of valve disease in the United States and Norway and studies of the elderly in the United Kingdom. Otherwise, work has focused on aortic stenosis, , mitral prolapse, or bicuspid aortic valve. ,
There are other limitations to our knowledge. Estimates of the prevalence of valve disease vary with the method of diagnosis, which for RhD was originally clinical but more recently includes echocardiography, which is approximately 10 times more sensitive than auscultation. The prevalence and types of valve disease differ whether assessment occurs by community screening, , , open access echocardiography, , or hospital services. For example, the frequency of bicuspid aortic valves is 0.5% to 0.8% in community surveys, , 2% in a postmortem study, and approximately 67% among patients in their 40s who have aortic valve replacement.
The limitations make it difficult to talk about the epidemiology of valve disease in general, but there are two broad patterns ( Table 1.1 ). , , The first occurs in economically disadvantaged areas of the world characterized by poor housing, sanitation, and nutrition and with inadequate access to medical care. These areas include parts of Africa, the Indian subcontinent, the Middle East, and South America.
| Prevalence in Economically Poorer Countries | Prevalence in Economically Richer Countries | References | |
| Causes | |||
| Chronic rheumatic heart disease | 0.9%–4.0% | 0.3% | 17–22 |
| Endomyocardial fibrosis | 20% a | 23 | |
| Calcific aortic stenosis | 0.4% b | 3 | |
| Bicuspid aortic valve | 0.5%–0.8% b | 9,10 | |
| Mitral regurgitation | 1.7% | 3 | |
| Mitral prolapse | 2%–3% | 8,23 | |
| Prolapse and mitral regurgitation | 0.2%–0.3% | 20,21 | |
| Secondary mitral regurgitation | 1.4% c | ||
| Failing biological replacement valves | |||
| Aortic dilation | |||
| Inflammatory conditions (e.g., SLE, rheumatoid arthritis) | |||
| Drugs, carcinoid, radiation | |||
| Incidence per 100,000 Population per Annum | |||
| Acute rheumatic fever, all attacks | 10–374 | 30 | 17–19 |
| Acute rheumatic fever, first attack | 8–51 | ≤10 | 17–19,22 |
| New rheumatic heart disease | 20 | 24 | |
| Infective endocarditis | 3.4 | 1.4–6.2 | 25,26 |
b The world population is 7,000,000,000. The proportion older than 60 years is about 10%, or 700,000,000. Assuming a prevalence of aortic stenosis of 1.74% in this age group gives a world prevalence of aortic stenosis of 12 million. The prevalence of bicuspid valve is 35 million based on a population prevalence of 0.5%. In a study of patients with bicuspid aortic valves with a mean age of 32 years at baseline and followed for 20 years, 28 (13%) of 212 needed aortic valve replacement for severe aortic stenosis. This suggests that about 4.5 million in the world could have severe aortic stenosis as a result of a bicuspid aortic valve and that the total as a result of nonrheumatic disease (calcific stenosis in those older than 65 years and bicuspid aortic valve in those younger than 65) could be 16.5 million.
c Estimated as U.S. population prevalence of mitral regurgitation minus prevalence of mitral regurgitation caused by mitral prolapse.
Valve disease is predominantly caused by rheumatic fever (RhF) and chronic RhD. Patients are typically young, and the morbidity and mortality rates are high. Acute RhF occurs mainly between the ages of 5 and 15 years, and heart failure, usually as a result of acute rheumatic mitral regurgitation, occurs most commonly within a year of the acute episode. , In Ethiopia, 20% with RhD die before the age of 5 years and 80% before 25 years. Endocarditis, stroke, and heart failure are common sequelae. Mitral prolapse occurs within the differential diagnosis of chronic RhD diagnosed by echocardiography. There are also a number of geographically localized diseases. Endomyocardial fibrosis (EMF) is second in incidence to RhD in Equatorial Africa. Subvalvar aneurysms are the third most common cause of mitral regurgitation after RhD and mitral prolapse in sub-Saharan Africa.
The second pattern occurs in economically richer areas, where the epidemiology of valve disease changed ( Box 1.1 ) in the second half of the 20th century, with a decrease in the incidence of rheumatic fever as a result of better housing to reduce overcrowding, better nutrition, and better health care. There also was a change in streptococcal serotypes. Rheumatogenic serotypes were found in 50% of children with pharyngitis in Chicago between 1961 and 1968, compared with 11% between 2000 and 2004. Calcific degeneration, secondary mitral regurgitation, and aortic dilation leading to aortic regurgitation predominantly affect older people; mitral prolapse and bicuspid aortic valve are found in younger people.
Reasons for reduced rheumatic disease with a decrease in acute rheumatic fever cases:
Better housing and reduced overcrowding
Better nutrition
Better health care (e.g., treatment of throat infections, better secondary prophylaxis)
Fall in rheumatogenic serotypes a
a There were rheumatogenic serotypes in 50% of children with pharyngitis in Chicago between 1961 and 1968, compared with 11% between 2000 and 2004.
Increasing age expectancy, leading to more degenerative disease
New types of valve disease requiring treatment (e.g., radiation, drugs)
More health care–related endocarditis
Biological replacement valves used in younger patients resulting in more reoperations, including valve-in-valve transcatheter procedures
More mild disease as a result of increased use of echocardiography
New types of valve disease are increasing in frequency as a result of drugs or radiation exposure. IE has a different pattern from that in the developing world. With older populations, more Staphylococcus aureus and more cases as a result of health care, mainly hemodialysis and implanted electrical devices. Tricuspid regurgitation as a result of endocardial pacing systems and defibrillators is also increasingly seen.
These two patterns are usually taken to apply broadly to the industrially underdeveloped and developed regions of the world. However, this is an oversimplification because pockets of poverty exist in the industrially developed world (e.g., indigenous populations of Australia and New Zealand). Conversely, richer people in the disadvantaged areas of the world are more likely to reflect patterns of valve disease usually seen in the industrially developed areas. Many countries, such as India and China, are in the process of rapid development and have a falling incidence of RhF while retaining a higher prevalence of chronic rheumatic heart disease among the more elderly population than seen in the United States or Western Europe.
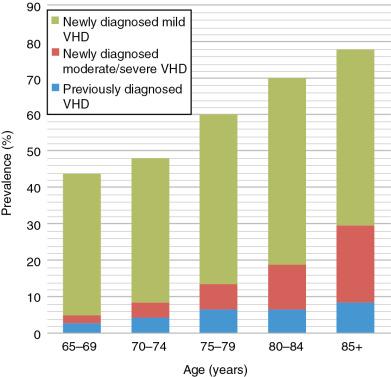
Valve disease remains underdetected , ( Fig. 1.1 ) in developed and developing countries. In the developing world, the detection rate is approximately 10% using clinical examination alone without systematic echocardiographic screening. , In the United States, the prevalence of moderate or severe valve disease is 1.8% when estimated from echocardiograms performed as clinically indicated compared with an age-corrected population prevalence of 2.5%. In the U.K. Oxford Valve (OxVALVE) study, the prevalence of previously undiagnosed moderate or severe valve disease was 6.4% for people aged 65 years of age or older and was already identified in another 4.9% (see Fig. 1.1 ).
A survey of open access studies found that significant valve disease was suspected from a murmur in 127 patients but was unsuspected in 177 cases. Postmortem examinations show that only about 50% of aortic stenosis cases may be diagnosed before death, and undetected or underinvestigated aortic stenosis is an important cause of perioperative and maternal deaths. Detection rates may increase in the future with improved access to point-of-care echocardiography, , perhaps aided by improved analysis of murmurs using artificial intelligence linked with mobile phone technology.
Access to valve surgery is restricted. In India and Africa, valve operations are performed at a rate of 1.8 per 100,000 people each year. This compares with 28 operations per 100,000 people in the Netherlands and 122 operations per 100,000 people in the United States. In Brazil, only 11,000 operations are performed each year, and 80% of those needing surgery remain on a waiting list. There is also variation in access to surgery in industrially developed countries. A comparison of rates of aortic valve replacement in the United Kingdom against estimated need found a variance between observed and expected ranging between −356 and +230. The causes of this variance have not been explored but may partly reflect the activity of community physicians. The elderly are particularly disadvantaged, and about one third with aortic stenosis are denied surgery inappropriately. Developing a transcatheter program leads to an increase in conventional surgical rates because clinically inappropriate perceptual barriers to referral are lifted.
The global burden of RhD was assessed in a World Health Organization (WHO) report in 2005, two reports in 2011, , and the global burden of disease study most recently updated in 2017. , All but the first of these used echocardiographic diagnosis, but the criteria for echocardiographic diagnosis have changed.
The annual incidence of all first or subsequent attacks of acute RhF worldwide is estimated at 471,000 cases based on a meta-analysis of regional reports. The median annual incidence ranges from 10 to 374 cases per 100,000 people. , The incidence is decreasing in all WHO regions for which data exist, except in the Americas, where the incidence is increasing slightly ( Fig, 1.2 ). , , It is higher in surveys with active surveillance (8 to 51 cases per 100,000) than in surveys using passive surveillance (5 to 35 per 100,000). Estimates of the incidence of first attack are based on few data but annual incidences are 23 per 100,000 people per annum (p.a.) in Kuwait, 35 per 100,000 in Iran, 51 per 100,000 in India, 80 per 100,000 in a Maori population in New Zealand, and 194 per 100,000 in an indigenous population in the Northern Territories of Australia.
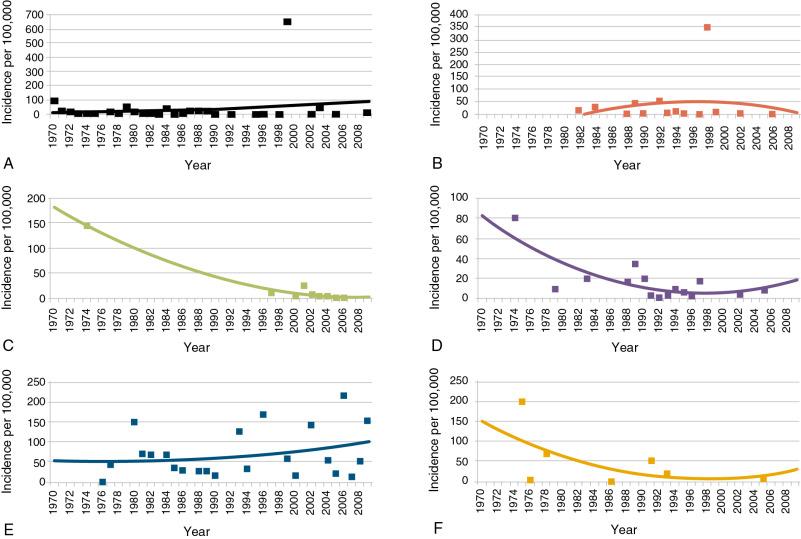
Males and females were equally affected by first attacks in one study, and females more frequently affected in another. The incidence peaks at age 5 to 14 at 150 per 100,000 men, falling to approximately 55 per 100,000 at 15 to 24 years of age and to 25 per 100,000 at 25 to 34 years of age. The recurrence rate is 4.5% during the first year and 12.5% at 5 years. There are no data on the incidence of first attack from Africa.
The incidence of new RhD is 27% to 35% at 1 year, 44% to 51% by 5 years, and 52% to 61% at 10 years. , Allowing that 60% of people with acute RhF develop chronic RhD, the annual incidence of new cases of RhD globally has been estimated at 282,000. However, there are major geographic differences, with a mean of 160 cases per 100,000 people (95% CI: 80–230) based on a meta-analysis of 37 population surveys. The estimated incidence is 23.5 cases per 100,000 people in Soweto, but with a J-shaped age-dependence curve and with 30 cases per 100,000 for those 15 to 19 years of age, 15 cases per 100,000 for those 19 years of age, and 53 cases per 100,000 for those older than 60 years.
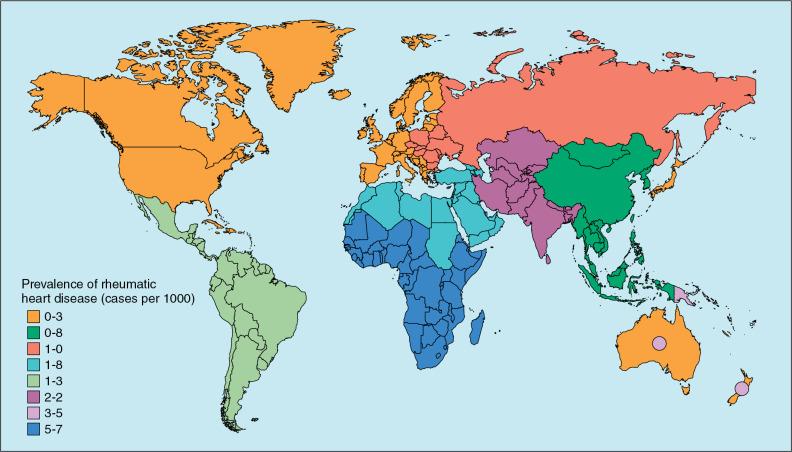
The global disease survey estimated a worldwide prevalence of chronic RhD of at just under 30 million in 2016, an increase of 3.4% compared with 2006. The prevalence is increasing in all WHO regions other than Europe ( Fig. 1.3 ). , , RhD is estimated to cause 4.3 million cases of heart failure, which is 6.9% of the total number of cases, and to produce an age-standardized mortality rate of 4.7 deaths per 100,000 people p.a.
In surveys concentrating on RhD, prevalence estimates have changed in recent years with the use of echocardiography and with the evolution of echocardiographic criteria for diagnosis. A seminal study in 2007 in Cambodia and Mozambique found a prevalence of 230 cases per 100,000 children as estimated by clinical examination but 2810 cases per 100,000 using echocardiography. The WHO 2006 criteria used a combination of clinical examination and echocardiography to give a prevalence of 2110 cases per 100,000 (95% CI: 1410–3140), compared with only 270 cases per 100,000 (95% CI: 160–440) based on clinical diagnosis.
There were concerns about the limitations of clinical examination and the specificity and variability of echocardiographic criteria. A retrospective re-evaluation of results gave a range in prevalence for the same population of 510 to 3040 cases per 100,000, depending on the echocardiographic criteria used. These concerns led to a World Heart Federation consensus statement in 2012 that categorized RhD as definite or borderline using a combination of regurgitation, gradient, and morphology ( Fig. 1.4 and Box 1.2 ), Using these criteria, the prevalence of borderline or definite RhD ranges from 910 to 4020 cases per 100,000 people in different populations ( Fig. 1.5 ).
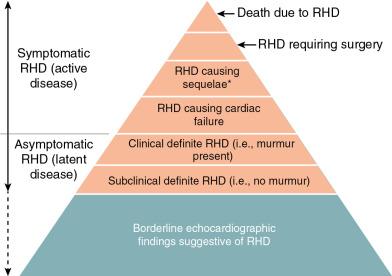
Definite diagnosis, one or more of the following criteria:
Pathologic mitral regurgitation a
a The pathologic regurgitation diagnosis must include all of the following: (1) seen in two views, (2) jet length in at least one view of ≥ 2 cm (for mitral) or ≥ 1 cm (for aorta, (3) peak velocity ≥ 3 m/s, and (4) pan systolic (mitral) or pandiastolic (aortic) in at least one signal.
and at least 2 morphologic features of rheumatic mitral valve disease b
b Morphologic features of the mitral valve: anterior leaflet thickening ≥ 3 mm, chordal thickening, restricted leaflet motion, and excessive leaflet tip motion in systole.
Mitral stenosis with mean gradient ≥4 mmHg
Pathologic aortic regurgitation a and at least two morphologic features of rheumatic aortic valve disease c
c Morphologic features of the aortic valve: irregular or focal thickening, coaptation defect, restricted leaflet motion, and prolapse.
Borderline disease of the aortic and mitral valves
Borderline diagnosis, any of the following criteria:
The echocardiographic criteria are relatively labor intensive to apply, and for the sake of screening children to give secondary prophylaxis, there is a move to handheld devices, nonphysician operators, and much simplified criteria, typically a 2-cm or larger mitral regurgitant jet or any aortic regurgitation. , This allows screening of 200 to 250 cases per day by one sonographer spending 2 minutes per patient. This approach has high sensitivity but reduced specificity, and a more expert review is required to confirm a diagnosis.
There is uncertainty about the clinical significance of echocardiographic borderline RhD. Small studies with a follow-up at a maximum of 24 months suggest that two thirds of mild lesions remain unchanged, one third improve or resolve completely, , and one third become worse, particularly if there is regurgitation plus a morphologic abnormality. A large, prospective, multicenter registry, the Global Rheumatic Heart Disease Registry (REMEDY), aims to determine the natural history of borderline disease more accurately and whether secondary prophylaxis is beneficial. New Zealand does not give secondary antibiotic prophylaxis to borderline RhD but uses active surveillance with intervention only if the echocardiographic appearance changes. Resource-scarce countries may have to weigh the cost of screening programs against the need to combat tuberculosis (TB) and human immunodeficiency virus (HIV) infections, but they may choose to prescreen with auscultation. Sadiq et al. screened 24,980 Pakistani children in this way.
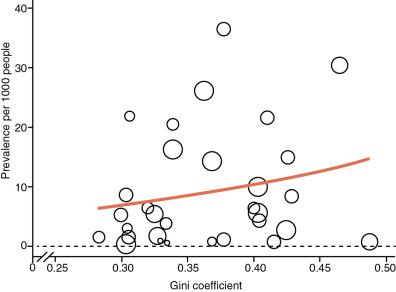
Prevalence increases with age in endemic areas from 470 cases per 100,000 children at age 5, about 1500 cases per 100,000 at age 10, and 2100 cases per 100,000 at age 16. Among Nicaraguan adults between the ages of 20 and 35 years, the prevalence based on echocardiography was 2200 cases per 100,000. The prevalence of RhD is higher in rural than urban populations and higher in low-income populations ( Fig, 1.6 ) because these factors affect living conditions and access to medical care, including secondary prophylaxis. In a lower-income area of Cape Town, the prevalence was 2700 cases per 100,000 school children compared with only 1250 cases per 100,000 in a higher-income area. In the same study, the average prevalence was 2000 cases per 100,000 in South Africa compared with 3000 cases per 100,000 in Ethiopia, where there is a poorer and more rural population. Similarly, in Addis Ababa the prevalence of RhD was seven times higher among low- than high-socioeconomic-class school children.
Throughout the industrially underdeveloped world, 10% to 35% of all cardiac admissions are a result of acute or chronic RhD. RhD is the dominant cause of heart failure, with 4.3 million cases worldwide, representing 6.9% of the totalcases. In a study from the Northern Territories of Australia, the 10-year cumulative incidence of heart failure was 18.6% after acute RhF, 3.4% for atrial fibrillation, 4% for IE, and 3.6% for stroke, and the death rate was 10.3%.
RhD, particularly mitral stenosis, is a major cause of mortality and morbidity in pregnancy. The prevalence among pregnant women is 230 cases per 100,000 in Fiji and South Africa. The maternal mortality rate for RhD was 34% in a study from Senegal and as high as 54% in the setting of severe mitral stenosis.
Few incidence figures exist, but it was estimated using hospital registries for the whole Swedish population between 2003 and 2010 as 63.9 cases per 100,000 patient-years. The incidence rose with age, with 69% of all valve disease diagnosed in people 65 years of age or older. The most commonly occurring lesions were aortic stenosis, mitral regurgitation, and aortic regurgitation.
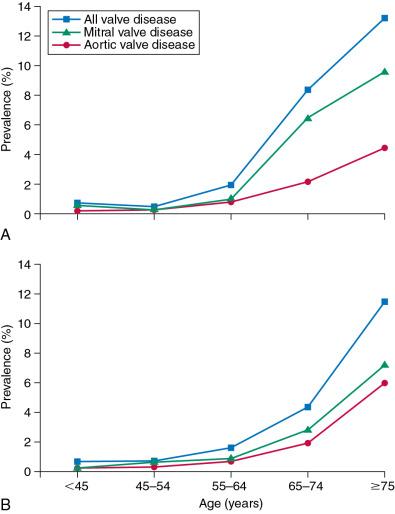
The population prevalence of valve disease was estimated at 2.5% in the United States ( Table 1.2 ) and 3.3% of those older than 25 years in Tromsø, Norway. The prevalence was age dependent, beginning to rise at age 55 and becoming steeper after age 65. After age 75, the prevalence was 13.2% in the United States but higher (18.7%) in the U.K. OxVALVE population ( Fig. 1.7 and Table 1.3 ). It is possible that patients volunteered for the U.K. OxVALVE study because of a known murmur or a cardiac symptom. The most common valve lesions were mitral regurgitation and aortic stenosis, and a similar prevalence of aortic stenosis was identified in Scandinavia. ,
| DATA BY AGE GROUP | |||||
| 18–44 | 45–54 | 55–64 | 65–74 | ≥75 | |
| Total participants | 4351 | 696 | 1240 | 3879 | 1745 |
| Mitral regurgitation | 23 (0.5%) a | 1 (0.1%) | 12 (1.0%) | 250 (6.4%) | 163 (9.3%) |
| Mitral stenosis | 0 | 1 (0.1%) | 3 (0.2%) | 7 (0.2%) | 4 (0.2%) |
| Aortic regurgitation | 10 (0.2%) | 1 (0.1%) | 8 (0.7%) | 37 (1.0%) | 34 (2%) |
| Aortic stenosis | 1 (0.02%) | 1 (0.1%) | 2 (0.2%) | 50 (1.3%) | 48 (2.8%) |
a Prevalence values are given as the number of cases (% of total participants in the age group).
| Valve Lesion | Mild | Moderate/Severe |
| Mitral regurgitation | 494 (19.8%) | 58 (2.3%) |
| Mitral stenosis | 7 (0.3%) | 2 (0.1%) |
| Aortic regurgitation | 341 (13.6%) | 41 (1.6%) |
| Aortic stenosis | 866 (34.6%) | 17 (0.7%) |
| Tricuspid regurgitation | 67 (2.7%) | |
| Pulmonary regurgitation | 7 (0.3%) |
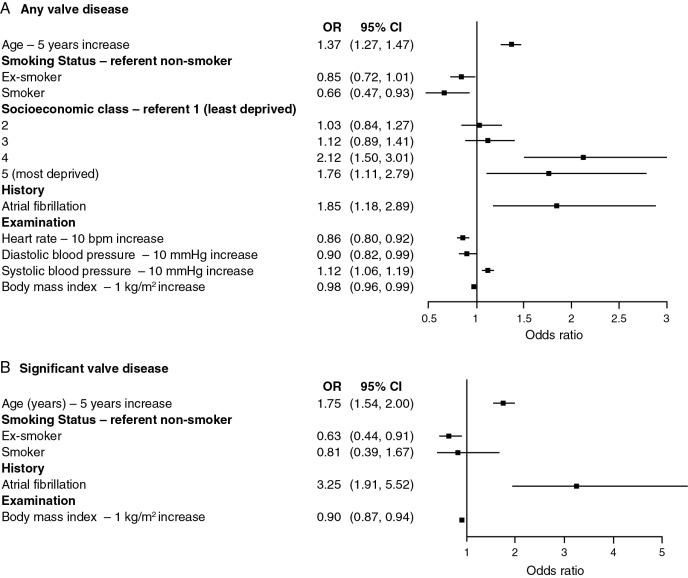
As expected, mild valve disease was more common, occurring in 44% of the OxVALVE population of 2500 community people (see Fig. 1.1 ). In the U.S. population-based studies, there were no gender differences for mitral valve disease or aortic regurgitation, but there was a trend ( P = 0.06) toward a higher prevalence of aortic stenosis among men, which became statistically significant ( P = 0.04) after adjustment for age (odds ratio = 1.52). This may be explained by bicuspid aortic valves being more common in men. The factors associated with valve disease included age, low socioeconomic class, and atrial fibrillation ( Fig. 1.7 ). , Atrial fibrillation was also associated with valve disease in a survey of open access echocardiograms.
Reports of selected patient groups inevitably record different results. In a study of 1797 men and women older than 60 years in long-term health care in the United States, there were 22 (1.2%) with mitral stenosis, 591 (33%) with mitral regurgitation, 301 (17%) with aortic stenosis, and 526 (29%) with aortic regurgitation. The EuroHeart Surveys I and II , recorded the causes of moderate and severe disease occurring mainly in Western and Eastern Europe in inpatients or in medical or surgical outpatient clinics. The mean age was 65 years (SD = 14 years), and age-related valve diseases were the most common causes ( Table 1.4 ). They included aortic stenosis (41% of the total), multiple valve disease (25%), and mitral regurgitation (21%) (see Table 1.4 ). Valve disease was reported for 29% of patients in the EuroHeart Failure survey.
| Cause | AS a ( n = 1197) | AR ( n = 369) | MS ( n = 336) | MR ( n = 877) |
| Degenerative (%) b | 81.9 | 50.3 | 12.5 | 61.3 |
| Rheumatic (%) | 11.2 | 15.2 | 85.4 | 14.2 |
| Endocarditis (%) | 0.8 | 7.5 | 0.6 | 3.5 |
| Inflammatory (%) | 0.1 | 4.1 | 0 | 0.8 |
| Congenital (%) | 5.4 | 15.2 | 0.6 | 4.8 |
| Ischemic (%) | 0 | 0 | 0 | 7.3 |
| Other (%) c | 0.6 | 7.7 | 0.9 | 8.1 |
a Aortic stenosis was defined by Vmax > 2.5 m/s, mitral stenosis by an orifice area < 2.0 cm 2 , and mitral or aortic regurgitation by the presence of ≥ 2/4 regurgitation.
b Defined as calcific aortic valve disease, mitral annulus calcification, and mitral prolapse. Subjects were recruited as inpatients or from medical or surgical outpatient clinics between April 1 and July 31, 2001, at participating hospitals, mainly located in western and eastern Europe. The mean age was 65 (SD = 14), and 16.8% were younger than 50, 50% were between 50 and 70, 30% were between 70 and 80, and 8.3% were older than 80 years.
c The overall frequency of right-sided disease was 42 (1.2%) and multiple valve disease was 713 (20%).
In the West, the frequency of chronic RhD fell after the 1950s. The proportion of aortic regurgitation of rheumatic origin was 62% in the period from 1932 to 1967, but it fell to 29% during the 1970–1974 period and 20% during the 1985–1989 period. Chronic RhD is still reported in residents of care homes in Russia and China, and the prevalence of RhD in nine urban and semi-urban provinces in China during the 2001–2002 period among middle-aged or older people using echocardiography was 186 cases in 100,000 people ( Fig. 1.8 ). Acute RhF continues to occur sporadically, such as in Utah, and the annual incidence is 30 cases per 100,000 p.a. for all attacks but 10 or fewer cases per 100,000 p.a. for first attacks.
The rational for detecting mild RhD is to give secondary antibiotic prophylaxis. However, there is no recognized treatment to reduce the rate of progression of mild nonrheumatic valve disease, which is increasingly detected when echocardiography is used for relatively nonspecific indications such as dizziness or palpitation or noncardiac chest pain. In a survey of open access echocardiography, mild valve disease of no hemodynamic significance was found in 8% of 1637 with no murmur. The natural history of mild disease is not as well known as that for moderate or severe disease, and there is a tendency for using surveillance, which carries the risk of causing health anxiety.
RhF occurs in children between the ages of 5 and 15 years. It usually results from the immune response to group A β-hemolytic streptococcal pharyngitis. The response occurs after 1 to 5 weeks and is caused by molecular mimicry of streptococcal M protein and human myosin and between the group A carbohydrate in the streptococci and valve tissue.
Genetically determined immune markers affect susceptibility to the initial infection and help determine the risk of developing chronic RhD. , HLA-DR7 has been the most frequently identified association. There is some evidence for a disordered signaling mechanisms and reactivation of the embryologic pathways. Some streptococcal serotypes ( emm- encoded types 3, 5, 6, 14, 18, 1 9, and 29) may be more likely than others to cause RhF. These host and bacterial factors vary geographically. There is evidence from native Australians that pyoderma (rather than pharyngitis) and group C and group G streptococci may have an important role.
RhF is uncommon after one episode of pharyngitis, but it occurs in up to 75% of people after recurrent episodes. Cardiac involvement occurs in 10% to 40% after the first attack of RhF and more frequently after multiple attacks.
A proliferative exudative inflammation of the collagen of the valve and annulus is characterized by modified histiocytes called Aschoff bodies. The valve, annulus, and chordae are edematous and inflamed, leading to annular dilation and chordal elongation and sometimes leading to mitral prolapse but otherwise to leaflet thickening and nodularity. These changes cause severe mitral regurgitation, which is the hallmark of acute RhF in the young. Much less is known about the mechanisms of aortic regurgitation, but there may be stretching of the cusps as a result of inflammation and subsequent prolapse.
The development of chronic RhD depends on the age at the time of the acute episodes, their severity and frequency, and the magnitude of the immunologic response. It is more likely with multiple valve involvement, failure to obtain medical help and lack of secondary prophylaxis ( Fig. 1.9 ). The mitral valve is affected in more than 90% of cases. Single valve involvement and mitral stenosis are more likely in older individuals with less active carditis. , Development of complications (e.g., atrial fibrillation, heart failure, stroke) occurs most commonly in the first year after an initial attack of RhF and more rapidly than after a second attack of RhF. This underlines the need for prevention. Complications and survival are also linked with comorbidities (e.g., alcohol use).
EMF is, after RhD, the second most frequent cause of acquired heart disease in Africa. It was originally described in the equatorial region of Africa, Uganda, Nigeria, and Ivory Coast, but with the use of echocardiography, it was identified more widely, including in Egypt and South Africa. EMF occurs predominantly in children and adolescents. It affects males and females in similar proportion, although there is a second peak in women of reproductive age. Echocardiography of a sample of 1063 people of all ages in coastal Mozambique found a prevalence of 20% (95% CI: 17.4%-22.2%).
EMF begins as a febrile illness that is followed by a latent phase of 2 to 10 years. Symptoms then reappear as left ventricular (LV) and right ventricular (RV) thrombi and fibrosis develop, leading to RV, LV, or biventricular restrictive cardiomyopathy and enmeshment of the posterior mitral leaflet or nonseptal tricuspid valve leaflets.
The pathology of EMF remains uncertain. There is evidence for the reactivation of embryologic pathways and postulated etiologic factors, which are not mutually exclusive and include the following:
Hypereosinophilia: Features are similar to hypereosinophilic syndromes, and the eosinophil count is transiently high in up to 30% of patients with EMF.
Infection: EMF may be associated with helminth infection, schistosomiasis, filariasis, and Mycoplasma pneumonia.
Autoimmunity: Immunoglobulin G (IgG) reactivity occurs to myocardial proteins.
Genetic predisposition: The incidence is high among some ethnic groups.
Diet: Eating uncooked cassava causes an EMF-like response in African green monkeys and may be relevant in humans, especially those eating protein-deficient diets.
Geochemistry: Increased levels of cerium are found in the hearts of some patients with EMF living near the coast of Mozambique.
The incidence of EMF in southern Nigeria has decreased. This is thought to result from improved health care delivery and better living conditions.
The incidence and severity of aortic stenosis increase with age by a process of active lipid deposition, inflammation, neoangiogenesis, and calcification (see Chapter 3 ). Aortic stenosis shares a number of risk factors with other atherosclerotic processes. Psoriasis has been shown to be an independent risk for coronary disease and aortic stenosis, possibly through inflammatory mechanisms.
Aortic valve sclerosis is defined by valve thickening and a peak transaortic velocity on echocardiography of less than 2.5 m/s. Approximately 20% of patients with sclerosis progress to stenosis within 10 years. , Aortic valve sclerosis is also associated with vascular disease and is a marker for a higher risk of myocardial infarction, particularly in patients with no established coronary disease or those with low conventional risk profiles, such as women or patients younger than 55 years (see Chapter 4 ).
Age-related calcification can affect the mitral annulus but rarely causes sufficient obstruction to require surgery, except occasionally in patients with chronic renal failure. If calcification is found at the aortic valve and mitral annulus and in the aorta, there is a significant likelihood of associated three-vessel coronary disease.
Aortic sclerosis is very common, with a population prevalence of 45% for those 65 years or older in Barcelona and 34% of those in Oxford. Prevalence is 73.5% among those older than 85 years. The population prevalence of moderate or severe aortic stenosis was 0.4% among those older than 18 years in the United States but 1.3% for those between the ages of 65 and 74 years and 2.4% for those older than 75 years. The Tromso study showed a higher prevalence of 1.3% for those between the ages of 60 and 69 years, 3.9% for those between 70 and 79 years, and 9.8% for those between 80 and 89 years of age. Two other studies , showed a prevalence rate between those in the Tromso and US studies. As expected from the age of the patients, aortic stenosis was identified in 8% of those admitted with a fractured hip.
The incidence is changing. In Sweden between 1989 and 2009, the crude incidence based on hospital coding remained approximately constant at between 19 and 26 cases per 100,000 people, but the age-adjusted incidence decreased from 15.0 to 11.4 cases per 100,000 men and 9.8 to 7.1 cases per 100,000 women. In contrast, an analysis of hospital coding over a 10-year period in Scotland showed an increased rate of admission, rising from 24.6 cases per 100,000 people in 1997 to 36.5 cases in 2005. These disparities may reflect differences in risk factor management.
Mitral prolapse is defined by abnormal systolic displacement into the left atrium of the whole or part of one or both leaflets. It is associated with myxomatous infiltration or fibroelastic deficiency of the leaflets. These histopathologic changes may coexist, , and although fibroelastic deficiency is more common in the elderly, it is not clear whether the two processes are genetically related or distinct.
Myxomatous degeneration causes leaflet thickening, annulus dilation, and abnormal chordae. The chordae are prone to stretching and rupture and may be deficient, particularly at the commissures or at the middle scallop of the posterior leaflet. Fibroelastic deficiency produces thin, smooth leaflets that are deficient in collagen, elastin, and proteoglycans and relatively mild annulus dilation and chordal lengthening.
Mitral prolapse may be sporadic or familial (i.e., syndromic and nonsyndromic). Genetic studies are based on small pedigrees that are subject to selection bias. Abnormalities of the gene encoding filamin A (FLNA) causes an X-linked mitral prolapse, but the genes responsible for the more common nonsyndromic autosomal dominant inheritance pattern are not known, although they are located on chromosomes 16, 11, and 13. Syndromic mitral prolapse is associated with Marfan syndrome and Ehlers-Danlos syndrome type IV, osteogenesis imperfecta, Loeys-Dietz syndrome, pseudoxanthoma elasticum, and aneurysms-osteoarthritis syndrome. It can be associated with mutations in several genes, including FBN1, TGFBR1 or TGFBR2 , and MADH3.
Methods of defining prolapse have been refined with advances in echocardiography and particularly with the realization that the mitral annulus is saddle shaped. Mitral prolapse is uncommon in children and young adults, but after the age of 30, the prevalence of prolapse using modern definitions is 2% to 3% , in unselected community populations, although the rate is higher in hospital series or for patients with syndromes associated with mitral valve prolapse or those with symptoms. Mitral prolapse is associated with tricuspid prolapse in 10% of cases or, rarely, with aortic valve prolapse.
Mitral regurgitation occurs in 6% to 9% of all patients with prolapse, , but 25% in the setting of leaflet thickening as a result of myxomatous degeneration. The grade of regurgitation depends on the degree of leaflet thickening and prolapse, and it is worse when ruptured chordae lead to flail or partially flail leaflet segments. In these cases, the mean 10-year survival rate without heart failure is only 37%. However, there is a low risk of progression if the prolapse is mild with no more than mild mitral regurgitation and a normal left atrial size in sinus rhythm, and for these patients, follow-up may not be required.
Use of the terms ischemic and secondary is not fully standardized. Ischemic regurgitation typically is used for acute ischemic mitral regurgitation as a result of papillary muscle rupture. This requires emergency surgery (see Chapter 17 ). Secondary mitral regurgitation is a chronic condition that primarily results from LV dysfunction, which causes altered stresses on the mitral valve apparatus and restriction of the leaflets. Dilation of the annulus may also occur. Leaflet restriction may be asymmetric when it affects predominantly the posterior leaflet. This is most commonly associated with an inferoposterior myocardial infarction. It may also be symmetric, which results from more generalized LV dysfunction.
LV dysfunction leading to functional mitral regurgitation results from the causes of heart failure, which vary geographically. Important causes are ischemic disease, hypertension, and alcohol. In the West, probably 20% of patients with chronic left ventricular systolic dysfunction have severe secondary mitral regurgitation, and 29% of patients in the EuroHeart Failure survey had valve disease with secondary mitral regurgitation as the most likely cause. Chagas disease and HIV are important in endemic areas. Chagas disease caused 383,900 cases of heart failure in 2013, which accounted for 0.6% of the global total and 11.4% in Brazil.
Functional aortic regurgitation results from dilation of the aortic root. Associated organic regurgitation often occurs as a result of a bicuspid aortic valve or arteriosclerosis. The risk factors for aortic dilation are age, weakness of the aortic wall, and the arteriosclerotic risk factors of hypertension, dyslipidemia, smoking, and diabetes. Weakness of the aortic wall as a result of medial necrosis occurs in Marfan syndrome and Ehlers-Danlos syndrome type IV.
Bicuspid aortic valve should be regarded as a general thoracic aortopathy, and it is associated with significant dilation of the aorta (to >40 mm) as a result of medial necrosis in about 20% of cases. Approximately one half affect the root, and the others affect the ascending aorta. Aortic dilation is more likely to be associated with coarctation, but dissection is relatively uncommon and has a relatively high operative success, probably because of the youth and underlying good health of the patient. Prophylactic surgery is necessary in about 5% of cases during a 20-year follow-up (see Chapter 11 ).
Vasculitides, especially giant cell arteritis and Takayasu arteritis, may weaken the arterial wall. Other causes of aortic dilation are trauma, cocaine, and amphetamines. In contradistinction to the usual symmetric fusiform dilation of a segment of aorta, a less common saccular aneurysm consisting of an outpouching of the aorta can result from inflammation from syphilis.
Tricuspid regurgitation is associated with rheumatic mitral disease or mitral prolapse. It may sometimes result from organic involvement of the valve or annulus, but more often, it is a result of RV dilation due to pulmonary hypertension. If not corrected, it progresses after left-sided surgery, recurring in 75% 3 years after repair for mitral prolapse. The 8-year mortality rate is 16% with tricuspid regurgitation, but it is only 5% without, and the effect of tricuspid regurgitation on survival is independent of LV systolic function. Secondary tricuspid regurgitation should therefore be repaired at the time of left-sided surgery, which provides freedom from severe tricuspid regurgitation for at least 10 years.
Tricuspid regurgitation induced by endocardially implanted electrical devices is a growing phenomenon as the use of pacemaker and defibrillator systems increases. New or worsening tricuspid regurgitation in the absence of infection of the system occurs in 20% to 32% of cases , and occurs more commonly with implantable defibrillators than pacemakers.
Significant, grade 2 or higher tricuspid regurgitation is associated with progressive enlargement of the right heart and a poorer outcome for patients over 1 to 1.5 years than for those with no or grade 1 tricuspid regurgitation. Tricuspid regurgitation does not resolve by changing the mode and does not depend on whether the RV lead is positioned at the apex or RV outflow tract. In a series of 41 patients requiring surgery for severe tricuspid regurgitation 6 years after pacemaker or defibrillator placement, tricuspid regurgitation was caused by lead impingement in 16, adherence in 14, perforation (usually of the septal leaflet) in 7, and entanglement in 4. Tricuspid replacement was required in 22, and annuloplasty was needed in 19. Fibrosis and adherence can occur as early as 17 days after implantation. Tricuspid regurgitation can also develop as a result of percutaneous removal of endocardial electrodes; a flail tricuspid leaflet occurred in 19 (9.1%) of 208 patients. Dense scarring causing tricuspid stenosis also occurs but is uncommon.
The estimated world incidence of IE in 2016 was 1,172,000 cases (95% CI: 1,068,000–1,280,000). The incidence is probably similar in poor and rich countries at 3.4 cases per 100,000 people. The estimated incidence in the West is 1.4 to 6.2 cases per 100,000. This increases to 14.5 episodes per 100,000 patients-years in those older than 70 to 80 years. , However, the epidemiology is different.
In the industrially underdeveloped regions, patients with endocarditis are young, and about one half to three fourths have rheumatic heart disease; the others have prosthetic valves. The risk is 1% at 1 year after diagnosis of RhD, 2% at 5 years, and 4% at 10 years. Health care–related IE is much less common than in the West. ,
In the West, the epidemiology is different from that in the underdeveloped world, and it has changed in the past 20 years. Patients are older and are more likely to have diabetes and immune suppression. There has been a reduction in the number with acquired heart disease and an increase in cases associated with health care, especially replacement heart valves, pacemakers, or hemodialysis. In the West, IE affects prosthetic valve in 20% to 30% of total cases, and the incidence of prosthetic valve endocarditis is 464 cases per 100,000 people in the United Kingdom (relative risk of 70, compared with the general population) and 600 cases per 100,000 people in Holland.
The major geographic variations depend largely on the frequency of medical devices used and on intravenous drug use. Hemodialysis-related IE is more common in the United States than in Europe (21% vs. 4%), with a lower use of native arteriovenous fistulas compared with Europe. , There is a male-to-female preponderance of 1.2 to 2.7:1, which is possibly related to bicuspid aortic valve being more common in men.
Oral streptococci are still the main infecting organisms in underdeveloped areas, but S. aureus is becoming more common. In the West, S. aureus was the predominant organism found in series reported from referral centers, which is likely related to a rise in health care–related IE and intravenous drug use. However, these series are biased to reporting severely ill patients, and population-based surveys of incidence report a gradual and statistically nonsignificant upward trend in the incidence of S. aureus –related IE from 0.52 in 1991 to 0.82 per 100,000 p.a. in 2008. At the same time, the incidence of oral streptococcal IE fell from 0.81 to 0.65 per 100,000 p.a. Some organisms have a regional focus. Brucella is rare outside the Mediterranean and Middle East. Bartonella occurs in 1% to 4.4% cases in France, Canada, and Brazil and in up to 7% of cases in India. Non-HACEK ( Haemophilus , Aggregatibacter , Cardiobacterium , Eikenella , and Kingella ), gram-negative bacilli are rare causes of IE globally but occur in 9% to 15% of cases in South Africa, India, and Pakistan.
A prospective cohort study of patients admitted to 58 hospitals in 25 countries allows comparison of the West and developing countries. Of 2781 adults with definite IE, most were from the United States ( n = 597 [21.5%]) or Europe ( n = 1213 43.6%), but there were 254 (9.1%) from South America and other countries ( n = 717 [25.8%]). The median age was 58, and 72.1% had native valve IE. Most patients (77.0%) presented within 30 days of disease onset and had few of the classic clinical hallmarks of IE. S. aureus was the most common pathogen (31.2%) overall but occurred in 43% of cases in the United States, compared with 17% in South America. Oral streptococcal infection occurred in 26% in South America and in only 9% in the United States.
In the same study, RhD occurred in less than 5%. Health care associated–IE occurred in 25% overall but 38% in the United States and only 20% in South America. The in-hospital mortality rate in the United States was 17.7%, similar to that in the underdeveloped world, and stroke occurred in 17% of patients and heart failure in 32%. Surgery was performed in 48% of patients, a similar rate to that in the underdeveloped world.
During recent years, use of antibiotic prophylaxis has diminished, with antibiotic prophylaxis before invasive dental procedures administered only in the setting of high-risk cardiac conditions such as replacement valves, prior endocarditis, or corrected adult congenital heart disease using valve conduits. These indications comply with the European Society of Cardiology (ESC), American Heart Association (AHA), and Australian guidelines, , , with the addition of RhD in indigenous Australians in the 2008 Australian guidelines and valve disease in transplanted hearts in the United States.
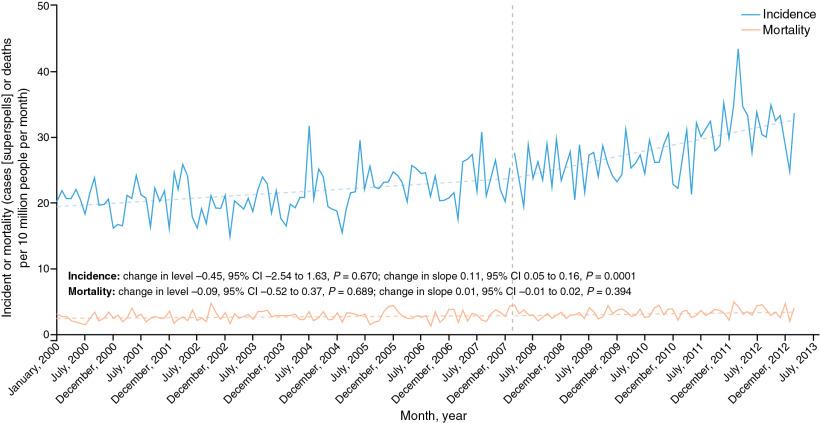
Although there has been a gradual increase in the incidence of IE, 192 changes in the ESC and AHA guidelines have not affected this, , but two studies in the United States and Germany show an increase in incidence since guidelines changes about antibiotic prophylaxis were made. The U.S. study suggested an increase in the incidence of streptococcal IE, but the study did not ascertain whether these cases were oral. In the United Kingdom, the 2008 National Institute for Health and Care Excellence (NICE) guidelines recommended the withdrawal of all antibiotic prophylaxis in all groups for all procedures. There is evidence of a rise in the incidence of IE beginning with the change in the guidelines, with an estimated 35 additional cases per month than would have been expected before the change in guidelines ( Fig. 1.9 ). , NICE has subsequently softened its recommendations and now advises that antibiotic prophylaxis can be given at the discretion of the physician, effectively bringing it in line with ESC and U.S. guidelines.
Replacement valves are a treatment for valve disease but introduce a risk of structural valve deterioration, nonstructural deterioration, thrombosis or thromboembolic events, infection, or bleeding. The pattern of use of replacement valves is different in developed and underdeveloped regions, and 85% of valves are used for 11% of the world population. In the underdeveloped world, safe anticoagulation may not be feasible. Repair is therefore performed even in patients with RhD in preference to the implantation of a replacement mechanical valve. Results may be good, with a 90% survival rate at 10 and 14 years, compared with a 79% survival rate at 10 years and only a 44% survival rate at 14 years for valve replacement using mainly mechanical valves.
Poor international normalized ratio (INR) control was thought to explain the 2.17 times increased risk of dying with a mechanical than a biological valve and the 8.45 times higher risk for a Maori or 6.54 times higher risk for a Pacific Islander compared with white patients in New Zealand. A biological valve may be used in a young person to avoid anticoagulation despite the known increased risk of early structural degeneration compared with use in older people. Reoperation for structural degeneration of biological replacement valves is a more common reason for surgery than in industrially developed areas, occurring in 41% of operations in Sao Paolo.
However, there is a strong trend toward using biological valves in younger patients in the United States and United Kingdom. Despite guideline recommendations to implant mainly mechanical replacement aortic valves in patients younger than 60 years (see Chapter 14 ), the proportion of isolated aortic valve replacements using biological valve in people between 56 and 60 years of age increased from 25% in 2004 to 40% in 2008. , This discrepancy partly reflects the perceived longer durability of third-generation biological valves coupled with the possibility of a valve-in-valve transcatheter procedure to treat primary failure.
In the United States, 31% of adult-sized children suitable for a mechanical valve between 2009 and 2013 elected to have a biological valve. However, durability is limited in younger patients, particularly those younger than 40 years. , In a series from Ottawa, the rate of 10-year freedom from reoperation for aortic replacements was 50.9% for patients younger than 40 years at implantation, compared with 99.7% for those older than 60 years. As a result, the need for redo procedures is increasing. In the United Kingdom, 7% of aortic valve operations are redo procedures. Defining valve deterioration echocardiographically rather than by the need for a redo procedure suggests that failure in some designs of valve is more common than previously thought.
Congenital lesions account for approximately 5% of valve operations throughout the world. Bicuspid aortic valve is the most common abnormality and affects up to 2.0% of the population based on autopsy series and 0.5% to 0.8% in larger population-based studies. , There is evidence of geographic clustering of cases, which is likely a result of genetic factors , because the risk of a bicuspid aortic valve or aortic disease is about 10% among first-degree relatives of probands. , The male-to-female ratio is approximately 2:1. The valve is anatomically or truly bicuspid in one third of cases and functionally bicuspid in two thirds as a result of incomplete separation of two cusps in utero. In 80% of cases, the pattern is failure of right-left separation, which is more likely to be associated with aortic dilation. Failure of separation of right and noncoronary cusps is more likely to be associated with mitral prolapse.
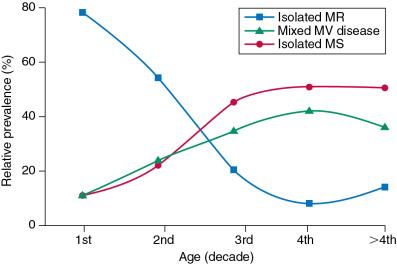
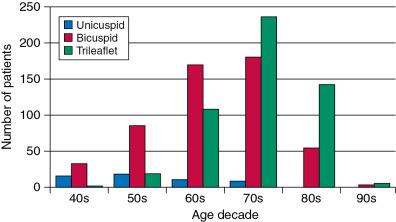
During a 20-year follow-up, 24% of patients with a bicuspid aortic valve developed severe stenosis or regurgitation requiring surgery. , Events are far more common in those with even mild valve thickening at baseline, with surgical rates of 75% at 12 years in the setting of thickening, compared with only 8% for those without valve thickening. Because the young are more likely to have surgery, the frequency of bicuspid disease is about one third of unselected surgical cases. However, detailed pathologic examinations of surgically excised valves reported that among patients with bicuspid valves and aortic stenosis having surgery, 67% were in their 40s and 28% were octogenarians ( Fig. 1.10 ).
Congenital mitral disease is uncommon. Congenital malformations , that affect the valve are double orifice, isolated cleft (without atrioventricular septal defect), dysplastic (associated with hypoplastic left heart), and Ebstein anomaly of the mitral valve. Those affecting the chordae are arcade mitral valve caused by unusually short chordae, and straddling mitral valve with chordae attached to both ventricles. In parachute mitral valves, the chordae tendineae are attached to only a single papillary muscle origin.
Submitral aneurysms occur in sub-Saharan African populations and are thought to be caused by a congenital weakness of the mitral annulus. The aneurysms arise in the posterior annulus and are often loculated and serpiginous, and they bulge in an extracardiac direction or into the left atrium. They occasionally occur under the aortic valve, and some may be tuberculous in origin.
Endocardial involvement is relatively common in systemic lupus erythematosus (SLE), particularly in patients with antiphospholipid antibodies. , However, it is usually subclinical. Symptomatic, significant valve disease occurs after recurrent valvulitis. Subendothelial deposition of immunoglobulins and complement cause proliferation of blood vessels, inflammation, thrombosis, and fibrosis.
Fusion of the mitral valve commissures may lead to stenosis, but more commonly, there is generalized thickening of the leaflets (30% to 70%) with regurgitation (30% to 50%). , , Libman-Sacks vegetations are usually less than 10 mm in diameter, sessile, of mixed echogenicity, and usually rounded. They may occur anywhere, but they most commonly occur at the leaflet edges on the atrial surface of the mitral valve and, less frequently, occur on the ventricular side of the aortic valve. The right-sided valves are rarely affected. Active vegetations have central fibrinoid degeneration with fibrosis and inflammatory infiltrate, whereas healed vegetations have central fibrosis with little or no inflammation.
Valve lesions may occur in the absence of features of SLE in the antiphospholipid syndrome. Antiphospholipid antibodies cause the following:
Activation of endothelial cells
Increased uptake of oxidized low-density lipoprotein (LDL), leading to macrophage activation
Interference with regulatory functions of prothrombin and decreased production of protein C and S
Rheumatoid arthritis causes an immune-complex valvulitis with infiltration of plasma cells, histiocytes, lymphocytes, and eosinophils that leads to fibrosis and retraction. Nodules consist of central fibrinoid necrosis surrounded by mononuclear cells, histiocytes, Langhans giant cells, and a border of fibrous tissue. The nodules are 4 to 12 mm in diameter and develop at the base of the mitral or aortic valves. Occasionally, there may be more generalized valvulitis. Healed valvulitis leads to leaflet fibrosis and retraction, causing regurgitation.
Ankylosing spondylitis is associated with HLA-B27–mediated chronic inflammation and proliferative endarteritis of the aortic root and left-sided valves. This commonly causes the following:
Aortitis of the aortic root leads to thickening, dilation, and functional aortic regurgitation.
Aortic valvulitis occurs with thickening of the leaflets and cusp retraction.
Downward displacement of the aortic root leads to a subaortic bulge at the base of the anterior mitral leaflet. This causes retraction of the anterior mitral leaflet with reduced coaptation.
The frequency of valve disease is uncertain because reported series are small and tend to be biased toward those with severe disease. Aortic valve thickening has been reported in 40%, mitral valve thickening in 34%, and significant aortic dilation in 25%.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here